Abstract
Background/Aim:
In addition to its cholesterol-lowering effect, atorvastatin (ATV) has been thought to have multiple cardiovascular benefits, including anti-inflammatory and anti-apoptotic properties. The present study was undertaken to determine whether ATV pretreatment could attenuate myocardial apoptosis and inflammation and improve cardiac function in a swine model of coronary microembolization (CME).
Methods:
Twenty-four swine were randomly and equally divided into a sham-operated (control) group, CME group, and CME plus ATV group. Swine CME was induced by intracoronary injection of inert plastic microspheres (diameter 42 μm) into the left anterior descending coronary, with or without pretreatment of ATV. Echocardiographic measurements, a pathological examination, terminal deoxynucleotidyl transferase–mediated nick end labeling staining, and Western blotting were performed to assess the functional, morphological, and molecular effects in CME.
Results:
The expression levels of caspase 3 and tumor necrosis factor-α (TNF-α) were aberrantly upregulated in cardiomyocytes following CME. Downregulation of caspase 3 and TNF-α with ATV pretreatment was associated with improved cardiac function and attenuated serum cardiac troponin I (cTnI) and high-sensitivity C-reactive protein. In addition, through a Pearson correlation analysis, the left ventricular ejection fraction negatively correlated with caspase 3, TNF-α, and cTnI.
Conclusion:
This study demonstrated that ATV pretreatment could significantly inhibit CME-induced myocardial apoptosis and inflammation and improve cardiac function. The data generated from this study provide a rationale for the development of myocardial apoptosis and inflammation-based therapeutic strategies for CME-induced myocardial injury.
Introduction
Coronary microembolization (CME), which can be induced by either a spontaneous rupture of a vulnerable coronary atherosclerotic plaque or therapeutic percutaneous coronary interventions (PCIs), has been found to be associated with adverse outcomes such as cardiac contractile dysfunction and reduced coronary reserve. 1,2 The typical consequences of CME include microinfarcts with an inflammatory response, contractile dysfunction, and reduced coronary reserve. 3 Coronary vasodilators protect against microembolization when their administration is initiated prior to PCIs. Distal protection devices can retrieve atherothrombotic debris and prevent its embolization into the microcirculation, but their effect on clinical outcome has been disappointing so far, except for saphenous vein bypass grafts. 4 Because of the grave consequences of CME, many investigations have sought to determine the underlying mechanisms of CME-induced myocardial injury and alleviate the deleterious effects of CME. There have been major advances in identifying factors of the myocardial inflammatory response and apoptosis that are involved in CME-induced myocardial injury, but the overall complexity of myocardial injury suggests that additional regulatory mechanisms remain to be elucidated. 5,6 Our previous studies demonstrated that cardiomyocyte inflammation and apoptosis played a vital role in the mechanism of CME-induced myocardial injury. 7 -12 Additionally, a previous study by Herrmann et al 13 showed the beneficial effect of statin against periprocedural CME. Furthermore, results of our recent studies revealed that atorvastatin (ATV) could lower the extent of myocardial injury after CME and improve cardiac function in mice, primarily through regulation of cardiomyocyte apoptosis, 7,14 although the potential molecular mechanisms remained unclear.
The aim of the present study was to determine the role of ATV pretreatment in CME-induced myocardial inflammation and apoptosis. In this study, we focused on the interactions of ATV with the proinflammatory protein tumor necrosis factor-α (TNF-α) and the proapoptotic protein caspase 3 in CME-induced myocardial inflammation and apoptosis. This study demonstrated that ATV pretreatment could significantly inhibit CME-induced myocardial apoptosis and inflammation and improve cardiac function. The data generated from this study provide a rationale for the development of myocardial apoptosis and inflammation-based therapeutic strategies for CME-induced myocardial injury.
Materials and Methods
Animal Preparation and Experimental Procedures
Healthy swine (25-30 kg) were purchased from the Animal Center of the Agriculture College of Guangxi University (Nanning, People’s Republic of China), and throughout all experimental stages, the animals were under controlled conditions of temperature, humidity, and light, with pig feed and water provided ad libitum. This investigation conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996). The Clinical and Animal Research Ethics Committees of Guangxi Medical University approved all procedures.
Modeling and Grouping
Twenty-four miniature swine were randomly assigned into 3 groups, including a sham-operated (control) group, CME group, and CME plus ATV (ATV) group, with 8 swine in each group. The ATV group was pretreated with ATV for 7 days (2.5 mg/kg/d) and a high loading dose of ATV (80 mg) 30 minutes before microsphere injection. The CME model was induced by manual unremitting injection of microspheres into the left anterior descending (LAD) artery, as previously described by Dorge et al, 15 Breuckmann et al, 16 and Carlsson et al. 17 The swine were initially sedated via an intramuscular injection of ketamine and atropine (10-15 mg/kg and 2 mg, respectively). After endotracheal intubation, anesthesia was maintained via an intravenous drip of diazepam into the ear vein. The right femoral artery was separated, and a 6F (Cordis Corporation, USA) vascular sheath was placed. Prior to the coronary cannulation, the animals were anticoagulated via an intravenous injection of 200 U/kg heparin followed by 100 U/kg/h to maintain heparinization. A 6F JL 4.0 guiding catheter was used for the coronary angiography. After the coronary angiography, a 1.8F infusion catheter (Cordis) was placed into the LAD artery, with the tip located between the second and third diagonal branches. Microspheres with a diameter of 42 μm (Dynospheres; Dyno Particles, Lillestrøm, Norway) at a mean dosage of 100 000 were selectively infused into the LAD within 40 minutes followed by a flush with 10 mL of saline. The sham-operated swine (sham group) were subjected to the same procedures, except that the injection was saline rather than microspheres. The systemic blood pressure and heart rate were continuously monitored during the procedure.
Statistical Analysis
All quantitative data are expressed as the means (standard deviation) and were analyzed using the SPSS 13.0 software. Two-tailed, unpaired Student t tests, 1-way analysis of variance, and Pearson correlation analysis were used for statistical evaluation of the data. Differences were considered statistically significant when P < .05.
Results
Animal Groups
No significant differences in the body weight or heart rate were observed among the 4 groups (Supplemental Table S1).
ATV Pretreatment Improved Cardiac Function Following CME
Results of echocardiographic examination (Table 1) showed that 12 hours after CME modeling, the CME group exhibited significantly decreased cardiac systolic function compared with the control group, as indicated by significantly reduced left ventricular ejection fraction (LVEF), fractional shortening, and cardiac output as well as increased left ventricular end-diastolic diameter in the CME group (P < .05). Additionally, ATV pretreatment was associated with improved cardiac function in the CME swine.
Effect of Atorvastatin on Cardiac Function Following CME.a

Abbreviations: ATV, atorvastatin; CME, coronary microembolization; CO, cardiac output; FS, fractional shortening; LVEDd, left ventricular end-diastolic diameter; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESD, left ventricular end-systolic diameter; LVESV, left ventricular end-systolic volume; PTEN, phosphatase and tensin homolog deleted on chromosome 10. Sham: sham group; CME: CME group; ATV: CME + ATV group.
aData are presented as the mean (SD).
b P < .05 compared with sham.
c P < .05 compared with CME.
ATV Pretreatment Attenuated Myocardial Injury Marker Following CME
Myocardial injury following CME could be assessed by levels of cardiac troponin I (cTnI) in blood obtained from the coronary sinus (Figure 1). At 12 hours after CME modeling, serum levels of cTnI in swine from the CME group were higher than the control group (0.207 [0.016] μg/L vs 0.053 [0.009] μg/L, P = .018). Moreover, ATV pretreatment attenuated myocardial injury following CME, as reflected by the reduced cTnI levels in the ATV group compared with the CME group (0.104[0.016] μg/L vs 0.207[0.016] μg/L, P = .037).
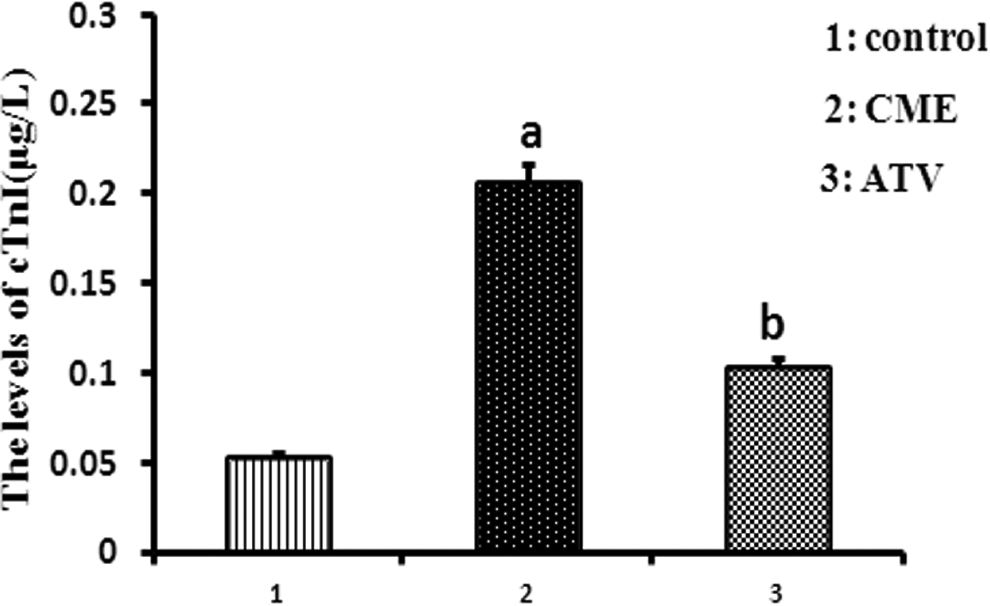
Atorvastatin (ATV) pretreatment attenuated the myocardial injury marker following coronary microembolization (CME). Serum cardiac troponin I expression was significantly increased in the CME group compared with that of the sham group (P < .05). The administration of ATV reduced the cTnI level. a P < .05 versus sham; b P < .05 versus CME. N = 8 for each group. Mean ± standard deviation. Sham: sham group; CME: CME group; ATV: CME + ATV group.
ATV Pretreatment Attenuated Inflammation Markers Following CME
The myocardial inflammatory response following CME could be assessed by measuring the levels of high-sensitivity C-reactive protein (hs-CRP) in the blood obtained from the coronary sinus (Figure 2). At 12 hours after CME modeling, serum levels of hs-CRP in swine from the CME group were higher than the control (4.58 [1.23] mg/L vs 0.56 [0.23] mg/L, P = .015). Moreover, ATV pretreatment attenuated the myocardial inflammatory response following CME, as reflected by the reduced hs-CRP levels in the ATV group compared with the CME group (2.16[0.86] mg/L vs 4.58 [1.23] mg/L, P = .042).

Atorvastatin (ATV) pretreatment attenuated the inflammation marker following coronary microembolization (CME). Serum high-sensitivity C-reactive protein (hs-CRP) expression was significantly increased in the CME group compared with that of the sham group (P < .05). The administration of ATV reduced the hs-CRP level. a P < .05 versus sham; b P < .05 versus CME. N = 8 for each group. Mean ± standard deviation. Sham: sham group; CME: CME group; ATV: CME + ATV group.
CME Histopathology
As revealed by Mayer hematoxylin and eosin (Figure S1A, B) and hematoxylin-basic fuchsin-picric acid (HBFP) staining (Figure S2A, B), the control animals exhibited subendocardial ischemia without infarction foci, whereas the CME animals exhibited multiple microinfarction foci. The administration of ATV reduced the microinfarct volume and inflammatory cell infiltration. The HBFP staining revealed myocardial karyolysis or hypochromatosis based on the red cytoplasmic staining of the microinfarction foci. In addition, peripheral cardiac muscle edema and denaturation, peripheral inflammatory cell infiltration, and erythrocyte effusion were detected. For each group, the infarct area was sham 0.00 (0.00)%, CME 6.24 (2.85)%, and ATV 3.45 (2.52)%, P = .023. (Figure S2C)
ATV Pretreatment Decreased Myocardial Apoptosis Following CME
Myocardial apoptosis was assessed using terminal-deoxynucleotidyl transferase-mediated nick end labeling (TUNEL) staining. Compared with the control group, more TUNEL-positive (brown) cardiomyocytes could be detected in swine from the CME groups (Figure S3A). Interestingly, ATV treatment significantly decreased the relative proportion of apoptotic cells following CME (Figure S3B). The percentages of myocardial apoptotic cells in the control, CME, and ATV were 0.32(0.18), 8.93(1.58), and 4.56(1.23), P = .032, respectively (Figure S3C).
ATV Pretreatment Inhibited Myocardial TNF-α and Caspase 3 in CME Swine
Western blotting showed significant upregulation of caspase 3 (Figure 3) and TNF-α (Figure 4) proteins following CME modeling compared with those from the control group (P < .05). However, ATV pretreatment was associated with reduced levels of TNF-α and caspase 3 proteins compared with the CME group (P < .05).
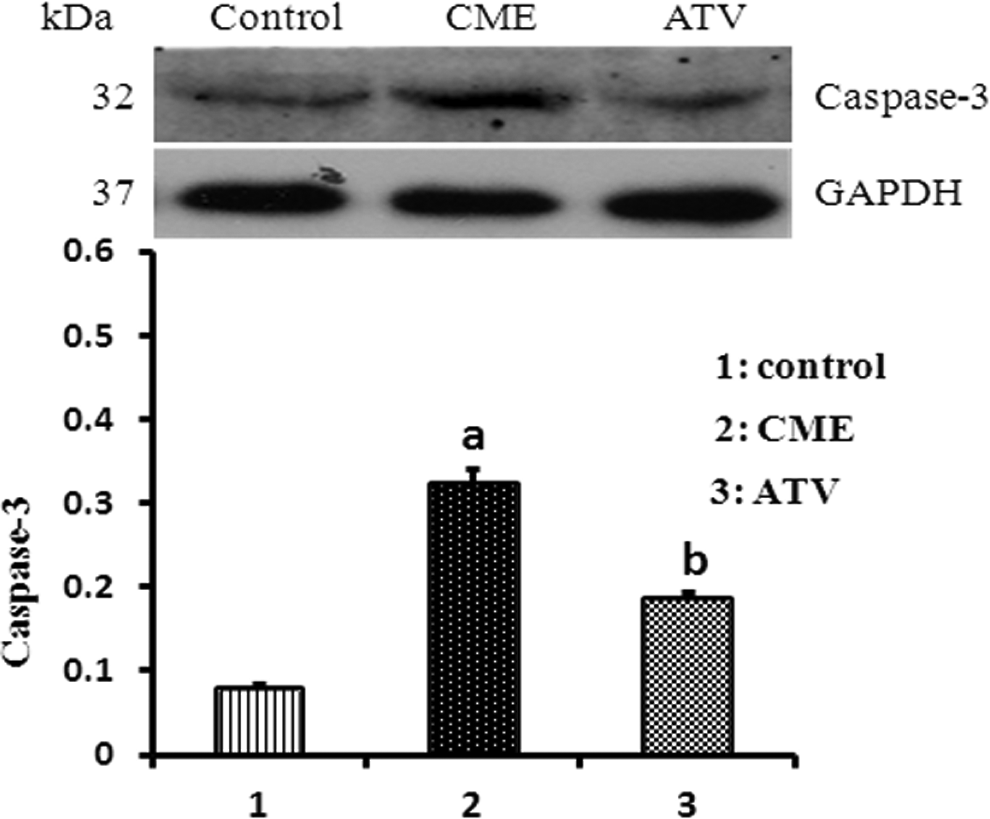
Effect of atorvastatin (ATV) on caspase 3 expression after coronary microembolization (CME). The relative protein level of cleaved caspase 3 was determined by Western blot, and the ratio of caspase 3 IA to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) IA was used to represent the relative level of caspase 3. a P < .05 versus sham; b P < .05 versus CME. N = 8 for each group. Mean ± standard deviation. Sham: sham group; CME: CME group; ATV: CME + ATV group.

Effect of atorvastatin (ATV) on tumor necrosis factor-α (TNF-α) expression after coronary microembolization (CME). The relative protein level of TNF-α was determined by Western blot, and the ratio of TNF-αIA to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) IA was used to represent the relative level of TNF-α. a P < .05 versus sham; b P < .05 versus CME. N = 8 for each group. Mean ± standard deviation. Sham: sham group; CME: CME group; ATV: CME + ATV group.
Pearson Correlation Analysis
Using a Pearson correlation analysis, the LVEF negatively correlated with caspase 3 (r = −.937, P < .001), TNF-α (r = −.852, P < .001), and cTnI (r = −.906, P < .001; Figure 5).

Pearson correlation analysis. Using a Pearson correlation analysis, the left ventricular ejection fraction (LVEF) negatively correlated with caspase 3 (r = −.937, P < .001), tumor necrosis factor-α (TNF-α; r = −.852, P < .001), and cardiac troponin I (cTnI; r = −.906, P < .001).
Discussion
The novel findings of the present study are summarized as follows. First, we demonstrated that the expression levels of caspase 3 and TNF-α were aberrantly upregulated in cardiomyocytes following CME. Second, downregulation of caspase 3 and TNF-α with ATV pretreatment was associated with improved cardiac function and attenuated cTnI and hs-CRP levels. These findings not only help us understand the mechanisms by which myocardial apoptosis and inflammation mediate myocardial injury but also support our hypothesis that TNF-α and caspase 3 proteins may represent 2 potential intervention targets. The data generated from this study provide a rationale for the development of myocardial apoptosis and inflammation-based therapeutic strategies for CME-induced myocardial injury.
Coronary microembolization is widely observed in acute coronary syndrome and is considered to be an iatrogenic complication following coronary interventions in clinical settings. 4 Coronary microembolization, caused by the embolization of thrombotic material and debris or the rupture of an atherosclerotic plaque, is believed to generate a transient decrease in coronary blood flow, followed by reactive hyperemia and myocardial systolic dysfunction. Rioufol et al 18 demonstrated that the formation of atherosclerosis frequently presented with the rupture and repair of plaques. Therefore, our data further confirm the pathophysiological manifestations of CME.
Previous studies demonstrate that the aggregate amount of infarction involved a small area (<5%) in the microembolized myocardium of pigs or dogs, as indicated by typical inflammatory responses, including increased TNF-α expression and leukocyte infiltration. 19 -22 Additionally, our previous studies demonstrated that an intense inflammatory response triggered by CME increases the protein level of TNF-α in cardiomyocytes. 9 Furthermore, our previous study revealed that cardiomyocyte apoptosis plays a significant role in CME-induced myocardial injury and that the expression of the proapoptotic protein caspase 3 in cardiomyocytes was elevated. 10
Atorvastatin had been proposed to have multiple cardiovascular benefits besides its cholesterol-lowering effect, including anti-inflammatory and antiapoptotic properties. In recent years, many studies validated its antiapoptotic effect in animal models of myocardial ischemia and reperfusion injury, 4 coronary atherosclerosis, 23 myocardial infarction, 5,24 and renal ischemia–reperfusion injury. 25 Additional studies have shown that ATV may regulate the mitochondrial apoptotic pathway and the inflammatory pathway. Caspases are a family of aspartic acid–specific proteases that have been considered to be key in the induction of apoptosis. 26 Caspase 3 is an initiator caspase in the mitochondrial apoptosis cascade. 26 Tumor necrosis factor-α is the most critical factor in inflammatory reaction pathways. Atorvastatin has been shown to modulate the cellular immune balance between activation and negative regulation of cells, thereby inhibiting expression of the proinflammatory factor TNF-α. 27 Atorvastatin could inhibit activation of the cell apoptotic signaling pathway. 28,29 In view of the aforementioned findings, we proposed that pretreatment of ATV in CME animals may protect against CME-related myocardial dysfunction by the blockade of the mitochondrial apoptotic pathway and the inflammatory pathway. In this study, we found that pretreatment with ATV (2.5 mg/kg/d) for 7 days was associated with substantially reduced myocardial apoptosis and inflammation and also preserved myocardial function. These results suggest that ATV can reduce CME-induced myocardial injury through inhibiting myocardial apoptosis and inflammation.
In summary, we revealed the important role of cardiac-specific caspase 3 and TNF-α in the CME-induced myocardial apoptosis and inflammation. The results of this study highlight the potential of caspase 3 and TNF-α as interventional targets for the treatment of CME-related myocardial apoptosis and inflammation. Moreover, the potential therapeutic role of ATV seems to have regulatory effects on the mitochondrial apoptotic pathway and the inflammatory pathway. It should be noted that our research was performed in swine models, and the findings may not be extrapolated directly to humans. Further research, especially translational research in humans, is needed to evaluate whether the potential regulatory effects of ATV on caspase 3 and TNF-α pathways could lead to a promising treatment strategy for CME-related cardiac dysfunction.
Footnotes
Author Contributions
Jiang-You Wang contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised manuscript; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Han Chen contributed to conception and contributed to analysis and interpretation. Xi Su contributed to conception, contributed to acquisition, and drafted the manuscript. You Zhou contributed to design, contributed to analysis, and drafted the manuscript. Lang Li contributed to conception and design; contributed to acquisition, analysis, and interpretation and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the National Natural Science Foundation of China (Grant No 81260042).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
