Abstract
The use of aspirin and a P2Y12 receptor antagonist as dual antiplatelet therapy (DAPT) has become the treatment of choice in patients with acute coronary syndrome (ACS) and percutaneous coronary intervention to prevent recurrent thrombotic events. Although DAPT is beneficial for most patients, few patients still experience recurrent thrombotic events and increased bleeding episodes. This article reviews current literature to identify various approaches that may enhance the DAPT benefit-risk ratio in patients with ACS. Three strategies addressed in this article include the following—(1) use of more potent antiplatelet agents other than clopidogrel; (2) addition of direct oral anticoagulants (DOACs) to DAPT; and (3) optimizing DAPT duration. Although the use of prasugrel or ticagrelor improves treatment efficacy, their use has been associated with increased risk of bleeding compared to clopidogrel. The combination of DOACs and DAPT may not be the most viable strategy because of its limited cardiovascular benefits and increased bleeding risks. The optimal duration of DAPT duration remains controversial. Most guidelines recommend 6 to 12 months of DAPT, whereas emerging studies have shown benefit for both shorter and longer duration of treatment. Risk stratification tools such as the DAPT score may play a role in individualizing DAPT duration according to patient’s ischemic and bleeding risks.
Keywords
Introduction
For many years, aspirin was found to significantly reduce cardiovascular events in patients diagnosed with a variety of coronary conditions. Its use as monotherapy following acute coronary syndrome (ACS) or percutaneous coronary intervention (PCI) has been associated with high rates of stent thrombosis and recurrent myocardial infarction (MI). The addition of clopidogrel to aspirin therapy has shown to significantly improve ACS outcomes when compared with aspirin alone. 1,2 Since 2001, the use of aspirin along with a P2Y12 receptor antagonist known as a dual antiplatelet therapy (DAPT) is the treatment of choice in patients with ACS and PCI to prevent recurrent thrombotic events. 3,4 Although DAPT has shown to have beneficial effects, few patients still experience recurrent thrombotic events and are at risk of increased bleeding episodes. 2,5 To improve DAPT efficacy and safety among patients with ACS with drug-eluting stents (DESs), several approaches have been investigated to achieve this goal. In this review article, 3 strategies will be discussed and supported with evidence on the usefulness, efficacy, and safety of DAPT (Table 1). The 3 strategies include the following—(1) use of potent antiplatelet agents other than clopidogrel, (2) addition of direct oral anticoagulants (DOACs) to DAPT, and (3) optimizing the duration of DAPT (Figure 1).
List of Clinical Studies That Investigated Efficacy/Safety of Potential Strategies to Improve DAPT.

Abbreviations: ACS, acute coronary syndrome; DAPT, dual antiplatelet therapy.

Antiplatelet treatment algorithm for patients with ACS treated with PCI. ACS indicates acute coronary syndrome; ASA, aspirin; DAPT, dual antiplatelet therapy with aspirin and clopidogrel; DES, drug-eluting stunt; DOACs, direct oral anticoagulants; PCI, percutaneous coronary intervention.
Use of More Potent Antiplatelet Agents Instead of Clopidogrel
Clopidogrel has been the mainstay P2Y12 receptor antagonist agent used with aspirin for years. Aspects such as delayed onset of action, drug resistance potential, and significant interpatient variability have placed limitations on using clopidogrel for all patients. 6 Recently, potent P2Y12 receptor antagonists (prasugrel and ticagrelor) were shown to be more appealing than clopidogrel in the setting of ACS and PCI. Prasugrel and ticagrelor generally attain higher degrees of platelet inhibition than clopidogrel and are not affected by CYP2C19 polymorphism. 7 ∓9 The CYP2C19 polymorphism is known to be one of the major contributing factors to clopidogrel resistance when used in patients with high-risk ACS. 10 Despite this effect, the utilization of an algorithm for genetic-guided P2Y12 receptor antagonist therapy has not yet been considered as standard clinical practice. 11
Prasugrel’s efficacy and safety have been evaluated in the TRITON-TIMI 38 trial where patients with ACS scheduled for PCI were randomized to either prasugrel or clopidogrel. 12 Prasugrel was shown to have superior efficacy outcomes (death from cardiovascular causes, nonfatal MI, or nonfatal stroke) when compared to clopidogrel (hazard ratio [HR]: 0.81; 95% confidence interval [CI]: 0.73-0.9; P < .001) but at the expense of increased rate of major bleeding (HR: 1.32; 95% CI: 1.03-1.68; P = .03). However, the net clinical benefit was still in favor of the prasugrel group which led to its approval in 2009. 13 Prasugrel is contraindicated in patients with high risk of bleeding and in those with a history of stroke or transient ischemic attack (TIA). It is also not recommended in patients with ACS who are not undergoing PCI, those with stable coronary artery disease (CAD) undergoing PCI, low-body weight (<60 kg), and elderly patients (>75 years). 7
Conversely, ticagrelor is a reversible inhibitor of P2Y12 receptor with a half-life of about 12 hours and requires twice daily dosing due to its fast offset of action. 7 Ticagrelor was examined against clopidogrel in the PLATO trial and was shown to have superior efficacy (HR: 0.84; 95% CI: 0.77-0.92; P < .001) and increased risk of major bleeding not related to coronary artery bypass grafting (CABG) (HR: 1.19; 95% CI: 1.02-1.38; P = .03) in patients with ACS. 14 Ticagrelor should be avoided in patients with high risk of bleeding and in patients with severe hepatic dysfunction. When used along with aspirin, the dose of aspirin should be 100 mg or less. 15 This precaution is based on a post hoc subgroup analysis of the PLATO trial where patients from North America performed better with the clopidogrel arm than the ticagrelor arm. The only significant difference noticed between patients from North America and the remaining cohort was the use of higher aspirin dose.
As prasugrel and ticagrelor have been shown to be more efficacious than clopidogrel when used in DAPT, switching clopidogrel to prasugrel or ticagrelor may be a plausible option in patients with high-risk ACS such as those with multiple recurrent events, extensive CAD, and diabetes mellitus. Careful assessment of the patient’s bleeding risk is important to consider prior to the initiation of these agents. Factors that may increase the risk of bleeding include history of bleeding, history of stroke/TIA, oral anticoagulation therapy, advanced age, diabetes mellitus, chronic kidney disease, anemia, and chronic use of steroids or nonsteroidal anti-inflammatory therapy. 11
Addition of DOACs to DAPT
The addition of oral anticoagulation to antiplatelet medications in patients with ACS and PCI and without an indication for oral anticoagulation has been studied extensively. The use of warfarin along with aspirin has been shown to reduce recurrent ischemic events when compared to aspirin alone or aspirin plus clopidogrel but at the expense of increased bleeding. 16 ∓18 New oral anticoagulants, which are known as DOACs, have been introduced into the market as an alternative to warfarin for stroke prevention in patients with atrial fibrillation and for the treatment and prevention of venous thromboembolism. Dabigatran, which acts by directly and selectively inhibiting thrombin, was the first approved DOAC in 2010 and was then followed by 3 factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). 19 ∓21 The addition of dabigatran, rivaroxaban, and apixaban to DAPT in patients with ACS has been explored given their optimal efficacy and safety profile. In the Re-DEEM study, the addition of a range of doses of dabigatran or placebo to DAPT post-ACS was assessed in 1861 patients. 22 This phase II study indicated dabigatran doses of 50, 75, 110, and 150 mg were associated with dose-dependent increases in bleeding events when compared to placebo (HR: 1.77; 95% CI: 0.7-4.5) for 50 mg; (HR: 2.17; 95% CI: 0.88-5.31) for 75 mg; (HR: 3.92; 95% CI: 1.72-8.95) for 110 mg; and (HR: 4.27; 95% CI: 1.86-9.81) for 150 mg. There was no significant difference in the cardiovascular efficacy outcome among the treatment groups. On the other hand, low-dose rivaroxaban (2.5 mg twice a day or 5 mg twice a day) was compared to placebo in 15 526 patients with ACS receiving DAPT in the ATLAS ACS 2–TIMI 51 study. 23 Both rivaroxaban doses were shown to significantly improve the primary cardiovascular efficacy outcomes (9.1% vs 10.7%, HR: 0.84, 95% CI: 0.72-0.97) for the 2.5-mg dose and for the 5-mg dose (8.8% vs 10.7%, HR: 0.85, 95% CI: 0.73-0.98). The rivaroxaban of 2.5-mg dose showed significant reductions in cardiovascular mortality (2.7% vs 4.1%, HR: 0.66, 95% CI: 0.51-0.86) and in all-cause mortality (2.9% vs 4.5%, HR: 0.83, 95% CI: 0.72-0.97). However, the rivaroxaban group had significantly higher intracranial hemorrhage (0.6% vs 0.2%, HR: 3.28, 95% CI: 1.28-8.42) and major TIMI bleeding (2.1% vs 0.6%, HR: 3.96, 95% CI: 2.46-6.38) with no significant increases in fatal bleeding. Based on these results, the use of rivaroxaban in patients with ACS has only been approved by the European Medicines Agency. 24 Reasons for lack of approval for rivaroxaban’s new indication by the Food and Drug Administration include the following—(1) the marginal P value in the primary outcome results (P = .02) for 2.5 mg and (P = .03) for 5 mg, (2) concerns about incomplete follow-up, (3) lack of genetic testing for CYP2C19 variants and use of proton pump inhibitors that are associated with impaired conversion of clopidogrel to its active metabolite, and (4) the absolute increase in TIMI major non-CABG bleeding in the rivaroxaban arm by about 1% per year. In addition, rivaroxaban did not address an unmet medical need. Even if rivaroxaban was approved, it still could not be used with the more potent antiplatelet agents because of the high risk of bleeding of such combination. While the use rivaroxaban along with clopidogrel may be an alternative to prasugrel or ticagrelor alone the superiority of this combination to the more potent antiplatelets is still questionable. 25
Finally, different doses of apixaban were compared to placebo in patients with ACS receiving DAPT in the APPRAISE trial. 26 In this study, both apixaban of dose 10 mg twice daily and apixaban of dose 20 mg once daily arms were discontinued due to excessive bleeding. The 2 other doses of apixaban (2.5 mg twice daily and 10 mg once daily) also showed a dose-related increase in bleeding but with a trend toward improvement in cardiovascular ischemic events. In the APPRAISE-2 study, apixaban of dose 5 mg twice daily failed to show significant improvements for the efficacy outcomes and resulted in excessive TIMI major bleeding (1.3% vs 0.5%, HR: 2.59, 95% CI: 1.5-4.46, P = .001). 27 A decision to end the trial prematurely was made based on these findings.
A meta-analysis of 7 randomized, placebo-controlled phase II and III studies of DOACs in more than 30 000 patients with ACS revealed the addition of DOACs to aspirin alone led to a 30% reduction in major adverse cardiovascular events (MACEs) (HR: 0.7, 95% CI: 0.59-0.84) but caused a substantial increase in bleeding (HR: 1.79, 95% CI: 1.54-2.09). 28 Additionally, when DOACs were combined with DAPT (aspirin and clopidogrel), there was slightly more reduction in MACE (HR: 0.87, 95% CI: 0.8-0.95), but the risk of major bleeding was more pronounced (HR: 2.34, 95% CI: 2.06-2.66). Therefore, the combination of DOACs and DAPT may not be the most viable strategy when optimizing DAPT. Furthermore, there is a possibility that a member of DOACs (dabigatran) increases the risk of MI. This was shown in the RE-LY study where dabigatran of dose 150 mg was associated with an increased incidence of MI compared to warfarin (HR: 1.38, 95% CI: 1.00-1.91, P = .048). There was also a similar trend with dabigatran of dose 110 mg arm (HR: 1.35, 95% CI: 0.98-1.87, P = .07). 29 However, these findings were never replicated in observational studies looking at the use of dabigatran in clinical practice. 30 In addition, none of the anti-Xa inhibitors (rivaroxaban and apixaban) showed similar associations. 31,32 It is not clear if findings from RE-LY studies were due to false-positive results or if increased incidence of MI is specific to direct thrombin inhibitors versus other anti-Xa inhibitors.
Dual Antiplatelet Therapy Duration
Determining the optimal duration of DAPT is another potential strategy that can enhance the benefit-risk ratio of DAPT among patients with ACS. Numerous studies have been published regarding the duration of DAPT. Twelve months of DAPT has been the standard of practice based on findings from the CREDO study. 5 Patients undergoing elective PCI were randomized to receive clopidogrel and aspirin for 1 year. Results revealed that 1-year duration with clopidogrel and aspirin therapy was associated with a 26.9% relative reduction in the combined risk of death, MI, or stroke (95% CI: 3.9%-44.4%; P = .02). Bleeding risk was increased at the end of 12 months; however, it was not shown to be significant (8.8% with clopidogrel vs 6.7% with placebo; P = .07). Canadian, European, US, UK, and Australian guidelines have adopted these findings and recommend 12 months of DAPT after placement of bare-metal stent or DES in patients with ACS. 11,33,34
The newer generation DESs are associated with lower risks of stent thrombosis. Based on this premise, short-term DAPT defined as 6 months or less of treatment has been explored to improve the efficacy and safety profile among patients in regard to ischemic and bleeding events. Several studies have shown these favorable effects with short duration of treatment but with limitations. One of the earliest short-duration studies was EXCELLENT. 35 Patients were randomized to either 6 months or 12 months of clopidogrel and aspirin therapy and further stratified to either everolimus- or sirolimus-eluting stents. Results showed 6 months of DAPT was noninferior to 12 months of DAPT for the primary end point of target vessel failure. These results should be interpreted with caution as the study did not assess death and MI as primary end points, the noninferiority margin was wide, safety was not addressed, the study design was open-labeled (unblinded), and many patients assigned to the 6-month group received clopidogrel for longer than 6 months.
Since EXCELLENT study, other short-duration DAPT studies have emerged investigating the noninferiority nature of short- versus long-term therapy. OPTIMIZE, RESET, ITALIC, and SECURITY studies showed 3 months or 6 months of DAPT was noninferior to 12 months. 36 ∓39 Despite these favorable results, these studies have shortcomings that need to be considered before applying them to practice. First, many studies had low event rates which lead to lack of power to detect small differences in the study outcomes. Additionally, high-risk patients were excluded from the studies which limit its applicability to few patients. Finally, the study design was not appropriate in the RESET study as the comparator group varied among each arm in terms of the DES, which may have affected the results of the study. As for the other studies, a potential for bias exists as they were open labeled. The SECURITY study had a small portion of patients who continued on clopidogrel despite the assigned 6-month period, which could have affected study findings.
Benefits of extending DAPT to greater than 12 months were explored after reports showing that late stent thrombosis and related death and MI events were higher among patients receiving 3 to 6 months of DAPT. 40 Long-duration studies such as DES-LATE, REAL-LATE, ZEST-LATE, and ARTIC-INTERRUPTION (Table 2) showed no significant difference in the reduction of ischemic events with the continuation of DAPT beyond 12 months compared to aspirin monotherapy. 41 ∓43 Most of these studies were an extension of previous studies and excluded high-risk patients. Additionally, these studies were open label and were underpowered to detect differences due to low event rates. Due to these limitations, the benefits of extended DAPT beyond 12 months remain uncertain.
Summary of Clinical Studies That Investigated Long DAPT Duration.
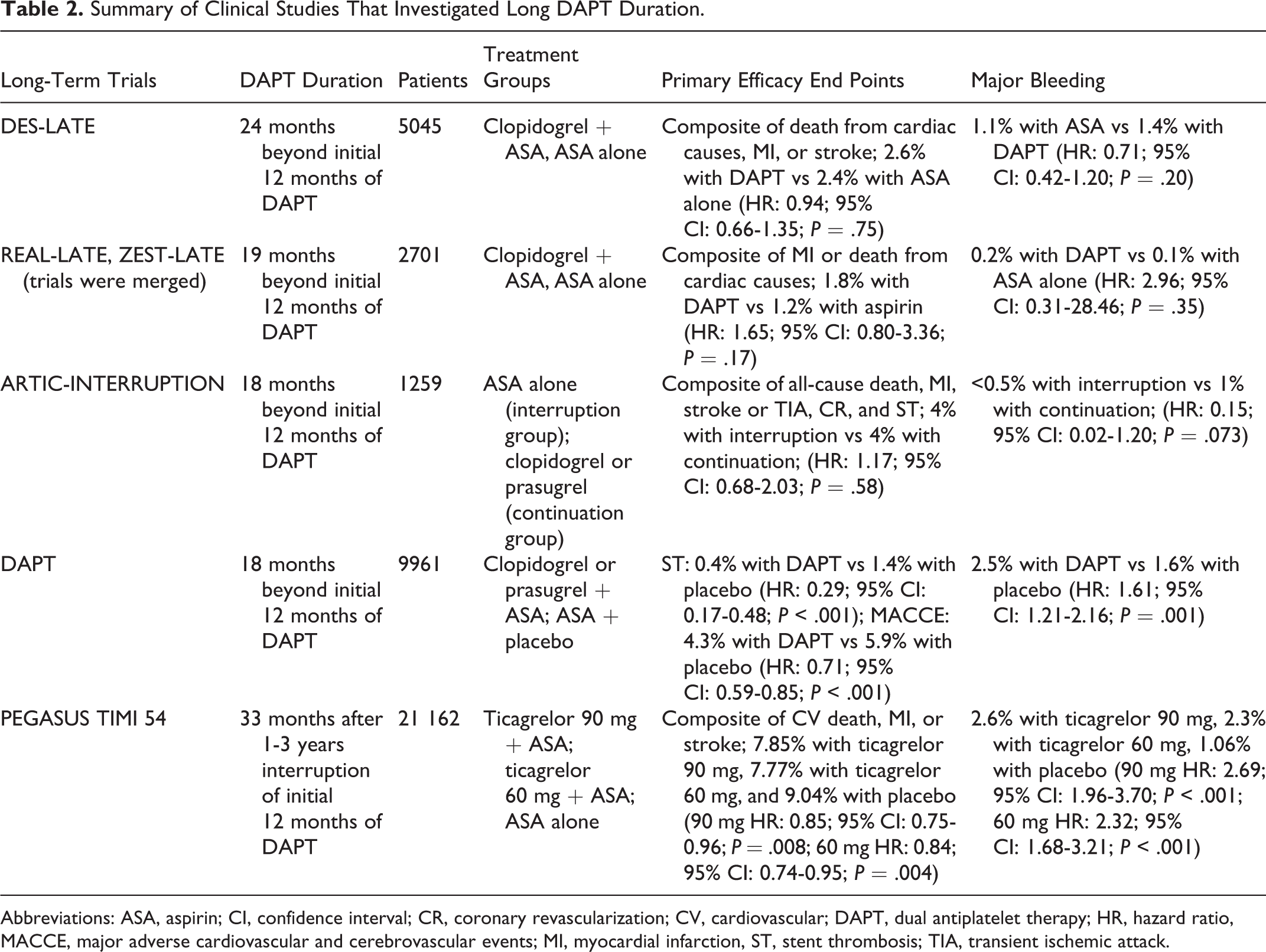
Abbreviations: ASA, aspirin; CI, confidence interval; CR, coronary revascularization; CV, cardiovascular; DAPT, dual antiplatelet therapy; HR, hazard ratio, MACCE, major adverse cardiovascular and cerebrovascular events; MI, myocardial infarction, ST, stent thrombosis; TIA, transient ischemic attack.
The DAPT study, a randomized placebo controlled trial, looked into the benefits of continuing DAPT beyond 1 year after stent placement. 39 Patients, who had not experienced MACE and were adherent to thienopyridine therapy after 12 months of DAPT, were randomized to another 18 months of either thienopyridine and aspirin or placebo and aspirin. Rate of stent thrombosis was 0.4% in the thienopyridine group compared to 1.4% in the placebo group (HR: 0.29; 95% CI: 0.17-0.48; P < .001), and MACE was 4.3% in the thienopyridine group compared to 5.9% in the placebo group (HR: 0.71; 95% CI: 0.59-0.85; P < .001). Moderate to severe bleeding was significantly increased in the thienopyridine group when compared to the placebo (2.5% vs 1.6%; P = .001). Therefore, extending DAPT beyond 12 months was shown to have significantly better results for MACE as compare to aspirin monotherapy but with an increased risk of bleeding. Unexpectedly, there was an increase in the number of deaths from any cause in the group that continued receiving thienopyridine than in the placebo group (HR: 1.36; 95% CI: 1.00-1.85; P = .05). This difference was driven primarily by deaths from noncardiovascular causes. Despite bleeding-related deaths that have contributed into this difference (11 deaths in the thienopyridine vs 3 deaths in the placebo group; P = .06), imbalance in patients with history of cancer at time of enrollment (22 more patients in the thienopyridine group) may have influenced this difference.
Although these study findings support DAPT beyond 12 months, it is important to note that participants included in the DAPT study were considered not at high risk for an ischemic event and did not experience any severe bleeding while on DAPT. Patients at high risk for stent restenosis would greatly benefit from this extended treatment period; however, this was not investigated in this study. Additionally, it is not very clear how long the beneficial effects of long-term DAPT will remain. 44
The PEGASUS-TMI 54, a randomized double-blind study, investigated the long-term use of ticagrelor and low-dose aspirin on MACE among patients 1 to 3 years after an MI. 45 All patients were taking aspirin of dose 75 to 150 mg and randomized to either ticagrelor of dose 90 mg twice daily, ticagrelor of dose 60 mg twice daily, or placebo. Kaplan-Meier rates at 3 years for ticagrelor of dose 90 mg were 7.85% (HR: 0.85; 95% CI: 0.75-0.96; P = .008), 7.7% for ticagrelor of dose 60 mg (HR: 0.84; 95% CI: 0.74-0.95; P = .004), and 9.04% in for the placebo group. Both ticagrelor doses significantly reduced the primary end points of cardiovascular death, MI, or stroke. Major bleeding was significantly higher at 2.6% for 90 mg of ticagrelor (HR: 2.69; 95% CI: 1.96-3.70; P < .001) and 2.3% for 60 mg of ticagrelor (HR: 2.32; 95% CI: 1.68-3.21; P < .001) when compared to 1.06% for the placebo group. Study findings support the addition of ticagrelor with low-dose aspirin in patients 1 to 3 years after an MI.
Although the PEGASUS study did not directly address the duration of DAPT, it revealed that ticagrelor has the potential to be utilized long term to reduce ischemic events. These findings should be interpreted carefully as PEGASUS and DAPT studies differed on a few aspects. In the PEGASUS study, there was a gap in using DAPT therapy for almost 2 years, whereas the DAPT study continued DAPT after the initial recommended duration of 12 months. Unlike the DAPT study, PEGASUS included patients who had bleeding episodes or cardiovascular events. Despite these differences, both studies have shown the benefits of prolonged P2Y12 receptor blocker and aspirin use in reducing ischemic events, but at the expense of increased bleeding episodes.
Despite the positive effects viewed in the above short- and long-term trials, the optimal duration of DAPT remains unclear. These studies enrolled mainly stable patients and utilized varying generations of DES leading to differing risks of stent thrombosis. These aspects contribute to disparity in determining the ideal DAPT duration.
Dual Antiplatelet Therapy Score
The DAPT score was developed by the DAPT study investigators to assist practitioners in deciding the patients that would benefit with extended treatment with DAPT beyond 1 year. 46 The DAPT score is available as an online calculator at http://www.daptstudy.org/for-clinicians/score_calculator.htm. The score determines those who are more likely to benefit from prevention of an ischemic event without bleeding risk as compared to those who will likely be at risk of bleeding. It is based on 8 clinical factors which include diabetes status, smoking status, age, history of MI or PCI, history of heart failure or LVEF <30%, MI at presentation, vein graft PCI, and stent diameter (Table 3). 47 Patients with a DAPT score of less than 2 were less likely to experience ischemic events and more likely to experience bleeding events. The number needed to treat (NNT) to prevent ischemia was 153, whereas the number needed to harm (NNH) to cause bleeding was 64. Patients with a DAPT score of 2 or more are most likely to benefit with extended DAPT. The NNT was 34 patients to prevent 1 ischemic event, whereas the NNH with 1 bleeding event would be 272 patients. 47,48
Clinical Factors Utilized to Calculate DAPT Score. 36

Abbreviations: DAPT, dual antiplatelet therapy; MI, myocardial infarction; PCI, percutaneous coronary intervention.
This scoring system is performed at the time of the PCI and is applicable to a selective patient population since the DAPT score was based on the DAPT trial results. This population included patients adherent to either clopidogrel or prasugrel and who did not experience moderate to severe bleeding, stent thrombosis, or major cardiovascular or cerebrovascular event within the first year of treatment with aspirin and thienopyridine after DES placement. The study excluded patients on anticoagulant therapy and a life expectancy of less than 3 years. The score may not be applicable to those on other antiplatelet regimens.
Practice Implications
The 2016 American College of Cardiology [ACC]/American Heart Association [AHA] guidelines favor prasugrel and ticagrelor over clopidogrel. 16 Despite these recommendations, the use of prasugrel and ticagrelor in real-life practice does not exceed 30%. 49 Clopidogrel, prasugrel, and ticagrelor have been all listed in the European Society of Cardiology (ESC) guidelines as viable options for use with aspirin therapy in patients with ACS undergoing stent placements. 50 Both guidelines state prasugrel is preferred over clopidogrel when the patient is not at high risk for bleeding and is without a history of stroke or TIA. As a practitioner, the choice of P2Y12 inhibitor should be dependent on the patient’s ischemic and bleeding risk. Despite the perceived risk of adverse cardiovascular outcomes in patients receiving clopidogrel concomitantly with proton pump inhibitors, randomized control trials showed no effect of omeprazole when added to DAPT.
For the addition of DOACs to DAPT, rivaroxaban (2.5 mg twice daily for about 1 year) is the only agent that may be considered with aspirin and clopidogrel therapy in patients with ACS after the acute phase based on the ESC 2015 guidelines. 50 Its use should be limited to patients with high risk of MACE and low risk of bleeding. Since the level of evidence for triple therapy in patients with PCI and no indication for oral anticoagulant is not robust, it is best to avoid this combination. When there is an indication for using triple therapy, clopidogrel is the P2Y12 inhibitor of choice for this combination. 11,51
The optimal duration of DAPT remains controversial with emerging studies showing benefit for both short and long-term therapy with DAPT. The 2016 ACC/AHA guideline recommends DAPT should be given for 12 months and that it may be reasonable to extend beyond this period. Shorter duration of DAPT is reasonable in patients with ACS treated with the newer generation stents who are at lower risk of ischemic events and high risk of bleeding (Table 4). 11 Similarly, the 2015 ESC guidelines are in line with the ACC/AHA recommendations. 50 For practitioners, tools such as the DAPT score can be used as a guide in clinical decision making for those patients benefiting from greater than 12 months of DAPT. However, the DAPT scoring tool should not be used to replace clinical judgment. The DAPT score is valid for a selected population. Aspects such as bleeding risk, ischemic risk, and patient factors should be carefully weighed when considering DAPT beyond 12 months. Bleeding scores, chronic nonsteroidal anti-inflammatory and anticoagulant therapy, anemia, and clinically significant bleeding while on the DAPT are a few considerations to discontinue DAPT after 1 year. Conversely, patients with recurrent ischemic events while on DAPT, ACS, prior ischemic stroke or MI, and stent-related complications are few factors to consider continuing DAPT beyond 12 months. Finally, patient factors such as short-life expectancy, poor socioeconomic status, and nonadherence to DAPT are reasons to discontinue DAPT after its required period. 34
2016 ACC/AHA Guideline Update on Duration of Dual Antiplatelet Therapy in Patients With Acute Coronary Syndrome. 11

Abbreviations: ACC, American College of Cardiology; ACS, acute coronary syndrome; AHA, American Heart Association; DAPT, dual antiplatelet therapy; PCI, percutaneous coronary intervention; RCT, randomized controlled trials; TIA, transient ischemic attack.
Summary
Amid the strategies outlined in this review, not 1 strategy stands out as the best without compromising efficacy or safety concerns with DAPT. It is important to emphasize that optimal therapy should be individualized based on the patient’s risk of ischemia and bleeding. Dual therapy with aspirin and potent P2Y12 inhibitors (prasugrel and ticagrelor) improve DAPT efficacy but with increased risk of bleeding. Among patients with ACS without atrial fibrillation, the addition of a DOAC to antiplatelet therapy led to minor reductions in cardiovascular events but at the expense of substantial bleeding risk. The only combination with potential for this population is rivaroxaban along with aspirin and clopidogrel. The optimal duration of DAPT remains controversial. However, standard therapy should be for 6 to 12 months with shorter and longer duration of treatment reserved for patients with high bleeding and high ischemic risk, respectively. The DAPT score is an additional tool that may help optimize DAPT duration.
Footnotes
Author Contributions
H. Elewa contributed to conception, design, acquisition, analysis, and interpretation. B. Paravattil contributed to design, acquisition, analysis, and interpretation. Both authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
