Abstract
Background:
Inappropriate sinus tachycardia (IST) is a clinical syndrome characterized by excessive resting heart rate (HR) or a disproportional increase in HR during exercise. β-blocker or calcium channel-blocker therapy is often noneffective or not well tolerated. The HR reduction on ivabradine is similar to β-blockers but in some patients its efficacy to resolve all IST-related symptoms is limited. The aim of the study was to assess the efficacy and safety of combining ivabradine with metoprolol succinate in patients with refractory highly symptomatic IST.
Methods:
Twenty patients (36 ± 10 years; 16 women) with IST were enrolled. All patients received metoprolol succinate 95 mg single dose during the first month of the study. After 4 weeks of treatment with metoprolol, ivabradine was administered as adjuvant therapy up to 7.5 mg twice daily. Holter monitoring and treadmill stress test were performed at baseline, after 4, and 8 weeks of the study, respectively.
Results:
We observed significant and similar reduction in resting HR both for metoprolol and for combined therapy compared to the baseline. The mean HR during daily activity was significantly lower on ivabradine and metoprolol compared to monotherapy with β-blocker. The combined treatment yielded a significant increase in exercise capacity as assessed by treadmill stress test. After 4 weeks of combined therapy a significant reduction in IST-related symptoms, measured by means of the European Heart Rhythm Association score, was observed.
Conclusion:
Combining ivabradine with metoprolol is an effective and well-tolerated treatment option for IST in patients with refractory to monotherapy.
Introduction
Inappropriate sinus tachycardia (IST) is a relatively uncommon form of tachyarrhythmia characterized by a persistent increase in resting sinus rate or disproportionate elevation of heart rate (HR) with minimal physiological effort. 1 Main complaints associated with IST include palpitations, fatigue, presyncope, and exercise intolerance.1,2 In some cases, the symptoms can be severe and lead to total disability. Autonomic dysfunction, abnormal automaticity of the sinus node, and atrial focal tachycardia originating near the sinus node have been proposed to be the underlying mechanisms.3,4 The syndrome is associated neither with structural heart disease nor with any reversible causes of sinus tachycardia (ie, fever, anemia, infection, hyperthyroidism, drug abuse). Therefore, the diagnosis is generally one of exclusion.
For pharmacological treatment of IST, high doses of β-blockers or nondihydropyridine calcium-channel blockers are recommended.1,2 However, some patients do not respond to such therapy or may have nontolerable side effects, most frequently symptomatic hypotension and/or fatigue. Nonpharmacologic interventions such as sinus node modification by means of catheter ablation have been tried but are limited by poor efficacy or relatively high risk of complications.5–7
Ivabradine is a specific HR lowering agent that inhibits pacemaker If current. It reduces HR at rest and during exercise without influence on intraventricular conduction, contractility, or hemodynamics. 8 The expression of If in the sinoatrial node makes ivabradine a promising treatment option in patients with IST. Several studies have suggested that monotherapy with ivabradine is a safe and effective second-line therapy in patient affected by IST; however, some patients may remain symptomatic.9-14 Recently published randomized trials in patients with coronary artery disease and heart failure showed that addition of ivabradine to β-blockers further reduced HR with a benefit in terms of major cardiovascular events. This therapeutic option produced additional efficacy without affecting safety or tolerability.15–19 Thus, combining ivabradine and a β-blocker may be a valuable therapeutic option in highly resistant cases of IST.20,21
The aim of this study was to evaluate the safety and efficacy of ivabradine in combination with metoprolol succinate in patients with highly symptomatic IST resistant to monotherapy with β-blockers.
Methods
Patient Population
The study population consisted of consecutive patients who were referred to the Autonomic Heart Dysfunction Laboratory due to highly symptomatic, persistent sinus tachycardia. All patients had been free of any previous antiarrhythmic treatment for at least 4 weeks at the time of screening. Those with a confirmed diagnosis of IST, defined as sinus tachycardia at rest (HR ≥ 100 beats per minute [bpm]) in sitting position or/and as an average HR ≥ 90 bpm during the 24-hour Holter monitoring, were considered to participate in the present study. 1 There were 16 women and 4 men (age 21-47 years; mean age 36±10 years). The patients mainly had palpitation (n = 20; 100%), dyspnea (n = 8; 40%), physical exercise intolerance (n = 12; 60%), or chest discomfort (n = 8; 40%). No patients had a history of orthostatic intolerance or postural orthostatic tachycardia during the orthostatic test. Secondary causes of tachycardia and structural heart disease had been excluded through careful examination or additional diagnostic tests (laboratory values including thyroid function, blood count, C-reactive protein, and echocardiography).
Three patients were on concomitant cardiovascular medication (ramipril, valsartan) due to a history of hypertension. All patients had been pretreated with β-blockers or verapamil prior to the study, without clinical effect or poor tolerance. The patient characteristics are depicted in Table 1. Patients gave informed consent prior to enrollment in the study. The study protocol had been approved by the local ethics committee.
Patient Characteristics.a

Abbreviations: ACE, angiotensin-converting enzyme; SD, standard deviation.
aValues are given as n (%) and mean ± SD.
Study Protocol
Patients with baseline clinical and Holter criteria for the diagnosis of IST were included in the study. In all the patients, previous antiarrhythmic and/or rate control medication had been discontinued 4 weeks prior to entry into the first treatment phase.
During the first 4 weeks of the study, the patients received metoprolol succinate 47.5 mg in the morning uptitrated to 95 mg once daily as a target dose. As per protocol, adjuvant therapy with ivabradine was introduced after 4 weeks of treatment with metoprolol. Initially, we administered ivabradine 5 mg twice daily (BID), up to 7.5 mg BID if well tolerated. The dose was augmented after 10 days. The patients were evaluated at baseline and at the end of each treatment period (after 4 and 8 weeks). During the visits, resting electrocardiogram (ECG) and 24-hour Holter monitoring (Medilog Darwin, Schiller AG, Switzerland) were recorded. Resting HR was obtained from ECG tracings. The following parameters were calculated from 24-hour Holter monitoring: mean HR, maximum HR, and daytime mean HR (defined as a mean HR between 6.00
Additionally, in order to assess physical activity, treadmill exercise tests were performed. The patients underwent fatigue-limited stress tests using the standard Bruce protocol (GE Marquette CASE T2100, GE Healthcare, Pollards Wood Nightingales Lane, United Kingdom) at baseline and after 4 weeks of each drug period. We recorded the maximal workload expressed in metabolic equivalent of task (MET) and total duration of test after both treatment phases. Although exercise capacity may somewhat improve with learning on each attempt done during the course of the study, we decided to use treadmill exercise testing, since it is an objective measure in routine clinical practice.
Subjective perception of symptoms in patients affected by IST was assessed with a semiquantitative questionnaire before and at the end of each treatment period. First, the patients were asked to define their general health status and frequency of selected symptoms. The frequency of IST-related symptoms was scored as 0: none, 1: very infrequent, 2: sometimes, 4: very frequent, and 5: incessant. The following symptoms were included in the check list: palpitation, exercise intolerance, dyspnea, presyncope, and chest discomfort. The total symptom score was expressed as the sum of all symptoms score (min -max 25 points). Additionally, the severity of IST-related symptoms was assessed using the European Heart Rhythm Association (EHRA) score. The EHRA score was previously developed and widely accepted as a useful tool to describe symptoms of atrial fibrillation, very similar to IST-related symptoms. 22 The usefulness of this score in symptom assessment in patients with IST has recently been confirmed. 13 The following items during presumed arrhythmia episodes were checked to determine the score: palpitation, exercise tolerance, dyspnea, dizziness, chest discomfort, and presyncope. The symptoms severity was classified as EHRA I: “no symptoms,” EHRA II: “mild symptoms,” EHRA III: “severe symptoms,” and EHRA IV: “disabling symptoms.”
Statistics
Statistical analysis was performed using Statistical software (version 9.0, StatSoft, Inc, Tulsa, Oklahoma). Continuous variables are expressed as mean ± standard deviation, and categorical variables are presented as frequency (%). As not all analyzed data had normal distribution (Shapiro-Wilk W test), the nonparametric analysis of variance Friedman test was used for multiple group in comparison with subsequent nonparametric post hoc test. The significance of the relationship between the categorical variables was tested with chi-square Pearson test and its modifications for 2 × 2 tables (Yates correction and Fischer exact test). The HR reduction defined as the difference between HR on treatment (metoprolol monotherapy or combined therapy) and baseline HR was calculated. Furthermore, Pearson coefficient was used to correlate HR reduction and baseline HR for resting HR and parameters derived from Holter monitoring. Values of P < .05 were considered statistically significant.
Results
The target daily dose of metoprolol (95 mg) was achieved in 16 (80%) patients during the first month of therapy. In 3 patients, we administered metoprolol at a lower dose of 47.5 mg due to symptomatic hypotension. No other side effects of β-blocker therapy occurred. Nineteen (95%) patients did tolerate the maximal daily dose of ivabradine. We observed significantly lower resting HR on each step of the treatment (114.4 ± 7.5 vs 97.3 ± 14.4 vs 90.5 ± 13.3 bpm; P < .001; Figure 1). The mean HR during Holter monitoring was significantly lower in patients treated with both drugs in comparison to baseline and monotherapy (103.3 ± 3.9 vs 84.1 ± 5.9 vs 77.6 ± 5.2 bpm; P < .001; Figure 2). The maximal HR during monitoring was 151 ± 8 bpm at pretreatment period, 131.5 ± 10 bpm on metoprolol, and 121.8 ± 7 bpm on combined therapy (P < .001). During daily activity, the mean HR on ivabradine and metoprolol was significantly lower than for metoprolol monotherapy (mean daytime HR 82.0 ± 7 bpm vs 90.6 ± 6 bpm; P < .001; Figure 3). The data analysis revealed correlations between HR reduction on combined treatment and baseline HR for mean 24 hours HR (r = ,75; P = .0002; Figure 4), daytime HR (r = .60; P = .007), and maximum HR (r = .65; P = .0028). A similar, however weaker, correlation was found on metoprolol monotherapy only for mean HR (r = .51; P = .024). There was no such correlation for resting HR neither in combined treatment (r = .40; P =.09) nor in metoprolol monotherapy (r = .15; P = .52). Representative HR trends recorded during 24-hour Holter monitoring in patients with IST treated with metoprolol or combination of metoprolol and ivabradine are presented in Figure 5. The combined treatment yielded a significant effect on exercise capacity as assessed during treadmill stress test. The maximal dose of exertion was 10.4 ± 1.0 METs at baseline, 11.9 ± 1.1 METs during metoprolol therapy, and 12.5 ± 1.0 METs on metoprolol in addition of ivabradine (P < .001). The maximal duration of exercise was longer in patients on combined therapy compared to baseline and to treatment with metoprolol (693.5 ± 45.8 seconds vs 524.3 ± 68.7 seconds vs 627.8 ± 53.9 seconds; P < .001). The analysis of events reported by patients during 24-hour Holter monitoring revealed significant reduction of symptoms associated with IST for both modes of treatment. Reduction of events was significant during combined drug therapy compared to monotherapy with metoprolol (mean events 5 ± 1 vs 16 ± 4; P < .05) and to baseline (mean 19 ± 4; P < .01).

Resting heart rate during metoprolol and combined therapy in comparison to baseline. Results are shown as mean ± standard deviation (SD). For multiple comparison P < .001.
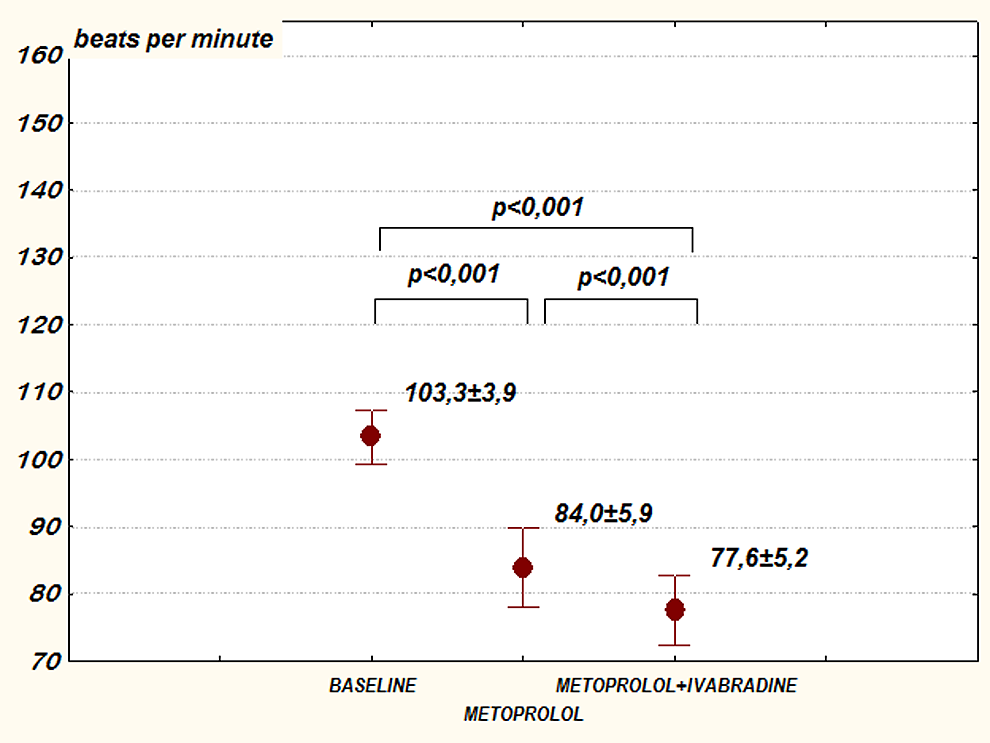
Mean heart rate (HR) during 24-hour Holter monitoring in patients treated with metoprolol or metoprolol plus ivabradine versus baseline. Results are shown as mean ± standard deviation (SD). For the multiple comparison P < .001.

Daytime mean heart rate (HR) on metoprolol and metoprolol plus ivabradine therapy compared to the baseline. Results shown as mean ± standard deviation (SD). For multiple comparison P < .001.
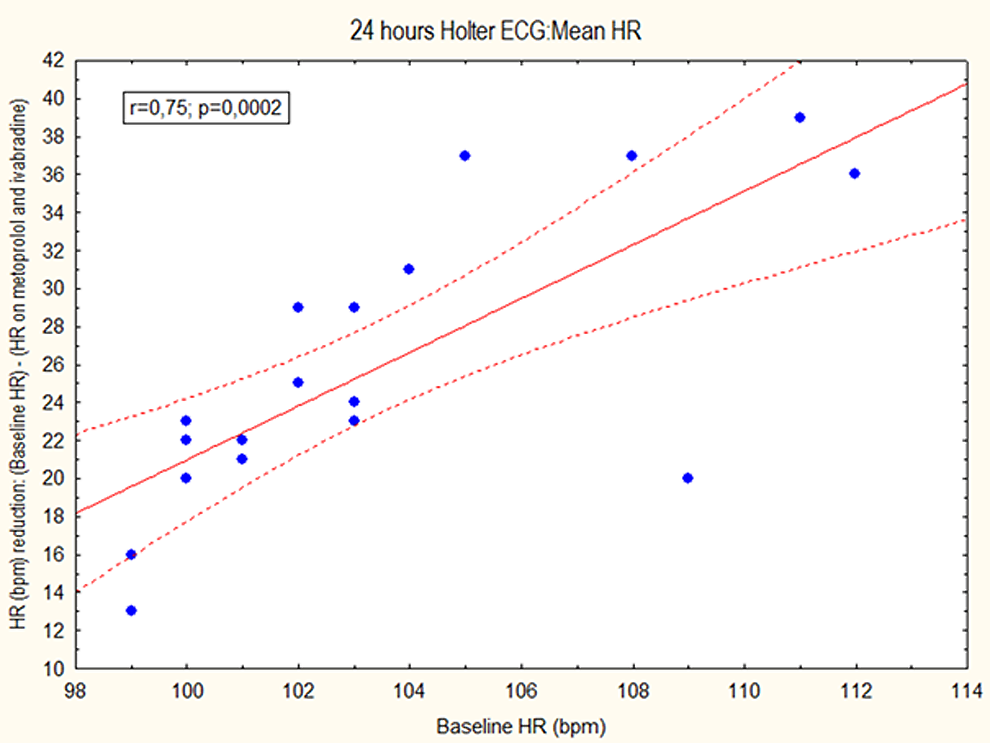
Correlation between heart rate (HR) reduction and mean HR on 24-hours Holter monitoring in patients treated with combined therapy.

Representative examples of heart rate (HR) trends recorded during 24-hour Holter monitoring in patients with inappropriate sinus tachycardia (IST) treated with metoprolol or combination of metoprolol and ivabradine versus baseline.
A significant reduction of symptoms, evaluated by means of EHRA score, was observed after 30 days of metoprolol treatment. This effect was even more pronounced after the next 30 days with ivabradine as adjuvant therapy (Table 2). After 1 month of combined treatment none of the studied patients reported episodes of IST-related symptoms (EHRA score III and IV: 12 patients—60% vs 4 patients—20% vs 0 patients—0%; chi-square 18.4, degrees of freedom = 2; P < .001).
The IST-Related Symptoms at Baseline, During Metoprolol Therapy, and Combined Treatment With Metoprolol and Ivabradine.a

Abbreviations: df, degrees of freedom; EHRA, European Heart Rhythm Association; IST, inappropriate sinus tachycardia; SD, standard deviation.
aValues are given as n (%) or mean ± SD. Definition of IST-related questionnaire and EHRA score, see Methods section. Mean scores in the IST-related questionnaire (min 0-max 5 points); total score (0-25 points).
bFor EHRA 1-4 multiple comparison table chi-square = 30.95; df = 2; P < .001; no significant difference (P > .05) when metoprolol monotherapy compared to combined therapy.
The combined treatment was well tolerated. We did not observe side effects such as severe bradycardia or hypotension during the therapy. In 1 female patient, transient phosphenes phenomena occurred on the maximal dose of ivabradine (15 mg daily), but it resolved after dose reduction (10 mg daily).
Discussion
Initial pharmacological therapy of IST is empiric and usually includes β-blockers. In cases of intolerance, nondihydropyridine calcium-channel blockers are considered as a second-line therapy.1,2 Some patients do not respond to pharmacological therapy or may have nontolerable side effects (hypotension, transient bradycardia). Interventional therapy using catheter ablation or modification of the sinus node is limited by a considerable risk of severe bradycardia requiring pacemaker implantation.5–7 In such circumstances a new selective inhibitor of If channels became an attractive alternative for patients with IST.1,8
Several studies have described the benefit of ivabradine in patients with drug refractory, highly symptomatic IST.9-13 A recently published study by Calò et al showed a significant reduction in medium HR and maximal HR (assessed by 24-hour ECG) after 3 to 6 months treatment with ivabradine in patients affected by IST. 10 The study also revealed an increased tolerance to physical stress, which was confirmed during exercise tests at 3 and 6 months of oral administration of ivabradine. No patient showed significant side effects of ivabradine, and the dose of 10 mg was sufficient to significantly reduce daily HR and symptoms. The beneficial effect of ivabradine on HR and symptoms reduction has been confirmed in a small randomized study by Cappato et al. The study showed that 1 month of monotherapy with ivabradine eliminates 70% of the variable symptoms associated with IST, but the complete elimination of symptoms can be expected in approximately half of the patients. 14 In a recent study from our group, metoprolol succinate was compared to ivabradine in a prospective, observational study design. The results showed that orally administered ivabradine is effective and safe in rate control in patients with IST, especially as a second-line therapy in cases of lack of efficacy or intolerance of β-blockers (metoprolol). 13
Recently published data suggest that ivabradine is beneficial when added to conventional treatment including a β-blocker, for example in patients with heart failure in sinus rhythm whose resting HR remained 70 bpm or greater and who had worse than moderate left ventricular impairment. 16 Thus, this drug combination seems to be an interesting alternative compared to conventional therapeutic strategy in patients with IST.
As with any other HR-lowering agent, the effect of β-blockers is proportional to the baseline HR. We did not observe such phenomenon in patients affected by IST treated with monotherapy of metoprolol. In contrast, a strong and significant correlation between HR reduction and mean, daytime and maximum Holter HR during combined therapy was observed. This confirms previous results showing an increased efficacy of ivabradine with greater pretreatment HR, even as adjuvant therapy.10,14
The present prospectively designed study yields important findings with respect to the clinical applicability of ivabradine in patients who do not tolerate or do not respond efficiently to standard doses of β-blocker monotherapy. It demonstrates that oral administration of ivabradine as adjuvant therapy to metoprolol reduced mean HR and resolved IST-related symptoms. A further reduction in HR was observed during daily activity. To our knowledge this is the first study assessing efficacy and safety of ivabradine in combination with β-blocker in a population of patients affected by IST. The study also revealed the positive effects of combination therapy on exercise capacity. The analysis revealed an increase in maximal workload and total duration of exercise during additional ivabradine therapy compared to baseline and metoprolol. The results are similar to previously published studies suggesting an increased tolerance to physical stress following ivabradine administration.9,11,13,14,17 The results of the present study generate the hypothesis that combined HR-lowering drug therapy is useful in selected patients with IST.
Although IST is considered to be a benign form of tachyarrhythmia, patients affected with this disorder may frequently remain highly symptomatic. There is no simple and easily applicable IST-specific score to measure symptoms in patients affected by IST. Therefore, we have applied especially designed questionnaires (IST-related score and total symptom score) and adopted EHRA score. The EHRA classification has originally been designed to assess symptoms related to atrial fibrillation. The checked symptoms of the EHRA score are palpitation, fatigue, dizziness, dyspnoe, chest pain, and anxiety. Likewise, these symptoms are characteristic of patients with IST. Therefore, we adapted this classification in our recent study. 13 Using this method, we revealed significant reduction of IST-related symptoms in both treatment options. During additional ivabradine administration, 70% of the patients were free of symptoms compared to 30% of the patients on β-blocker only. In the present study, we also observed a significant reduction in IST-related symptoms reported by patients during 24-hour Holter monitoring on ivabradine compared to baseline and to metoprolol therapy.
Combined therapy was very well tolerated. We did not observe the important side effects such as severe or symptomatic bradycardia requiring reduction or discontinuation of therapy. Previously reported ivabradine-specific visual disturbances (phosphenes, diplopia) occurred only in 1 patient. A phosphene is an entoptic phenomenon characterized by light sensations without actual light exposure. Phosphenes can be directly induced by mechanical, electrical, or magnetic stimulation of the retina or visual cortex as well as random firing of cells in the visual system. Phosphenes have also been reported in patients during drugs administration, especially a hallucinogen or ivabradine.8,15,16 The important advantage of ivabradine over β-blockers is the lack of influence on systolic ventricular function, blood pressure, and lipid or glucose metabolism.8,15,16 In most patients, therefore, the maximal daily dose of ivabradine can be administered for HR control. Including ivabradine as part of a combined rate-lowering drug therapy may thus be a very promising and safe treatment option in patients not tolerating high doses of β-blockers.
Study Limitation
The sample size of the present study is small; however, the syndrome is relatively rare in a general population. Another limitation is also the design of the study, which is an observational, off-label, nonrandomized study without crossover and only 2-month follow-up. Since ivabradine is a novel therapeutic option for IST, we aimed to systematically test this therapeutic approach in patients in who for some reason the gold standard treatment with “stand-alone” β-blockers had been failed. Thus, the results of the present study need confirmation in a trial with a blinded crossover design. Noteworthy, so far no treatment option in IST has been subjected to large, randomized trials, mainly due to specific characteristics of this disorder.
We adopted arbitrarily 95 mg as a target dose for metoprolol succinate, since this is the generally tolerated average dose in many cardiovascular disorders. It cannot be excluded that some patients may have tolerated a higher dose of metoprolol. However, this mean dose was chosen to facilitate titration of ivabradine to the maximal daily dose.
Conclusion and Clinical Implications
The results of the present study indicate that orally administered ivabradine in combination with metoprolol succinate represents an effective and safe means of rate control in patients with IST, especially as a second-line therapy in cases of lack of efficacy or intolerance of β-blocker monotherapy. This treatment option seems to be more effective especially to relieve symptoms during exercise or daily activity. It is also well tolerated even at the maximal recommended dose of ivabradine. Future controlled randomized trials are needed to examine the efficacy and safety of this combined therapy in the long-term treatment of IST.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
