Abstract
Background:
Several clinical trials have documented clinical benefits associated with prophylactic corticosteroid administration at the time of coronary artery bypass graft (CABG) surgery, including a reduction in the risk of atrial fibrillation and hospital length of stay. Despite the published data, the extent to which providers have adopted the perioperative use of corticosteroids remains unknown.
Objectives:
To assess temporal trends, between-hospital variation, and determinants of perioperative intravenous corticosteroid use during CABG surgery.
Methods:
We identified all patients admitted for CABG surgery in the Premier Healthcare Database (2003-2014), a large US-based inpatient database. We determined the proportion of patients administered prophylactic corticosteroids on the day of CABG surgery. Linear time-series models were used to estimate the rate and trend of corticosteroid use over time. Separate multivariable generalized estimating equation models were used to quantify the variation in and determinants of perioperative corticosteroid use.
Results:
Of 401 788 eligible patients who underwent a CABG surgery between 2003 and 2014, 20% (n = 80 681) were administered intravenous prophylactic perioperative corticosteroids (methylprednisolone, dexamethasone, or hydrocortisone). Corticosteroid use increased from 17.5% in 2003 to 22.6% in 2014 (annual rate = 0.42%; P < .001). Individual hospitals accounted for >50% of variation in corticosteroid use. High between-hospital variation was also observed, and the probability of utilization was ≥32.4% in the upper versus ≤3.4% in the bottom quartiles of hospitals.
Conclusion:
Prophylactic corticosteroid administration during CABG has increased gradually since 2003. To further evaluate the risk–benefit trade-off associated with their use, we believe a large-scale outcomes study is warranted to assess this highly variable practice.
Introduction
Nearly half a million coronary artery bypass graft (CABG) surgery procedures are performed annually in the United States. 1 Cardiopulmonary bypass and injury related to ischemia and reperfusion in cardiac surgery can induce a severe systemic inflammatory response. 2,3,4 This inflammatory process is associated with an extensive release of pro- and anti-inflammatory cytokines 5,6 and can lead to respiratory and multiple organ failure. 7,8
The prophylactic administration of corticosteroids has been studied and debated for more than 3 decades as a means to attenuate the acute phase response in cardiac surgery. 9,10 Corticosteroids have potent anti-inflammatory effects and have been shown to reduce the release of pro-inflammatory cytokines, inhibit macrophage production, and have favorable effects on C-reactive protein. 11,12 Existing evidence suggests that corticosteroids during cardiac surgery significantly reduce the risk of atrial fibrillation and shorten hospital length of stay, without increasing the risk of infection or other complications. 13 Importantly, corticosteroids increase blood sugar levels through gluconeogenesis, alter fluid and electrolyte balance, and suppress the immune system. 14,15 Moreover, the impact of corticosteroids on perioperative mortality is unclear, and confirmation regarding the safety of their perioperative use in cardiac surgery is lacking. 10,16 In 2004, the American College of Cardiology/American Heart Association (ACC/AHA) guidelines for CABG supported the “liberal prophylactic use” of corticosteroids for the reduction of systemic inflammation among patients undergoing CABG, especially for those receiving on-pump CABG with cardiopulmonary bypass (ie, extracorporeal circulatory support). 17 However, the most recent update, published in 2011, no longer recommended prophylactic corticosteroids. 18
There are currently no national data available regarding the trends and patterns of utilization of perioperative prophylactic corticosteroids. Furthermore, it remains uncertain as to which factors influence the administration of corticosteroids during CABG surgery. Therefore, we sought to assess trends in the use of perioperative intravenous corticosteroids in CABG surgery in the United States and to identify patient- and hospital-level factors that determine their use. We aimed to explore between-hospital variation in the prophylactic perioperative administration of intravenous corticosteroids.
Methods
Data Source and Cohort Selection
We generated a cohort of patients who underwent CABG surgery (both on-pump and off-pump) using 2003 to 2014 data from the Premier Healthcare Database. This database captures data from over 700 geographically diverse hospitals and health systems in the United States, including community and teaching hospitals in both rural and urban settings. A large inpatient database, Premier Healthcare Database contains information on ∼1 in 6 hospitalizations in the United States, including data for ∼148 million patients. 19 The database contains a record of daily charges for all medications, procedures, and diagnostic tests during each patient’s hospitalization, as well as patient demographic and hospital characteristics, discharge diagnoses, and discharge status.
Figure 1 is a schematic of how patients were selected for analyses. The analyses included all patients who underwent CABG surgery. To focus on patients who received corticosteroids prophylactically, rather than for the treatment of other preexisting conditions, we excluded those who received oral or inhaled corticosteroids on any hospital day prior to or on the day of CABG surgery and those who received intravenous corticosteroids on any hospital day prior to surgery.

Sample selection of patients undergoing coronary artery bypass graft (CABG) surgery.
Outcome
The study outcome was intravenous corticosteroid use on the day of surgery, which was ascertained through hospital charge codes. For the time trend analysis, we calculated the proportion of CABG surgeries in which patients received intravenous corticosteroids on the day of surgery. We calculated yearly proportions both overall and separately for each intravenous corticosteroid. For the utilization prediction analysis, we modeled intravenous corticosteroid use as a binary outcome.
Study Variables
We ascertained both patient- and facility-level characteristics using a combination of International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) discharge diagnosis codes, procedure codes, and hospital charge codes. All charge codes used to define covariates were assessed in the period between initial hospitalization and the date of CABG surgery (inclusive). All patient characteristics, demographic information, preexisting medical conditions, medical procedures, and medication use were defined based on available preoperative data, except for valve repair/replacement and on-/off-pump procedures, which were assessed on the day of CABG. The hospital-level characteristics included number of hospital beds, regional location, teaching hospital status, and whether the hospital was located in a rural or urban area. A full list of study variables is provided in Table 1. All charge and ICD-9 codes are provided in Appendix A.
Distribution of Patient- and Hospital-Level Characteristics by Perioperative Intravenous Corticosteroid Administration Status.

Abbreviations: ACE, Angiotensin converting enzyme; ARB, Angiotensin II receptor blocker; CABG, coronary artery bypass graft; ICU, Intensive care unit; IV, intravenous.
aRhythm control drugs: amiodarone, dronedarone, sotalol, procainamide, or propafenone.
Statistical Analysis
Statistical analyses were performed to describe the preoperative characteristics of patients who did and did not receive corticosteroids, to evaluate temporal trends in perioperative corticosteroid use, and to explore sources of variation in perioperative corticosteroid use. Analyses were performed with SAS v 9.4 (SAS Institute Inc, Cary, North Carolina) statistical software.
Descriptive and temporal trend analysis
We characterized those who did and did not receive corticosteroids based on baseline demographics, clinical conditions, preexisting chronic conditions, perioperative corticosteroid use, and hospital-level characteristics. Trends in annual utilization of corticosteroids were evaluated visually with time-series plots of the proportion of CABG patients who received these agents prior to surgery. Linear time-series analyses were used to model annual rates of perioperative intravenous corticosteroids utilization between 2003 and 2014. Specifically, an autoregressive model was used to correct for potential first-order autocorrelation among annual data points, since we modeled aggregated intravenous corticosteroid use per yearly intervals. The models were further stratified by geographical region to assess trends in the perioperative corticosteroid administration by region.
Between-hospital variation
We plotted the probability of intravenous corticosteroid administration (percentage of CABG patients administered corticosteroids) across hospitals. Multilevel hierarchical models were 20 used to quantify the proportion of variation in intravenous corticosteroid use explained by the clustering of patients within hospitals as well as to identify patient- and hospital-level factors associated with intravenous corticosteroid use. Specifically, these models were built with multivariable generalized linear mixed models (PROC GLIMMIX function in SAS v 9.4) with a binary indicator of intravenous corticosteroid use as the dependent variable and hospitals as the clustering variable. Models were built in the following sequential fashion to quantify the incremental explanatory effects of patient- and hospital-level factors: model 1: hospital (random effects intercept) plus calendar year of intravenous corticosteroid use (random effects); model 2: model 1 plus patient demographics (fixed effects); model 3: model 2 plus patient clinical characteristics (fixed effects); model 4: model 3 plus hospital-level factors (fixed effects). The intracluster correlation coefficient (ICC), a measure of how much of the total variation in the probability of intravenous corticosteroid use was accounted for by the hospitals, was estimated from each model as: (intercept variance estimate)/(intercept variance estimate + 3.29 [residual variance]). 21,22 Additionally, we created caterpillar plots of the variation in intravenous corticosteroid use to visualize the extent to which each group of prespecified factors explained the observed variation.
Determinants of perioperative corticosteroid use
We estimated multivariable-adjusted odds ratios (ORs) for the association between intravenous corticosteroid use and each patient- and hospital-level characteristic in model 4 and described above.
Results
We identified and included 401 788 eligible patients who underwent CABG between 2003 and 2014. Of these, 80 681 were administered intravenous methylprednisolone (n = 39 291), dexamethasone (n = 26 705), or hydrocortisone (n = 14 685). We restricted our analyses to these 3 corticosteroids because <1% of patients were administered other intravenous corticosteroids. The median ages of both who received or did not receive intravenous corticosteroid were similar at 66 years (interquartile range: 58-74 years). Table 1 shows patient- and hospital-level characteristics for those who did and did not receive intravenous corticosteroids.
Temporal Trends in Intravenous Corticosteroid Administration
The proportion of patients administered methylprednisolone, dexamethasone, or hydrocortisone intravenously on the day of CABG surgery increased from 17.5% in 2003 to 22.6% in 2014 at an annual rate of 4.2 additional exposed patients per every 1000 CABG surgeries each year (annual rate = 0.42%; 95% confidence interval [CI]: 0.29%-0.55%; P < .001; Figure 2). This increase over time was largely driven by an increase in methylprednisolone use (annual increase = 0.46%; 95% CI: 0.36%-0.50%; P < .001). There was also substantial regional variation in corticosteroid use, with the highest and lowest observed in Midwest (23.6%) and the Northeast regions (13.2%), P < .001, respectively (Supplemental Figure 1).
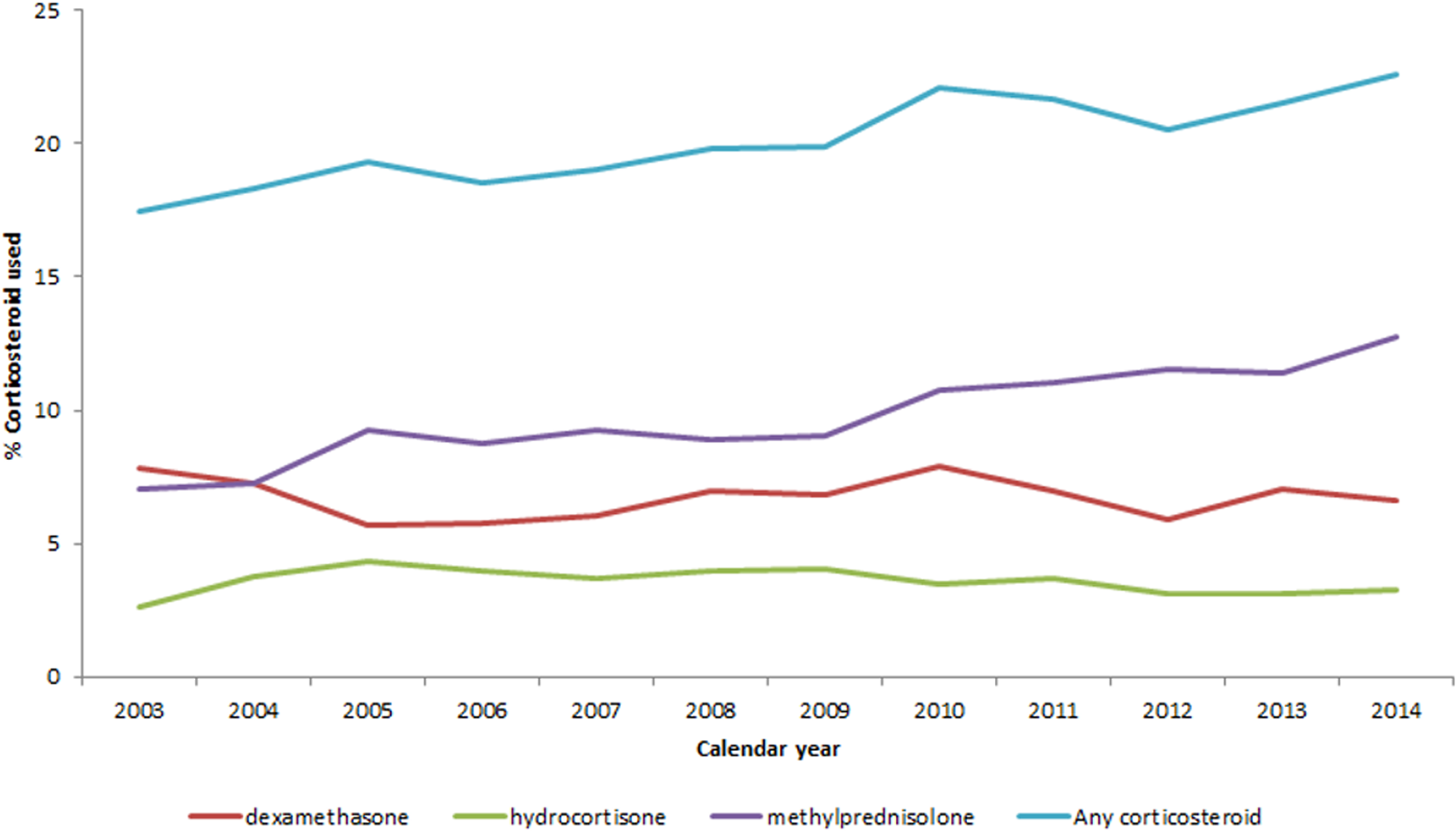
Perioperative intravenous corticosteroid utilization over time by total and individual medication use.
Determinants of Intravenous Corticosteroid Administration
Figure 3 shows the multivariable-adjusted OR and 95% CIs for the association between perioperative intravenous corticosteroid administration and preoperative patient- and hospital-level characteristics. Hispanic and black patients, as well as patients of other ethnicities, were more likely to be administered intravenous corticosteroids, as compared to white patients. Males were more likely to be administered these medications than females. The use of several medications during the perioperative period was predictive of intravenous corticosteroid administration: warfarin, statins, aspirin, fibrates, angiotensin-converting enzyme inhibitors, and β-blockers were positively associated with intravenous corticosteroid administration, whereas clopidogrel, heparin, proton pump inhibitors, aspirin/extended-release dipyridamole, digoxin, loop diuretics, vitamin K, heart rhythm control drugs, nicotine replacement therapy, and H2 blockers were inversely associated with intravenous corticosteroid exposure. Prior medical procedures such as oxygen use, valve repair/replacement, intensive care unit (ICU) stay, and previous PCI were inversely associated with use of intravenous corticosteroid. Patients undergoing CABG surgery in hospitals located in the South and Midwest (vs West) were more likely to be administered intravenous corticosteroids, while patients admitted to hospitals in urban areas (vs rural) were less likely to be administered an intravenous corticosteroid.

Determinants of perioperative intravenous corticosteroid use adjusted for patient clusters within hospitals.
Between-Hospital Variation in Intravenous Corticosteroid Use
Figure 4 shows that there was substantial variability in intravenous corticosteroid administration across the 503 hospitals where patients underwent CABG surgery. Among the upper quartile of hospitals with highest use of intravenous corticosteroid administration during CABG surgery, ≥32.4% of patients were administered an intravenous corticosteroid compared to no more than 3.4% of patients in the lower quartile. From the generalized linear mixed-effects models, hospitals alone, conditioned on calendar year, accounted for over half (ICC = 51%) of all observed variation in perioperative utilization of intravenous corticosteroids between 2003 and 2014. Plots of the variance in intravenous corticosteroid administration (Figure 5) show that the observed variability in intravenous corticosteroid administration attributable to hospitals did not change with the addition of patient demographics (ICC = 51%), clinical conditions (ICC = 52%), and hospital-level (ICC = 52%) characteristics. Hence, about half of the variation in intravenous corticosteroid administration between hospitals could not be explained by patient- and hospital-level characteristics.

Interhospital variation in perioperative intravenous corticosteroid utilization.

Panel plot of the proportion of variability in perioperative intravenous corticosteroid utilization explained by hospitals, patient demographic and clinical factors, and hospital characteristics.
Discussion
We observed a modest but steady rise in perioperative administration of intravenous corticosteroid among patients undergoing CABG surgery from 17.5% in 2003 to 22.6% in 2014. Our analyses found that prophylactic perioperative intravenous corticosteroid administration varied considerably across hospitals and by geographical location of these hospitals. Our study also provides insights into patient- and hospital-level characteristics that are significantly associated with the use of intravenous corticosteroids during CABG. However, patient characteristics appear to explain little variation in intravenous corticosteroid administration in CABG above and beyond variation across hospitals and geographic regions.
There may be several explanations for the increase in intravenous corticosteroid use over time. First, there was an increase between 2004 and 2005 that corresponded with the timing of the publication of the 2004 ACC/AHA guidelines for CABG, which recommended use of corticosteroids for the reduction of systemic inflammation. There was another increase between 2009 and 2010, which occurred following the publication of a meta-analysis of randomized trials by Whitlock et al, which suggested that corticosteroids reduced the incidence of atrial fibrillation and postoperative bleeding and reduced duration of ICU stay and length of total hospitalization. 10
Second, we observed differences in trends in intravenous corticosteroid administration across geographic regions. Notably, intravenous corticosteroid administration increased dramatically in the South between 2003 and 2014. Many studies have previously documented wide regional variation in use of surgery and other medical practices, even within fairly small geographic units, such as states. 18 Large differences in corticosteroid use across regions persisted even after accounting for hospital characteristics in multivariable regression models.
Third, about half of the proportion of variation in intravenous corticosteroid administration was accounted for by hospitals alone. Neither the addition of hospital-level nor patient-level characteristics explained much additional variation. A potential explanation for the observed variability in intravenous corticosteroid administration across hospitals may relate to institution-based differences in knowledge of, beliefs about, and preferences toward use of these medications in CABG. In the absence of clear guidelines about the role of intravenous corticosteroids in CABG, different hospitals may make different decisions on the basis of the same evidence in the literature. Providers in different hospitals likely also have different degrees of familiarity with guidelines and published literature. This may explain why the prevalence of intravenous corticosteroid administration was below 34.6% in three-quarters of the hospitals, even after the AHA/ACC endorsed the use of these medications in 2004. Differences in formularies could also explain across-hospital variation. Also, the individual providers can be an important source of variability when there is substantial clustering of patients within providers. For example, different providers could have different experiences with these drugs. However, we could not assess the role of individual surgeons, anesthesiologists, or care teams on corticosteroid use. Other unobserved hospital-level traits, such as institutional practice culture, could also have accounted for the residual unexplained variation in intravenous corticosteroid use. Future studies should explore these factors when assessing variability in perioperative intravenous corticosteroid use across hospitals.
Our findings are strengthened by the large sample of patients undergoing CABG surgery drawn from 503 hospitals across the United States during a 12-year period. This makes our findings generalizable to patients undergoing CABG surgery in the United States. We were able to show that hospitals are a major determinant for perioperative corticosteroid use. This information is important and needs to be factored into the design of future studies evaluating the effect of corticosteroid use on health outcomes. Using inpatient data captured during the perioperative period allowed us to accurately capture prophylactic use of methylprednisolone, dexamethasone, and hydrocortisone around the time of CABG surgery. We were also able to measure and examine a large number of potential predictors of corticosteroid use.
Our findings are limited by the unavailability of some important potential sources of variation in perioperative intravenous corticosteroid, such as formulary and prescriber preferences. The Premier Healthcare Database does not contain information on the reason for perioperative corticosteroid use of these corticosteroids. Theoretically, it is possible that some of the observed intravenous corticosteroid use may have been for underlying clinical issues. Also, given that we tested for statistical significance for the associations between several potential predictors and perioperative corticosteroid administration, it is possible that some of the observed statistically significant associations were due to chance. Finally, the Premier database contains only inpatient data. As such, we were not able to fully capture comorbid conditions that may have been present upon admission but were not recorded among the discharge diagnoses.
Conclusion
Perioperative corticosteroid use among patients undergoing CABG surgery in the United States increased steadily between 2003 and 2014. Utilization differed by geographic region. Individual hospital was identified as the single major source of the variation, accounting for more than half of the observed variation. Nearly a quarter of patients undergoing CABG in the United States now receive perioperative intravenous corticosteroids. Given the substantial variation in practice patterns, we are currently conducting a large-scale study to assess the associations between the perioperative prophylactic use of corticosteroids and multiple postoperative safety end points.
Supplemental Material
Supplemental Material, Supplemental_data - Trends and Patterns of Corticosteroid Use During Coronary Artery Bypass Grafting Surgery in the United States
Supplemental Material, Supplemental_data for Trends and Patterns of Corticosteroid Use During Coronary Artery Bypass Grafting Surgery in the United States by Macarius M. Donneyong, Alexander Kulik, and Joshua J. Gagne in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Appendix A
List of Covariates

| Covariates | Codes (Charge/ICD-9 diagnosis) | Period Assessed |
|---|---|---|
| Diabetes drugs | CAP | |
| COPD drugs | CAP | |
| Antibiotics | CAP | |
| Date of hospital admission | ||
| Age | Day of CABG | |
| Sex | CAP | |
| Race | CAP | |
| Marital status | CAP | |
| Region | CAP | |
| Year | Day of CABG | |
| Indicator for hospital day of CABG | Day of CABG | |
| Hospital ID | Day of CABG | |
| IV hydrocortisone | Day of CABG | |
| IV methylprednisolone | Day of CABG | |
| IV dexamethasone | Day of CABG | |
| History of CABG | ICD-9 diagnosis = V4581 | CAP |
| Valve repair or replacement on day of surgery | Day of CABG | |
| On-pump surgery | Day of CABG | |
| Off-pump surgery | Day of CABG | |
| Chronic renal disease or end-stage renal disease (hemodialysis, peritoneal dialysis, erythropoietin, and darbepoetin) | CAP | |
| PCI (PTCA or stent) on the index admission | CAP | |
| Rhythm-control drugs (amiodarone, dronedarone, sotalol, procainamide, and propafenone) | CAP | |
| Calcium channel blocker | CAP | |
| β-blocker | CAP | |
| Thiazide diuretic | CAP | |
| Loop diuretic | CAP | |
| ACE inhibitor | CAP | |
| Angiotensin receptor blocker | CAP | |
| Warfarin | CAP | |
| IV or low-molecular-weight heparin (bemiparin, nadroparin, reviparin, enoxaparin, parnaparin, certoparin, dalteparin, and tinzaparin) | CAP | |
| Proton pump inhibitors (charge codes available in spreadsheet) | CAP | |
| H2 blockers (charge codes available in spreadsheet) | CAP | |
| Vitamin K (charge codes available in spreadsheet) | CAP | |
| Aldosterone antagonist | CAP | |
| Digoxin | CAP | |
| Aspirin | CAP | |
| Clopidogrel | CAP | |
| Dipyridamole | CAP | |
| Statin | CAP | |
| Fibrate | CAP | |
| Corticosteroid | CAP | |
| Telemetry | CAP | |
| Echocardiogram | CAP | |
| Oxygen use | CAP | |
| Intensive nursing care | CAP | |
| Intensive physician visits | CAP | |
| Nicotine replacement therapy | CAP | |
| Varenicline | CAP |
Abbreviations: ACE, angiotensin-converting enzyme; CABG, coronary artery bypass graft; CAP, covariates assessment period; COPD, chronic obstructive pulmonary disease; ICD, International Classification of Diseases; IV, intravenous; PCI, percutaneous intervention; PTCA, percutaneous transluminal coronary angioplasty.
aCovariates assessment period was defined as the period between hospital admission date to date of CABG (inclusive). Also assess and define covariates occurring only on day of CABG.
Authors’ Note
Dr Gagne is principal investigator of an investigator initiated, unrestricted research grant from Novartis Pharmaceuticals Corporation to the Brigham and Women’s Hospital for unrelated work.
Author Contributions
M. Donneyong contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; and gave final approval. A. Kulik contributed to design, contributed to interpretation, drafted the manuscript, critically revised the manuscript, and gave final approval. J. Gagne contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; and gave final approval. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Gagne is a consultant to Aetion, Inc and to Optum, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
