Abstract
Ivabradine is a selective
Introduction
Hyperpolarization-activated cyclic nucleotide (HCN)-gated channels play a significant role in the pacemaker activity of immature cardiomyocytes and cardiac sinoatrial node cells. 1 This mechanism helps maintain normal heart rate (HR). However, some cardiac pathogeneses are due to dysfunctional HCN channels and lead to rhythm disorders. Therefore, blockade of these channels helps balance HR. 1 Blockade of HCN channels also leads to a reduction in the HR to normal levels because reducing elevated HR is important to reduce the risk of mortality and morbidity. Several studies found that elevated resting HR was a major risk factor for morbidity and mortality, especially all-cause and cardiovascular mortality. 1 -4 This blockade also improves cardiac performance and the remodeling of cardiac muscles in cases of cardiac hypertrophy. 5 One example of an HCN blocker is ivabradine (Procoralan, Corlanor). Ivabradine is approved and registered for use by several countries, including the United Kingdom, the United States, Australia, and other European and Middle Eastern countries. 6
The European Medicines Agency (EMA) approved ivabradine for use under the trade name of Procoralan in October 2005. It was approved for use in patients with chronic stable angina pectoris as a symptomatic treatment for patients with coronary artery disease (CAD) with normal sinus rhythm. It is used in patients who do not tolerate β-blockers or in patients in whom β-blockers are contraindicated. It is also used in combination with β-blockers in patients who cannot be controlled with maximally tolerated β-blocker doses and in patients whose HR is >60 beats per minute (bpm) in sinus rhythm. It is also used in patients with chronic heart failure (CHF), New York Heart Association (NYHA) classes II to IV with systolic dysfunction, and patients whose HR is at or above 75 bpm in sinus rhythm with or without β-blockers. 7
The drug was approved in January 2006 in the United Kingdom under the trade name Procoralan for use in patients with chronic stable angina pectoris as a symptomatic treatment for patients with an HR ≥70 bpm and normal sinus rhythm. Ivabradine may be used in adult patients who do not tolerate β-blockers or when β-blocker-only therapy is inadequate to alleviate disease symptoms. Ivabradine is also indicated for inclusion in standard therapy with or without β-blockers for patients with CHF NYHA classes II to IV with systolic dysfunction and patients whose HR is at or above 75 bpm in sinus rhythm. 8
The drug was approved in Australia in October 2006 under the trade name Corlanor for use in chronic stable angina that results from atherosclerotic CAD, as a symptomatic treatment in patients with an HR ≥70 bpm and normal sinus rhythm, in combination with atenolol 50 mg once daily, and in patients who are unable to tolerate β-blockers or have a contraindication to their use. Ivabradine is also indicated for the treatment of symptomatic NYHA Class II or III CHF with left ventricular ejection fraction (LVEF) ≤35% and adult patients with an HR ≥77 bpm with normal sinus rhythm in combination with standard CHF treatment. 9
The US Food and Drug Administration (FDA) approved Corlanor for use in 2015 in adults having stable CHF with LVEF ≤35% who are in sinus rhythm with a resting HR at or above 70 bpm and who have contraindications to β-blockers or are using the maximally tolerated doses of β-blockers to reduce the hospitalization rate from worsening in these heart failure (HF) cases. 10
Ivabradine Approval Status
The drug has been approved in 108 countries worldwide, and it is marketed in 93 countries. Most of these countries are located in the European Union. Twelve countries in the Middle East have approved the drug, including Saudi Arabia. The 5 and 7.5 mg doses (twice daily) as a film-coated tablet are approved in these countries. 6
Objective of This Review
This review assesses the benefit–risk profile of ivabradine. The primary interest in performing this review was the debate that arose from the Study AssessInG the Morbidity-Mortality BeNefits of the
Effectiveness of Ivabradine
The drug was evaluated by 5 clinical trials. The SHIFT, BEAUTIFUL, and SIGNIFY trials were the largest trials with significant results.
The SHIFT Study
The SHIFT was a large multicenter, international, randomized, controlled, double-blind placebo study conducted with 6558 patients in which 49.9% of the patients were treated with ivabradine and 50.1% of the patients were given placebo. 12 To be eligible for participation, all patients had stable chronic NYHA classes II to IV, CHF for more than 4 weeks with reduced LVEF (≤35%) and a resting HR ≥70 bpm, and were hospitalized at least once in the last 12 months prior to the study. The patients had a stable clinical regimen that was usually the maximally tolerated dose of β-blockers, angiotensin-converting enzyme inhibitors (ACEIs), or angiotensin receptor blockers (ARBs), spironolactone, and diuretics (Table 1).
Comparison Between the SHIFT, BEAUTIFUL, and SIGNIFY Trials.
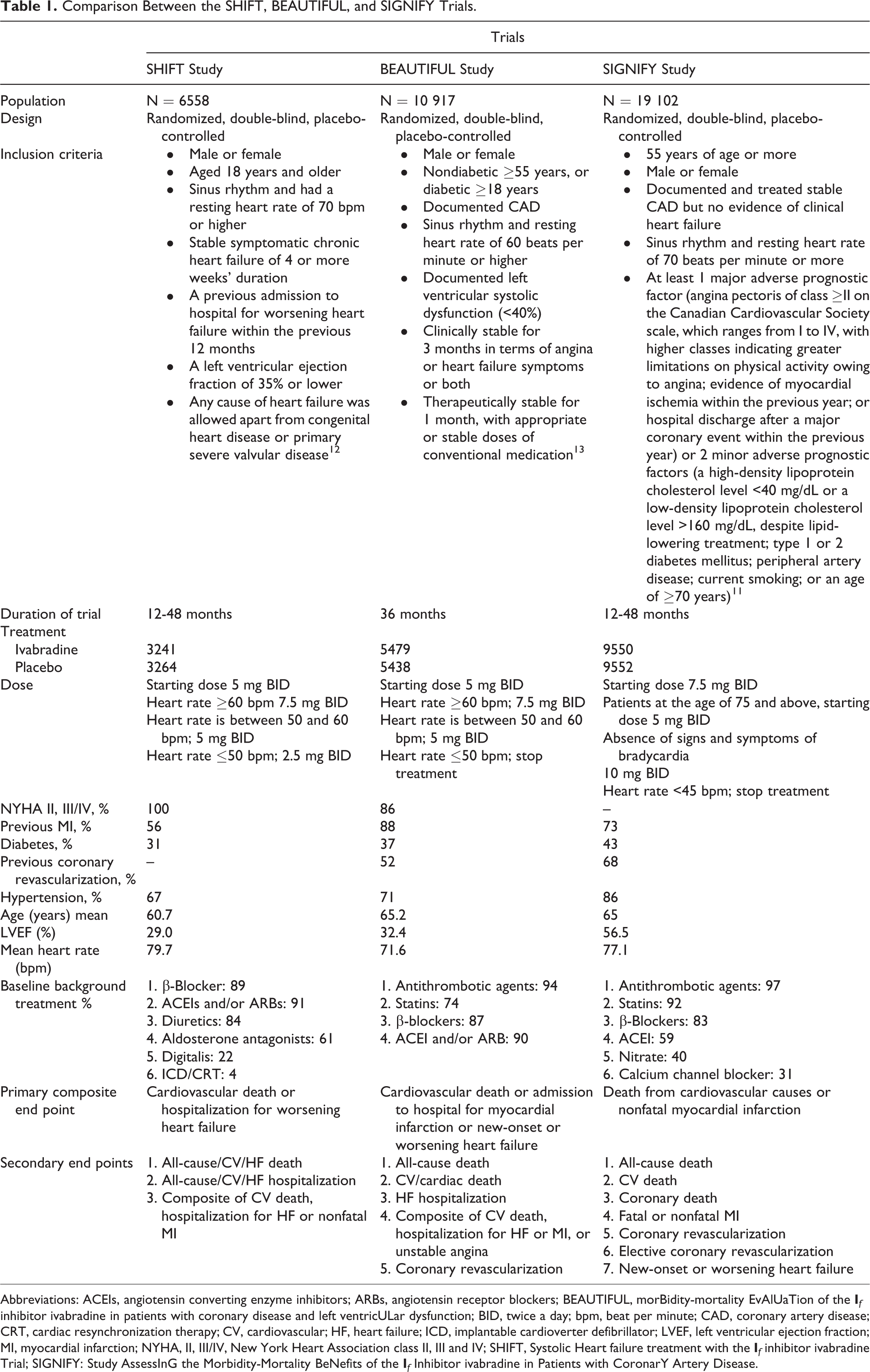
Abbreviations: ACEIs, angiotensin converting enzyme inhibitors; ARBs, angiotensin receptor blockers; BEAUTIFUL, morBidity-mortality EvAlUaTion of the
The patients in this study exhibited the following baseline characteristics—49% of the participants were CHF NYHA class II, 50% were CHF NYHA class III, and 2% were CHF NYHA class IV. The mean LVEF was 29%. β-Blockers (89%) were the most frequent drug group in these patients. The use of ACEIs and/or ARBs was reported by 91% of the patients, followed by diuretics (84%) and antialdosterone agents (61%). The median follow-up duration was 22.9 months (Table 1). During the first month, 67% of the treated group received 7.5 mg ivabradine twice daily. The mean HR was reduced by 15 bpm in this treatment group from the baseline HR of 80 bpm. In addition, HR was reduced by 10.8 bpm after 28 days of treatment, 9.1 bpm after 12 months, and 8.3 bpm after 24 months of treatment compared to the placebo group.
Statistically significant differences between the ivabradine and placebo groups with respect to the primary aim of the study were evident. Ivabradine reduced the rate of the primary composite end point of cardiovascular mortality and hospitalization for worsening HF by 18% (hazard ratio [HR]: 0.82, 95% confidence interval [CI]: 0.75-0.90, P < .0001). However, this reduction was primarily reflected by the end point of hospitalization for worsening HF.
Figure 1 shows the Kaplan-Meier curve that reflects the time to the first occurrence of hospitalization for worsening HF or cardiovascular death in the study. For subgroup analyses, patients with NYHA classes II and III exhibited reductions in the primary end point, but this reduction was not observed in patients with NYHA class VI. In addition, patients on standard doses of β-blockers exhibited significant reductions in the primary end point, but this reduction was not observed in patients on higher doses of β-blockers.

The Kaplan-Meier curve of time to first event of primary composite end point. Adapted from Swedberg et al.12
The BEAUTIFUL Trial
The BEAUTIFUL trial (2008) was a large, randomized, double-blind, placebo-controlled trial study of 10 917 adult patients with CAD with impaired LVEF of <40% and a resting HR ≥60 bpm. 13 The trial evaluated the primary end point of the composite time to first hospitalization for new-onset or worsening HF, hospitalization for acute myocardial infarction (MI), and cardiovascular death. Based on tolerability and resting HR, 5479 patients received 5 mg ivabradine with an increased dose of up to 7.5 mg twice daily and 5438 patients received a matched placebo. As inclusion criteria, patients had to have stable symptoms of HF and/or angina for at least 3 months and have received conventional cardiovascular medications at stable doses for at least 1 month.
Treatment with β-blockers was not required as inclusion criteria. However, 87% of the patients were receiving β-blockers. The median follow-up was 19 months (Table 1). There was no difference in the rate of the primary composite outcome between the ivabradine and placebo groups (HR: 1.00, 95% CI: 0.91-1.10, P = .94).
However, 1 major concern was the inclusion criteria of an HR ≥60 bpm. Inclusion of patients with an HR equal to 60 could lead to patient discontinuation because 86% of the patients were already on β-blockers. These results should not be generalized to patients whose HR was over 70, 75, or 80 bpm.
Nevertheless, the study demonstrated a benefit to taking ivabradine as a treatment for CAD. Ivabradine reduced the secondary end point of admission to hospitals for fatal or nonfatal MI (HR: 0.64, 95% CI: 0.49-0.84, P = .001) and coronary revascularization (HR: 0.70, 95% CI: 0.52-0.93, P =.016). 13 These results were reported in a subgroup of the study population with an HR of 70 bpm or greater.
The SIGNIFY Trial
The SIGNIFY study (2014) was a randomized, double-blind, placebo-controlled trial examining ivabradine in 19 102 patients (ivabradine = 9550 and placebo = 9552) with stable CAD.
11
This study assessed the morbidity and mortality benefits of the
Participants treated with ivabradine began the study at a dose of 7.5 mg twice daily, and the dose could be increased to 10 mg twice daily (unapproved dose) or reduced to 5.0 mg twice daily with a mean dose of ivabradine of 8.2 mg twice daily. The targeted HR was 55 to 60 bpm. Treatment with β-blockers was not required, but 83% of the sample was on β-blockers. The median follow-up duration was 24 months (Table 1).
The main study outcome was no difference between the 2 arms with respect to the primary composite end point (HR: 1.08, 95% CI: 0.96-1.20, P = .20). Subgroup analyses revealed that ivabradine was associated with an increased incidence of the primary composite end point in patients who had angina of Canadian Cardiovascular Society class II or higher (7.6% vs 6.5% with placebo; HR, 1.18; 95% CI: 1.03-1.35; P = .02). However, this association was not observed in patients without angina or patients who had class I angina (HR: 0.89, 95% CI: 0.74-1.08, P = .25).
Safety
Clinical trials are an initial point in determining safety because the nature of clinical trials (eg, low number of patients, short duration, type of patients, etc) cannot provide full safety information. The most common adverse events associated with ivabradine were bradycardia, atrial fibrillation, phosphenes (luminous phenomena), and hypertension. Postmarketing information revealed that ivabradine was associated with rash, diplopia, angioedema, pruritus, urticaria, visual impairment, erythema, and vertigo.
Patients with a risk of bradycardia should be monitored. Therefore, ivabradine should not be started in patients with an HR lower than 70 bpm. Patients with an atrioventricular block of the second degree should avoid this drug. Ivabradine should not be used during pregnancy because it exhibited reproductive toxicity in animal studies.
The BEAUTIFUL Study
Bradycardia was a major adverse event in the ivabradine group, which was the reason for discontinuation of the study in some cases (13% vs 2% placebo). There was a safety concern of increased risk of cardiovascular death in the ivabradine group, despite the small and insignificant difference. However, this risk was higher in patients with an HR less than 70 bpm. 13
The SHIFT Study
This trial found that the incidence of all serious side effects was lower in the ivabradine group compared to the placebo group (45% vs 48%, P = .025). Bradycardia was the most common adverse event that led to discontinuation in 48 (1%) patients in the ivabradine group and 10 (<1%) patients in the placebo group (Tables 2 and 3). 12
Incidence of Serious Adverse Events in the SHIFT Study Arms. 12

Abbreviation: SHIFT, Systolic Heart failure treatment with the
Most Common Adverse Events in the SHIFT Study. 12

Abbreviation: SHIFT, Systolic Heart failure treatment with the
The SIGNIFY Trial
There was a significantly greater incidence of bradycardia in the ivabradine group compared to the placebo group for symptomatic bradycardia (7.9% vs 1.2%) and asymptomatic bradycardia (11.0% vs 1.3%; Table 4). In all, 37.6% of the ivabradine group experienced serious adverse events compared to 35.4% of the placebo group (P = .001). These events were considered cardiac disorders in 19.0% of the ivabradine group and 16.7% of the placebo group. 11
Adverse Events in Both SIGNIFY Study Arms. 11

Abbreviation: SIGNIFY, Study AssessInG the Morbidity-Mortality BeNefits of the
World Health Organization Database “VigiBase”
VigiBase is a database of the World Health Organization that includes adverse drug reactions (ADRs) sent by National Pharmacovigilance Centers worldwide (over 110 countries). This database contains over 10 million individual case safety reports. VigiBase revealed that the total number of reports for ivabradine was 1449, with the most events reported in 2014 (25%) and 2015 (21%). For gender distribution, 51% of the reports were of males. The most frequent events reported with ivabradine use affected the cardiac system, and there were 578 (40%) reports in the database. A total of 483 (84%) of these reports were associated with cardiac arrhythmias. The second organ system reported in the database was nervous system disorders with 315 (22%) reports. Most of these reports (50%) occurred in patients aged 65 and older, and 29% of the reports were of patients between 45 and 64 years.
Notably, these events were not necessarily associated with drug use because they are ADR reports, and causality and association cannot be concluded. In addition, 50% of the VigiBase reports came from the FDA’s adverse event reporting system, and there were no reports from the FDA because this drug was recently approved. 14
International Regulatory Action
Ivabradine is approved in 108 countries worldwide, including 12 countries in the Middle East. Most of these countries approved 5 and 7.5 mg twice daily. Several regulatory bodies reviewed the safety profile after the SIGNIFY results to assess the benefit–risk balance of the drug.
US FDA
The US FDA recently approved ivabradine in April 2015. The 3 trials were reviewed during the approval process. Both doses (ie, 5 and 7.5 mg twice daily) were approved for use in adults having stable CHF with LVEF ≤35% and a resting HR ≥70 bpm who have contraindications to β-blockers or are taking the maximally tolerated doses of β-blockers to reduce hospitalization rates from worsening in these HF cases. The HR should be ≥70 bpm, which is a very important point to remember when treating patients. There was no black box warning on the US FDA label, but there was a medication guide for the drug. 15
European Medicines Agency
The EMA reviewed the available data on ivabradine and assessed the results of SIGNIFY. The EMA updated their recommendations and e-drug profile on June 02, 2015, based on their review. “The revised recommendations include 2 indications which read as follows: Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in adults with CAD having normal sinus rhythm and HR ≥70 bpm. Ivabradine is indicated: in adults unable to tolerate or with a contraindication to the use of β-blockers or in combination with β-blockers in patients inadequately controlled with an optimal β-blocker dose. Ivabradine is indicated in CHF NYHA II to IV classes with systolic dysfunction, in patients in sinus rhythm and whose HR is ≥75 bpm, in combination with standard therapy including β-blocker therapy, or when β-blocker therapy is contraindicated or not tolerated.”
8,16
Medicines and Healthcare Products Regulatory Agency
The Medicines and Healthcare Products Regulatory Agency (MHRA) is the regulatory body of the United Kingdom that also assessed the safety profile and the benefit–risk balance for ivabradine following the SIGNIFY trial. The agency published similar recommendations as the EMA, and these recommendations were incorporated into the summary product characteristics (SPC). The new indications for ivabradine are described below.
Controlling symptoms of chronic stable angina pectoris
Ivabradine may be prescribed for patients with CAD having normal sinus rhythm and an HR at or above 70 bpm to control symptoms of chronic stable angina pectoris. It is suitable for use in adult patients with contraindications to β-blockers or when the maximum β-blocker dose is inadequate to manage disease symptoms. 17
Treatment of CHF
Patients in sinus rhythm with an HR that is ≥75 bpm and who have NYHA classes II to IV and CHF with systolic dysfunction may use ivabradine alone or in combination with β-blockers as a standard therapy. 8,17
Australian Therapeutics Goods Administration
The Therapeutics Goods Administration (TGA) also assessed the safety profile of ivabradine and provided new recommendations. These recommendations are similar to the EMA and MHRA, but there are slight differences. These recommendations were added to the SPC for the drug, which indicate ivabradine use for: “Symptomatic treatment of chronic stable angina due to atherosclerotic CAD in patients with normal sinus rhythm and HR at or above 70 bpm, who are unable to tolerate or have a contraindication to the use of β-blockers, or in combination with atenolol 50 mg once daily when angina is inadequately controlled. Treatment of symptomatic CHF of NYHA classes II or III and with documented LVEF ≤35% in adult patients in sinus rhythm and with HR at or above 77 bpm, in combination with optimal standard CHF treatment.”
9
Saudi FDA
The Saudi FDA also assessed the safety profile of ivabradine after the SIGNIFY trial. The drug was withdrawn from the market in 2015 following their evaluation of a negative benefit–risk balance. 18
Ivabradine Benefit–Risk Balance
Ivabradine was approved in 108 countries since the first approval of ivabradine in October 2005 in France, and the United States and China are the most recent countries to approve it for use. There are 2 doses of ivabradine, 5 and 7.5 mg twice daily, and all of these countries approved both doses. 6 Ivabradine has 2 indications: (1) symptomatic treatment of chronic stable angina pectoris and (2) treatment of CHF. Previous guidelines mandated that patients who use this drug have an HR of 60 bpm or greater, but several regulatory agencies altered their recommendations for ivabradine use only in patients with an HR of 70 bpm or greater based on the new evidence of a risk for patients if their HR is less than 70 bpm. These recommendations vary between agencies.
The drug was studied in several clinical trials after approval, and these trials were funded by the pharmaceutical company that produces the drug. The BEAUTIFUL, SHIFT, and SIGNIFY trials examined different types of populations. The BEAUTIFUL trial was conducted in patients with CHF and CAD having a resting HR ≥60 bpm, and the SHIFT trial was conducted in patients with CHF and an HR ≥70 bpm. The recent SIGNIFY trial was conducted in patients with CAD and an HR ≥70 bpm without clinical HF (LVEF > 40%). Therefore, some similarities in the types of populations in the SIGNIFY and BEAUTIFUL trials with respect to CAD were noted, but there were differences in HR and the inclusion of patients with CHF. Similarly, there was a similarity between the BEAUTIFUL and SHIFT trials because both trials included patients with CHF, but there was a difference in HR and the inclusion of CAD in the BEAUTIFUL trial (Table 1). These differences in the types of trial populations were the primary reason for some of the conflicting results in the trials, and these differences confounded readers and reviewers in the assessment of the SIGNIFY trial results.
Ivabradine exhibited beneficial effects in patients with CHF having an HR of ≥70 bpm in the 3 trials, which was also found in the SHIFT trial. This beneficial effect was not supported in the BEAUTIFUL trial, which included patients with CHF. However, patients with an HR ≥60 bpm were included in the study, which may have affected the results and trial conclusions. The BEAUTIFUL trial also included patients with CAD, but the SHIFT trial investigators found that approximately 56% of the patients had a medical history of MI but not angina. The SIGNIFY trial was conducted in patients with CAD but without CHF, and therefore, the results of this trial should only be applied to similar populations and not patients with CHF. The SIGNIFY trial examined a new dose regimen of 10 mg (not an approved dose), which may have affected the results, particularly because the rate of bradycardia was higher in the ivabradine group than the placebo group (18.0% vs 2.3%, P < .001).
Ferrari and Fox discussed this difference. They stressed that the SIGNIFY trial used higher doses (10 mg twice daily), which led to a higher HR-lowering effect (<55 bpm) than the SHIFT trial (60% vs 48%, respectively) and more bradycardia (18% vs 10%, respectively). They mentioned that the primary difference between the 2 trials, which may explain the difference in the effect of ivabradine, was that the reduction in HR was associated with an improvement in patients in the SHIFT trial because it included patients with left ventricular (LV) dysfunction, but normal LV function as in SIGNIFY. Therefore, ivabradine exhibited an effect in patients with low LVEF but not with normal LVEF because the primary effect of ivabradine as an
They also discussed the role of HR in CAD. Ivabradine improves angina symptoms by reducing HR, and it is used with β-blockers for this purpose. This beneficial effect was clearly demonstrated in improving the class of angina in the SIGNIFY trial, but it did not demonstrate a prognostic benefit. Furthermore, β-blockers never demonstrated a prognostic benefit in patients with stable CAD. 19
The SIGNIFY trial reported a statistically significant difference between ivabradine and the placebo because ivabradine was associated with an increased combined risk of cardiovascular death and nonfatal heart attack (7.6% vs 6.5% in the placebo group; HR: 1.18, 95% CI: 1.03-1.35, P = .02). This analysis was performed on a subgroup of patients, which was not sufficiently powerful to detect differences. However, this risk should be considered.
Nevertheless, there was evidence that β-blockers could affect the results of the SHIFT trial because ivabradine exhibited beneficial effects on cardiovascular death and hospital admission for worsening HF as 89% of the participants were using β-blockers. β-Blockers affect patients with CHF, which is observed in ventricular remodeling. However, this result should be considered for the treatment of cases using β-blockers together with ivabradine, if needed.
A review by McMurray discussed that the interaction between the cause of left ventricular systolic dysfunction (LVSD) and ivabradine use helped explain the conflict between the 3 trials. However, this review suggested that the results of the BEAUTIFUL and SHIFT trials might be weighed more heavily than SIGNIFY due to the greater similarity of patient populations in BEAUTIFUL and SHIFT. However, there is a difference in the reason for LVSD because one-third of the SHIFT population with HF and reduced ejection fraction was due to nonischemic reasons. This difference made this review stress that ivabradine has little or no effect on patients with CAD. Furthermore, ivabradine usually has an effect in patients with reduced ejection fraction and without CAD. The author mentioned that other medications exhibit the same beneficial effect of ivabradine on nonischemic HF with reduced ejection fraction. Examples of these medications are β-blockers, digoxin, and amiodarone. Their effect on nonischemic CHF occurs because this condition is reversible, but ischemia is not. 20
Ferrari and Fox discuss the role that HR reduction plays in CAD and HF in another review. This review addresses the role and mechanism of HR in CAD in humans and animals. Ferrari and Fox also evaluated these attributes in patients with HF. The authors note that elevated HR affects the progression of atherosclerosis, which may lead to damage in the coronary arteries. This effect is not limited to the coronary arteries, but the results of this damage (eg, MI or angina) may lead to LV dysfunction. Therefore, HR reduction is essential to avoid any deterioration in LV function. Data show that HR reduction improves microcirculation and coronary flow reserve, and this effect was observed with ivabradine rather than β-blockers. Therefore, HR reduction should improve prognosis. 21
Ferrari and Fox also mention that the BEAUTIFUL trial data indicate that elevations in HR (≥70) are associated with an increased risk of cardiovascular death compared to HR <70 bpm, and reduction in HR may significantly reduce the risk of MI and coronary revascularization. This was further tested in the SIGNIFY trial, but the SIGNIFY population included normal LV function in patients with CAD. Moreover, there was no effect on the primary or secondary end points. The authors compared these results with the results of the SHIFT trial, which reported a significant effect of ivabradine in patients with HF compared to the placebo. Ferrari and Fox also explain the difference between the trials using a similar explanation as McMurray. The first explanation was based on the results of the SHIFT and the BEAUTIFUL trials and concluded that the difference occurred because one-third of the SHIFT population included patients with HF and reduced ejection fraction that resulted from nonischemic reasons. The second explanation was due to the SHIFT and the SIGNIFY trials, which indicated that elevated HR was a risk factor, but only in cases of reduced LV. 21
The aforementioned importance of LVEF was emphasized by the US FDA decision to approve ivabradine only in adults with stable CHF having LVEF ≤35% and a resting HR ≥70 bpm. Ivabradine may not be beneficial when LVEF >40%.
The drug manufacturer funded all of these trials, and clearly this influenced the differences in these populations because the manufacturer has investigated this drug in new populations. For example, SIGNIFY only tested patients with CAD and used a new higher dose regimen of 10 mg. Therefore, each trial should be analyzed alone based on the conditions performed. Table 1 provides a comparison between the trials.
All the studies were clinical trials, which are generally conducted and monitored in strict environments. However, an epidemiological study should be performed to assess real-life situations, especially in patients who generally exhibit other comorbidities and use multiple medications.
Most actions from regulatory bodies considered the benefit–risk balance, and all the aforementioned information and clinical and statistical evidence indicate a positive benefit–risk balance for ivabradine, especially in HF indicators. This benefit was noted in the recent approval by the US FDA and the labeling changes by EMA, MHRA, and TGA.
Conclusion
Ivabradine is an option in patients with HF and some patients with symptomatic angina. The aforementioned information suggests that the drug exhibits a positive benefit–risk balance, and it will be of good value to the patients. However, there is the need to provide minimization tools to mitigate the risk, such as risk management plans, including a medication guide, and a need to conduct further postauthorization safety studies to assess the benefit–risk balance using real-world data.
Footnotes
Author Contributions
TMA contributed to conception and design, contributed to acquisition and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
