Abstract
Background:
Cardiac troponins (cTn) are reliable and the most sensitive biomarker in the setting of acute decompensated heart failure (ADHF). Acute decompensated heart failure is usually associated with worsening chronic heart failure, and it may be caused by ongoing minor myocardial cell damage that may occur without any reported precipitating factors.
Methods:
We compared the short-term effect of levosimendan (LEV), dobutamine (DOB), and vasodilator treatment (nitroglycerin [NTG]) on myocardial injury with hemodynamic, neurohumoral, and inflammatory indicators. One hundred twenty-two patients with a mean age of 66 ± 9 years were treated with LEV (n = 40), DOB (n = 42), and NTG (n = 40) and examined retrospectively. Blood samples (cTnI, N-terminal probrain natriuretic peptide [NT-proBNP], highly sensitive C-reactive protein [HsCRP], and others), left ventricular ejection fraction (LVEF), systolic pulmonary artery pressure (sPAP), and 6-minute walk distance (6MWD) were compared before and after treatment.
Results:
At admission, detectable levels of cTnI were observed in 53% of patients (≥0.05 ng/mL). Serial changes in the mean cTnI levels were not significantly different between the groups (LEV 0.04 ± 0.01 to 0.03 ± 0.01 ng/mL; DOB 0.145 ± 0.08 to 0.08 ± 0.03 ng/mL; NTG 0.1 ± 0.03 to 0.09 ± 0.02 ng/mL; overall P = .859). Favourable effects on the NT-proBNP, sPAP values, LVEF, 6MWD, and HsCRP were observed overall, especially in the LEV groups.
Conclusion:
Beneficial effects of short-term use of LEV, DOB, and NTG on ongoing myocardial injury were demonstrated. These findings can be attributed to the anti-ischemic properties as well as the hemodynamic, neurohumoral, and functional benefits from the positive inotropes, especially LEV, in patients with ADHF.
Introduction
There is increasing evidence that cardiac troponins (cTn) are reliable and sensitive biomarkers for identifying high-risk patients and good prognostic markers in the setting of acute decompensated heart failure (ADHF). 1 -4 Ongoing myocardial ischemia, inflammation, and apoptosis are associated with myocyte degeneration in the cardiomyopathic myocardium, and they may be responsible for worsening heart failure and cTn release. 5
Positive inotropic treatment may be given to patients who are refractory to optimal medical treatment and intravenous diuretics or who have evidence of low cardiac output, such as hypotension or hypoperfusion in patients presenting with ADHF.
Dobutamine (DOB) is a potent inotropic and weak chronotropic agent that acts on the β1-2 and α1 adrenergic receptors. 6 Levosimendan (LEV) is a parenteral inodilatory and so-called calcium-sensitizing agent used for treating ADHF. In contrast to DOB, it is suggested that LEV does not change the intracellular calcium levels or oxygen consumption, and it may also exhibit protective properties against cardiac ischemia, inflammation, and apoptosis. 7 -10 On the other hand, positive inotropic therapy, including LEV and DOB treatment, may have adverse hemodynamic, intracellular, and arrhythmogenic effects and they should be used carefully in this patient-group. 11 -13
This retrospective study was designed to elucidate the effects of LEV, DOB, and nitroglycerin (NTG) treatment on ongoing myocardial injury as well as hemodynamic, neurohumoral, and inflammatory indicators in patients with ADHF.
Methods
Study Population
The patients who were admitted to the hospital of Osmangazi University (Eskişehir, Turkey) between June 2008 and May 2010 and those examined retrospectively were included in the study. A total of 122 patients with ADHF included in the study with a mean age of 66 ± 9 years were treated with LEV, DOB, or NTG; all patients had received standard heart failure therapy including optimal oral medication, diuretics and left ventricular ejection fraction (LVEF) of 35% or less as measured by transthoracic echocardiography. LEV or DOB infusion was administered to patients who have evidence of low cardiac output such as peripheral hypoperfusion and hypotension or who are refractory to optimal medical treatment according to the clinician’s decision. NTG was administered preferably to patients who have had hypertension or normotension with pulmonary and/or peripheral edema in patient with ADHF. In patients presenting with restrictive or hypertrophic cardiomyopathy, uncorrected valvular disease, cardiogenic shock, sepsis, severe hepatic impairment, severe renal impairment (glomerular filtration rate <30 mL/kg/1.73 m2), chest pain or electrocardiogram changes with increased cTn values in serial measurement, cTnI levels of ≥5× cutoff, second and third-degree atrioventricular block, coexistence of acute respiratory distress syndrome, recently with a history of cardiac arrest, and any positive inotropes received last 4 weeks were excluded.
Study Design and Protocol
In all patients, cTnI, N-terminal probrain natriuretic peptide (NT-proBNP), highly sensitive C-reactive protein (HsCRP), echocardiographic estimation of LVEF, systolic pulmonary artery pressure (sPAP) estimated by Doppler echocardiography, 6-minute walk distance (6MWD), and other laboratory findings were recorded before and after treatment. The mean time of follow-up postinitiation of treatment was 48 hours. In the all patients had been received of blood plasma samples before treatment and postinitiation of 3 days. LEV with a loading dose of 6 to 12 µg/kg over 10 minutes and followed by a continuous infusion of 0.1 to 0.2 µg/kg/min was administered. When there were adverse clinical effects, the dose was reduced to 0.05 µg/kg/min. In the patients who have had received LEV, 5 patients of those had reduced dose of 0.05 µg/kg/min temporary. Dobutamine was infused at a rate of 10 µg/kg/min for 48 hours. Initial dose of NTG was 25 µg/kg/min and followed by a continuous infusion up to 100 µg/kg/min when the dosage adjusted according to blood pressure for 48 hours.
The baseline demographic, clinical, and hemodynamic characteristics were recorded. Noninvasive measurements of the blood pressure and heart rhythm during treatment were noted. The results of invasive procedures that were performed at the coronary care unit or catheterization laboratory were recorded and compared.
Cardiac Troponin I Analysis
The control group consisted of healthy volunteers who were the same age and gender in our clinic. Values of multiple cTnI analyses of patients (Siemens Dimension cTnI Heterogeneous Immunoassay Module) were recorded. The 99th percentiles of the cTnI levels were established for optimal precision, and the cTnI reference ranges were determined. 14 Maximum 10% coefficients of variation were applied to this level; in this way, the cTnI reference intervals were determined with a lower limit of detection of 0.05 ng/mL and upper reference limit of 0.1 ng/mL (cutoff).
Statistical Analysis
The statistical analysis was performed using IBM SPSS Statistics for Windows (version 20). The distribution forms of the data were tested by Kolmogorov-Smirnov and Shapiro-Wilk tests. Data for cross-sample comparisons were analyzed by χ2 tests and correlations. Comparison of the average of the data was examined by t tests, and 1-way analysis of variance and Tukey or Dunn test were used for post hoc analysis. Mean ± standard error values were used to summarize the data, and P < .05 was considered as statistically significant.
Results
LEV (n = 40), DOB (n = 42), and NTG (n = 40) groups were compared in the study. Baseline clinical and demographic characteristics are shown in Table 1. At admission, no significant differences were observed between the groups in terms of these characteristics.
Baseline and Clinical Characteristics of Patients at Admission.
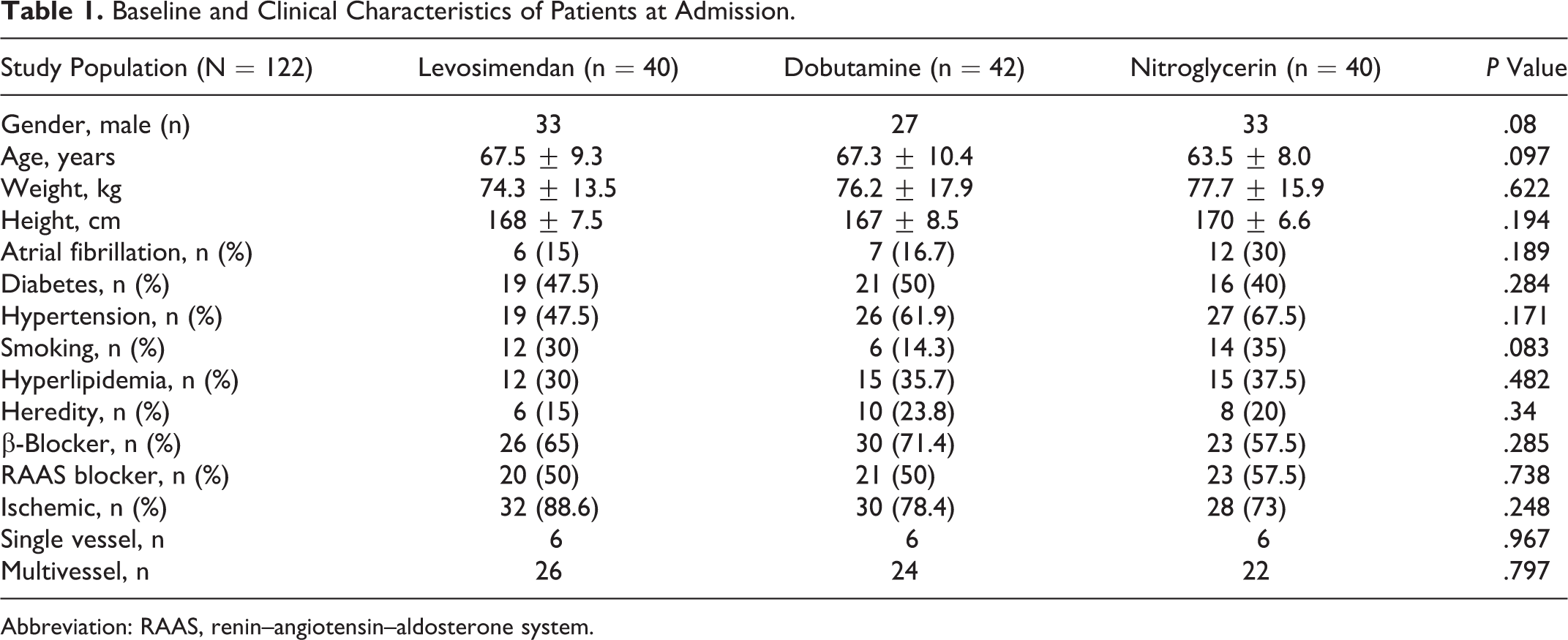
Abbreviation: RAAS, renin–angiotensin–aldosterone system.
Cardiac troponin Levels at Admission and After Treatment
At admission, 53% of patients were found to exceed the lower limit of detection of cTnI. The percentages of patients with increased, decreased, and unchanged cTnI levels after treatment were 36%, 44%, and 20%, respectively. After treatment in all patients, decreasing trends were observed in the mean cTnI levels, but statistically significant differences were not found. After the treatment, no statistically significant effect was observed with respect to serial changes in the cTnI between the groups (overall P = .859; Figure 1).
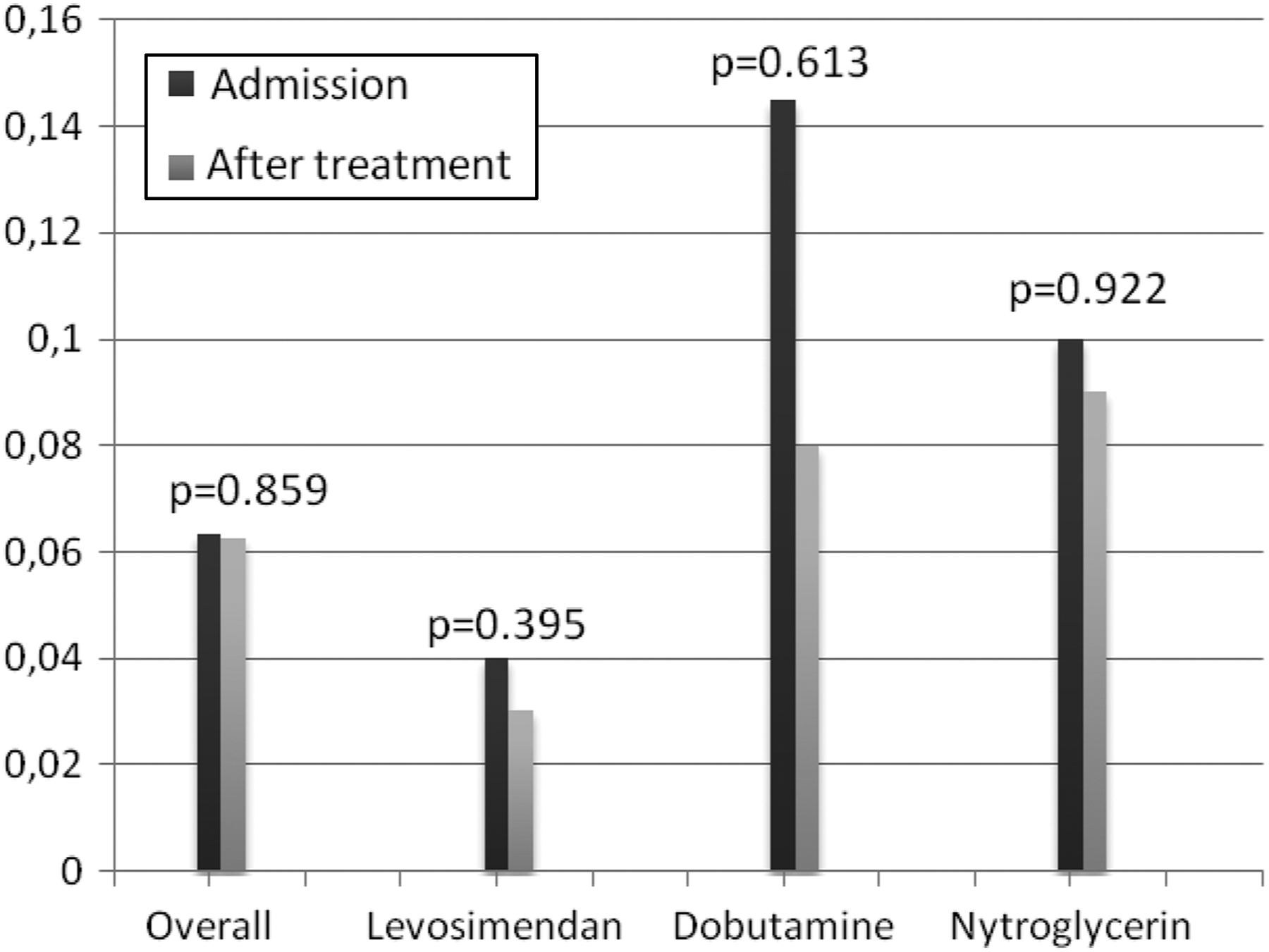
Mean cTnI levels at admission and after treatment in all groups. cTnI indicates cardiac troponin I.
Neurohumoral and Inflammatory Response
Overall, the mean NT-proBNP levels at admission of 9057 ± 1052 pg/mL and after treatment of 5930 ± 804 pg/mL were observed (P < .001). In the post hoc analysis, no statistically significant differences were found between the groups (P = .207). Similarly, no significant differences were found in terms of the mean change in HsCRP (P = .487) as well as total white blood cell count and its subtypes (P = .173).
Hemodynamic and Functional Indicators
In each group with more pronounced LEV, statistically significant increases were found in the LVEF after treatment. In post hoc analysis, the LVEF increase was significantly higher for LEV and DOB compared to NTG (LEV ≡ DOB > NTG, <.001). In each group, the sPAP values were significantly decreased after treatment. This decrease tended to be greater in the LEV group. Overall, statistically significant increases were observed in terms of 6MWD after treatment, but no statistically significant difference was found between the groups (P = .087; Table 2). In all patients, statistically significant increases were observed in the mean heart rate after treatment (77.5 ± 15 vs 84 ± 16, P < .001), but no statistically significant differences were found between the groups (P = .124).
Changes in Clinical and Laboratory Variables Following Heart Failure Therapy.

Abbrevaitions: HF, heart failure; HsCRP, highly sensitive C-reactive protein; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal probrain natriuretic peptide; PLT, platelets; sPAP, systolic pulmonary arterial pressure; WBC, white blood cell; 6MWD, 6-minute walk distance.
Discussion
ADHF is a clinical syndrome with increasing incidence that is associated with frequent hospitalizations and high morbidity and mortality. Although many different medications are used in the effective treatment of acute and chronic heart failure, inotropes may be required to improve hemodynamic stabilization, peripheral perfusion, and cardiac output. 15 -17 However, they should be used carefully because of safety concerns, such as myocardial ischemia, atrial and ventricular tachyarrhythmias, hypotension, and mortality risk. 18 -23
Many clinical studies on ADHF indicate that LEV improves myocardial function, reduces the cTn levels, and suppresses inflammation and apoptotic indicators of both intense myocardial ischemia and stunned myocardium after myocardial infarction. 24 -27 In addition, lower levels of cTn release during LEV administration and improving myocardial performance indicators have been reported. 28 -30
An association between DOB and myocardial injury has rarely been studied in patients with ADHF. DOB increases the myocardial contractility, reducing the peripheral vascular resistance and thus improving the cardiac output without seriously affecting the blood pressure or sympathetic tone. 31 In the pilot randomized study of nesiritide versus dobutamine in heart failure (PRESERVED-HF) study, some patients developed cTn release during hospitalization with DOB treatment. 5 In several studies that used DOB, there was no significant release of cTn even for patients with a diffuse coronary artery disease or previous myocardial infarction. 32 -34
At admission, detectable cTnI levels were found in 53% of our patients. No significant changes were observed with the mean cTnI levels after positive inotropic therapy or NTG. In the present study, we observed that high levels of cTnI can initially be detected regardless of the etiology of cardiomyopathy and they tend to decrease, but did not change significantly, after ADHF treatment. In inotropes, but more pronounced in the LEV group, statistically significant improvements were observed in the LVEF and 6MWD, and the NT-proBNP and sPAP levels were more effectively decreased. Overall, no significant differences were observed in the inflammation indicators after treatment.
Recent studies suggest that positive inotropic therapy is not associated with adverse events if eligible patients are treated. 35 This may be associated with different causes, such as anti-ischemic properties, hemodynamic, neurohumoral, and functional benefits of positive inotropes, especially LEV. 36 Furthermore, in previously reported studies, associations between adverse events and using positive inotropes have not inspired widespread use of implantable cardioverter defibrillators (ICDs) or the fact that the vast majority of mortality depends on the sudden cardiac death. 37 In the vicious cycle of congestive heart failure and its progressive nature, it is already associated with adverse outcomes in the absence of optimal medical therapy, complete revascularization, and treatment of comorbidities, which is exacerbated if an ICD is not implanted per the current recommendations. Moreover, hospital admission with serious clinical conditions, such as cardiogenic shock, pulmonary edema, or end-organ dysfunctions caused by low cardiac output, can be treated with commonly used positive inotropes, which may be considered a lifesaving feature of ADHF. 38
Conclusion
The present results indicate that beneficial effects were shown by the short-term infusion of LEV, DOB, and NTG on ongoing myocardial cell injury, which is considered as the main reason of worsening heart failure. However, improvement in hemodynamic, neurohumoral, and functional parameters is more pronounced with LEV than those with DOB and NTG. In addition, LEV, DOB, and vasodilator treatment might be reliable options in the short term with eligible patients having ADHF are treated. Further studies are needed to support this conclusion.
Footnotes
Author Contributions
Volkan Doğan contributed to conception, design, and analysis. Müjgan Tek Öztürk, Aydın Nadir, Ahmet Musmul, and Yüksel Cavuşoğlu contributed to conception. Yüksel Cavuşoğlu contributed to design. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
