Abstract
Background:
Whether the reduction of heart rate with ivabradine (IVA) could affect sympathetic activation and cardiac innervation in heart failure (HF) remains unknown.
Purpose:
The present study assessed the chronic effects of IVA and β-blocker on the systemic and local sympathetic nervous systems of hypertensive animals with HF.
Methods and Results:
The Dahl salt-sensitive rats received chronic IVA, bisoprolol (BIS), or placebo (CTL) therapy. The survival of the animal models with IVA and BIS significantly improved (median; 19.7 in IVA and 19.7 in BIS vs 17.0 weeks in CTL, P < .001). A similar decrease in 24-hour heart rate (mean; 305 in IVA and 329 in BIS vs 388 beats/min in CTL, P < .001) without effect on blood pressure, and an improvement in the left ventricular dysfunction (mean fractional shortening; 56.7% in IVA and 47.8% in BIS vs 39.0% in CTL, P < .001) were observed in the animals with IVA and BIS. However, a negative inotropic effect was only observed in the animals with BIS. Excessive urinary noradrenaline excretion in animals with CTL was only suppressed with the use of IVA (mean; 1.35 μg/d in IVA and 1.95 μg/d in BIS vs 2.27 μg/d in CTL, P = .002). In contrast, atrial noradrenaline and acetylcholine depletion in the animals with CTL improved and the tyrosine hydroxylase expression in the both atria were restored with the use of both IVA and BIS.
Conclusions:
IVA therapy improved the survival of hypertensive animals with HF. Furthermore, it was associated with the amelioration of systemic sympathetic activation and cardiac sympathetic and parasympathetic nerve innervations. Chronic β-blocker therapy with negative inotropic effects had beneficial effects only on cardiac innervations.
Introduction
The sympathetic nervous system (SNS) is important for compensatory mechanisms that maintain cardiovascular homeostasis. Heart failure (HF) has been thought to be a condition associated with SNS activation, and it is a reflex reaction to changes in cardiac and peripheral hemodynamics, which is initially compensatory one, but ultimately pathological one. 1 Chronic β-blocker therapy ameliorates left ventricular (LV) dysfunction, reverses LV remodeling, and decreases the risk of hospitalization and mortality of individuals who experienced HF, and it is now one of the established treatments for HF with reduced ejection fraction. 2 A variety of cardioprotective mechanisms have been attributed to the use of β-blockers. They include the protection against the deleterious myocardial effects of catecholamines and the pharmacological actions, such as negative inotropic and chronotropic effects associated with the reduction in myocardial oxygen consumption. 3
Heart rate (HR) is associated with clinical outcomes in a variety of cardiovascular diseases, such as hypertension and HF. Ivabradine (IVA) is an HR-lowering agent that acts specifically on the sinoatrial node by inhibiting the If (funny channel) current of the pacemaker cells. 4 In comparison with β-blockers, it does not have negative inotropic effects, and it has no considerable impact on blood pressure. In the SHIFT trial, reducing HR by 11 beats/min with the use of IVA decreased the primary endpoint (cardiovascular death and hospitalized HF). 5 The HR may be a modifiable factor, or IVA itself may have some protective effects against HF in addition to HR reduction (pleiotropic action). Although HR is regulated by autonomic balance and autonomic activity plays an essential role as a pathogenetic factor of and a prognostic index of HF, the effect of the therapeutic agent on the autonomic nervous system is not fully understood. 6 Therefore, the present study utilized the Dahl salt-sensitive (DS) rats with HF, which have been associated with the activation of SNS. 7 Furthermore, the chronic effects of IVA on sympathetic activity and cardiac innervation were assessed and compared with those of β-blockers in this animal model with HF.
Methods and Materials
Experimental Protocol
Male inbred DS rats were fed with 8% high-salt (HS) diet after 6 weeks of age 8 and 0.3% low-salt (LS) diet was given throughout the experiment to only the rats of the LS group (n = 16), which have no hypertension or no left ventricular hypertrophy (LVH) and dysfunction throughout the study. After 11 weeks (LVH stage), the animals that were on HS diet received chronic IVA (IVA group, n = 39), bisoprolol (BIS group, n = 38), or placebo (CTL group, n = 38) treatment. 10 mg/kg/d of IVA was given in drinking water based on a previous study. 9 Moreover, 4 mg/kg/d of BIS was incorporated into the diet. This dosage was selected to lower HR without affecting blood pressure. 7
In the first experiment, the rats (n = 66) were monitored, and death was recorded till 21 weeks. Serial blood pressure and echocardiographic measurements were obtained. In the second series, they (n = 65) were sacrificed at 17 to 18 weeks of age (HF stage), to obtain blood samples and heart tissues (Supplementary Figure 1). The animals were treated in accordance with the guidelines of the Kindai University Faculty of Medicine.
Blood Pressure/HR Measurements and Echocardiography
Systolic blood pressure (SBP) and HR were cautiously measured by the tail-cuff method at 10, 13, and 16 weeks. The telemetry (TA11PA-C10, Data Sciences International, St Paul, ML ) was performed for measuring the BP and HR in conscious rats over 24 hours at approximately 15 weeks (n = 6 in each group), as described previously. 7 The LV geometry and function was evaluated by echocardiography under light anesthesia with 1.5% isoflurane at 16 weeks. 8 The LV end-diastolic dimension (LVDd), end-systolic dimension (LVDs), posterior wall thickness (PWT), intraventricular septal wall thickness (IVST), and left atrial (LA) dimension (LAD) were measured by short-axis view. The LV fractional shortening (FS), ejection fraction (EF), and meridional end-systolic wall stress (SWS) were calculated (LV SWS; SBP × LVDs × 1.35/4 × PWTsystole × [1 + PWTsystole]/LVDs). 8 The HR-corrected mean velocity of circumferential fiber shortening (cVcf) was calculated and LV SWS–cVcf relationship as an index of HR- and load-independent systolic function was examined. 10 Diastolic functional parameters such as early-to-late transmitral filling ratio (E/A) and the ratio of peak velocity of early mitral inflow to early diastolic velocity of the mitral annulus (E/E´) were also calculated by transmitral Doppler flows and Doppler tissue signal from the lateral mitral valve annulus. 11
Serum, Urinary, and Tissue Biochemical Measurements
Urine samples were collected for 24 hours in metabolic cages in the rats of 16 weeks old. Urinary parameters such as urinary sodium, potassium, creatinine, nitrogen, total protein, and albumin or noradrenaline (NAd) and normetanephrine were measured. The rats were sacrificed at 17 to 18 weeks, and serum sodium, potassium, creatinine, and urea nitrogen were measured by commercially available kits. The LV and atrial tissues were homogenized in 0.4 or 0.1 M perchloric acid containing ethylenediaminetetraacetic acid for the measurement of NAd or acetylcholine (ACh) concentrations, respectively. The NAd, normetanephrine, and ACh were determined with automated high-performance liquid chromatography. 7,12
Histology
The formalin-fixed and paraffin-embedded tissues (LV and atria) were sliced into 5 μm sections. Hematoxylin–eosin and Masson’s trichrome stainings were performed for the assessment of the myocyte diameters and myocardial interstitial fibrosis, as previously described. 7 Immunofluorescence was performed using anti-tyrosine hydroxylase (TH) antibody (1:300, Abcam, Cambridge, UK/Merck, Darmstadt, Germany), followed by Alexa 594-conjugated anti-rabbit immunoglobulin G (1:1000, Invitrogen, Carlsbad, CA), and the nuclei were counterstained using 4′,6-diamidino-2-phenylindole. Immunohistochemical staining for TH was performed using the UltraTek HRP Anti-Polyvalent (DAB) Staining System (Scytek Laboratories, Logan, UT) according to the manufacturer’s instructions.
Gene and Protein Expression Analysis
RNA was isolated from LV and atria using TRIzol followed by treatment with DNase I (Invitrogen). First-strand complementary DNAs were synthesized using the SuperScript first-strand synthesis system kit (Invitrogen) for quantitative real-time polymerase chain reaction (PCR) in StepOnePlus system (Applied Biosystems, Carlsbad, CA). The messenger RNA (mRNA) levels were measured using specific primers (Supplementary Table) and normalized to an endogenous control, 18S ribosomal RNA mRNA.
Forty milligrams of the right atrium (RA) and LA and LV myocardium were homogenized in a 1 mL lysis buffer (CST, Danvers, MA). Each isolated protein sample (40 μg) was electrophoresed on 4% to 12% Bis–Tris gels and was transferred to polyvinylidene difluoride membranes. They were incubated with rabbit anti-TH antibody (1:10 000, Abcam) or rabbit anti-GAPDH (1:10 000, CST). The proteins were detected with ECL Prime (GE Healthcare, Little Chalfront, UK).
Statistical Analyses
The groups were compared using analysis of variance (ANOVA) and the post hoc Tukey honestly significant difference test for the individual group comparison was performed only when there is a significant difference by ANOVA. Two-way repeated-measures ANOVA was used to compare the serial measurements in body weight (BW), HR, or SBP. The survival was analyzed by the Kaplan-Meier method, and a comparison was performed using the log-rank test. All data were reported as mean (standard deviation). A P value less than .05 was considered statistically significant.
Results
Survival Analysis
All animals in the CTL group died from severe LV dysfunction followed by pulmonary congestion between 14.7 and 20.7 weeks (median survival: 17.0 weeks). The IVA (19.7 weeks) and BIS groups (19.7 weeks) had a significantly prolonged survival (P < .01) than the CTL group (Figure 1). There was no significant difference in terms of survival time of the IVA and BIS groups, which indicated that IVA and β-blocker had comparable efficacies.

Effects of ivabradine and bisoprolol treatment on survival. BIS indicates bisoprolol group; CTL, control group; IVA, ivabradine group.
Blood Pressure/HR and Echocardiographic Measurements
The BWs of the IVA and BIS groups increased (both P < .05 vs the CTL group). No differences were observed among the 3 groups in terms of SBP. The IVA and BIS groups had a similar decrease in HR during the entire study period (Figure 2A). Moreover, telemetric analysis showed no significant differences in terms of the mean blood pressures of the animals from the 3 groups that were approximately 15 weeks old over 24 hours (Figure 2B). Both the IVA and BIS groups have comparable reduction in HR. Mean HRs differed between at daytime and nighttime in the CTL and IVA groups. However, they were conflicting. Echocardiography revealed improved LV FS and EF, and the significantly suppressed progression of LV hypertrophy (LV mass) and SWS at 16 weeks (the HF stage) in both the IVA and BIS rats (Table 1). Also, the LV SWS–cVcf relationship showed that IVA had preserved systolic function and that BIS had slightly suppressed systolic function as compared with LS group, independent of HR and loading condition (Figure 2C). In the diastolic parameters, IVA showed decreased both E/A and E/E´ and BIS showed decreased E/A. In the BIS group, LV enlargement and the decrease in systolic parameter and the increase in diastolic functional parameter were admitted as compared with the IVA group, suggesting BIS had some negative inotropic/lusitropic effects.

Changes in hemodynamics and echocardiographic parameters. (A) Body weight, blood pressure, HR at 10 to 16 weeks, (B) 24-hour mean HR, mean blood pressure at 15 W. (C) LV SWS–cVcf relationship and (D) diastolic functional parameters in echocardiography at 16 weeks. The values were presented as mean (SD). BIS indicates bisoprolol group; BP, blood pressure; CTL, control group; cVcf, HR-corrected mean velocity of circumferential fiber shortening; E/A, early-to-late transmitral filling ratio; E/E', the ratio of peak velocity of early mitral inflow to early diastolic velocity of the mitral annulus; HR, heart rate; IVA, ivabradine group; LS, low-salt group; LV, left ventricle; SD, standard deviation; SWS, end-systolic wall stress.
Echocardiographic Parameters at 16 Weeks.a

Abbreviations: BIS, bisoprolol group; CTL, control group; Dd, dimension at diastole; Ds, dimension at systole; EF; ejection fraction; FS, fractional shortening; HR, heart rate; IVA, ivabradine group; IVSTd, intraventricular septal thickness at diastole; LAD; left atrial dimension; LS, low-salt diet group; LV, left ventricle; PWTd, posterior wall thickness at diastole; RDN, renal sympathetic denervation; SBP, systolic blood pressure; SWS, end-systolic wall stress.
aValues were presented as mean (SD).
bP < .05 versus the CTL group.
cP < .05 versus IVA group.
Serum, Urinary, and Tissue Biochemical Data
The urinary NAd and its metabolite normetanephrine excretions were significantly increased in the CTL group than in the LS group at 15 weeks, suggesting sympathetic overactivity in the CTL animals. By contrast, only the IVA group had significantly decreased urine NAd/normetanephrine excretion (Figure 3A). The high serum NAd and adrenaline concentrations observed in the CTL group were significantly inhibited in the IVA and BIS groups at 17 weeks. The reduced LV NAd amount in the CTL group was significantly restored only with the use of BIS, which indicated the preserved LV sympathetic function. By contrast, the NAd amount in the RA and LA was significantly restored in both the IVA and BIS groups (Figure 3B). Furthermore, the decreased ACh content in the RA was restored in both the IVA and BIS groups, whereas that in the LA was restored only in the BIS group (Figure 3C).

Changes in catecholamines and acetylcholine levels in the urine and cardiac tissues. A, Noradrenaline and normetanephrine levels in the urine. B, Noradrenaline levels in the tissues; left ventricle and right and left atria. C, Acetylcholine levels in the right and left atria. The values were presented as mean (SD). BIS indicates bisoprolol group; CTL, control group; IVA, ivabradine group; LS, low-salt group; SD, standard deviation.
At 17 weeks, the BIS group had higher creatinine clearance than the CTL group. By contrast, the IVA group showed a significantly lower urine albumin excretion than the CTL group (Table 2).
Serum/urine Measurements and Heart/Lung/Kidney Weights at 17 Weeks.a

Abbreviations: BIS, bisoprolol group; BW, body weight; Ccr, creatinine clearance; CTL, control group; IVA, ivabradine group; LA, left atrium; LS, low-salt diet group; LV, left ventricle; RA, right atrium.
aValues were presented as mean (SD).
bP < .05 versus the CTL group.
Histological Analysis of the LV and Atria at 17 Weeks
The IVA and BIS groups had a significantly reduced lung weight to BW ratio than the CTL group, which indicated the decrease in pulmonary congestion (Table 2). Moreover, significant reductions in both LV/BW and LA/BW ratios were observed. The CTL group presented with myocyte hypertrophy and myocardial fibrosis in the LV. The IVA and BIS groups had significantly less myocyte diameters (14.5 [0.7], 20.7 [0.9], 17.6 [0.7], and 17.8 [0.9] μm for LS, CTL, IVA, and BIS groups, respectively) and significantly less interstitial fibrosis area (2.48% [0.70%], 5.55% [0.93%], 3.75% [0.61%], and 4.05% [0.72%] for LS, CTL, IVA, and BIS groups, respectively) than the CTL group (Supplementary Figure 2).
mRNA Expression in the LV and Atria
Table 3 depicts the changes in the mRNA in the LV and both atria. In LV myocardium, the mRNA expression levels of the atrial natriuretic peptide (ANP), angiotensin-converting enzyme, arginine vasopressin, and endothelin-1 were significantly suppressed in the IVA and BIS groups compared with the CTL group. There were no differences in the mRNA levels of the β-myosin heavy chain and collagen-type Ia (Col1a1) among the 3 groups. Although the β1-adrenergic receptor and nerve growth factor mRNA expressions were reduced in the CTL group in comparison with the LS group, they were restored in the IVA and BIS groups (P < .05). An increase in both HCN2 and HCN4 mRNA was observed in the BIS group. However, only HCN4 mRNA increased in the IVA group. In RA, HCN2 mRNA was decreased in the IVA and BIS groups compared with the LS group. In LA, ANP and Col1a1 were significantly suppressed in the IVA and BIS groups compared with the CTL group. There were no differences in the both HCN2 and HCN4 mRNAs.
mRNA Expressions via Quantitative Real-Time PCR Analysis.a

Abbreviations: ACE, angiotensin-converting enzyme; ADR, adrenergic receptor; ANP, atrial natriuretic peptide; AVP, arginine vasopressin; BIS, bisoprolol group; β MHC, β-myosin heavy chain; Col1a1, collagen-type Ia; CTL, control group; ET-1, endothelin-1; HCN, hyperpolarization-activated cyclic nucleotide-gated channel; IVA, ivabradine group; LA, left atrium; LS, low-salt diet group; LV, left ventricle; mRNA, messenger RNA; PCR, polymerase chain reaction; NGF, nerve growth factor; RA, right atrium.
a Values were presented as mean (SD). The mRNA levels were normalized with an endogenous control (18S rRNA mRNA) and were expressed as arbitrary units. The values for the LS rats were set at 1.0, and the remaining values were adjusted accordingly.
bP < .05 versus the LS group.
cP < .05 versus the CTL group.
Tyrosine Hydroxylase Expressions in the LV and Atria
In Western blot analysis, the CTL group revealed a significantly reduced expression of TH in the LV, RA, and LA compared with the LS group, which suggested sympathetic nerve dysfunction in the LV and both atria (Figure 4A). A significant recovery in this TH underexpression was observed in the IVA and BIS groups. The representative images of immunofluorescence/immunohistochemical staining to detect TH are shown in Figure 4B and Supplementary Figure 2. The TH-positive cell density in the LV, RA, and LA reduced in the CTL group compared with the LS group. These expression levels (innervation) were recovered in the IVA and BIS groups.
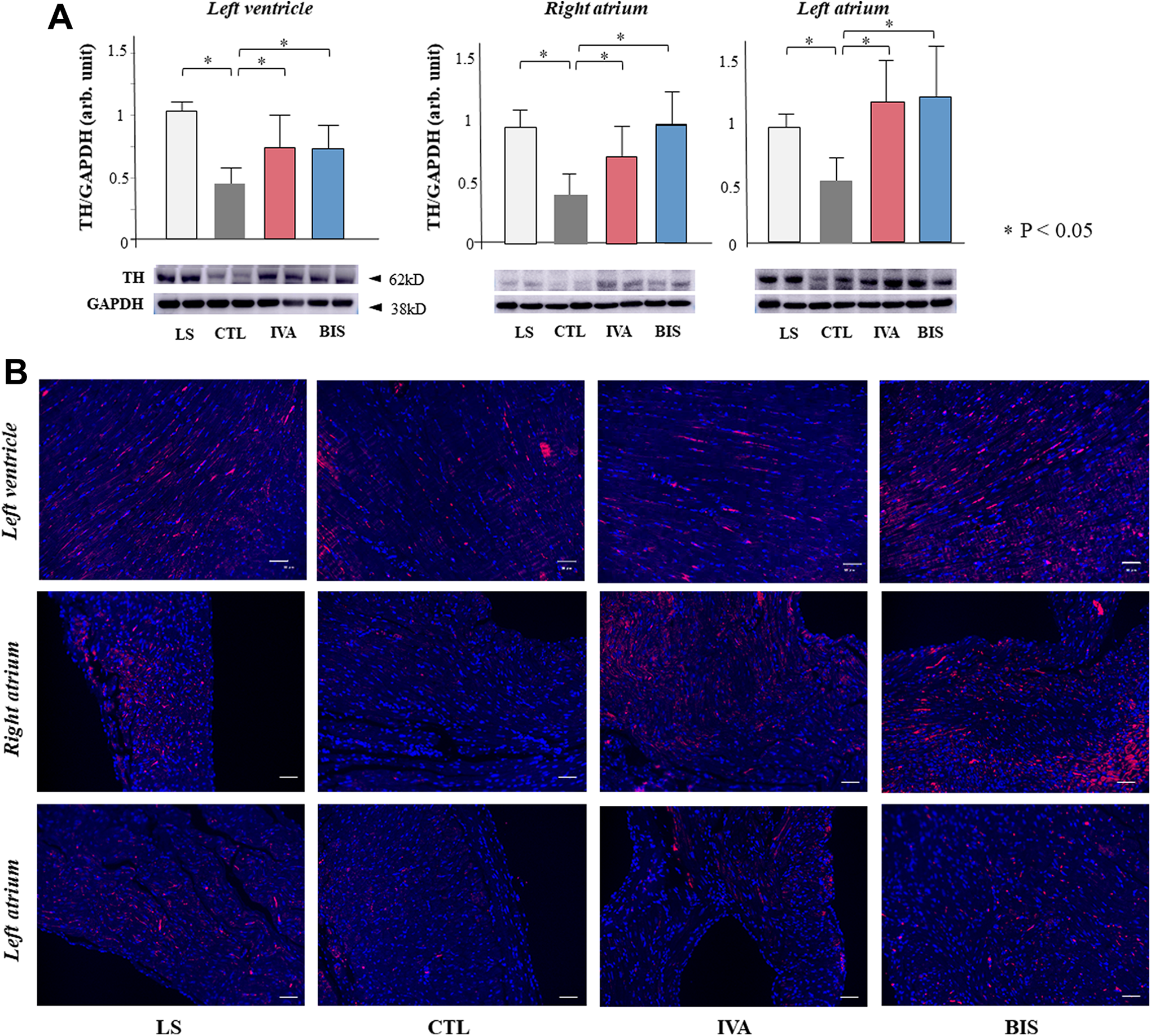
Tyrosine hydroxylase (TH) expression in the cardiac tissues. A, Quantification of TH protein via Western blotting. B, Representative images of immunofluorescence staining with anti-TH antibody. The protein levels were expressed relative to the LS group. The scale bar indicates 50 μm. Data were presented as mean (SD). BIS indicates bisoprolol group; CTL, control group; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; IVA, ivabradine group; LS, low-salt group; SD, standard deviation.
Discussion
HR Reduction and Use of IVA in Animals With HF
Prior studies have analyzed that HR reduction with IVA or digoxin prolonged life span in normal mice. 13,14 They could not determine whether the improved longevity was secondary to HR reduction or to pleiotropic effects of the drugs per se. The prolonged survival of hypertensive HF animals by chronic IVA therapy is one of the key novel findings in the present study. Although an increase in resting HR is an independent prognostic factor of HF and the mechanisms correlating HR and HF are not fully elucidated, 15 the beneficial effects of IVA for HF as shown in the present study may suggest a significant pathophysiological association between HR and HF, as well as the benefits of β-blockers in HF.
Because the decrease in HR lengthens the diastolic filling time, ameliorates the atrioventricular coupling, and decreases myocardial ischemia, it could improve the diastolic filling. Other mechanisms than a simple prolongation of diastolic period in the beneficial effect of IVA on diastolic function have been reported in the animal models. The IVA increased the phosphorylation of phospholamban, decreased the N2B isoform expression of titin and collagen amount, or ameliorated arterial stiffness and endothelial dysfunction. 16,17 Recently, HR reduction with the use of IVA decreased the period of isovolumic contraction and relaxation both in resting and under dobutamine-stimulated conditions and contributed to favorable LV filling and preservation of ejection in conscious pigs with LVH induced by hypertension. 18 The histological findings in the improvement of LV fibrosis and myocyte hypertrophy and the restoration of decreased TH expression suggest that the improvement in diastolic properties may be associated with the amelioration of HF and survival benefit in this model.
Ivabradine and SNS in HF
The IVA does not modify atrioventricular or intraventricular conduction or myocardial contractility in contrast to β-blockers. 19 Thus, the effects on the autonomic nervous system are presumably different between them. In the present study, chronic IVA therapy was associated with the amelioration of systemic sympathetic activation and the preservation of atrial/ventricular sympathetic nerve activities. The findings in the telemetry, lower HR in the nighttime (active time in rodents) and higher HR in the daytime (sleeping time) in IVA group, may support the strong inhibitory effect on systemic SNS. With regard to the use of β-blocker, the suppressive effects were not observed at a systemic level but at a local cardiac level. The difference may be associated with the findings that IVA does not, but BIS has negative inotropic/lusitropic effects, as supported by the echo data.
Recently, we demonstrated that IVA did not acutely affect sympathetic arterial pressure regulation by baroreflex, and it also spared the magnitude of the sympathetic HR response in the vagotomized rats. 20 Silva et al have reported that a 7-day treatment with IVA reduced HR without compromising the cardiovascular reflexes and tonic regulation of the cardiac and renal sympathetic nerve activity in normal rats. 21 Other human studies have supported the evidence, showing that the acute administration of IVA does not impair sympathetic or parasympathetic baroreflex function. 22 The use of β-blockers does not cause preserved sympathetic HR response. Thus, these acute effects might lead to chronic effects in the present study, and only the IVA (but not the BIS) group had an amelioration of the systemic sympathetic nerve overactivity, as indicated by urine NAd/normetanephrine excretion.
Analysis of heart rate variability (HRV) is carried out for assessing the cardiac autonomic activity and balance. A reduced HRV, which is characterized by sympathetic overactivity and parasympathetic withdrawal, has been observed in patients with HF, which is in accordance with the severity of HF and the poor prognosis. 23 Several studies have shown that chronic treatment with IVA in addition to β-blockers improved the reduced HRV. 24,25 In the present study, decreased TH expressions and NAd contents and decreased ACh contents in the RAs were restored with IVA treatment, indicating the improvement in local sympathetic and parasympathetic nerve innervations. These findings may be used as a basis in improving HRV in clinical studies. Possible mechanisms for the improvement might be as follows: reduced sympathetic stimulation leading to the amelioration of adrenoreceptor-mediated cytotoxicity, apoptosis, and hypertrophy; decreased myocardial oxygen demand and ischemia; and ameliorated diastolic filling and myocardial blood supply by prolonged diastole period. 9 However, the present findings that chronic IVA administration ameliorated systemic sympathetic activation and cardiac sympathetic innervations might be indirect effects or accompanying events.
Pleiotropic Actions of IVA in HF
A number of studies have explored the pleiotropic effects of IVA, that is, cardioprotective effects beyond HR reduction. 26 They include the amelioration of cardiac inflammatory response, antifibrotic, vascular protective, and antiatherosclerotic effects. 27 The IVA was reported to reduce myocardial ischemia/reperfusion injury and to improve the viability of cardiomyocytes. 28 Reduced reactive oxygen species (ROS) formation is suggested as a key mechanism. The modulation of the SNS by IVA documented in this study may be associated with these pleiotropic actions. For example, amelioration of activated SNS might suppress cardiac inflammation, including increased ROS production in HF. 29 Further studies are necessary to examine a direct effect of IVA on sympathetic/parasympathetic nerve systems and a relationship between sympathetic/parasympathetic nerve systems and the pleiotropic actions in IVA treatment.
Study Limitation
This study has several limitations. We used a somewhat special HF model of DS hypertensive rats with progressive cardiac and kidney damages. The chronic effects of IVA on sympathetic activity and innervation must be compared with those in other HF models that are not related to hypertension, such as myocardial infarction, myocarditis, and aortic constriction models. Although we showed an improvement in survival time with the use of IVA and BIS, we did not find any definite difference in the histology or PCR analysis results other than alterations in SNS activation among the 2 treatment groups. Further analysis must be performed to validate other differences in the mechanisms between IVA and BIS treatments, particularly in terms of LV dysfunction or damage. Moreover, the assessment of the synergistic effects of IVA and β-blocker may be useful in this regard. Lastly, we used light anesthesia with 1.5% isoflurane in echocardiography. Previous studies have suggested that isoflurane anesthesia may exert modest cardiovascular effects, such as negative inotropic and lusiotropic effects, and decrease in HR. 30
Conclusion
The chronic effects of IVA on sympathetic activity and innervation were assessed and compared with those of β-blockers in hypertensive animals with HF. The IVA therapy improved the survival time of the animals without negative inotropic effects, which was mainly associated with the amelioration of systemic sympathetic activation and cardiac sympathetic/parasympathetic innervations, followed by the prevention of progressive myocardial damages and dysfunction. The overall effect was similar to that of chronic β-blocker therapy with negative inotropic effects. However, it had suppressive effects only on the cardiac sympathetic/parasympathetic innervations.
Supplemental Material
Supplemental_material - Modulation of Sympathetic Activity and Innervation With Chronic Ivabradine and β-Blocker Therapies: Analysis of Hypertensive Rats With Heart Failure
Supplemental_material for Modulation of Sympathetic Activity and Innervation With Chronic Ivabradine and β-Blocker Therapies: Analysis of Hypertensive Rats With Heart Failure by Kazuyoshi Kakehi, Yoshitaka Iwanaga, Heitaro Watanabe, Takashi Sonobe, Tsuyoshi Akiyama, Shuji Shimizu, Hiromi Yamamoto and Shunichi Miyazaki in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
Kazuyoshi Kakehi, MD, and Yoshitaka Iwanaga, MD, contributed equally to this work.
Acknowledgments
We would like to thank Kumi Ohmi and Yasumitsu Akahoshi for their technical assistance. Bisoprolol was kindly provided by the Mitsubishi Tanabe Pharma Corporation.
Author Contribution
All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Y.I. and S.S. contributed to acquisition, analysis, and interpretation. K.K. contributed to acquisition and analysis. H.W. and T.S. contributed to acquisition; T.A. contributed to analysis. H.Y. contributed to analysis and interpretation. S.M. contributed to interpretation. Y.I., H.Y., and S.M. contributed to conception and design. K.K. and S.S. contributed to conception. H.W., T.S., and T.A. contributed to design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant-in-aid for scientific research (C) from the Japan Society for the Promotion of Science (16K09465).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
