Abstract
Background:
The administration of aspirin is traditionally discontinued prior to coronary artery bypass grafting (CABG), given a potential risk of excessive postoperative bleeding. Few studies have previously suggested the benefits of continuing aspirin until the time of surgery. The primary aim of this review is to evaluate the effects of preoperative aspirin therapy on several clinically important outcomes in patients undergoing CABG.
Methods:
A meta-analysis of eligible studies of patients undergoing CABG, reporting preoperative aspirin in comparison with no aspirin/placebo and our outcomes, was carried out. The safety outcomes included postoperative bleeding, packed red blood cell (PRBC) transfusion requirements, and reoperation for bleeding. The efficacy outcomes included perioperative myocardial infarction (MI), cerebrovascular accidents (CVAs), and mortality.
Results:
In 8 randomized controlled trials (RCTs; n = 1538), preoperative aspirin increased postoperative bleeding (difference in means = 132.30 mL; 95 % confidence interval [CI] 47.10-217.51; P = .002), PRBC transfusion requirements (difference in means = 0.67 units; 95% CI 0.10-1.24; P = .02), and reoperation for bleeding (odds ratio [OR] = 1.76; 95% CI 1.05-2.93; P = .03). In 19 observational studies (n = 19551), preoperative aspirin increased postoperative bleeding (difference in means = 132.74 mL; 95% CI 45.77-219.72; P = .003) and PRBC transfusion requirements (difference in means = 0.19 units; 95% CI 0.02-0.35; P = .02) but not reoperation for bleeding (OR = 1.13; 95% CI 0.91-1.42; P = .27). Subgroup analyses for RCTs demonstrated that aspirin given at doses ≤ 100 mg/d might not increase the postoperative bleeding, and the dose of 325 mg/d might not be a cutoff value that has clinical and statistical significance. No statistically significant differences in the rate of perioperative MI, CVAs, or mortality were seen between the 2 groups.
Conclusions:
Preoperative aspirin therapy is associated with increased postoperative bleeding, PRBC transfusion requirements, and reoperation for bleeding in patients undergoing CABG. Doses lower than 100 mg/d may minimize the risk of bleeding. Additional RCTs are needed to assess the effects of preoperative aspirin on the safety and efficacy outcomes in patients undergoing CABG.
Introduction
Aspirin (acetylsalicylic acid [ASA]) is an integral pharmacological therapy for patients with coronary artery disease. More than 200 clinical trials presented the evidence that aspirin therapy reduced the risk of the combined outcome of serious vascular events by about one-fourth; the risk of nonfatal myocardial infarction (MI) was reduced by one-third, nonfatal stroke by one-fourth, and vascular mortality by one-sixth. 1 The administration of early aspirin after coronary artery bypass grafting (CABG) is indicated to prevent graft occlusion, reduce ischemic events (MI and stroke), and improve survival. 2 The 2011 American College of Cardiology Foundation (ACC)/American Heart Association (AHA) guidelines recommend that all patients receive daily aspirin therapy after CABG (Class I indication). 3 However, there has been controversy regarding preoperative aspirin use due to a potential increased risk of bleeding complications. 4 Most centers of cardiac surgery are currently in favor of aspirin withdrawal prior to CABG in patients without acute coronary syndrome, 5 in accordance with current guidelines from the Society of Thoracic Surgeons 6 and ACC/AHA. 7 Concerns still exist that abrupt discontinuation of chronic aspirin therapy may trigger a prothrombotic rebound phenomenon, potentially placing patients with CABG at excess risk of ischemic events in the perioperative period. 8 Evidence from randomized trials and observational studies has suggested the potential for increased postoperative bleeding, transfusion requirements, and reoperation for bleeding attributable to preoperative aspirin therapy. 9,10 However, several reports support the benefits of continuing aspirin until the day of surgery to decrease graft occlusion 11 and to improve survival 12,13 and that low-dose aspirin does not increase postoperative bleeding. 14,15
We undertook the present systematic review and meta-analysis of the relevant literature to evaluate the effects of preoperative aspirin therapy on the safety outcomes of the postoperative bleeding, packed red blood cell (PRBC) transfusion requirements, reoperation for bleeding, and the efficacy outcomes of perioperative MI, cerebrovascular accidents (CVAs), and mortality in patients undergoing CABG.
Methods
Eligibility Criteria
We sought eligible studies which met the following criteria: (1) published randomized controlled trials (RCTs) or observational studies; (2) involving patients older than 18 years; (3) undergoing first-time elective CABG, either on- or off-pump, with or without combined procedures; (4) treatment groups discontinuing aspirin less than 7 days before CABG versus no aspirin/placebo; and (5) end points included in the studies (safety end points: postoperative bleeding, reoperation for bleeding, and PRBC transfusion requirements; efficacy endpoints: perioperative MI, CVAs, and mortality). Non-English language studies were excluded as were case reports, editorial comments, and review articles. Only studies or subgroups of patients with stable coronary artery disease were included in the present analysis. Studies or subgroups of patients who were preoperatively exposed to other anticoagulants (such as heparin and warfarin) or antiplatelet drugs (such as clopidogrel and dipyridamole) were excluded in order to eliminate the potential for confounding.
Search Strategy
MEDLINE, EMBASE, and Cochrane libraries were independently searched by 2 reviewers (Xiaochun Ma and Chi Ma) for potentially eligible studies from the earliest available date to May 2013. The search terms used either alone or in combination included CABG, coronary artery bypass graft, coronary artery bypass surgery, aspirin, acetylsalicylic acid, ASA, retrospective, prospective, RCTs, observational studies, patency, occlusion, and bleeding. Reference lists from retrieved articles were also manually searched for articles meeting our criteria.
Abstracts identified using our search strategies were reviewed separately by 2 reviewers (Xiaochun Ma and Yan Yun). The full-text articles that potentially met the criteria were then reviewed in duplicate to determine the inclusion in the analysis. Disagreements between reviewers were resolved by consensus among all the authors.
Data Extraction
Data extraction was performed independently by 2 reviewers (Xiaochun Ma and Chi Ma). Extracted data included study design, assessment of study quality, baseline characteristics, patient demographics, aspirin doses, and safety and efficacy end points. The quality of studies was assessed with the Newcastle-Ottawa scale for observational studies 16 and with the Jadad scale for RCTs. 17 The Newcastle-Ottawa scale assesses the study on 3 aspects, selection of the study cohort, comparability of the cases and controls on the basis of study design, and the methods used to determine exposure to the event studied. The Jadad scale uses a set of 5 criteria to determine the quality of a RCT with a score ≥3 denoting high quality of the study. The meta-analysis has been reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Data Analysis
Review Manager 5.1 and Stata 12.0 were used for performing the meta-analysis. Random-effect models were chosen for pooling the results from included trials. 18 Differences in postoperative bleeding and transfusions were summarized as the mean differences (MDs) of the amounts of postoperative mediastinal drainage of blood (mL) and transfused PRBC (units) between treatment groups, respectively. Reoperation for bleeding, perioperative MI, CVAs, and mortality were summarized in the form of odds ratios (ORs) with 95% confidence intervals (CIs). A 2-sided P value of <.05 was considered statistically significant. Chi-square test of heterogeneity with statistical significance set at α = .10 was used to test for heterogeneity. We also used the I2 statistic, which estimates the percentage of total variation across studies, to estimate the inconsistencies between included studies. Publication bias was assessed using the Funnel plots and Egger test with a P value of <.05 considered as the level of significance. Agreement assessment between the reviewers was evaluated using the Cohen k statistic.
Subgroup analysis was carried out to explore heterogeneity between the groups. In the present analysis, the included studies were conducted over a period of more than 30 years and the sample sizes of these studies varied. It was hypothesized that there would be heterogeneity between studies by date of publication and sample sizes. Thus, subgroup analyses for RCTs or observational studies were performed on the basis of sample sizes (large sample size: ≥100 patients; small sample size: <100 patients) and publication year (before 1990; 1990 and after). We also hypothesized a priori that patients given higher doses of aspirin would have increased amounts of postoperative bleeding. 19 Thus, we also explored heterogeneity for RCTs by aspirin doses (cutoff doses: 325 mg/d or 100 mg/d) and by quality based on Jadad scale (high quality: ≥3; low quality: <3). 17 The intervention of aspirin groups was the preoperative intake of aspirin within 7 days of surgery. For individual trials with no events in 1 or both the groups, a continuity correction of .5 was added to each cell for each effect measure. 20 When multiple groups were present in a single study, those groups solely receiving aspirin were allowed for inclusion in the present study, and the sample size of the control group was split evenly by the number of included aspirin groups. 21
Results
Eligible Studies
A total of 5468 potential eligible studies were identified with the literature search. The titles of these studies were reviewed, and 5237 were rejected leaving 231 potentially eligible studies. After review of abstracts, an additional 188 were rejected and 43 studies (12 RCTs and 31 observational studies) were then retrieved in full text to determine the inclusion. Initially, assessment agreement of study selection of the full-text articles between the 2 reviewers led to a k score of .83 (95% CI 0.77-0.89). Then, a further discussion was conducted among all the authors with regard to the studies upon which the 2 reviewers disagreed. Finally, corporate review of full-text articles excluded 3 studies for not reporting our end points, 3 studies for inclusion of other studies within their manuscript, and 10 studies for not providing adequate and detailed statistics (Figure 1).

Flow diagram depicting the methodology of article selection. RCTs indicate randomized controlled trials.
Study Characteristics
Ultimately, 8 RCTs 22 –29 involving a total of 1538 patients and 19 observational studies 12,14,15,30 –45 comprising a total of 19 551 patients met our criteria and were included in this meta-analytical review. Characteristics of the included studies are detailed in Tables 1 and 2. Based on the Jadad scores, 5 RCTs were of high quality while the other 3 were of low quality. All the 5 high-quality RCTs were published after 1990 with large sample sizes. The 3 low-quality RCTs were published before 1990 with small sample sizes. Of the observational studies, 13 with large sample sizes were published after 1990. The other 6 observational studies had small sample sizes. Of the RCTs, 4 reported aspirin doses ≥325 mg/d, while the remaining 4 RCTs reported doses <325 mg/d; 2 RCTs reported dosages ≤100 mg/d. Aspirin doses in the RCTs differed from 80 to 2600 mg/d, and aspirin doses in the observational studies ranged from 75 to 2400 mg/d.
Outline of RCTs Included in the Meta-Analysis.a

Abbreviations: ASA, aspirin; RCT, randomized controlled trial; MI, myocardial infarction; CVAs, cerebrovascular accidents.
a Study quality has been assessed using the Newcastle-Ottawa scale (Selection = S; Comparability = C; Exposure = E) and Jadad scale for observational and randomized controlled trials, respectively. Subgroups of patients who were preoperatively exposed to other anticoagulants or antiplatelet drugs were excluded.
Outlines of Observational Studies Included in the Meta-Analysis.a

(Continued) Large Sample Size and After 1990.
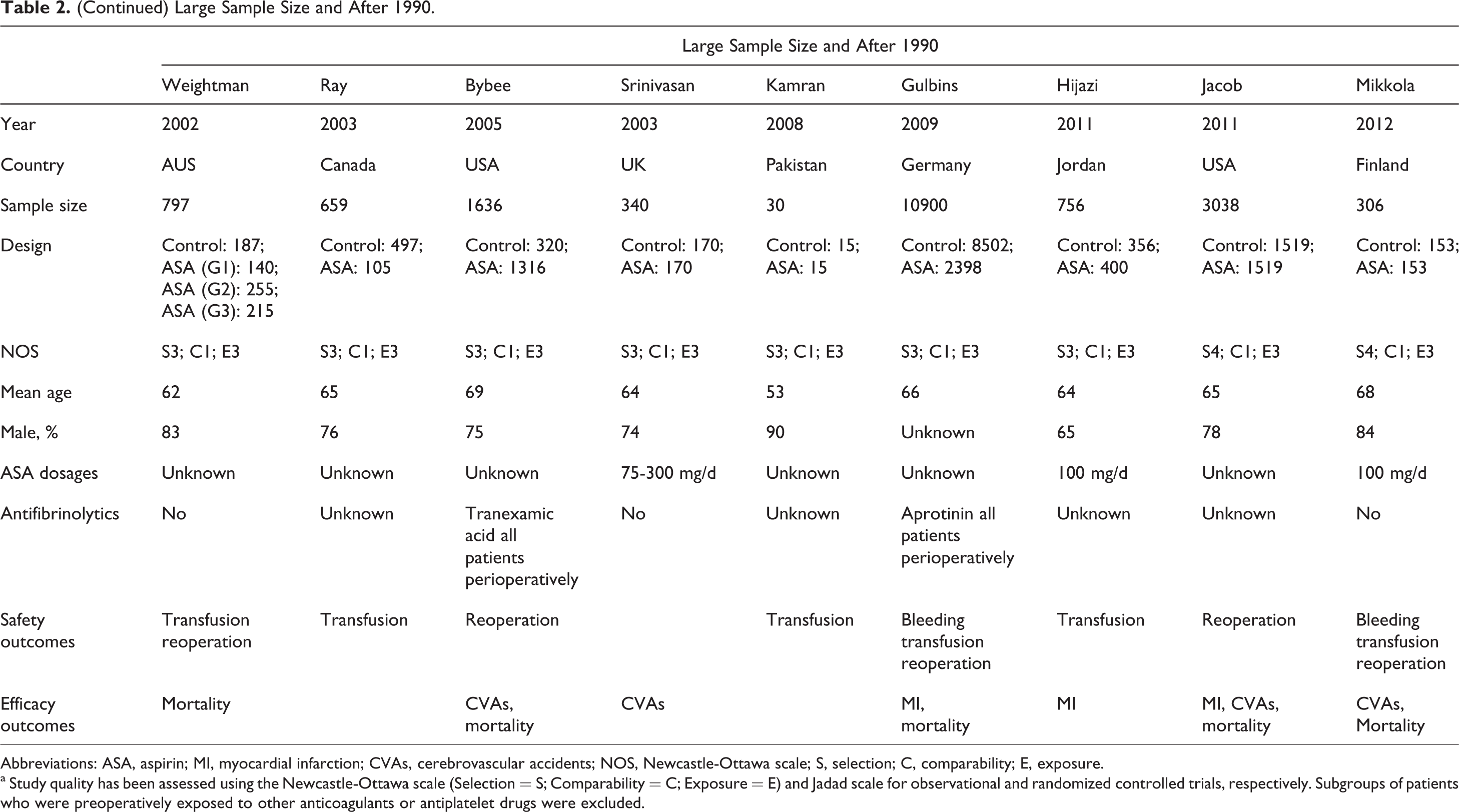
Abbreviations: ASA, aspirin; MI, myocardial infarction; CVAs, cerebrovascular accidents; NOS, Newcastle-Ottawa scale; S, selection; C, comparability; E, exposure.
a Study quality has been assessed using the Newcastle-Ottawa scale (Selection = S; Comparability = C; Exposure = E) and Jadad scale for observational and randomized controlled trials, respectively. Subgroups of patients who were preoperatively exposed to other anticoagulants or antiplatelet drugs were excluded.
Safety Outcomes
Postoperative Bleeding
Randomized controlled trials
Pooling the results of 7 studies showed that preoperative aspirin increased postoperative bleeding (MD = 132.30 mL; 95% CI 47.10-217.51; P = .002). Heterogeneity was not statistically significant (P = .21; I2 = 27.5%) and remained not evident when studies were analyzed separately in subgroup analysis by sample size, date of publication, and Jadad score. The 4 high-quality studies, published after 1990 and with large sample sizes, showed a significant increase in postoperative bleeding in the aspirin groups (MD = 105.15 mL; 95% CI 28.40-181.91; P = .01; P heterogeneity = .29; I2 = 20.2%). The other 4 low-quality studies published before 1990 and with small sample sizes demonstrated a trend toward increased postoperative blood loss in the aspirin groups (MD = 233.25 mL; 95% CI −15.04-481.53; P = .07; P heterogeneity = .16; I2 = 41.5%). But this finding did not reach statistical significance. Heterogeneity between the subgroups was not significant (P = .68; Figure 2). In addition, sensitivity analysis was performed for the RCTs excluding Fuller et al’s that reported an aspirin dose of more than 2000 mg/d. The results were consistent before and after excluding the study of Fuller et al (MD = 122.67 mL; 95% CI 33.28-212.05; P = .01; P heterogeneity = .19; I2 = 33.3%).

Meta-analysis of postoperative bleeding for RCTs (including subgroup analysis according to date of publication, sample size, and Jadad scale). RCTs indicate randomized controlled trials; CI, confidence interval.
Additionally, the studies were analyzed separately according to aspirin dose. For the cutoff value of 325 mg/d, the studies receiving aspirin doses ≥325 mg/d showed a significant increase in postoperative bleeding (MD = 229.61 mL; 95% CI 18.73-440.49; P = .03; P heterogeneity = .22; I2 = 32.1%). The studies of patients treated with doses <325 mg/d also demonstrated a significant increase in postoperative blood loss (MD = 101.61 mL; 95% CI 15.80-187.43; P = .03; P heterogeneity = .28; I2 = 21.4%). Heterogeneity between the subgroups was significant (P = .02; Figure 3). For the cutoff value of 100 mg/d, the studies receiving doses >100 mg/d showed an increased postoperative bleeding (MD = 160.70 mL; 95% CI 43.13-278.27; P = .01; P heterogeneity = .02; I2 = 43.5%). However, the studies on patients treated with doses ≤100 mg/d did not show any significant difference between the 2 groups (MD = 103.08 mL; 95% CI −65.95-272.11; P = .23; P heterogeneity = .38; I2 = 0%). Heterogeneity between the subgroups was not significant (P = .92).
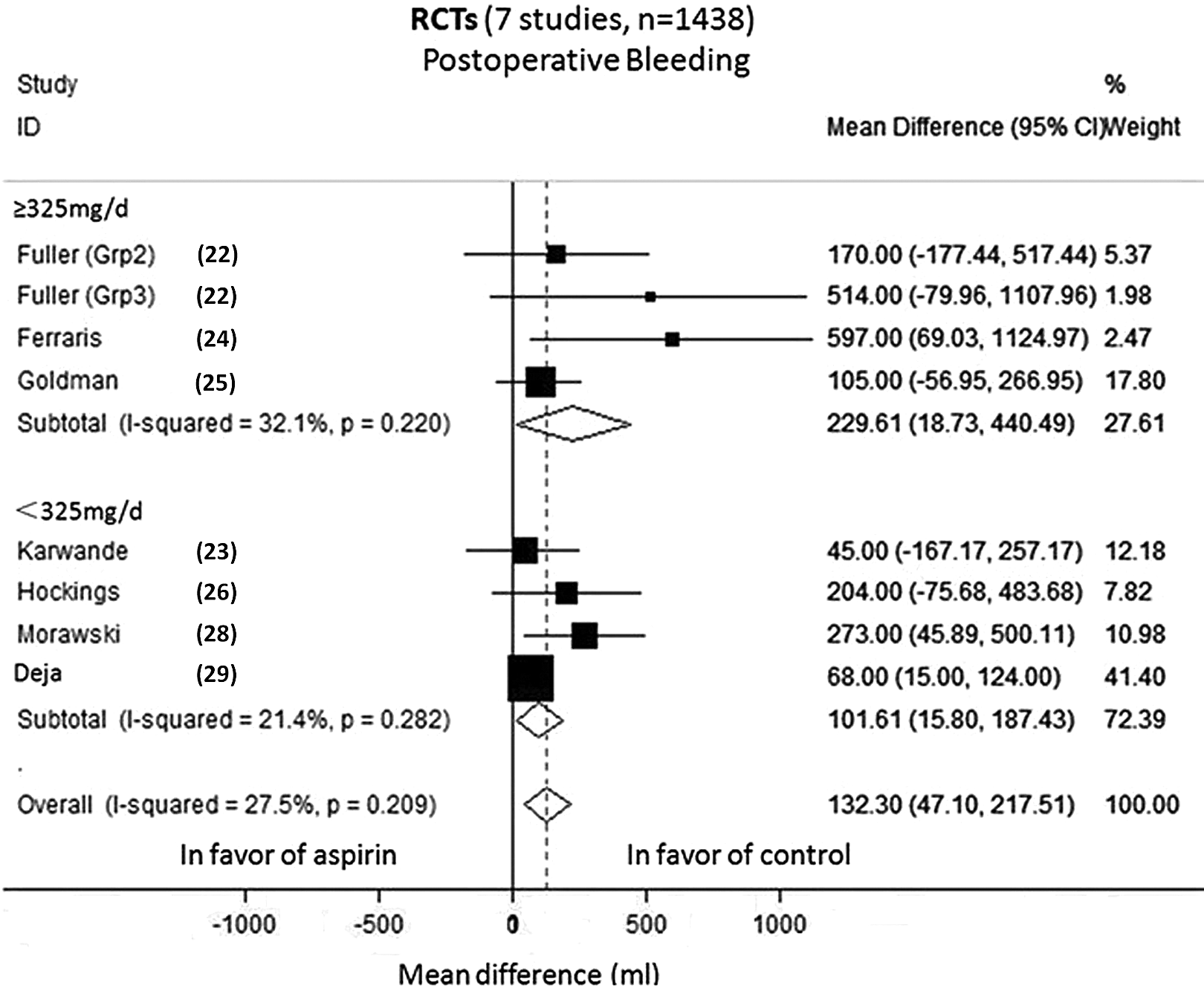
Subgroup analysis of postoperative bleeding for RCTs according to aspirin doses (325 mg/d). RCTs indicate randomized controlled trials; CI, confidence interval.
Observational studies
Pooling the results of 11 studies showed significantly greater postoperative bleeding in the aspirin groups (MD = 132.74 mL; 95% CI 45.77-219.72; P = .003). High heterogeneity was noted (P < .001; I2 = 88.3%), thus the subgroup analysis was performed according to sample size and date of publication. In all, 5 studies were published before 1990 and had small sample sizes. The pooled analysis demonstrated an increase in postoperative blood loss in the aspirin groups without heterogeneity (MD = 232.57 mL; 95% CI 147.74-317.41; P < .001; P heterogeneity = .23; I2 = 28.4%). The other 6 studies were published after 1990 and had large sample sizes. However, the pooled analysis did not show a significant increase in postoperative bleeding in the aspirin groups (MD = 46.53 mL; 95% CI −39.85-132.92; P = .29; P heterogeneity < .001; I2 = 86.1%: Figure 4). Heterogeneity was still statistically significant, and sensitivity analysis was thus performed excluding that of Gulbins et al’s, because the calculated MD for this study was extremely lower (MD = −75.00 mL, 95% CI −109.60-−40.40). The pooled analysis showed that the end point and heterogeneity changed greatly before and after exclusion of Gulbins et al’s study (MD = 75.92 mL; 95% CI 37.60-114.24; P < .001; P heterogeneity = .54; I2 = 0%), suggesting the instability of the original results. Heterogeneity between the subgroups was significant (P < .001). In addition, sensitivity analysis was performed for the observational studies excluding the studies of Michelson et al and Torosian et al that reported aspirin doses of more than 2000 mg/d. The results were consistent before and after excluding the studies of Michelson et al and Torosian et al (MD = 97.75 mL; 95% CI 12.62-182.87; P = .02; P heterogeneity < .001; I2 = 88.7%).

Meta-analysis of postoperative bleeding for observational studies (including subgroup analysis according to date of publication and sample size). CI indicates confidence interval.
Packed Red Blood Cell Transfusion Requirements
Pooling the results of 4 RCTs showed significantly greater PRBC transfusion requirements in the aspirin groups (MD = 0.67 units; 95% CI 0.10-1.24; P = .02). Heterogeneity was evident (P heterogeneity = .01; I2 = 76.7%) and was not eliminated when the subgroup analysis was carried out by date of publication, sample size, and Jadad score (data not shown). Pooling the results of 14 observational studies demonstrated that preoperative aspirin increased PRBC transfusion requirements in the aspirin groups (MD = 0.19 units; 95% CI 0.02-0.35; P = .02). Heterogeneity was statistically significant (P heterogeneity = .01; I2 = 53.4%; Figure 5) and remained evident when studies were analyzed separately according to date of publication and sample size (data not shown).

Meta-analysis of packed red blood cell transfusion for observational studies. CI indicates confidence interval.
Reoperation for Bleeding
Randomized controlled trials
The results of 7 studies were pooled and showed that preoperative aspirin increased the rates of reoperation for bleeding in the aspirin groups (OR = 1.76; 95% CI 1.05-2.93; P = .03). Heterogeneity was not statistically significant (P = .80; I2 = 0%) and remained not evident when studies were analyzed separately in a subgroup analysis according to sample size, date of publication, and Jadad score. The 5 high-quality studies, published after 1990 and with large sample sizes, showed a significant increase in the rates of reoperation for bleeding in the aspirin groups (OR = 1.74; 95% CI 1.03-2.96; P = .04; P heterogeneity = .61; I2 = 0%). The other 3 low-quality studies, published before 1990 and with small sample sizes, did not demonstrate any difference (OR = 1.99; 95% CI 0.27-14.49; P = .50; P heterogeneity = .57; I2 = 0%), and the heterogeneity between subgroups was not significant (.63; Figure 6).

Meta-analysis of reoperation for bleeding for RCTs (including subgroup analysis according to date of publication, sample size, and Jadad scale). RCTs indicate randomized controlled trials; CI, confidence interval; OR, odds ratio.
Observational studies
Pooling the results of 11 studies showed no difference in the rates of reoperation for bleeding between the aspirin groups and the control groups (OR = 1.13; 95% CI 0.91-1.42; P = .27; P heterogeneity = .94; I2 = 0%; Figure 7).

Meta-analysis of reoperation for bleeding for observational studies. CI indicates confidence interval; OR, odds ratio.
Efficacy Outcomes
Myocardial Infarction
Pooling the results of either 4 RCTs or 4 observational studies alone showed no significant difference in the rates of perioperative MI between the 2 groups (RCTs: OR = 0.73; 95% CI 0.37-1.44; P = .36; P heterogeneity = .88; I2 = 0%; observational studies: OR = 1.24; 95% CI 0.92-1.66; P = .16; P heterogeneity = 1.00; I2 = 0%; Figure 8).

Meta-analysis of myocardial infarction for RCTs and observational studies. RCTs indicate randomized controlled trials; CI, confidence interval; OR, odds ratio.
Cerebrovascular Events
Preoperative aspirin did not significantly decrease the rates of perioperative CVAs, showed by a pooled analysis of 5 studies (OR = 0.80; 95% CI 0.40-1.59; P = .52; P heterogeneity = .19; I2 = 34.8%; Figure 9).
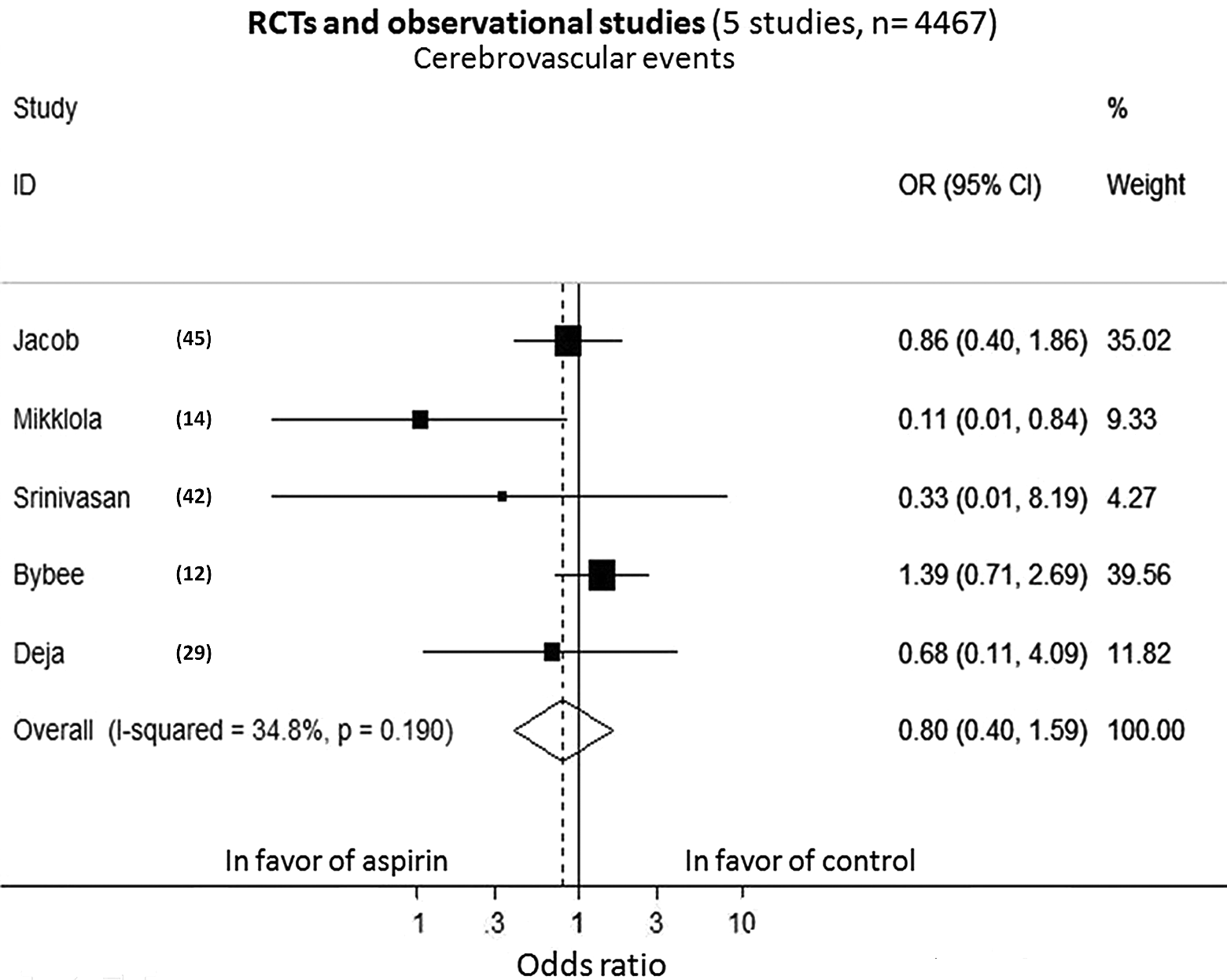
Meta-analysis of cerebrovascular events for RCTs and observational studies. RCTs indicate randomized controlled trials; CI, confidence interval; OR, odds ratio.
Mortality
Pooling the results of either 5 RCTs or 8 observational studies alone did not demonstrate any significant difference in the rates of postoperative mortality (RCTs: OR = 1.04; 95% CI 0.39-2.74; P = .94; P heterogeneity = .99; I2 = 0%; observational studies: OR = 0.87; 95% CI 0.66-1.14; P = .32; P heterogeneity = .48; I2 = 0%; Figure 10).

Meta-analysis of mortality for RCTs and observational studies. RCTs indicate randomized controlled trials; CI, confidence interval; OR, odds ratio.
Publication Bias Analysis
The funnel plots and results of Egger tests showed that there was no statistical publication bias observed in all our analyses except for the outcome of MI in the RCTs (Egger test P = .02). It is difficult to interpret this in regard to MI, because there was no significant difference in MI likely due to lack of power.
Discussion
The present systematic review and meta-analysis were performed to assess the effects of preoperative aspirin therapy on the safety and efficacy outcomes in patients undergoing CABG. For safety outcomes, our analysis demonstrated that preoperative aspirin increased postoperative bleeding, transfusion requirements, and reoperation for bleeding, which were demonstrated by pooling the results of RCTs. In 19 observational studies, preoperative aspirin increased postoperative bleeding and PRBC transfusion requirements but not reoperation for bleeding. Subgroup analysis for RCTs by date of publication, sample size, and Jadad score yielded consistent results for postoperative bleeding, suggesting the robustness of the initial analysis. Sensitivity analysis for observational studies was performed excluding the study of Gulbins et al after subgroup analysis by date of publication and sample size in the analysis of postoperative bleeding. The pooled estimate and heterogeneity altered significantly before and after it was excluded, suggesting that the robustness of the subgroup analysis (for all the 6 recent observational studies with large sample sizes) was limited and the results were greatly influenced by Gulbins et al. We failed to completely eliminate the heterogeneity. The difference between Gulbins et al’s and the others accounted for a portion of heterogeneity that could not be removed by simply excluding the study of Gulbins et al. Further investigation demonstrated that doses of aspirin ≤100 mg/d did not appear to increase postoperative bleeding. However, only 2 eligible studies were included in our study, thus more studies should be pooled to further verify the conclusion. The dose of 325 mg/d may not be a cutoff value that has clinical and statistical significance. Furthermore, sensitivity analyses were performed excluding 3 studies that reported aspirin doses more than 2000 mg/d for the RCTs and observational studies, respectively, because such high doses would be very likely to increase postoperative bleeding and are no longer used in clinical practice for antiplatelet therapy. We showed that despite the differences in effect sizes, the results were consistent before and after excluding the study of Fuller et al for the RCTs and that of Michelson et al and Torosian et al for the observational studies, respectively, suggesting the robust stability of our initial results. For efficacy outcomes, no difference in the rates of perioperative MI, CVAs, and mortality was statistically significant between the aspirin and the control groups. We noted publication bias for the outcome of MI in the RCTs. It is perplexing, and asymmetry among the RCTs was probably due to the absence of enough studies and the fact that Egger regression test has been challenged because of its high type I error rates (the proportion of false-positive results) when ORs are used. 46
Despite substantial advances in surgical technique in the recent years, complications of CABG continue to be troubling. Aimed at reducing the risks of MI and death from coronary artery disease, CABG correlates with its own risks of major adverse ischemic events (such as MI and CVAs). 2 Although its administration immediately before CABG may reduce thrombotic events and improve postoperative outcomes, the beneficial effects of preoperative aspirin may be counteracted by a competing risk of excessive intraoperative and postoperative bleeding. 47 Preoperative aspirin therapy for CABG is certainly one of the most important issues to be investigated in cardiovascular surgery because of disparate guidelines from professional organizations, conflicting practices of cardiac surgeons, and inconsistent results from the clinical trials.
It has long been established that the antiplatelet effects of aspirin are primarily mediated by its ability to irreversibly inhibit platelet cyclooxygenase 1 (COX-1) and cyclooxygenase 2 (COX-2), thereby impairing platelet aggregation. 47 Inhibition of COX-2 at higher aspirin doses may favor prothrombotic events, but it is 170-fold more potent in inhibiting COX-1 than COX-2. 48 Endothelial injuries of grafts resulting from operative procedures and utilization of cardiopulmonary bypass technology are believed to increase the risks of thrombotic events in CABG. 49,50 Within 1 month after surgery, 3% to 12% of saphenous vein grafts occlude, which is associated with 9% of mortality. 49,51 Early postoperative aspirin has been shown to, short-term and long-term, prevent graft occlusion, reduce ischemic events, and improve survival. 2 There is class 1A evidence that early aspirin administration after CABG reduces thrombotic saphenous vein grafts occlusion as well as overall morbidity and mortality. The AHA/ACC and the European Association of Cardiothoracic Surgeons recommend that 150 to 325 mg aspirin be administered within 6 hours following CABG. 3 In addition, aspirin continues to be effective in preventing vein-graft occlusion late after the operation. 52 Preoperative aspirin therapy might also be beneficial for patients with perioperative CABG. Furthermore, recent evidence has suggested that sudden withdrawal of long-term antiplatelet therapy may initiate a prothrombotic state, leading to excess risks of ischemic events perioperatively. 8,53 This is the most common case for those awaiting elective CABG. However, it remains to be clearly determined whether the benefits of preoperative aspirin may outweigh the hazards of excessive bleeding attributable to its use prior to CABG. Low doses of aspirin have been suggested to be as effective as higher doses for antiplatelet therapy while minimizing the risk of bleeding. A daily dose of 100 mg is commonly recommended for primary prevention of coronary artery disease. 1 Our analysis demonstrated that a daily dose of ≤100 mg before CABG may not be associated with an excess of bleeding. Sun et al 9 demonstrated that doses < 325 mg/d before CABG did not appear to increase postoperative bleeding. However, the effects of low-dose preoperative aspirin on patients with CABG need to be further clarified. Our meta-analysis suggested that preoperative aspirin was associated with increased transfusions in RCTs and observational studies and increased reoperation for bleeding in RCTs. Moreover, excessive postoperative bleeding leads to postoperative hypovolemia and hypotension, postoperative tamponade, and higher rates of blood transfusion. 54 –56 However, it is not well established that an increase in postoperative bleeding of around 200 mL is clinically relevant because of a lack of solid evidence. Preoperative aspirin did not increase reoperation for bleeding in the observational studies, and moreover, there was heterogeneity that we failed to eliminate for the results of transfusions. Thus, the clinical relevance of increased postoperative drainage in the aspirin groups with regard to some significant outcomes remains to be further elucidated.
In the past 30 years, many great advances in CABG surgery have taken place, and their links to our end points should be taken into consideration. As we included historical studies conducted for more than 30 years, it was critical to distinguish those outdated studies of low quality. Hence, we carried out subgroup analyses by Jadad score (for RCTs only), date of publication, and sample size in order to not only explore the effects of these factors on heterogeneity but also pool those relatively high-quality studies to obtain more robust estimates of our end points. A 2009 study by Gulbins et al 15 reviewed a sum of more than 10 000 patients with CABG from 1995 to 2007 and showed that preoperative aspirin therapy did not seem to influence the postoperative bleeding. The authors did not provide us with detailed information regarding aspirin doses. All patients received antifibrinolytic therapy using aprotinin, which might explain why postoperative bleeding did not differ between the 2 groups. In our analysis, postoperative bleeding was shown to be increased in the aspirin groups in both early and late era, but the effect size of the latter was smaller. The use of blood conservation strategies and improvements in cardiopulmonary bypass technology, which lessens the damage to cellular components, may account for such a difference. 10 In addition, we failed to resolve the problem of heterogeneity when we analyzed the results of transfusion requirements. The lack of homogeneity could be attributed to interhospital differences in the use of blood products and adherence to transfusion protocols. 57,58 Moreover, Dacey et al 13 and Bybee et al 12 showed lower risks of fatal outcomes or mortality in the preoperative aspirin groups. Mikkola et al 14 demonstrated that preoperative aspirin lowered the risk of postoperative stroke. However, our analysis had insufficient power to evaluate the effects of preoperative aspirin on the efficacy end points.
Inhibition of platelet activity by aspirin is not always achieved, and recent evidence has noted a wide variability in the response to aspirin. Residual platelet reactivity or aspirin resistance, measured by platelet function tests after aspirin administration, might be an important risk factor for adverse ischemic events in CABG. 59 Inversely, a pronounced response to aspirin potentially places perioperative patients at an excess risk of bleeding. 55 Although still in the early stage of research and development, individually tailored dose-dependent aspirin therapy based on platelet function tests is likely to be clinically employed in the near future. 60 Since the reported incidence of aspirin resistance varied widely in the literature, the lack of platelet response to aspirin quantification may constitute a major drawback of current RCTs and observational studies, and it is critical to adjust for this variable in the future trials. The duration of aspirin effects is also individually variable. Therefore, most studies had inherently diluted intergroup comparisons and reduced capacity to define safety and efficacy outcomes of preoperative aspirin when dichotomizing their participants into early and late discontinuation groups using a cutoff value of 3, 5, or 7 days. 61 Another confounder we wish to point out is the administration of other antiplatelet or antifibrinolytic drugs in patients with CABG. Intraoperative use of antifibrinolytics has been revealed to significantly reduce postoperative blood loss in cardiac surgeries, and it might lower or even eliminate the bleeding risk of preoperative aspirin. 62 Dual antiplatelet therapy is commonly prescribed for a portion of patients awaiting CABG, especially ones receiving percutaneous coronary intervention before surgery. 63 Thus, inclusion of studies either using antifibrinolytic drugs or not reporting detailed data for use of drugs rather than aspirin possibly has the potential for confounding.
In 2012, Deja et al 29 reported a single-center, double-blind RCT involving patients undergoing first-time elective CABG. The patients were assigned to receive either 300 mg of aspirin (n = 387) or placebo (n = 396) the night before the coronary bypass surgery. The trial revealed that performing coronary grafts on aspirin is associated with increased postoperative bleeding but may decrease the long-term hazard of coronary events. Our analysis was consistent with the results from Deja et al with regard to postoperative bleeding. However, high-level evidence regarding preoperative aspirin administration in patients with CBAG is poorly sourced and currently limited to observational studies with a few RCTs. A lack of high-quality prospective studies may account for the divergent opinions on preoperative aspirin in CABG at present. Sun et al and Alghamdi et al called for high-quality prospective trials to assess the association between preoperative aspirin therapy and important postoperative outcomes in the settings of contemporary cardiac surgical practice. 9,10
Limitations
There are several limitations to our meta-analysis. First, we pooled the results of RCTs and observational studies in the analysis of CVAs and MI. Although there has been controversy regarding pooling the results of different study designs, we noted no significant difference caused by study design. Second, inclusion of observational studies inevitably introduced a source of potential bias that inherently exists in nonrandomized, unblinded design. Third, differences between treatment groups in the doses of preoperative aspirin, use of postoperative aspirin, perioperative antifibrinolytics, and other antiplatelet drugs have been several confounders. Fourth, individual variability in response to aspirin and the duration of aspirin may dilute any effects of preoperative aspirin on our end points. Fifth, we failed to eliminate the heterogeneity in the analysis of transfusion requirements. Sixth, included studies were underpowered to determine significant difference in our efficacy outcomes. Finally, our studies did not have an analysis of only on-pump CABG, thus the risk benefit is unclear for this group.
Conclusions
Preoperative aspirin therapy increases postoperative bleeding, PRBC transfusion requirements, and reoperation for bleeding in patients undergoing CABG, which may be resolved by doses ≤100 mg/d. High-quality RCTs are required to obtain conclusive results for the effects of preoperative aspirin therapy on the safety and efficacy end points in patients with CABG.
Footnotes
Authors’ Note
The study was performed at the School of Medicine, Shandong University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
