Abstract
Aims:
This study aims to evaluate atherosclerosis, oxidative stress, and arterial stiffness attenuation by simvastatin and ivabradine in hyperlipidemic rabbits.
Methods and Results:
Forty rabbits were randomly divided into 4 groups: atherogenic diet (group C), atherogenic diet plus simvastatin (group S), atherogenic diet plus ivabradine (group I), and atherogenic diet plus simvastatin and ivabradine (group S + I). After 9 weeks, rabbits were euthanized and descending aortas excised for mechanical testing. Atherogenic diet induced the development of significant atherosclerotic lesions in group C animals but in none of groups S, I, and S + I. RAM-11 and HHF-35–positive cells were significantly reduced in groups S, I, and S + I compared with group C (P < .001). A significant neointimal hyperplasia and intima–media ratio reduction was demonstrated in groups S (P = .015 and P < .001), I (P = .021 and P < .001), and S + I (P = .019 and P < .001) compared with group C. Protein nitrotyrosine levels were significantly decreased in group S compared with group C (P = .009), and reactive oxygen species levels were decreased in group I compared with group C (P = .011). Aortic stiffness was significantly reduced in groups S, I, and S + I compared with group C (P = .003, P = .011, and P = .029).
Conclusion:
Simvastatin and ivabradine significantly inhibited intimal hyperplasia and oxidative stress contributing to aortic stiffness reduction in hyperlipidemic rabbits.
Introduction
Atherosclerotic cardiovascular disease remains the leading cause of death in developed countries. 1 Arterial stiffness increasingly constitutes an important end point for cardiovascular disease, as it is correlated with the presence of cardiovascular risk factors and atherosclerosis. 2 –4 Recently, arterial stiffness measured by the aortic pulse wave velocity has been suggested as a powerful predictor of future cardiovascular events and all-cause mortality. 2
Statins or 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors demonstrate favorable effects on atherosclerosis progression and plaque instability, extending beyond their lipid-lowering activity. 5 –7 Pleiotropic antiatherogenic actions of statins, such as endothelial function amelioration, plaque stabilization, inhibition of vascular smooth muscle cell (SMC) proliferation, reduction in vascular inflammatory response, antithrombotic function, and antioxidant activity, may translate into an improvement in arterial stiffness, 5,8,9 reducing further cardiovascular morbidity. 10,11
Heart rate elevation as an integral part of pulsatile mechanical stress can modify arterial wall compliance, resulting in structural alterations and atherosclerosis enhancement. 12,13 Ivabradine, a selective If current inhibitor, reduces resting and exercise heart rate and exerts antianginal and anti-ischemic effects without affecting cardiac contractility or blood pressure. 12 Two recent experimental studies suggest that ivabradine inhibited vascular oxidative stress, endothelial dysfunction, atherosclerotic plaque formation, and preserved aortic compliance in apoE-deficient mice. 12,14
Atherosclerosis alters the mechanical properties of the arterial wall through highly variable changes in its cellular composition and histological structure. 15 The importance of arterial compliance for cardiovascular performance highlights the role of arterial stiffness as a potential treatment target.
Taken together, elevated heart rate and atherosclerosis may reduce arterial wall compliance, thereby rendering arterial stiffness a potential treatment target. This study aims to evaluate the effectiveness of simvastatin and ivabradine regarding atherosclerosis prevention, oxidative stress reduction, and arterial stiffness attenuation in thoracic aortas of hyperlipidemic rabbits.
Materials and Methods
Animal Model
Forty male New Zealand white rabbits were housed individually 15 and allowed to fed on regular rabbit chow for 7 days to acclimate to their environment. After the week of acclimation, atherogenic diet consisting of regular rabbit chow supplemented with 4% cholesterol (ELPEN Pharmaceutical, Athens, Greece) was initiated. The 40 rabbits were randomly but not in a blinded way divided into 4 groups of 10 animals as follows: atherogenic diet (group C), atherogenic diet plus simvastatin 5 mg/kg/d (group S), 16,17 atherogenic diet plus ivabradine 5 mg/kg/d (group I), 12,14 and atherogenic diet in combination with simvastatin 5 mg/kg/d and ivabradine 5 mg/kg/d (group S + I) for 9 weeks. Simultaneously, 8 animals fed regular rabbit chow were used as long-term control. At the end of this period, rabbits were anesthetized with ketamine (50 mg/kg) plus xylazine (10 mg/kg) intramuscularly and euthanized by intravenous injection of a saturated KCl solution. The descending thoracic aortas were exposed, and a 50-mm segment was marked and excised so as to retain aortic physiological prestrain in vitro. 18 Aortas’ segments were stored in normal saline 0.9% at 4°C prior to mechanical testing. All mechanical tests were performed immediately after animal killing. At the end of the mechanical testing, aortas were isolated and harvested for histological, immunohistochemical, and biochemical analysis.
The study was conducted in accordance to the Institutional “Guide for the Care and Use of Laboratory Animals” and was approved by the Institutional Animal Care and Use Committee of the West Greece Prefecture and the University of Patras. All experiments were performed in the Animal House of the Medical School of Patras University.
Mechanical Tests
The apparatus and the function of our experimental system have been already described. 15 The aortic specimens, after physiological prestraining, were subjected to pulsatile flow of 400 mL/min (mean arterial flow), at 60 cycles/min, with peak systolic/diastolic pressure values of 180/0+ mm Hg. The diastolic pressure was nonphysiologically low (approximately 0+ mm Hg) to maximize the pressure dilation range of mechanical testing. It should be noticed that although the heart rate in vivo is much higher than 60 cycles/min used during the mechanical tests, the stiffness (the actual elastic modulus of the tissue materials) is considered to be insensitive to loading rate, especially in large arteries such as aorta. 19
Pressure was measured with physiologic pressure transducers directly at the input point of the tested aortic segment via a T-tube connector, whereas external diameter was calculated with an accurate laser micrometer. Experimental data of pressure and external diameter were stored in an appropriate computer system and further analyzed to calculate arterial stiffness. A modified arterial stiffness EPm was calculated instead of the significant Peterson modulus EP. 18 External diameter (D) measurements were divided by the diameter at a reference pressure (DS, at P = 100 mm Hg), 20 so as to compute normalized radial arterial dilatation (RAD) according to the formula: RAD = D/Ds. EPm at each pressure was finally computed from the slope of P − RAD curves, according to the following equation: EPm = ΔP/(ΔD/DS). 15
Biochemical Assays
Total serum cholesterol (TC), triglyceride (TG), and high-density lipoprotein (HDL) levels were assessed. Renal and liver functions were monitored using creatinine and serum hepatic enzymes, respectively.
Histological Analysis
Hematoxylin/eosin stain was performed for histopathology analysis. Collagen components were detected by Masson trichrome stain, and fragmentation and/or disorientation of elastic tissue in atherosclerotic lesions were detected by Van Gieson stain (Histo-Line Laboratories, Italy). Atherosclerotic lesions were classified as initial (type I-II), intermediate (type III), and advanced (type IV-VI). 21 Intermediate and advanced atherosclerotic lesions were defined as significant.
Immunohistochemistry
Macrophages and SMCs were detected by immunohistochemistry using monoclonal antibodies against RAM-11 (1:200 dilution; Dako Corp, California) and α-actin (HHF-35, 1:100 dilution; Dako A/S, Denmark), respectively, and graded as described previously. 22
Morphometry
Image J software was used to quantify aortas cross-sectional areas. 23 For each aorta, measurements of luminal area, area bounded by the internal elastic lamina (IEL), area encircled by the external elastic lamina, neointimal area, medial area, total vessel area, and intima–media (I/M) ratio were performed. 22
Oxidative Stress Analysis
In order to test for signs of oxidative stress, we used 2 end points: nitrotyrosine formation as a biomarker for nitrating species and 6-carboxy-2′,7′-dichlorodihydrofluorescein diacetate (carboxy-H2DCFDA) oxidation as a general oxidative stress indicator. In the first assay, descending aortas were homogenized in radioimmunoprecipitation assay buffer and 100 μg of total proteins from each sample was analyzed by Western blot for 3′-nitrotyrosine, as described previously. 22 Quantification of the protein levels that corresponded to all immunoreactive bands for 3’-nitrotyrosine in each lane was quantified using the Image PC image analysis software (Scion Corp, Frederick, Maryland). In the second assay, 30 μg of total proteins from each sample was incubated in the dark for 15 minutes with carboxy-H2DCFDA at a final concentration of 50 μmol/L. Fluorescence intensity of the carboxy-dichlorofluorescein (DCF) product was determined spectrophotometrically using an excitation wavelength of 485 nm and an emission wavelength of 500 nm.
Statistical Analysis
Continuous and categorical data are presented as means ± standard deviation (SD) or standard error (SE) and n (%), respectively. For continuous variables, 1-way analysis of variance was used for comparisons between groups at the end of the treatment period. Tamhane or Bonferroni method was used for post hoc test with respect to homogeneity of variances. Data in box plots represent medians (first to third quartile), whereas whiskers’ ends represent minimum and maximum. Fisher exact test was used for comparisons of categorical variables between groups. Paired samples t test was used for comparisons between 2 specific groups at different time points of the study. All tests were 2 tailed, and statistical significance was considered for P values <.05. Statistical analyses were performed using SPSS for Windows (version 19.0; SPSS Inc, Chicago, Illinois).
Results
Blood Chemistry and Electrocardiogram Evaluation
Baseline lipid and heart rate levels did not differ between groups. Post hoc analysis revealed that atherogenic diet induced severe hypercholesterolemia in group C (4423 ± 493.39 mg/dL), which was significantly prevented in groups S, I, and S + I (1094 ± 167.41 mg/dL, 2691 ± 575.71 mg/dL, and 1200 ± 179.76 mg/dL, respectively, P < .001; Table 1). Simvastatin demonstrated a larger decrease in TC, TG, and HDL levels (P < .001 according to post hoc analysis; Table 1) in comparison with ivabradine. Ivabradine contributed to significant reduction in TC compared to group C, effect that was not present when the HDL–TC ratio was assessed (0.11 ± 0.02 vs 0.09 ± 0.01, P = .837; Table 1).
Lipid Profile, Weights, and Heart Rates of Dyslipidemic Rabbits.a

Abbreviations: ANOVA, analysis of variance; dfb, degrees of freedom between groups; dfw, degrees of freedom within groups; GGT, γ glutamyltransferase; HDL, high-density lipoprotein; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine; SGPT, serum glutamic pyruvic transaminase; TC, total serum cholesterol; TG, triglyceride.
a P values according to Tamhane or Bonferroni post hoc ANOVA.
b P < .001 versus groups S, I, S + I, and controls.
c P < .001 versus group I and controls.
d P = .875 versus group S + I.
e P < .001 versus group S + I and controls.
f P < .001 versus controls.
g P < .001 versus groups S, I, S + I, and controls.
h P < .001 versus group I and controls.
i P = 1 versus group S + I.
j P = .01 versus group S + I.
k P < .001 versus controls.
l P < .001 versus controls.
m P < .001 versus group S and controls.
n P = .837 versus group I.
o P = .04 versus group S + I.
p P < .001 versus group I and controls.
q P = 1 versus group S + I.
r P = .01 versus group S + I.
s P < .001 versus controls.
t P < .001 versus controls.
u P < .001 versus groups S, S + I, and controls.
v P = .047 versus group I.
w P = .001 versus group I.
x P = .765 versus group S + I.
y P = .003 versus group S + I.
z P < .001 versus controls.
aa P < .001 versus controls.
bb P = .004 versus group S.
cc P = 1 versus group I and controls.
dd P < .001 versus group S + I.
ee P = .192 versus group I.
ff P = 1 versus group S + I.
gg P = .032 versus controls.
hh P = .014 versus group S + I.
ii P = 1 versus controls.
jj P = .002 versus controls.
kk P = 1 versus groups S and controls.
ll P < .001 versus groups I and S + I.
mm P < .001 versus groups I and S + I.
nn P = 1 versus controls.
oo P = 1 versus group S + I.
pp P < .001 versus controls.
qq P < .001 versus controls.
Simvastatin administration was associated with serum glutamic pyruvic transaminase (SGPT) elevation, with no evidence of renal impairment as demonstrated in Table 1. No effect of ivabradine on creatinine and hepatic enzymes was seen.
Group I animals demonstrated a significant heart rate reduction after 9 weeks compared with 5 weeks of ivabradine administration (141 ± 21.45 bpm vs 167 ± 26.88 bpm; P = .019). Similarly, group S + I revealed a marginally significant heart rate decrease at the end of 9 weeks compared with 5 weeks (143 ± 19.04 bpm vs 170 ± 27.43 bpm; P = .051; Table 1). At the end of the 9-week period, group I and S + I rabbits showed a significantly reduced heart rate compared to group C (245 ± 9.43 bpm; P < .001, post hoc analysis).
Signs of toxicity were observed in groups S and S + I based on the respective weights. Group S rabbits demonstrated a significant weight reduction compared to groups C and controls (3260 ± 275.7 kg vs 3650 ± 150.9 kg and 3580 ± 82.3 kg; P = .004 and P = .032, respectively). Accordingly, group S + I revealed a significant weight reduction compared to groups C and controls (3160 ± 263.3 kg vs 3650 ± 150.9 kg and 3580 ± 82.3 kg; P < .001 and P = .002, respectively). It should be noted that there was a mortality rate of 23% in group S + I (3 animals died and were further replaced, overall rabbits = 13), fact that is in accordance with the significant weight reduction in group S + I.
Histological Evaluation
Hematoxylin and eosin-stained sections of descending thoracic aortas were examined for signs of atherosclerosis. Atherogenic diet induced the development of significant atherosclerotic lesions in all group C animals but in none of groups S, I, and S + I.
Specifically, atherosclerotic lesions of group C thoracic aortas were classified as intermediate type III (n = 5 animals) or advanced type IV (atheroma, n = 5 animals). Thoracic aortas of group S demonstrated absence of atherosclerotic lesions (n = 10). Thoracic aortas of group I animals revealed no signs of atherosclerosis (n = 4), initial type I atherosclerotic lesions (n = 2), and type II lesions (n = 4). Group S + I thoracic aortas presented inhibition of atherosclerotic lesion formation (n = 8), except for 2 cases that showed initial type I atherosclerotic lesions (Figure 1).

Hematoxylin–eosin, Masson trichrome, and Van Gieson staining. A, Hematoxylin–eosin staining of type IV atherosclerotic lesion. B, Masson trichrome demonstrates moderate collagen tissue deposition in type IV atherosclerotic lesion. C, Van Gieson staining reveals local fiber disruption and disorientation in type IV lesion. D, Hematoxylin–eosin staining reveals no atherosclerotic lesion formation. E, Masson trichrome staining demonstrates no collagen tissue deposition in the aorta wall. F, Van Gieson stating shows normal elastic fiber orientation of aortic wall.
Masson and Van Gieson staining revealed slight to moderate collagen tissue deposition in tunica media and intima as well as local disruption of elastic fibers in IEL of group C and group I atherosclerotic aortas. No elastic fiber disruption or collagen deposition was observed in group S and S + I aortas (Figure 1).
Immunohistochemistry Evaluation
The atherogenic diet was accompanied by a significant increase in lipid deposition and foam cell formation, indicated by the increase in RAM-11 immunoreactivity in group C animals compared with groups S, I, and S + I, respectively (P < .001; Figure 2). Post hoc analysis revealed a significant foam cell percentage reduction in groups S, I, and S + I compared with group C atherosclerotic lesions, respectively (P < .001; Figure 2). A similar reduction effect on macrophage cell deposition between groups S and I was also found (P = .32), although there was no difference in RAM-11–positive cells in group S + I compared to group S (P = 1) and group I (P = .95).

RAM-11 and HHF-35–positive cell distribution in individual groups (n = 10). A, Simvastatin and ivabradine reduced foam cell deposition in aortic atherosclerotic lesions as expressed by the percentage of RAM-11 positive cells. a P < .001 versus groups S, I, and S + I, b P = .32 versus group I, c P = 1 versus group S + I, and d P = .95 versus group S + I. Lines represent medians, and error bars represent interquartile range. P values were derived by Bonferroni post hoc ANOVA. B, Both simvastatin and ivabradine decreased smooth muscle cell proliferation in atherosclerotic lesions of thoracic aortas as indicated by HHF-35–positive cell percentage. e P < .001 versus groups S, I, and S + I, g P = .92 versus group I, hP not defined versus group S + I, and i P = .92 versus group S + I. Lines represent medians, and error bars represent interquartile range. P values were derived by Tamhane post hoc ANOVA. ANOVA indicates analysis of variance; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine.
Specifically, group C thoracic aortas were strongly positive (n = 9 animals) for RAM-11 staining, except for 1 case that was moderately positive (Figure 3). Group S thoracic aortas revealed no RAM-11 staining, except for 2 cases that showed mildly positive RAM-11 immunoreactivity. Group I thoracic aortas showed either no staining (n = 4) or mildly positive RAM-11 immunoreactivity (n = 5), except for 1 case that was moderately positive. Similarly, group S + I rabbits revealed no RAM-11 immunoreactivity (n = 8) as presented in Figure 3, and only 2 cases demonstrated mildly and moderately positive immunoreactivity. It should be noticed that the percentage of macrophages was in accordance with the respective histological severity of atherosclerotic lesions.
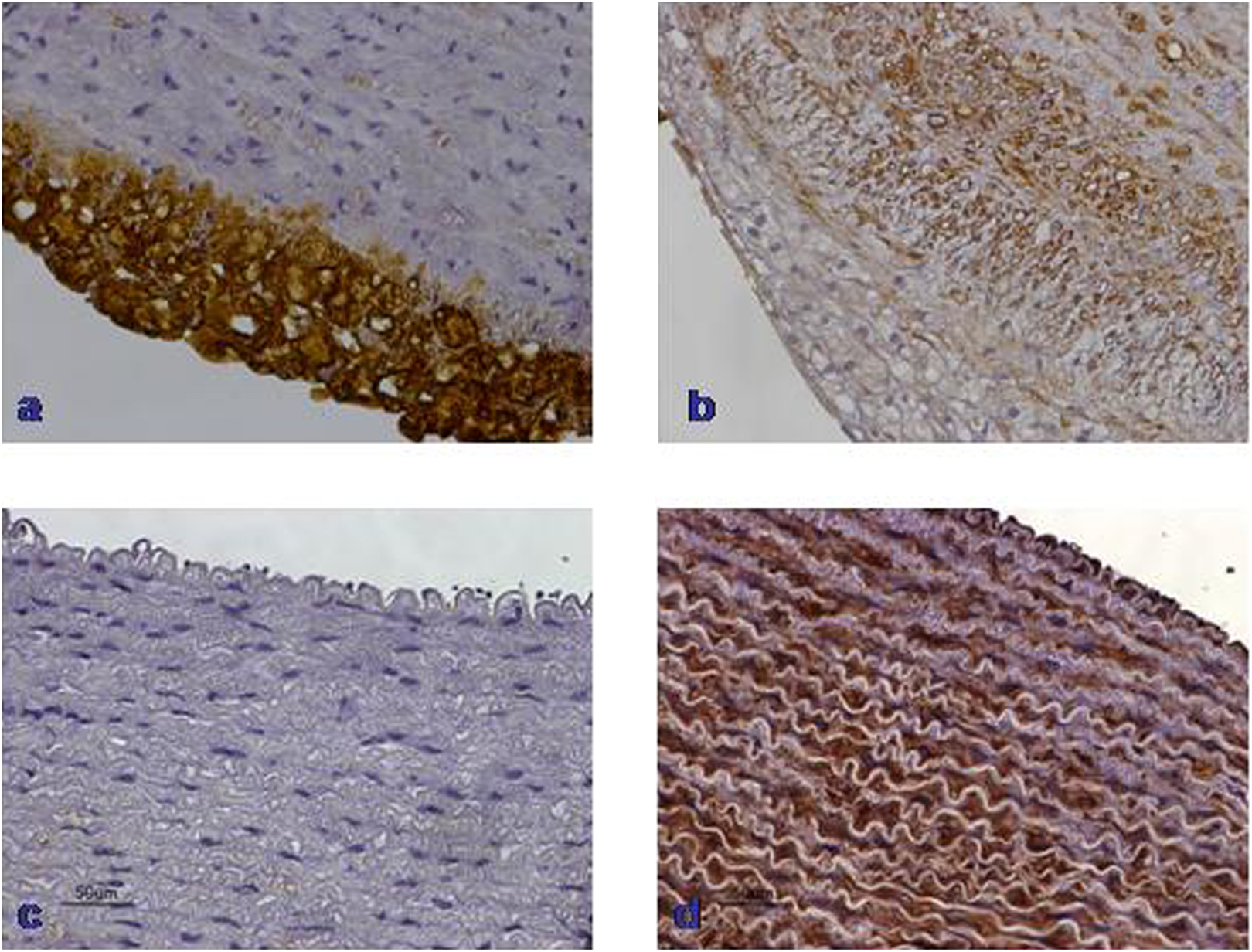
RAM-11 and HHF-35 staining of thoracic aortas. A, Plethora RAM-11 positive foam cells in type IV atherosclerotic lesion. B, Several HHF-35–positive SMCs between foam cells in atherosclerotic plaque type IV lesion. C, Lack of foam cell deposition. D, Normal presence of HHF-35–positive SMCs in the media of nonatherosclerotic aortic wall. SMC indicates smooth muscle cell.
The percentage of SMCs in atherosclerotic lesions of group C thoracic aortas was significant compared with groups S, I, and S + I, respectively (P < .001; Figure 2). Post hoc analysis revealed a significant decrease in SMC percentage, indicated by the reduced HHF-35 immunoreactivity in groups S, I, and S + I aortic atherosclerotic lesions compared with group C (P < .001). There was a similar reduction effect on HHF-35–positive cells between groups S and I (P = .920), as demonstrated in Figure 2.
Particularly, group C aortic atherosclerotic lesions revealed either moderately positive (n = 6) or mildly positive (n = 4) HHF-35 immunoreactivity (Figure 3). Groups S and S + I thoracic aortas demonstrated no HHF-35 immunoreactivity (n = 20). Group I aortic atherosclerotic lesions showed no HHF-35 immunoreactivity (n = 9), except 1 case that indicated mildly positive staining.
Morphometry
Atherogenic diet induced significant intimal hyperplasia in atherosclerotic lesions of group C thoracic aortas compared with groups S, I and S + I, as demonstrated in Table 2 and Figure 4. Post hoc analysis revealed a significant neointimal hyperplasia inhibition in groups S, I, and S + I compared with group C (P = .015, P = .021, and P = .019, respectively). In addition, group S demonstrated a marginally significant inhibition of intimal hyperplasia compared with group I (P = .059), whereas neointimal formation was significantly lower in group S compared to group S + I (P = .032). Medial area in group C thoracic aortas demonstrated no difference compared to groups S, I, and S + I (P = .680, P = .525, and P = .539, respectively) as reported in Table 2 and Figure 5.
Quantification of Morphometric Parameters in Aortic Atherosclerotic Lesions.a

Abbreviations: ANOVA, analysis of variance; C, atherogenic diet; dfb, degrees of freedom between groups; dfw, degrees of freedom within groups; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine.
a P values calculated by 1-way ANOVA.

Neointimal hyperplasia and intima–media ratio in the thoracic aortas’ atherosclerotic lesions by treatment group (n = 10). A, Both simvastatin and ivabradine inhibited significantly the intimal hyperplasia in aortic atherosclerotic lesions. Simvastatin revealed a marginally superior effect compared to ivabradine. a P = .015 versus group S, b P = .021 versus group I, c P = .019 versus group S + I, d P = .059 versus group I, e P = .032 versus group S + I, and z P = .903 versus group S + I. B, Both simvastatin and ivabradine demonstrated a significant reduction in intima–media ratio of atherosclerotic aortas, whereas simvastatin proved to be significantly superior compared to ivabradine. α P < .001 versus groups S, I, and S + I, β P = .048 versus group I, γ P = .022 versus group S + I,δ and P = .92 versus group S + I. Lines represent medians, and error bars represent interquartile range. P values were derived by Tamhane post hoc ANOVA. ANOVA indicates analysis of variance; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine.

Media, lumen, and total vessel area of atherosclerotic thoracic aortas by treatment group (n = 10). A, Lumen area of thoracic aortas revealed no difference in groups C, S, I, and S + I. i P = 1 versus groups S, I, and S + I, k P = .855 versus group I, l P = .877 versus group S + I, and m P = 1 versus group S + I. B, Medial area of aortic wall was similar in groups C, S, I, and S + I. h P = .680 versus group S, t P = .525 versus group I, f P = .539 versus group S + I, g P = .791 versus group I, j P = .920 versus group S + I, and qP = 1 versus group S + I. C, Total vessel area of atherosclerotic aortas showed no difference in groups C, S, I, and S + I. n P = .781 versus group S, x P = .646 versus groups I and S + I, o P = .908 versus group I, p P = .926 versus group S + I, and r P = 1 versus group S + I. Lines represent medians, and error bars represent interquartile range. P values were derived by Tamhane post hoc ANOVA. ANOVA indicates analysis of variance; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine.
Similarly, group C thoracic aortas revealed a significant increase in I/M ratio compared with groups S, I, and S + I (P < .001; Table 2, Figure 4). Post hoc analysis showed a significantly lower I/M ratio in groups S, I, and S + I compared with group C (P < .001). Interestingly, simvastatin was superior to ivabradine in reducing the I/M ratio, as post hoc analysis demonstrated a significant I/M ratio reduction in group S thoracic aortas compared to group I (P = .048). Moreover, the S + I group presented similar I/M ratio reduction compared to group I (P = .92) but significantly higher I/M ratio compared to group S (P = .022).
No difference in lumen area of group C thoracic aortas compared with groups S (P = 1), I (P = 1), and S + I (P = 1) was found, as reported in Table 2 and Figure 5. Similarly, total vessel area of group C thoracic aortas showed no difference compared with groups S (P = .781), I (P = .646), and S + I (P = .646; Figure 5). Consequently, atherogenic diet did not induce severe lumen stenosis and total vessel remodeling.
Oxidative Stress Analysis
As shown in Figure 6, atherogenic diet significantly increased protein tyrosine nitration and carboxy-DCF fluorescence in thoracic aortas of cholesterol-fed rabbits. Simvastatin significantly reduced protein nitrotyrosine levels (P = .009), while ivabradine significantly reduced carboxy-DCF fluorescence (P = .011), suggesting that both drugs inhibited oxidative stress in hyperlipidemic rabbits, although the mechanisms involved seem to be different.
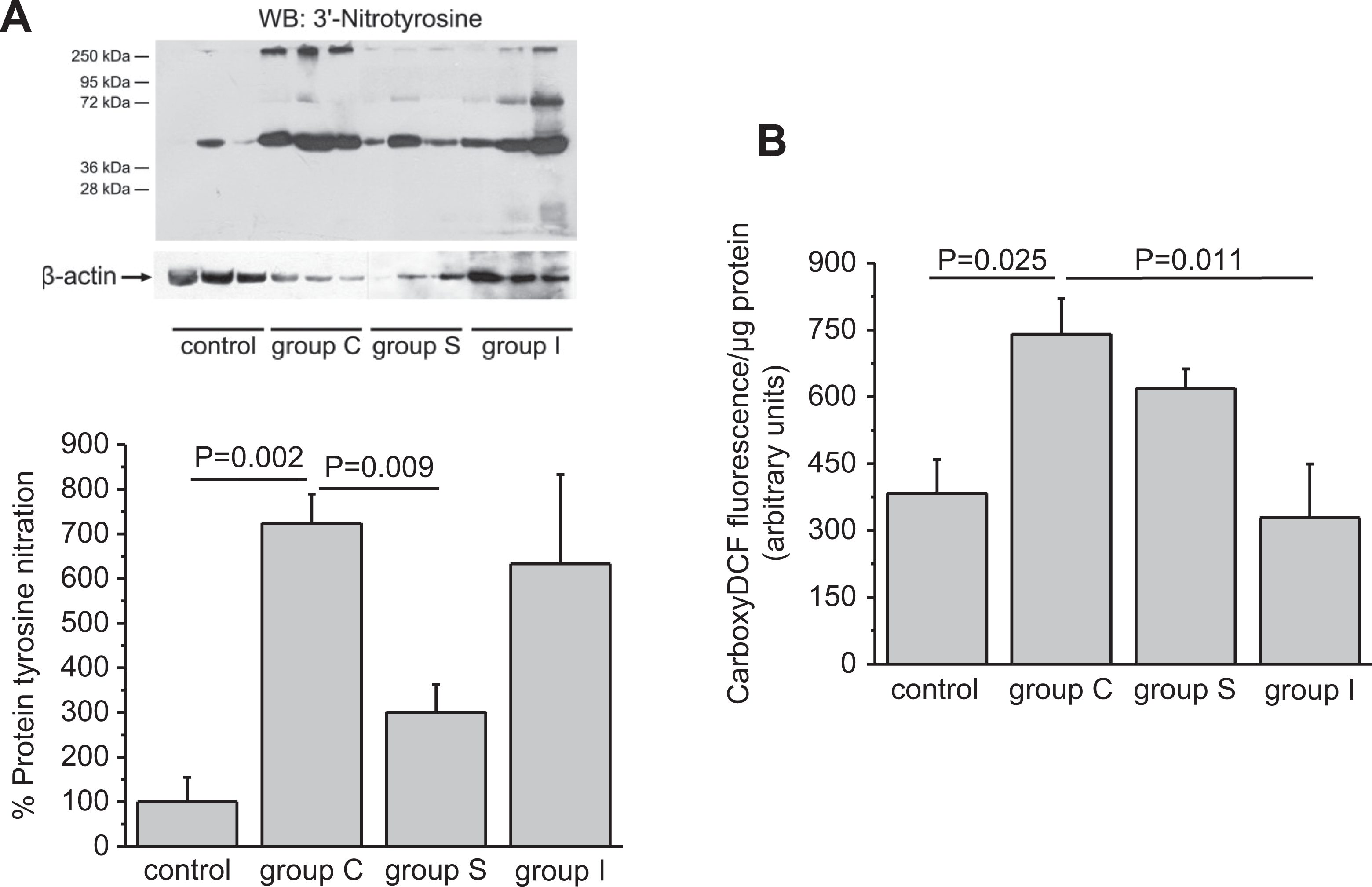
Simvastatin and ivabradine have antioxidant effects. A, Simvastatin decreased protein tyrosine nitration caused by high cholesterol diet. Quantitative data are expressed as mean ± SE (n = 3 in each group) of the percentage change in protein tyrosine nitration compared with the control group (set as default = 100). β Actin was used as a loading control. B, Ivabradine decreased carboxy-DCF fluorescence elevation caused by high cholesterol diet. Results are expressed as mean ± SE (n = 3 in each group) of carboxy-DCF fluorescence/μg total protein. DCF represents dichlorofluorescein; SE, standard error.
Mechanical Tests
Aortic stiffness EPm was reduced significantly by simvastatin, ivabradine, and their combination, [F(dfb,dfw) = 6.211(3,28); P = .002], as presented in Figure 7. Post hoc analysis revealed a statistically significant aortic stiffness reduction in groups S (35.91 ± 8.37 KPa), I (37.70 ± 7.16 KPa), and S + I (39.21 ± 7.80 KPa) compared with group C (51.52 ± 8.76 KPa; P = .003, P = .011, and P = .029, respectively). No difference in aortic stiffness was observed in group S compared to groups I and S + I (P = 1 and P = 1, respectively; Figure 5).
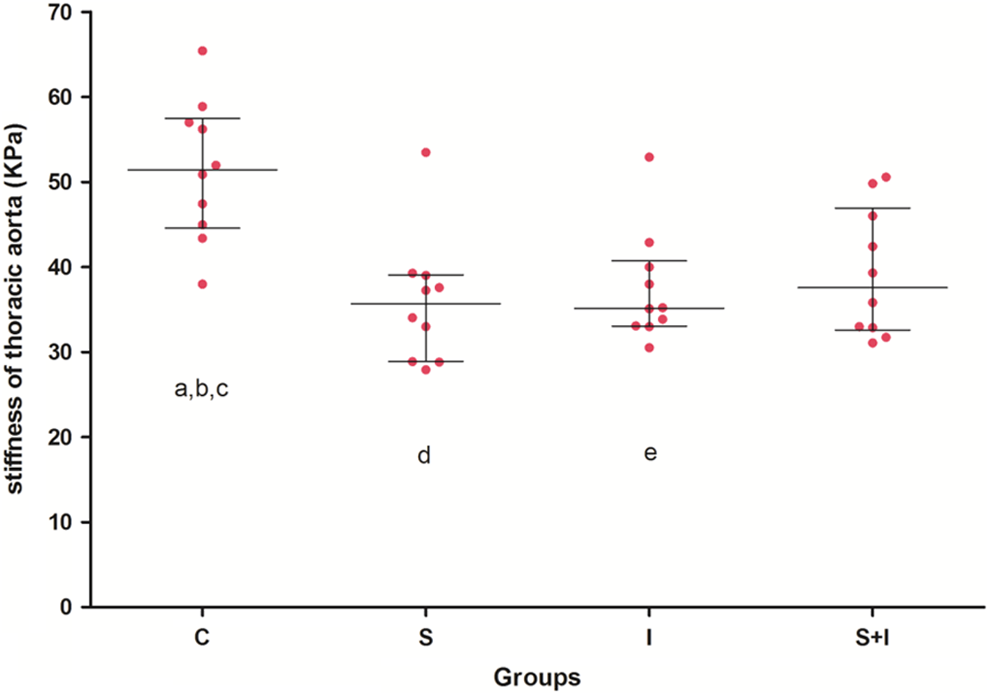
Both simvastatin and ivabradine decrease significantly wall stiffness of atherosclerotic aortas in individual group (n = 10). a P = .003 versus group S, b P = .011 versus group I, c P = .029 versus group S + I, d P = 1 versus group I, and e P = 1 versus group S + I. Lines represent medians, and error bars represent interquartile range. P values were derived by Bonferroni post hoc ANOVA. ANOVA indicates analysis of variance; I, atherogenic diet plus ivabradine; S, atherogenic diet plus simvastatin; S + I, atherogenic diet plus simvastatin and ivabradine.
Discussion
We used quasi-physiologic conditions and a closed-loop flow system to assess the aortic wall stiffness as expressed by the pressure-diameter elastic modulus in hypercholesterolemic rabbits treated with simvastatin and/or ivabradine. The novel finding of the study is that the marked reduction of intimal hyperplasia in aortic atherosclerotic lesions combined with oxidative stress attenuation following both simvastatin and ivabradine treatment resulted in significant improvement in aortic stiffness.
In our study, simvastatin significantly decreased serum lipid levels and inhibited atherosclerotic lesion formation. Simvastatin-treated rabbits revealed absence of histological atherosclerotic lesions as well as reduced macrophage accumulation and SMC proliferation in thoracic aortas. Interestingly, the significant reduction of intimal hyperplasia and the respective I/M ratio in thoracic aortas of simvastatin-treated hypercholesterolemic animals was accompanied by remarkable inhibition of protein tyrosine nitration, indicative of vascular oxidative stress diminution. These beneficial effects of simvastatin may be implicated in the significant reduction of arterial stiffness in dyslipidemic rabbits, in line with previous clinical and experimental studies that have demonstrated that statins decrease the incidence of acute coronary events, 10,24 improve endothelial function by enhancing nitric oxide bioavailability, 25 reduce inflammatory status by altering circulating cytokines and adhesion molecules as well as inhibit vascular SMC proliferation 26 and blood thrombogenicity. 27
Clinical studies designed to investigate the potential impact of statins on arterial stiffness have demonstrated conflicting results. 9,28 –39 In our model, simvastatin significantly decreased the aortic wall stiffness, inhibiting intimal hyperplasia and nitrating species-mediated oxidative stress in atherosclerotic lesions. These results confirm previous ones from 3 recent experimental animal studies, revealing that statins prevent hypertension and hypercholesterolemia-induced structural alterations in arterial wall and therefore improve arterial compliance. 4,40,41 Enhancement of endothelial function via vascular oxidative stress reduction constitutes a potentially foremost mechanism of statin-mediated arterial stiffness amelioration, 5,42,43 beyond atherosclerosis inhibition. It is notable that ivabradine demonstrated similar pleiotropic benefits to simvastatin, displaying significant antiatherogenic and antioxidant effects, although its impact on atherosclerosis reduction was not as pronounced as that of simvastatin. Ivabradine significantly inhibited atherosclerosis progression and had a beneficial impact on aortic wall stiffness, in line with previous studies showing that selective heart rate reduction by ivabradine is accompanied by a marked reduction in oxidative stress through inhibition of nicotinamide adenine dinucleotide phosphate oxidase activity and prevention of endothelial nitric oxide synthase uncoupling, 12,44,45 both leading to decreased reactive oxygen species levels. Such an effect is also in line with an improvement in endothelial function and reduced atherosclerotic plaques formation, 12,46 as well as decreased hyperlipidemia-mediated arterial stiffness. 14,47,48 It is interesting to note that in our study, ivabradine demonstrated a “surprising” lipid-lowering effect, inducing significant reduction in serum lipid levels of treated animals. Such an effect of ivabradine has never been previously shown nor can it be explained by the known mechanisms through which ivabradine might act. However, further assessment of the HDL–TC ratio, which constitutes an important risk marker of the coronary heart disease in individuals with metabolic disorders, 49 seems not to support a significant antilipidemic activity of ivabradine. Further studies should explore this possible effect of ivabradine, for example, by measuring lipid levels in patients under treatment with the drug, to estimate its possible usefulness in case of simvastatin contraindication, such as severe renal impairment or active liver disease. Moreover, the clarification of any potential synergistic effect between simvastatin and ivabradine, regarding atherosclerosis inhibition, oxidative stress reduction, and arterial wall compliance amelioration, should be further explored. Although in our study there is a lack of a synergistic effect, this cannot be totally excluded based on the observation, for example, that the 2 drugs seem to have antioxidant effects through different mechanisms. Therefore, similar studies in a significantly larger sample size could answer this question and verify our conclusions.
Conclusion
The present study demonstrated that both simvastatin and ivabradine treatment significantly inhibited atherosclerosis progression, intimal hyperplasia, foam cell accumulation, smooth muscle proliferation, and oxidative stress in thoracic aortas of high cholesterol-fed rabbits, contributing to aortic stiffness reduction and further improvement in aortic wall compliance. The beneficial effects of simvastatin and ivabradine on arterial stiffness, achieved by the prevention of atherosclerosis- and high heart rate-induced mechanical stress, may offer an additional mechanism of cardiovascular risk reduction.
Footnotes
Author Contributions
I. Koniari, D. Mavrilas, E. Apostolakis, and E. Papadimitriou contributed to conception or design, contributed to acquisition, analysis, or interpretation, drafted the manuscript, and critically revised the manuscript. E. Papadaki contributed to acquisition, analysis, or interpretation and critically revised the manuscript. A. Papalois contributed to conception or design and critically revised the manuscript. E. Poimenidi and I. Xanthopoulou contributed to acquisition, analysis, or interpretation, drafted the manuscript, and critically revised the manuscript. G. Hahalis and D. Alexopoulos contributed to conception or design, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
