Abstract
In the search for innovative solutions to treat ischemic heart disease, recent basic science and clinical approaches have focused on remote ischemic preconditioning (RIPC). Remote ischemic preconditioning involves short intervals of limb blood flow occlusion by the application of a blood pressure cuff inflated to a suprasystolic pressure. The promise of RIPC in the development of new cardioprotective therapies is founded on the premise that it is cost-effective, technically simple, and overcomes many logistical and biochemical hurdles associated with other ischemic preconditioning approaches. However, RIPC as a research subarea is still in its infancy and clinical applications for individuals at high risk of cardiovascular disease remain elusive. The thesis of the current review is that observational and mechanistic similarities between exercise-induced preconditioning and RIPC may reveal novel therapeutic links to cardioprotection. While reductionist understanding of the exercised heart is still in the formative stages, available mechanistic knowledge of exercise-induced cardioprotection is juxtaposed to RIPC and potential implications discussed. In total, additional research is needed in order to fully appreciate the mechanistic and translative connections between exercise and RIPC. Nonetheless, existing rationale are strong and suggest that RIPC approaches may be helpful in the development and application to pharmacologic interventions in those with ischemic heart disease.
Introduction
Heart disease, including ischemic heart disease, remains the leading cause of morbidity and mortality in industrialized nations. 1 Despite the significant advances in statin applications, revascularization, and other strategies to combat ischemic heart disease, persistent morbidity has created a medical need to preempt coronary events when they occur. To date, more than 4 decades of collective effort have been marshaled in the search for therapeutic countermeasures. These efforts include a wealth of research based upon ischemic preconditioning—cellular protection exerted by nonpathologic ischemia applied prior to a major coronary event. 2 The landmark experiment in this regard was the observation that intervals of sublethal cardiac ischemia–reperfusion (repetitions of surgical left anterior descending coronary artery (LAD) ligation) in canines conferred a robustly protected phenotype against subsequent extended duration ischemia. 3 In the decades since this discovery, efforts to “reverse engineer” a pharmacologic approach to cardioprotection have not delivered. Barriers to progress have been significant and are perhaps most notably highlighted in a 2004 scientific call to arms where eminent physicians and scientists concluded, “hundreds of experimental interventions protect the ischemic myocardium in experimental animals; however, with the exception of early reperfusion, none has been translated into clinical practice.” 4 In the years since this now infamous statement, several important fields of scientific inquiry have matured and offer better understanding of potential solutions for cardiac preconditioning: (1) exercise-induced cardioprotection and (2) remote ischemic preconditioning (RIPC) through intermittent cycles of limb ischemia as applied through a blood pressure cuff inflated to suprasystolic pressures. This review summarizes recent findings of RIPC research and juxtaposes related findings from exercise preconditioning as an informative stimulus.
An Overview of RIPC: Promise and Limitations
The concept of limb-based RIPC to cardioprotect against ischemic injury has been studied experimentally for several decades. 5,6 Similar to ischemic preconditioning, intervals of limb ischemia are performed hours to days prior to surgically induced ischemia of a pathological duration. 7 While numerous methodological approaches to RIPC exist, most include somewhere between 2 and 8 cycles of limb ischemia maintained for time durations between 2 and 10 minutes. 8 The fact that various RIPC approaches confer roughly similar levels of cardioprotection highlights the threshold nature of ischemic preconditioning within (heart-to-heart) 9 or between organs (limb-to-heart). 8 Moreover, limb muscle mass does not appear to be a mitigating factor for RIPC efficacy, 8 a fact that sheds important insight on the majority of recent clinical investigations which employ arm occlusion. As variations of common physiologic phenomenon, both ischemic and remote preconditioning confer biphasic cardioprotection with early-phase (minutes) and late-phase (hours to days) protective time windows. 9,10 Given these similarities, it is not surprising that remote preconditioning and ischemic preconditioning share many mechanisms of protection, as will be discussed later in comparison with an exercise stimulus.
Based on a wealth of experimental promise, the clinical potential of RIPC as a means of conferring cardioprotection was recognized immediately as an adjuvant to cardiothoracic procedures in children. 11 Representative studies of RIPC in multiple species, including humans, are provided in Table 1. Early evidence of therapeutic potential is highlighted by the fact that RIPC is noninvasive, technically simple, and inexpensive. Preemptive RIPC to elicit ischemic resistance in those at high risk of a cardiovascular event, however, remains uncertain. Logistical hurdles in the clinical translation of RIPC are limited by the fact that cardiac events are almost invariably unpredictable, even in high-risk individuals. Further complicating matters, the aged heart is insensitive to the stimuli of ischemic, pharmacologic, and remote preconditioning, 12,13 This unfortunate fact contrasts the proportional relationship between the incidence of myocardial infarction and advancing age. 1 Importantly, findings from individual studies suggest that the clinical promise of RIPC is most apparent when associated with late-phase preconditioning window outcomes. 14 –16 In contrast, important findings from the Effect of Remote Ischaemic preConditioning on clinical outcomes in patients undergoing Coronary Artery Bypass Graft surgery (ERICCA) and Remote Ischemic Preconditioning for Heart Surgery (RIPHeart) clinical trials, encompassing observations from over 3000 patient interventions, found no convincing evidence of RIPC-derived benefits based on short- and long-term outcomes. Moreover, acute outcome markers observed in ERICCA and RIPHeart trials, including the examination of cardiac-specific troponin levels in blood, did not indicate RIPC efficacy. 17,18 In this regard, several authoritative reviews on RIPC attend to scientific and philosophical considerations of RIPC. 19,20 Weighing in the balance is an important scientific debate about RIPC as a clinical approach. Specifically, is efficacy limited by the clinically unsuccessful field of cardiac preconditioning or would refined dependent measures and scope of study reveal benefits of RIPC that were not observed in ERICCA and RIPHeart investigations?
RIPC Outcomes in Biomedical and Clinically Directed Investigations.
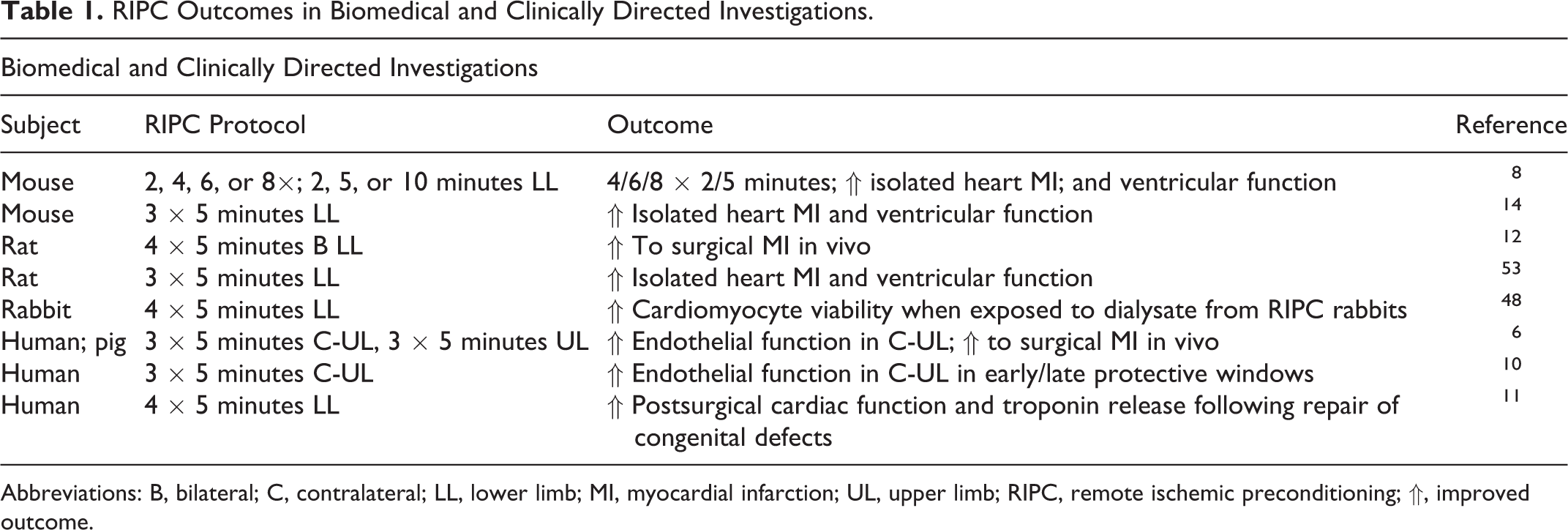
Abbreviations: B, bilateral; C, contralateral; LL, lower limb; MI, myocardial infarction; UL, upper limb; RIPC, remote ischemic preconditioning; ⇑, improved outcome.
Uncertainties aside, many leading physicians and scientists remain hopeful that RIPC can be translated clinically. 7 Additional experimental and clinical research is needed to resolve to what extent RIPC may prove essential for conferring ischemic resistance in the heart and perhaps other organs. Foundational to this review is the premise that exercise biology is among the most sustainable, pragmatic, and cost-effective means of inducing an ischemia-resistant heart. As such, the discussion will now turn to novel understanding of exercise-induced cardioprotection in order to understand mechanistic underpinnings that may also translate to RIPC.
Exercise-Induced Cardioprotection and Implications for RIPC
Experimental understanding of exercise preconditioning prior to myocardial ischemia is well defined, consistently observed, and very reproducible across multiple species. The reader is directed to several extensive reviews by several leading research groups. 21 –24 Briefly, both long-duration training (months) and short-duration exercise exposure (days) confer cardioprotection against ischemic injury. 25,26 In the latter instance, observations of cardioprotection afforded by a few days of exercise support the notion that exercise preconditions the myocardium via acute upregulation of numerous endogenous protective mediators. 27 Observational similarities exist in the onset duration of preconditioning due to exercise and ischemic and pharmacologic approaches. All 3 preconditioning modes are characterized by the aforementioned transient early cardioprotective phase and more robust late-phase window of protection. 27 –29
In contrast to ischemic and pharmacologic methodologies, exercise preconditioning is unique in many respects, and confers a unique profile of cellular mediators of cardioprotection (Figure 1). The cardioprotective duration due to exercise is sustained for at least 9 days following the last aerobic exercise session, 30 roughly double the protective window observed following ischemic stimuli. 31 Even more importantly, the aging heart eventually becomes unresponsive to ischemic 13 and pharmacologic preconditioning, 32 while a few days of exercise preconditioning confers potent cardioprotection in hearts from senescent animals. 33 –35 These observational differences between exercise and ischemic stimuli were central to subsequent mechanistic investigations of exercise-induced cardioprotection and debunked prior notions that exercise was simply “another” form of cardiac preconditioning. 29,36 As a generalized summarization of the 3 preconditioning modes, ischemic and RIPC have a nearly identical phenotype and mechanistic profile, while exercise preconditioning is characterized by phenotypic and mechanistic differences.

Mechanisms of exercise-induced cardioprotection against ischemic injury. Short-term exercise elicits pleiotropic mechanisms of protection against ischemic injury. Upregulated factors include fortification of endogenous antioxidants, activation of ATP-sensitive K+ channels in the mitochondria and sarcolemma, and preservation of Ca2+ handling. Moreover, receptor-based cardioprotection likely occurs through δ-opioid and IL-6 (soluble and insoluble) activation. Given the nature of the exercise stimulus, this biochemical phenotype is important in understanding sustainable mechanisms of cardioprotection. Ca2+ indicates sarcoplasmic endoplasmic calcium ATPase (SERCA2A); δ, delta opioid receptor; GR, glutathione reductase; K+, ATP-sensitive potassium channels in the mitochondria and sarcolemma; IL-6, soluble and membrane bound interleukin-6 receptors; SOD2, manganese isoform of superoxide dismutase.
Based on the known mechanistic differences in the exercise stimulus versus ischemic and RIPC, several give particular insight into the fact that exercise is a unique physiologic and biochemical stressor. Perhaps most notable are differences in the inflammatory mediators across the various stimuli. For instance, inducible nitric oxide synthase (iNOS) and prostanoids, including cyclooxygenase-2 (COX-2), are hierarchically activated by ischemic ligation 37,38 and RIPC stimuli. 16,39 In contrast, neither iNOS nor COX-2 is essential to exercise-induced ischemic resistance. 34,40 This latter point is very important and reinforces a well-articulated biological position that inflammatory-derived cytoprotection is temporarily beneficial but untenable to long-term viability. 41 Based on the limited available evidence, the reader is cautioned against the formation of sweeping conclusions relative to iNOS and COX-2 in the context of chronic application of RIPC. Nonetheless, the concept remains a compelling avenue for future investigation and an important point of contrast between exercise and ischemic stimuli.
Alternately, a few cellular mediators of cardioprotection are common to exercise and ischemic preconditioning approaches, including RIPC. Overlapping protective mediators include KATP channel opening in the mitochondria and sarcolemma, 42 –45 endogenous opioid pathway activation, 46 –48 and release of circulating interleukins (including interleukin 6). 14,40,49,50 In this latter instance, however, activation of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway downstream of the IL-6 receptor appears to be fundamentally different for exercise 40 versus surgical ischemic 15,49 and RIPC 51 approaches. Comparisons between exercise and ischemic stimuli are complicated by the fact that a clinically viable means of pharmacologic KATP channel activation remains elusive. 43 Further complicating potential pharmacologic solutions to confer cardioprotection in at-risk hearts, application of opioid agonists evokes cellular habituation and a loss of ischemia resistance within a matter of days. 52 Based on existing data, 53 there is reason to suspect that similar habituation to prolonged opioid use may also occur relative to RIPC, although this outcome is currently untested. In contrast to these observations, exercise appears to offer unmitigated preconditioning so long as the stimulus is reintroduced in semiregular intervals.
Perhaps the most important implication exercise holds for RIPC is that circulating endogenous opioids (enkephalins specifically) and IL-6 appear to exert cytoprotective effects via their respective cardiac receptors. 40,47 The next logical experimental concept is to verify whether exercise confers ischemic resistance via remote factors. In this regard, available evidence is limited and conclusions tentative. In terms of opioid-mediated protection, the delta opioid receptor appears to be essential for exercise preconditioning, a point that is probably moot in comparison with RIPC because circulating enkephalins due to exercise are of cardiac origin. 46,47 In contrast, early findings indicate that postexercise spikes in circulating IL-6 come exclusively from skeletal muscle. 40 If correct, this finding serves as important first evidence that myokines mediate exercise-induced cardioprotection.
Given the multifaceted protection afforded by exercise, it seems likely that future research will uncover additional circulating protective factors, including myokines. One possibility includes adenosine, a well-established trigger for RIPC-associated benefits. 54 A wealth of experimentation is required, and the exercise literature lags far behind ischemic, pharmacologic, and RIPC subareas. Metabolomic understanding of exercise-induced cardioprotection is among the obvious next step approaches and would be an interesting comparison to recent RIPC findings. 55 Examination of microRNAs released by skeletal muscle in exercised and occluded limbs, however, may prove to be the most promising avenue by which a common mechanism of cardioprotection is discovered. Candidate microRNAs are being identified in response to exercise 56 and RIPC 57 stimuli, but integrative understanding of muscle-derived microRNAs is currently all but unknown.
Lessons From RIPC Applications for Improved Exercise Performance
In recent years, RIPC has been used with the intent of improving exercise performance. A quick survey of the literature reveals that, unlike clinical approaches in ischemic preconditioning/RIPC and emphases on tissue-to-tissue cross talk, exercise performance improvements are purportedly linked to acute alterations in limb blood flow. Remote ischemic preconditioning evokes a well-defined hyperreactivity response in the affected limbs. 58 –60 For all the speculation of underlying physiologic mechanisms, very little is currently confirmed about how transient limb occlusion may improve exercise performance. The reader is directed to an authoritative review of hyperemia during exercise as a means of reinforcing foundational physiologic concepts. 61 Suffice it to state that the hyperemia phenomenon in response to RIPC and exercise is complicated and includes numerous factors. Contributing physiologic factors include, but are not limited to, increased systolic blood pressure balanced against local and regional vasodilation. Other factors likely include vasodilation in conduit arteries, the potential influence of sympathetic drive and related neural feedback, circulating vasoactive factors, and local metabolic influences. Integrated understanding of these physiologic factors is essential to better understand RIPC influences on exercise outcomes, in addition to health and cardioprotection.
As with prior discussion, the reader is cautioned against sweeping generalizations of RIPC and exercise performance because of the likelihood that key dependent outcome measures are variably influenced by methodology. Interpretation of findings should incorporate understanding that hyperemic blood flow responses are impacted differently depending on which limbs are affected during RIPC and exercise, the total muscle mass that is engaged during exercise, and the physical condition of the subject (eg, elite athlete, highly fit, healthy unfit, diseased). 61 Indeed, recent investigations give important insight on subtle differences in RIPC methodology, including the influence of peak systolic cuff pressure on limbs of the upper and lower body. Key findings suggest blood flow changes are transient and that upper limbs are more responsive to a spectrum of occlusive pressures than lower limbs with a greater muscle mass. 62,63 Given these complexities, and the fact that many published RIPC and exercise studies are limited to performance outcomes without investigation of mechanistic cause and effect, it is not surprising that findings are sometimes equivocal. 59,64 –66
Nonetheless, RIPC is now well investigated and some general conclusions can safely be drawn. Fortunate for this summative interpretation, many studies report slight variations in common protocols, whereby 3 to 4 intervals of 3- to-5 minute ischemia–reperfusion and suprasystolic occlusion pressures 50 to 100 mm Hg above baseline are employed. Moreover, exercise performance is generally examined within 5 to 30 minutes of limb occlusion. The first conclusion that can be drawn is that matching of limb occlusion (lower vs upper body) to limb exercise is most often associated with improvements in exercise performance, 59,64,65,67,68 a collective finding that reinforces acute increases in limb blood flow as a likely contributing factor to improved exercise performance. Several comprehensive investigations support this notion with associative evidence of simultaneous improvements in exercise performance and increased limb blood flow. 58 –60 Future research that incorporates direct measures of vascular function is needed to better define physiologic control of exercise improvements due to RIPC. Next, overall improvements in aerobic exercise outcomes 59,64,65,67,68 are more frequently reported than alterations in VO2max 59,65 or anaerobic exercise performance. 60,69 These summarized findings sample a wealth of data that largely support the idea that RIPC offers some benefit (∼2%-5%) in acute exercise performance, a conclusion that is tentatively linked to manipulation of limb blood flow in healthy individuals.
The implications of RIPC and exercise on at-risk populations are less certain, although findings from recently published studies are revealing. A well-controlled examination of upper and lower limb RIPC prior to time trial cycling reported no benefit on exercise performance, but exercise-induced elevation in circulating cardiac troponin (cTnI) was mitigated following RIPC. 70 A related investigation of cTnI and cardiac troponin C (cTnC) quantified by high-sensitivity assay techniques was compared following endurance running. In contrast to prior findings, RIPC did not impact postexercise-induced spikes in circulating levels of either troponin isoform, but the time course of cTnI and cTnC profiles was reversed from that observed following myocardial infarction. 71 This understanding provides important insight for next-generation investigations in diseased and at-risk populations. Another well-controlled study in healthy participants examined electrocardiogram (ECG) changes to a steady-state exercise protocol following RIPC. Examination of ECG QT intervals revealed that, independent of heart rate, RIPC was associated with accelerated ventricular repolarization. 72 Thus, clinically directed outcome measures from exercise and RIPC investigations in humans appear to support experimental findings from animal studies and suggest that RIPC may confer some degree of cardioprotection, a conclusion that requires scientific verification. Less promising, however, are early findings of RIPC and exercise as applied to diseased populations. A recent investigation of clinical exercise test outcomes in claudicating patients found no benefit of RIPC. 73 These findings contrast the potent vasodilatory responses in healthy individuals and suggest that physiologic improvements to RIPC may be limited in those with compromised vascular tone or autonomic function. Whether physiologic and/or biochemical effects of RIPC can be afforded to diseased populations remains understudied and highlights an exciting avenue for future investigations.
Conclusion
The search for sustainable, pragmatic, and cost-effective solutions to cardiovascular disease is ongoing. Remote ischemic preconditioning has emerged in recent years as an adjuvant to clinical interventions. Furthermore, RIPC has emerged as a novel scientific approach to better understand the power of ischemic preconditioning in high-risk individuals. Limb-based RIPC exerts a potent cardioprotective phenotype, on par with preconditioning induced by coronary artery ligation. While viable therapies derived from coronary artery ischemic preconditioning and RIPC remain elusive, the latter approach overcomes many hurdles related to cost and treatment logistics. Central to this review, descriptive and mechanistic understanding of exercise preconditioning sheds new light on potential cardioprotective solutions. Findings from the exercise preconditioned heart are in stark contrast to the 2 forms of ischemic preconditioning. The cardioprotective benefits of exercise are multifaceted and include both physiologic and biochemical advantages that span multiple organ systems. This latter point is punctuated by established evidence of RIPC and improved exercise performance. Additional basic science and clinical research should be directed at better understanding the benefits of exercise and RIPC. Underpinning the importance of these future research efforts, exercise-based therapies stand alone in the conference of robust protection and anti-ischemic attributes in both aged and diseased hearts. Thus, exercise itself may serve as an adjuvant to current and yet-to-be-developed pharmacologic therapies to improve drug potency and/or mitigate untoward side effects.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
