Abstract
It is not known whether there are positive or negative interactions on ventricular function when a calcium-sensitizing inotrope is added to a phosphodiesterase inhibitor in the clinical setting of acute left ventricular (LV) dysfunction. We hypothesized that when levosimendan is added to milrinone treatment, there will be synergetic inotropic and lusitropic effects. This was tested in an anesthetized porcine postischemic global LV injury model, where ventricular pressures and volumes (conductance volumetry) were measured. A global ischemic injury was induced by repetitive left main stem coronary artery occlusions. Load-independent indices of LV function were assessed before and after ventricular injury, after milrinone treatment, and finally after addition of levosimendan to the milrinone treatment. Nonparametric, within-group comparisons were made. The protocol was completed in 12 pigs, 7 of which received the inotrope treatment and 5 of which served as controls. Milrinone led to positive lusitropic effects seen by improvement in tau after myocardial stunning. The addition of levosimendan to milrinone further increased lusitropic state. The latter effect could however not be attributed solely to levosimendan, since lusitropic state also improved spontaneously in time-matched controls at the same rate during the corresponding period. When levosimendan was added to milrinone infusion, there was no increase in systolic function (preload recruitable stroke work) compared to milrinone treatment alone. We conclude that in this model of postischemic LV dysfunction, there appears to be no clear improvement in systolic or diastolic function after addition of levosimendan to established milrinone treatment but also no negative effects of levosimendan in this context.
Keywords
Introduction
Patients with myocardial ischemia or postischemic ventricular stunning may require inotropic support during critical illness or in connection with cardiac surgery in the postcardiopulmonary bypass period. β-adrenergic drugs are commonly used in this situation, 1,2 even though they increase myocardial oxygen consumption 3 and may be unfavorable for patients with ongoing ischemia. The majority of patients with preexisting cardiovascular disease are however treated chronically with β-adrenergic antagonists. In those patients, β-adrenergic drugs are less effective and non–β-adrenergic receptor-stimulating agents may be needed to treat acute heart failure or dysfunction. 4 –7 The 2 drug classes currently available for that purpose are phosphodiesterase III (PDE III) inhibitors and calcium-sensitizing drugs. 8 –10 When used as monotherapy these inodilators have very similar cardiovascular effects but are thought to have different mechanisms of action, therefore expected to have in theory additive effects. To our knowledge, there are no clinical or experimental studies on cardiac function effects when these 2 drugs are combined following ischemic myocardial stunning. It is unknown whether these drugs interact in an additive, synergistic, or other way. In clinical practice, one type of inotrope is often chosen and dosed primarily, and a different type of inotrope is added if further inotropic stimulation is desired. Our hypothesis was that additional systolic and diastolic improvement would occur when levosimendan is added to milrinone treatment in the setting of global LV dysfunction. The specific aim was to measure load-independent cardiac systolic and diastolic effects of milrinone alone and then after the addition of levosimendan in a porcine model of acute postischemic left ventricular (LV) stunning.
Methods
This study was performed after approval from the Regional Animal Research Ethics Committee in Umeå, Sweden, and was conducted in line with the “Humane Care and Use of Laboratory Animals and the Guide for the Care and Use of Laboratory Animals” (1996), from the National Academy of Sciences’ Institute for Laboratory Animal Research, United States.
Preparation
Swedish land-race pigs were premedicated with intramuscular ketamine 10 mg
An arterial catheter with the tip in the aorta, a central venous catheter (Arrow-Howe Multi-Lumen Central Venous Catheter; Vingmed, Järfälla, Sweden), and a 7F 110 cm pulmonary artery catheter (4 lumen thermodilution balloon catheter; Arrow International, Vingmed, Järfälla, Sweden) were placed via neck vessels. Fluoroscopy was used to confirm correct catheter position. Cardiac output was measured using the pulmonary artery thermodilution technique, injecting a cold saline bolus at end-expiration with an automatic injector device and recording a mean of 3 measurements.
Hemodynamic Measurements and Calculations
Hemodynamic measurements were recorded together with conductance and LV pressure measurement (see below), and all data were continuously saved in digital format (AcqKnowledge; Biopac, Goleta, California). Systemic vascular resistance was calculated using standard formulae.
Conductance Catheter Measurements
Following a carotid artery cut down on the right side of the neck, a combined tip manometer and conductance catheter (7-F, CA-71083-PN; CD Leycom, Zoetermeer, the Netherlands) was advanced to a position in the LV long axis, with its tip in the apex, using fluoroscopic guidance. This catheter has been described previously. 11,12 Briefly, it houses 12 electrodes with 8-mm spacing between them to create an electric field that is used for electrical conductance measurement in the surrounding tissue. During balloon occlusion of the main stem of the left coronary artery to achieve myocardial stunning (described below), the conductance catheter was withdrawn and kept in a saline bath. Following myocardial injury, the conductance catheter was repositioned using fluoroscopy and the volumetric measurements recalibrated.
Parallel conductance and flow reference ratio 13 –18 were determined for volume signal calibration by injecting 3 mL of 10% hypertonic saline into the distal lumen of the pulmonary artery catheter, before each conductance catheter parameter collection. The volume signal was further calibrated from relative to absolute values using thermodilution cardiac output and stroke volume measurements. 13 –16,19,20 A signal conditioning amplifier (Leycom Sigma 5DF; CD Leycom, Zoetermeer, the Netherlands) was used in dual-field mode to generate volume signals. Volume and pressure data were recorded at 250 Hz.
In order to record consecutive heart cycles with controlled alteration in end-diastolic volumes, a 7.5F balloon occlusion catheter (Vascular Technologies, Solna, Sweden) was placed, guided by fluoroscopy, with the balloon tip in the inferior vena cava. Ventricular pressure and volume data were recorded during controlled and gradual restriction of venous return by injection of 2 to 3 mL saline into the balloon-tipped catheter, leading to transient partial obstruction of vena cava flow during apnoea.
Left ventricular systolic stroke work, maximal LV power (PWRmax), maximal LV pressure increase and decrease over time (dp/dtmax and dp/dtmin, respectively), end-systolic elastance (Ees), preload recruitable LV stroke work (PRSW), 21 –25 diastolic LV pressure half-time, time constant of LV pressure decay during isovolumetric relaxation (tau), end-diastolic LV pressure and volume were recorded, and end-diastolic LV stiffness (end-diastolic pressure/volume single loop) was calculated from a single end-diastolic point from apnoeic heart cycles. Arterial elastance (Ea) was calculated as end-systolic pressure/stroke volume. Myocardial efficiency was calculated as stroke work/pressure volume area (SW/PVA) as well as with arterial–ventricular coupling ratio Ea/Ees (Ea calculated as end-systolic pressure/stroke volume).
Myocardial Stunning
A standardized global LV ischemia–reperfusion (I/R) injury, resulting in diffuse LV myocardial stunning was induced by a total time period of 20 minutes of intermittent left main stem coronary occlusions with a balloon-tipped catheter (balloon atrioseptostomy catheter SPT002, 9.5 mm; NuMED, Hopkinton, New York). 26,27 Coronary angiography was done immediately before inducing coronary ischemia in order to adequately position the occlusion balloon in the left coronary artery main stem. First, there were 2 one-minute sequences of left main stem occlusion, separated by 1 to 2 minutes of reperfusion. This was followed by 9 sequences of balloon occlusions, up to all of 2 minutes duration, interrupted by 1 to 2 minutes of recovery, back to approximately preocclusion blood pressure. Mean arterial pressure (MAP) was allowed to drop to a momentary minimum 30 mm Hg where upon reperfusion was instituted if the MAP remained below 30 mm Hg. Defibrillation was performed if ventricular fibrillation occurred. Contrast medium was injected in the aortic root during the procedure in order to ensure total occlusion of both branches of the left coronary artery.
Protocol
Baseline measurements of all circulatory parameters were recorded after preparation and stabilization, baseline 1, (B1). In the first stage, all animals were exposed to intermittent left main stem coronary occlusion to cause global LV I/R injury leading to LV stunning. Hemodynamic measurements following myocardial stunning were collected when the animal had developed myocardial stunning but clearly reestablished normal blood pressure so that they could tolerate the subsequent transient vena cava occlusions to assess load-independent measurements of contractility, baseline 2 (B2). The noninotrope-treated animals had hemodynamic measurements at hourly intervals for 2 hours, (C1, C2). In the inotrope treatment group, inotropic support was started with the first agent, IV milrinone, (Corotrope; Sanofi, Bromma, Sweden) immediately after the B2 measurement. An initial milrinone bolus of 50 µg
Analysis
The study design included within-group changes using paired tests and each animal as its own control. We performed sample size and power estimations in the following way: We anticipated an approximate 50% increase in improvement of systolic and diastolic function in response to levosimendan treatment, from the level already achieved by milrinone treatment. We used a paired test, power of 80% and an α value of .05, and determined a minimal sample size of 7. A smaller number of noninotrope-treated animals were also analyzed in the same way in order to confirm the injury model. Treatment effects of this drug combination in this injured heart model had not been reported before the study was performed.
Based on the small number of observations, nonparametric statistical analysis was used for within-group comparisons. A Wilcoxon signed-rank test was used to identify differences in paired comparisons for before versus after I/R, post-I/R vs with milrinone, and then finally (the main hypothesis) post-I/R with milrinone vs postinjury with milrinone plus levosimendan added. The primary outcome variables were PRSW and tau. A P value < .05 was considered to be statistically significant.
Results
The Injury Model
A total of 22 animals were prepared and entered the protocol of brief coronary occlusions. Fourteen animals completed the protocol to create an injured left ventricle successfully (received a total of 20 accumulated minutes of coronary occlusion), whereas 8 did not survive the intervention due to malignant dysrhythmia or complications related to the coronary occlusion (no recovery or deterioration between occlusions). All animals received additional fluid during the preparation, Voluven 500 mL. Of the 14 animals (32.1 ± 8.1 kg, mean ± standard deviation) that successfully completed the inotrope treatment/nontreatment protocol, 2 were subsequently excluded due to technical problems so that 7 animals successfully completed the inotropic drug protocol and 5 completed the noninotrope treatment protocol. Ventricular fibrillation episodes during the coronary occlusion sequences with successful defibrillation occurred in 3 of the 5 noninotrope-treated animals and in 2 of the 7 inotrope-treated animals. No malignant dysrhythmias (or defibrillations) occurred after the injury preparation period. The time aspects of the protocol, the coronary occlusions as well as postreperfusion measurement points are shown in Figure 1.
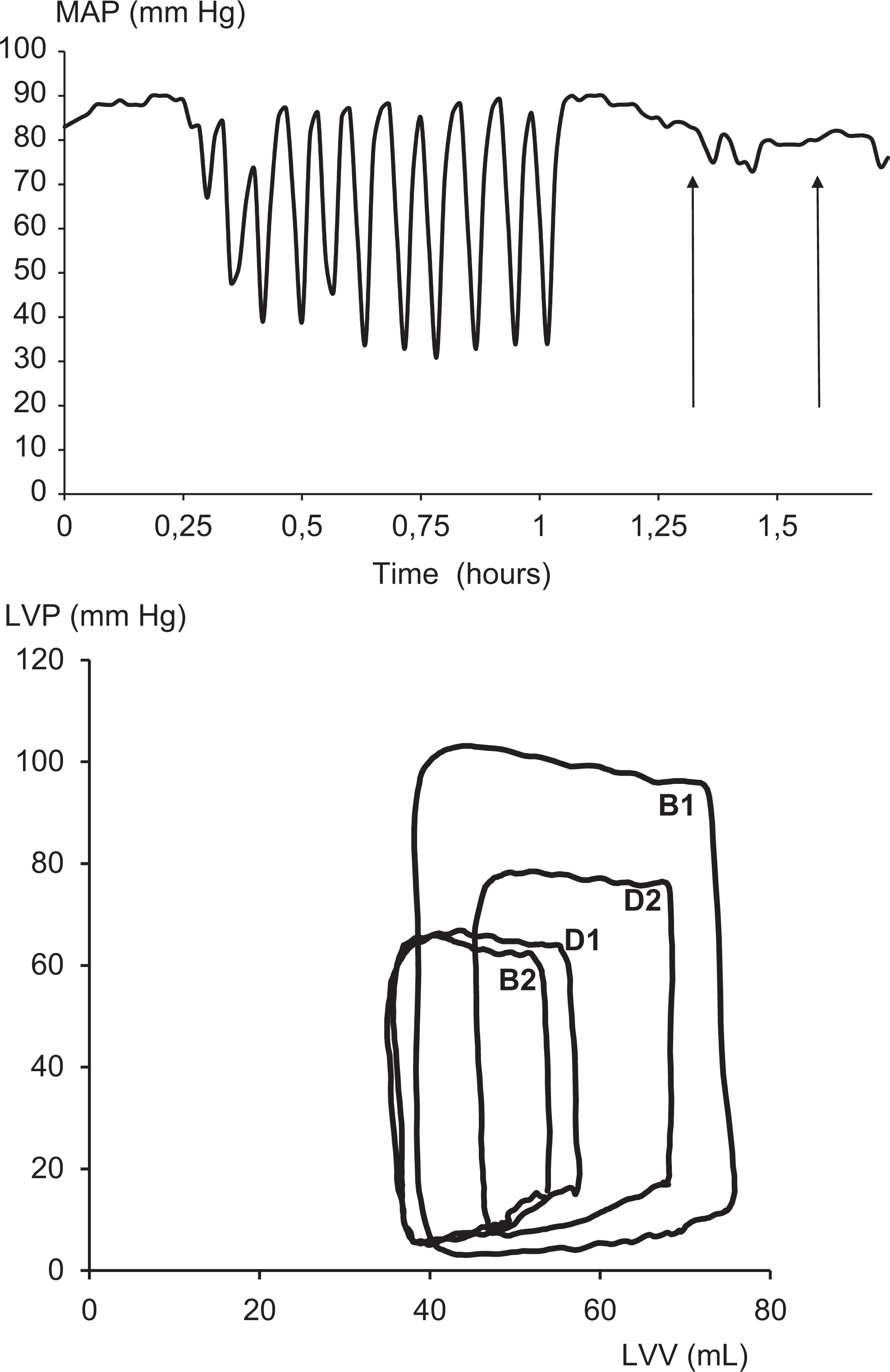
Hemodynamic illustration of the injury model and treatment protocol. In the upper panel, the injury model is illustrated showing mean arterial blood pressure (MAP) valleys that correspond to the coronary occlusions and release. The arrows indicate points first for postinjury baseline measurement and then for start of the 1 hour milrinone infusion. The lower panel demonstrates single heart cycle pressure–volume measurements throughout the protocol. Baseline hemodynamic measurements were performed before coronary occlusions (B1). A postischemia–reperfusion injury measurement (B2) and then after 1 hour milrinone treatment (D1) are shown. Measurement of milrinone + levosimendan at 1 hour of levosimendan infusion (with milrinone at second hour of infusion) is shown by D2.
The cardiac and vascular effects of myocardial I/R are shown in Tables 1 and 2, where the model of impaired ventricular performance is shown with baseline (B1) and post-I/R measurements (B2). The primary load-independent contractility indicator PRSW decreased with the I/R injury, B2 vs B1 (nondrug treatment group P =.04, drug treatment group P =.02), corroborated by SW, PWRmax, and dp/dtmax observations. End-systolic elastance is also shown at the same point. The primary load-independent variable for ventricular relaxation, tau, was prolonged after the I/R injury, B2 versus B1 (nondrug treatment group P =.04, drug treatment group P =.02).
Inotrope Treatment Group.a

Abbreviations: CO, cardiac output; CVP, central venous pressure; dp/dtmax, maximal rate of pressure increase; dp/dtmin, maximal rate of pressure decrease; Ea, arterial elastance; Ea/Ees, ventricular–arterial coupling ratio; EDP, end-diastolic pressure; EDPVR, end-diastolic pressure/volume single loop; EDV, end-diastolic volume; Ees, end-systolic pressure-volume relation slope; ESP, end-systolic pressure; ESV, end-systolic volume; HR, heart rate; I/R, ischemia/reperfusion; MAP, mean arterial pressure; PRSW, preload recruitable stroke work; PWRmax, maximal power; SVR, systemic vascular resistance; SW, stroke work; SW/PVA, stroke work/pressure volume area ratio; Tau, isovolumic relaxation time constant; min, minimum; max, maximum.
aData are presented as median (min-max).
Wilcoxon Signed-Rank within group test:
bP < .05 B2 vs B1.
cP < .05 D1 vs B2.
dP < .05 D2 vs D1.
Noninotrope Treatment Group.a

Abbreviations: CO, cardiac output; CVP, central venous pressure; dp/dtmax, maximal rate of pressure increase; dp/dtmin, maximal rate of pressure decrease; Ea, arterial elastance; Ea/Ees, ventricular–arterial coupling ratio; EDP, end-diastolic pressure; EDPVR, end-diastolic pressure/volume single loop; EDV, end-diastolic volume; Ees, end-systolic pressure-volume relation slope; ESP, end-systolic pressure; ESV, end-systolic volume; HR, heart rate; I/R, ischemia/reperfusion; MAP, mean arterial pressure; MPAP, mean pulmonary artery pressure; PRSW, preload recruitable stroke work; PWRmax, maximal power; SVR, systemic vascular resistance; SW, stroke work; SW/PVA, stroke work/pressure volume area ratio; tau, isovolumic relaxation time constant.
aData are presented as median (min-max).
Wilcoxon Signed-Rank within group test:
bP < .05 B2 vs B1.
cP < .05 C1 vs B2.
dP < .05 C2 vs C1.
Post-I/R Injury, Milrinone at One Hour
After 1 hour of milrinone infusion, an improvement in ventricular relaxation occurred as indicated in tau (and dp/dtmin; Table 1, D1. Milrinone also increased blood pressure, SW, PWRmax, and dp/dtmax load-dependent systolic performance) and the load-independent variable, PRSW, although the latter improvement did not reach statistical significance (P = .06; Table 1, D1). At the same time point after I/R for the noninotrope-treated group (Table 2, C1), neither PRSW nor tau had improved.
Levosimendan Effects, When added to Milrinone
The primary outcome variable of this study concerned the effects of adding levosimendan to already existing milrinone treatment effects (Table 1, D2 vs D1). While one secondary load-dependent systolic performance variable, PWRmax, increased, all other systolic variables remained unchanged and particularly the primary systolic outcome measurement, PRSW (P = .24). Concerning ventricular relaxation (tau), there was an observed decrease for D2 when levosimendan was added to milrinone (D2 vs D1, P = .02). In the nondrug-treated group, there was however a spontaneous recovery of tau in this same time period (C2 vs C1, P = .04), which was not observed during the first post-I/R hour in the nondrug treatment group (C1 vs B2, P = .23), corresponding with the milrinone treatment period. This spontaneous recovery in tau in the second post-I/R recovery hour coincides with the time that levosimendan effect was being assessed.
Discussion
In this animal model of postischemic LV dysfunction, adding levosimendan to milrinone led to no increase in systolic function compared to milrinone alone. Milrinone was shown to be a positive lusitropic agent. Diastolic effects of levosimendan, when added to milrinone, were more difficult to identify with confidence. This was because there could have been confounding effects of spontaneous recovery as well as additional late milrinone effects during this second hour of inotropic support. The noninotrope-treated animals demonstrated a prolonged and stable systolic dysfunction, though diastolic post I/R function assessed as tau, in contrast to systolic function, showed some spontaneous recovery during the second post I/R hour. This made the diastolic effects of levosimendan when added to milrinone, difficult to identify with precision. It is possible that levosimendan had some minor lusitropic effects in addition to those expected from milrinone and spontaneous recovery, although this study design did not allow a specific test of this. We interpret that levosimendan did not impaire lusitropic recovery when milrinone effect was present.
This study design and findings were unique in several ways. First, we established and validated a closed-chest global myocardial ischemia porcine model, which was a further development of an open thorax model with the same goal of controlled global LV ischemia and recovery (open-chest left main coronary artery snare ligation 27,28 ). In our closed-chest model, intermittent and repeated balloon occlusions of the main stem of the left coronary artery were performed, and a reproducible global LV postischemic dysfunction or stunning was demonstrated. In the second part of this study, the effects of milrinone alone and subsequently milrinone plus levosimendan on ventricular function were measured successfully in a single group using within-group comparisons. A wide range of variables and results demonstrated a mild positive inotropic effect, particularly in the load-dependent variables, for comparisons between postischemic myocardium (B2) and postmilrinone infusion (D1). The load-independent systolic parameters, including PRSW, are among the most robust for demonstrating changes in contractile status for serial measures, particularly when loading conditions change.
No recovery was seen in PRSW in the nontreatment animals (C1 vs B2 and C2 vs C1). While PRSW did not show a conclusive positive load-independent inotropic effect for milrinone (D1 vs B2), there was a trend. When levosimendan was added to milrinone, there was no improvement in PRSW (D2 vs D1). An inotropic effect was however observed when milrinone and levosimendan were combined together compared to post-I/R with no inotrope (within groups, D2 vs B2), though much of this effect was already present with milrinone alone (D1).
Milrinone by itself did produce a strong lusitropic effect, indicated by a decrease in tau. Although there was a measurable difference in tau (D2 vs D1) with the drug combination, the nontreatment group showed a similar trend (C2 vs C1) during the same measurement period, to which levosimendan may have contributed. However, a specific lusitropic effect of levosimendan in this context could not be identified with certainty. Levosimendan clearly did not worsen the lusitropic status.
Reports of milrinone effects on LV diastolic function are not in agreement concerning lusitropic effects. 29 –31 From results (Table 2) in the nontreated group, where there was no milrinone and levosimendan administered, some spontaneous recovery of diastolic function was observed at 2 hours after the injury though not for systolic function. Diastolic recovery was not observed during the first hour after injury in the untreated group. This means that in the treatment group, it is reasonable to interpret that there was a positive lusitropic effect with milrinone treatment, which has been reported earlier. This observation from the model of the pattern of spontaneous diastolic recovery between post-I/R hours 1 and 2 means that it was not possible to clearly determine whether the improvement in diastolic function occurred specifically and solely due to the addition of levosimendan to milrinone.
Milrinone has been demonstrated to increase PRSW together with a decrease in tau in an experimental sheep model made ischemic by aortic cross clamping, 32 and was shown to increase PRSW in an open chest dog heart model. 33 No effect of milrinone on PRSW was, however, found in rabbits, 34 while a reduction in tau in patients with severe congestive heart failure was seen. 35 These reports are consistent with our present findings when milrinone alone was used.
Levosimendan has been shown to improve systolic function by increasing PRSW in earlier reports, 33 but the effect on diastolic function has been less clear. In one study in patients undergoing percutaneous coronary angioplasty, levosimendan improved systolic function of stunned myocardium without obvious impairment of diastolic function. 36 Other groups have suggested that “unmasking” of the PDE-inhibiting effect of levosimendan has been shown with higher doses, 37 when using an isolated rat model and 45 minutes of normothermic ischemia, and that calcium sensitizers, such as levosimendan, should be used with caution because of its potentially negative lusitropic effects. 38 In one clinical report, levosimendan in patients with aortic stenosis undergoing cardiac surgery, assessed with isovolumetric relaxation time as a surrogate measure of tau, reported improved lusitropy. 39 Another study in patients undergoing coronary angioplasty showed an impairment of lusitropic state by levosimendan. 40
One highly possible explanation for no additional effect of levosimendan to milrinone in our study might be that the cardiac effects of levosimendan, and also its long-acting metabolite OR-1896 could be mediated mainly or totally through PDE III inhibition, enhancing cyclic adenosine monophosphate. Presuming that the milrinone effect is already significantly inhibiting PDE III activity, then one would not expect much effect from additional PDE III inhibitor treatment. Experimental studies in failing human myocardium from the explanted heart in cardiac transplantation 41 and from rats 42 support this, since no inotropic or lusitropic effects were seen in the pre-PDE III inhibited myocardial strips when levosimendan was added. The authors concluded that only a minor, if any, inotropic or lusitropic effect of levosimendan is mediated via calcium sensitizing.
Possible clinical implications of our findings include that while direct systolic and diastolic function effects of the drugs were the focus of this study, both drugs have clinically relevant vascular effects, which reduce both afterload and preload, and this can positively influence ventricular performance in patients with myocardial dysfunction, if treatment is needed.
Limitations of the Study Design
One important limitation is the relatively small sample size. Since the effect sizes were not as large as anticipated in the study design, there were limits for identifying positive effects of the drug interventions. A future study could potentially include additional treatment arms for milrinone alone and levosimendan alone over the same time period. Also, observation of recovery time over a longer period, both with and without drug might be relevant in a future study. Another limitation is that we did not perform a dose–response study, which may be important. Doses in animals were extrapolated from human pharmacological reports and from previous porcine studies. 43,44 Therefore, future studies should also incorporate dose–response effect when investigating inotropic drugs in combination. Finally, this form of within-group nonparametric analysis makes it difficult to present effect sizes. A larger study would be needed for this.
In summary, in this experimental study using a postischemic myocardial porcine model and milrinone treatment as a starting point after myocardial stunning, and where levosimendan was added to milrinone infusion, there was no increase in systolic function with levosimendan (together with milrinone) compared to milrinone treatment alone. Similarly, diastolic function did improve when levosimendan was added to milrinone treatment, although the isolated diastolic effects of levosimendan need to be interpreted together with the findings of spontaneous recovery during that same time period in the nontreatment group.
Based on these findings, we conclude that levosimendan appears to provide no clear additional systolic support when milrinone treatment is already in effect, in the setting of postischemic ventricular systolic dysfunction. We further conclude that levosimendan does not appear to add additional lusitropic effect when milrinone treatment is present, though neither does levosimendan appear to impair diastolic effect in this setting. Further clinical study of possible inotropic drug interactions to support ventricular dysfunction is needed.
Footnotes
Authors’ Note
The animal experimental work was carried out at the large animal experimental facility at Umeå University, Umeå, Sweden.
Author Contribution
Axelsson, B, Häggmark, S, Svenmarker, S, Johansson, G, and Haney, M contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Gupta, A, Tydén, H, and Wouters, P contributed to conception and design, analysis and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Örebro County Council, Örebro; Umeå University, Umeå, Sweden and also the Swedish Heart-Lung Foundation.
