Abstract

We read with interest the study performed by Ma and associates, which failed to demonstrate any impact on the 30-day, all-cause mortality in patients receiving aspirin (ASA) prior to coronary artery bypass grafting (CABG). 1 However, recent studies have shown that the preoperative administration of ASA may be of benefit not only to patients undergoing CABG but also to patients undergoing other cardiac procedures. 2 –4 In order to determine whether a preoperative ASA continuation has any impact on the 30-day, all-cause mortality, we performed an updated meta-analysis of preoperative ASA use in patients undergoing all cardiac surgical procedures.
Current through May 2015, all articles concerning preoperative ASA and postoperative all-cause mortality were identified through OVID SP and a manual search of MEDLINE, EMBASE, and COCHRANE Central Registry of Controlled Trials. The exclusion criteria were as follows: the use of clopidogrel, dipyridamole, integrilin, ticagrelor, heparin, or vitamin K antagonists prior to the surgery; age less than 18; studies using per-protocol analysis; and those not presenting mortality within 30 days following surgery. Finally, 17 trials (12 observational nonrandomized and 5 randomized controlled trials) comparing patients undergoing cardiac surgery, in whom preoperative ASA had been continued until the surgery, with patients in whom aspirin had been withdrawn no less than 7 days prior to the surgery or who had been randomized to receive placebo, were included. Presented analysis involved altogether 30 786 patients, the majority of whom underwent CABG (89.4%, n = 27 533), while the remaining underwent valve surgery (4.9%, n = 1523), CABG with concomitant valve surgery (3.1%, n = 982), or other cardiac procedures (2.4%, n = 748).
Pooled analysis showed a 27% statistically significant reduction in 30-day all-cause mortality in patients receiving ASA preoperatively (odds ratio: 0.73 [95%confidence interval: 0.57-0.94]; P = .0127; Figure 1). There was no significant between-studies heterogeneity (I 2 = 11.51%, Q = 19.21, P = .32). No difference was seen on a fixed-effect analysis, neither when any single study was omitted.
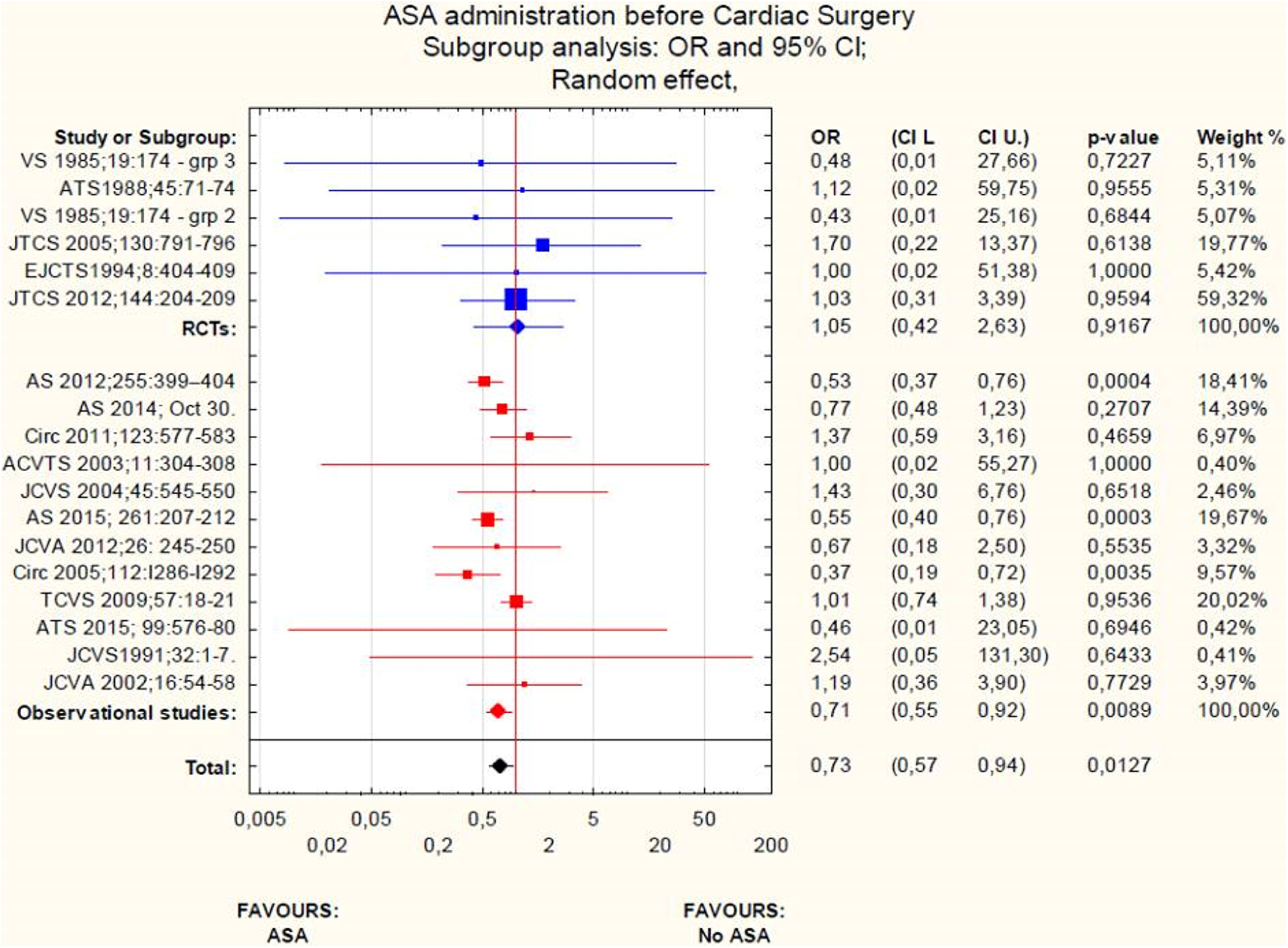
Early mortality after Cardiac Surgery among patients receiving preoperative aspirin versus placebo or no aspirin. AS indicates Annals of Surgery; VS, Vascular Surgery, JTCS, Journal of Thoracic and Cardiovascular Surgery; ATS, Annals of Thoracic Surgery, Journal of Cardiothoracic and Vascular Anesthesia; TCVS, Thoracic and Cardiovascular Surgeon; JCVS, Journal of Cardiovascular Surgery, Circ, Circulation, ACVTA, Asian Cardiovascular and Thoracic Annals; RCT, randomized controlled trial.
Our analysis differs from the previous report in 2 aspects. First, it involves patients undergoing all cardiac procedures with the great majority receiving CABG. Second, it gathers the most up-to-date evidence. Despite the results of Ma et al, we conclude that there might be an approximately 30% reduction in mortality in patients undergoing cardiac surgery when using ASA within the 7 days preoperatively. Most of the included patients underwent CABG (92.5%), therefore preoperative continuation of ASA may reduce 30-day mortality in patients undergoing surgical coronary revascularization. Although there is a statistically significant reduction in early mortality in our analysis, this conclusion is substantially limited by the absence of any support from the randomized controlled trials and the sole reliance on nonhypothesis testing, nonrandomized observational studies. In this letter, we do not address the bleeding risk reports by Ma et al, with preoperative aspirin use in patients undergoing cardiac surgery. We should thus await conclusive results of large, well-powered randomized clinical trials and a careful risk–benefit analysis of an early mortality benefit, despite any excess bleeding risk before forming any judgments or making recommendations to use preoperative aspirin in patients undergoing cardiac surgery.
Footnotes
Author Contribution
Sleiman Aboul-Hassan contributed to conception and design, acquisition, and interpretation; drafted the manuscript; critically revised the manuscript; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Stankowski, T contributed to design and acquisition, critically revised the manuscript, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Marczak, J contributed to design, contributed to conception and design, acquisition, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Cichon, R contributed to design and interpretation, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
