Abstract
Reperfusion in the setting of acute ischemia is essential in limiting tissue necrosis. However, reperfusion itself is associated with significant adverse effects. There is animal evidence that platelets play a role in the adverse effects of ischemia and reperfusion (IR) injury. We examined whether clopidogrel would have favorable effects on endothelial dysfunction induced by an episode of IR. Using a parallel design, we administered clopidogrel 600 mg or matching placebo to normal volunteers (n = 20) 24 hours before an episode of IR. Flow-mediated dilatation (FMD, radial artery) was assessed before and after 20 minutes of upper arm ischemia. Following IR, there was a highly significant decrease in FMD in the placebo group (7.6% ± 1.3% vs 3.4% ± 0.1%;
Introduction
Timely tissue reperfusion is a primary goal in the management of infarction. However, it is now understood that reperfusion therapy exacerbates tissue damage caused by ischemia and that the mechanism(s) behind this injurious process are multifactorial. A variety of interventions, prior to an episode of ischemia and reperfusion (IR), can limit tissue damage, a phenomenon termed preconditioning. Murry et al were the first to describe ischemic preconditioning, in which brief episodes of ischemia, prior to an episode of more severe IR, limit myocardial infarct size. 1 Since that time, it has been recognized that a number of pharmacologic interventions given prior to an episode of IR can also limit tissue injury associated with ischemia/infarction, an effect termed pharmacologic preconditioning. 2
The success of antiplatelet agents in the therapy of acute coronary syndromes emphasizes the importance of intravascular thrombosis in its pathogenesis. However, the role that platelets play as mediators of IR injury remains poorly understood. The concept that platelets participate in IR injury is supported by the fact that platelet activity is enhanced during IR 3 ∓6 as well as the observation that platelet deposition increases during reperfusion to a greater extent than that observed during ischemia alone. 3,5 These observations suggest that platelets may be involved in IR injury. Furthermore, studies have documented that ischemic preconditioning prevents the activation of platelets associated with IR, with associated increases in coronary artery patency rates. 7,8 Consistent with these findings are animal studies that suggest antiplatelet agents can favorably modify the response to IR. For example, prior treatment with clopidogrel has been shown to provide protection against the adverse consequences of IR injury in animals. 9,10 The mechanism of protection is thought to involve a reduction in reactive oxygen species (ROS) production associated with an increase in nitric oxide bioavailability. More recent studies have suggested that the P2Y12 adenosine diphosphate (ADP) receptor blocker cangrelor as well as clopidogrel have protective effects in the setting of IR that involve signal transduction mechanisms similar to those observed with ischemic preconditioning and are independent of their antithrombotic effects. 11 To date, no human studies have been performed to evaluate whether antiplatelet therapy is protective against IR injury. The following study utilizes a well-established forearm model of IR injury to demonstrate that a single dose of clopidogrel, administered prior to IR, can protect the endothelium against injury.
Methods
The Mount Sinai Hospital Research Ethics Board approved the study, and written informed consent was obtained from all participants prior to their enrollment in the study.
Participants
Healthy, nonsmoking volunteers (18-30 years old) were recruited for this study. Each participant’s state of health was evaluated using a brief questionnaire that was completed during a consultation visit prior to enrollment. Exclusion criteria were any current disease or illness and risk factors for cardiovascular disease such as hypercholesterolemia, hypertension, atherosclerosis, and smoking. All participants were asked to abstain from caffeine on the day of each study visit and from any drugs, including supplemental vitamins, for at least 2 weeks prior to and during the study.
Materials
Both the study drug (clopidogrel 600 mg, Apotex, Ontario, Canada) and placebo (lactose tablets; Odan) were purchased from the pharmacy at Mount Sinai Hospital (Toronto, Ontario, Canada).
Study Protocol
Visit 1
After screening for admission into the study, participants returned to the laboratory at a later date for administration of either the study drug (clopidogrel 600 mg) or placebo. Participants were randomized to either study arm by a registered nurse who was otherwise not involved with the study.
Visit 2
Twenty-four hours after treatment administration, participants returned to the laboratory for measures of standing blood pressure and heart rate. This was followed by assessment of flow-mediated dilation (FMD) in the radial artery before and after induction of IR. A venous blood sample was drawn before and after induction of IR (from the arm undergoing IR) for the evaluation of specific biomarkers.
Inducing IR Injury in the Forearm Vasculature
Forearm ischemia was induced by inflating a sphygmomanometer (Welch Allyn) placed at the brachial artery to 280 mm Hg for 20 minutes. At the end of this ischemia period, the cuff was deflated to allow for 15 minutes of reperfusion. Flow-mediated dilation was assessed before and after this process.
Flow-Mediated Dilation Protocol
Radial artery FMD was measured using a protocol that has been described and validated by our laboratory in a number of previous studies. 12 ∓15 Briefly, participants rested in the supine position on a partially inclined bed for a minimum of 20 minutes to allow for proper acclimatization to the study environment. The participant’s left arm was then positioned onto a pillow and a small pneumatic pressure cuff was placed around the wrist. Using an ultrasound probe running on Doppler mode (Vivid 7; General Electric), an image of the radial artery was captured and recorded onto an external laptop device for a total running time of 10 minutes. The 10-minute period was initiated by a minute of baseline imaging, followed by inflation of the pneumatic wrist cuff to 220 mm Hg for 4.5 minutes to inhibit antegrade blood flow and cause temporary ischemia. Imaging was continued for an additional 4.5 minutes after deflation of the pressure cuff. This procedure was performed before and after inducing IR in the forearm to evaluate changes in endothelial function afforded by IR injury.
Vascular Imaging
Imaging of the radial artery was recorded onto an external laptop device at ∼30 frames/second, with the actual image being captured at the peak of the R-wave during each cardiac cycle. Because arterial diameter fluctuates during a single cardiac cycle, triggering the image to an external electrocardiogram (termed “ECG gating”) and capturing at a single point avoid considerable fluctuations in diameter. 16
Data Analysis
An edge detection software program (SpLiNeS; HeartWorks, Italy) developed specifically for FMD analyses was used to measure arterial diameters (from intima to intima) and subsequent percentage dilation values. Radial artery FMD was calculated as follows: percent FMD = (peak diameter − baseline diameter)/baseline diameter.
Blinding
An investigator blind approach was used. Participants were not blind to their treatment allocation, but all study personnel involved in data acquisition and analysis remained blind to which treatment participants received. Although baseline ultrasound images were reviewed for quality, the final analysis of all FMD measurements was performed blind. Ultrasound images of each participant were then coded and subsequently analyzed by one of us (W.K.) who remained blinded to both the sequence of imaging (pre- vs post-IR) and the study medication received.
Other End Points
Blood flow
Measurements of blood flow were made using a pulsed-wave Doppler system (Vivid 7; General Electric) at 3 different time points during the FMD protocol—at baseline and immediately prior to cuff inflation and after cuff deflation. Blood flow was calculated as the cross-sectional area of the radial artery multiplied by the velocity–time integral of the Doppler spectrum (taken as an average of 5 cardiac cycles) multiplied by the heart rate.
Blood pressure/heart rate
A vital signs monitor (Carescape V100; General Electric) equipped with a blood pressure cuff (Critikon, Dura-Cuf, GE Healthcare, UK) was used to measure various blood pressure parameters (systolic, diastolic, mean arterial) and heart rate (taken as an average of 3 measurements).
Biomarkers
Before and after IR, a venous blood sample was obtained for the analysis of intercellular adhesion molecule 1 (ICAM-1), chemokine ligand 5 (RANTES), and interleukin 6 (IL-6). Plasma concentrations were determined using quantitative sandwich enzyme immunoassay techniques (R&D Systems, Minneapolis, Minnesota).
Statistical Analysis
Sample size calculations were made based on a previous study conducted in our laboratory, where IR injury diminished FMD from 7.9% ± 3.3% to 1.2% ± 2.3%.
17
To prevent 50% of this impairment in endothelial function with clopidogrel, 10 participants per treatment arm is required (1 − β = 2, 2-sided α = .05). All data analyses were performed with SAS statistical software (version 9.4). Tests for normality by the group were done using the Shapiro-Wilk test. Comparison of baseline values and changes within groups was made using the Student
Results
Twenty normal male volunteers participated in the study. Their age ranged from 21 to 28 years. Of this group, 10 were randomized to receive placebo and 10 received clopidogrel.
Heart Rate and Blood Pressure Responses
There were no significant differences in baseline heart rate blood pressures between groups or between visits 1 and 2 (Table 1).
Heart Rate and Blood Pressure on Visits 1 and 2.

Abbreviations: bpm, beats per minute; BP, blood pressure
Blood Flow and Arterial Diameters
Baseline radial artery diameters were not significantly different when measured both before and after IR. Radial artery blood flow at baseline (both pre- and post-IR) was similar in all groups (Table 2). The hyperemic flow responses following cuff deflation were also similar, with no differences observed between the placebo and clopidogrel groups both before and after IR (Table 3).
Radial Artery Diameters (mm).

a
b
Radial Blood Flow (mL/min).

Flow-Mediated Dilation
At baseline, there was no difference in FMD between the 2 groups (7.6% ± 1.3% vs 8.3% ± 0.8%,
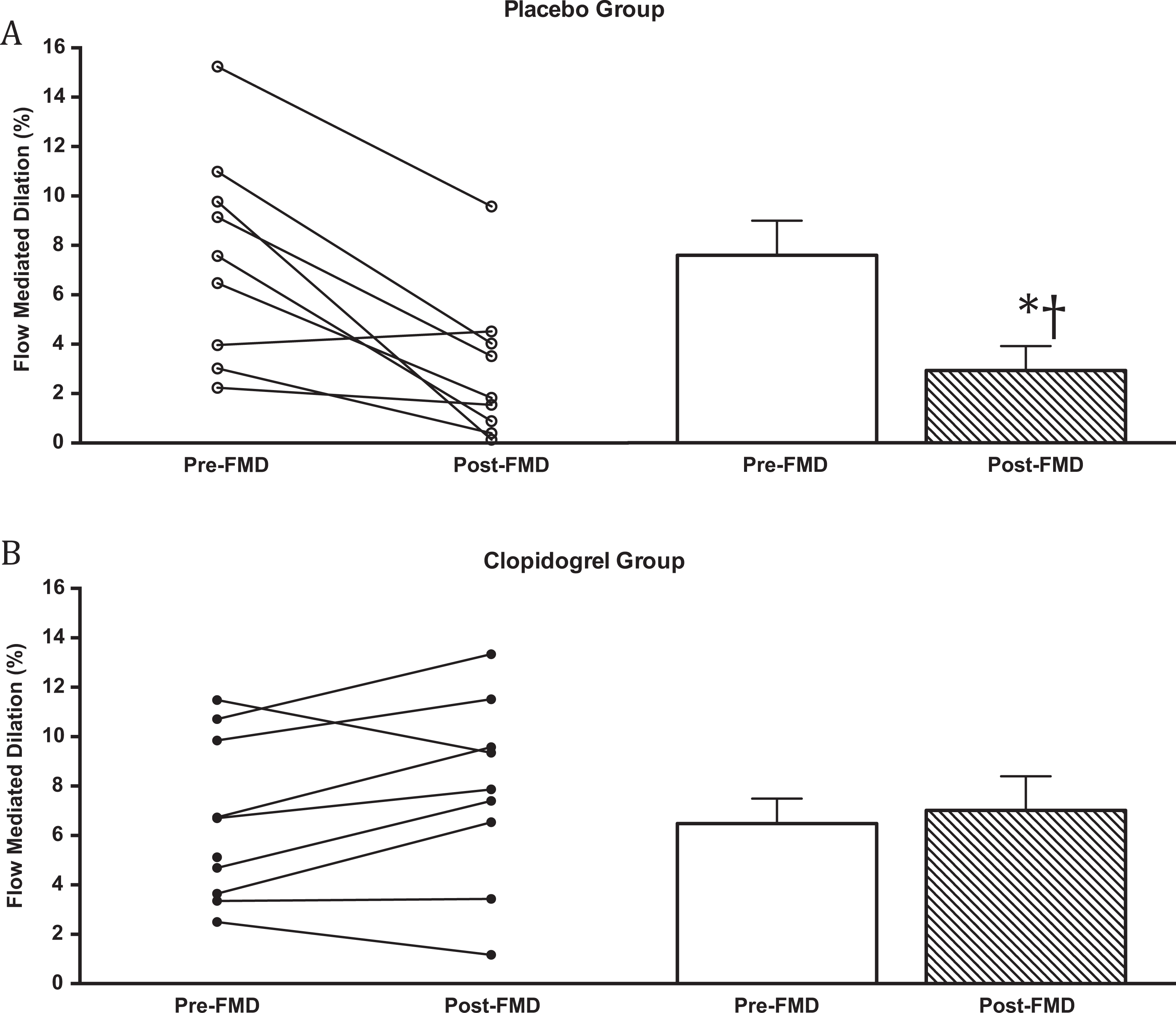
Flow-mediated dilatation of the radial artery before and after ischemia reperfusion (IR). A, Placebo group. B, Clopidogrel group. Individual responses are shown on the left and group means on the right of each panel. *
Biomarkers
There was no statistically significant difference in baseline biomarker measurements between the 2 treatment groups (Table 4). Before and after IR, there was no significant change in the values of ICAM-1, RANTES, or IL-6 (Table 4).
Biomarkers.

Abbreviations: ICAM-1, intercellular adhesion molecule 1; IL-6, interleukin 6.
Discussion
Platelets play a central role in the development of acute coronary syndromes and other vascular ischemic events. The role that platelets play in thrombosis has been the focus of attention as new strategies to prevent and modify the natural history of cardiovascular ischemic events have been developed. In recent years, a variety of new antiplatelet agents have been developed with broad application in the therapy of acute coronary syndromes and percutaneous interventions. Glycoprotein IIA/IIIB antagonists and the P2Y12 receptor antagonists clopidogrel, prasugrel, and ticagrelor have all been shown to reduce recurrent cardiac events and improve long-term outcome in patients with acute coronary syndromes and those undergoing percutaneous intervention. 18 Although the mechanism of their beneficial effects is likely complex, it is generally assumed to be secondary to their antithrombotic effects. However, the clinical situations in which antiplatelet agents have beneficial effects all involve 1 or more episodes of IR. Furthermore, some studies have suggested that antiplatelet agents, including clopidogrel, have the ability to limit infarct size via a mechanism that is independent of their antithrombotic effects. 19,20 However, whether antiplatelet agents have clear preconditioning effects in humans has not yet been described.
It is now recognized that platelets have biologic roles that go beyond their role in hemostasis and acute coronary syndromes. Platelets have important interactions with the endothelium, other blood cell elements, the complement system, and a host of other biologic systems. As such, platelets are now recognized to play both local and systemic roles as mediators of atherosclerosis and inflammatory responses. A number of lines of evidence demonstrate that platelets play important roles in many aspects of IR injury. Although direct mechanistic information concerning a nonthrombotic role for platelets in IR-mediated injury is limited, there is increasing evidence in support of this phenomenon. In a rat model of hind limb IR, clopidogrel has been shown to reduce the bioavailability of free radicals while decreasing plasma and tissue nitric oxide concentrations. 9,10 A number of studies have now documented that P2Y12 ADP receptor blocking agents are effective in reducing infarct size in animal models of IR. 11,21,22 For example, Yang et al have documented that cangrelor (as well as clopidogrel) was protective in a rabbit model of myocardial infarction by mechanisms not related to their effects on platelet aggregation. 11 Results with clopidogrel have been somewhat inconsistent. In 2 recent reports, ticagrelor but not clopidogrel limited infarct size in rat models of IR. 21,22 Although some differences between P2Y12 ADP-receptor blocking agents and the response to IR have been found in animal models, it remains unclear whether there are any clinically significant differences between these agents and the response to IR.
The current investigation examines the effect of clopidogrel on abnormal endothelial function responses to IR in the human forearm. The model employed has been used in multiple studies exploring the impact of IR and preconditioning interventions. It is acknowledged that this model of IR is only a surrogate and may not reflect what occurs in response to IR in the setting of clinical infarction. For this reason, beneficial effects observed with an intervention using this model may not translate to an actual reduction in infarct size following myocardial infarction. We demonstrate that a single dose of clopidogrel (600 mg) provides protection from the adverse effects of IR on conduit artery endothelial function as assessed by FMD. The observation that IR caused a significant decrease in FMD in the placebo group is consistent with a number of previous observations. We did not examine the impact of a nonendothelium-dependent vasodilator on radial artery responses in this study, as prior studies have demonstrated that IR does not impair nonendothelium-dependent responses. To the best of our knowledge, this is the first human study to address the impact of clopidogrel on abnormalities induced by IR injury. Two prior studies have documented that clopidogrel therapy is associated with improvements in endothelial function in patients with coronary artery disease. 23,24 In the current study, we found no effect on endothelial function prior to IR, although the population involved normal volunteers and one might not expect an increase in endothelial responses in those with normal baseline responses. We cannot address the mechanism of the preconditioning effect of clopidogrel in this study. As suggested by observations in animal models, drugs such as clopidogrel may cause an upregulation in signaling systems involved in the late window of preconditioning (such as increased expression of nitric oxide synthase or cyclooxygenase 2). 11,21 Alternatively, the antiplatelet effect of clopidogrel may prevent platelet deposition/adherence to the endothelium and in so doing modify the inflammatory response to IR with resulting salutary effects on endothelial function.
The biomarkers we measured have all been shown to be involved in the response to IR. 25 ∓27 However, in the current study, clopidogrel had no effect on biomarkers of inflammation either at baseline or after IR. Previously, clopidogrel was shown to decrease markers of inflammation, including RANTES, CRP, and CD40, in patients with coronary artery disease and type 2 diabetes. 23,28,29 Furthermore, in a rat model, Kanko et al demonstrated that clopidogrel protected against injury by preventing an increase in malondialdehyde and decreases in glutathione and superoxide dismutase. 9 In that report, clopidogrel was administered for 10 days and the hind limb was rendered ischemic for 4 hours prior to reperfusion. In the current study, biomarkers of inflammation did not change following IR and were not modified by clopidogrel. Methodologic differences likely explain the differences between these prior results and what we report here. We used a normal volunteer model and only gave 1 dose of clopidogrel. Further, we measured systemic venous plasma concentrations of these biomarkers that may not reflect what is occurring at a tissue level. Of note, consistent with our results, Hu et al found in a mouse model that a single dose of clopidogrel was protective in the setting of renal ischemia followed by reperfusion. 30 They used a variety of functional and biochemical markers to document protection but not the biomarkers we used in the current study.
Therefore, for the first time in humans, we document that clopidogrel provides protection from the adverse effects of IR on conduit artery endothelial function. The relevance of this to improved clinical outcome observed when P2Y12 ADP-receptor blocking agents are administered to patients with acute coronary syndromes cannot be determined from the present observation. It does however suggest 1 mechanism by which clopidogrel may exert its beneficial effects.
Footnotes
Authors’ Contributions
WK and JP both contributed to the study conception, study design and writing of the manuscript. WK conducted the study protocol on health volunteers.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
