Abstract
Myocardial ischemia is one of the main causes of sudden cardiac death. Autophagy has been demonstrated to protect cardiomyocytes from ischemia/reperfusion (I/R)-induced damage. A small molecule compound 5-(3-(4-(2-(4-fluorophenyl)ethoxy)phenyl)propyl)furan-2-carboxylic acid (D942) has been previously shown to specifically activate adenosine monophosphate-activated protein kinase (AMPK) in cancer cells. Another reagent, curcumin, has been shown to inhibit mammalian target of rapamycin (mTOR) signal pathway in tumor cells. Since AMPK signaling induces autophagy, while mTOR signaling inhibits autophagy, here we tested the potential protective efficacy of D942 with curcumin for cardiomyocytes under oxygen-glucose deprivation and reoxygenation (OGD/R). Mouse neonatal cardiomyocytes were treated with D942 and curcumin after being subjected to OGD/R. Cell survival and autophagy-related signal pathways were measured after treatment. Our data indicated both D942 and curcumin enhanced cell survival after OGD/R. The D942 and curcumin induced autophagy in cardiomyocytes through activating AMPK pathway or inhibiting mTOR signaling. Induction of autophagy by D942 and curcumin was the cause of cardioprotection, since inhibition of autophagy abolished the protective efficacy. Furthermore, combination treatment with D942 and curcumin profoundly upregulated autophagy after OGD/R and significantly promoted cell survival. Treatment with D942 and curcumin significantly upregulated autophagy in a murine myocardial I/R model. Taken together, our research suggests that D942 and curcumin could be promising therapeutic agents for myocardial I/R.
Introduction
Myocardial ischemia has been one of the main causes of sudden cardiac death during the past decades. Minimizing ischemia/reperfusion (I/R)-induced injury in cardiomyocytes has appropriately received a great deal of attention owing to the lack of regeneration of cardiomyocytes. The mechanisms underlying I/R damage in cardiomyocytes are yet to be fully understood. Molecular and cellular events during and after I/R are complex representing the confluence of divergent biological pathways. Increased intracellular calcium and sodium, dissipation of mitochondrial membrane potential, free radical formation/reactive oxygen species, dysregulated nitric oxide metabolism, endothelial dysfunction, platelet aggregation and microembolization, immune activation, and apoptosis are all considered to contribute to postischemic injury. 1–3
Autophagy has been a research focus of cell biology for the past decade. Emerging evidence suggests that it is a stringently regulated procedure that plays roles in cell growth, differentiation, and homeostasis as well as in maintaining a balance between synthesis, degradation, and recycling of cellular products. 4,5 Recent evidence indicates autophagy is critical for maintaining homeostasis and functions of cardiac myocytes. 6,7 Furthermore, autophagy has been implicated in enhancing cardiac myocyte survival after I/R. 8,9 Autophagy is a strictly regulated biological process. Although several signaling pathways have been shown to be involved in autophagy regulation, 2 signaling events are believed to be key modulators, the adenosine monophosphate-activated protein kinase (AMPK) pathway and mammalian target of rapamycin (mTOR) pathway. The AMPK plays a key role as a master regulator of cellular energy homeostasis. The kinase is activated in response to stresses that deplete cellular adenosine-5′-triphosphate (ATP) supplies such as low glucose, hypoxia, ischemia, and heat shock. The AMPK signaling regulates autophagy directly through phosphorylation of Ulk1 or indirectly through inhibition mTOR signaling. 5,10 The mTOR acts as a negative regulator for autophagy by inhibiting Ulk1, which is critical for membrane nucleation. 5-(3-(4-(2-(4-Fluorophenyl)ethoxy)phenyl)propyl)furan-2-carboxylic acid (D942) is a selective AMPK activator and has been demonstrated to induce autophagy in human prostate cancer PC3 cells. 11 However, its effect on cardiomyocytes has never been studied. Since AMPK signaling potently regulates autophagy and metabolism under nutrient insufficiency conditions, we speculated that it could be beneficial to protect cardiomyocytes from I/R damage. Curcumin is a major active component of turmeric Curcuma longa. Modern scientific research has demonstrated its anti-inflammatory, antioxidant, anticarcinogenic, antithrombotic, and cardiovascular protective effects. 12,13 Curcumin prevents and reverses murine cardiac hypertrophy. 14 It also promotes cardiac repair and ameliorates cardiac dysfunction following myocardial infarction. 15 Recently, its capability of inducing autophagy has been considered one of the mechanisms underlying its anticancer effects. 16 However, its effect on cardiomyocytes is still unclear. Other studies have demonstrated the inhibitory effect of curcumin on α-serine/threonine kinases (Akt)/mTOR signaling in several tumor cell lines. 17–19 Curcumin is able to dissociate raptor from mTOR, leading to inhibition of mTORC1 activity. 17 Since mTOR signaling is believed to negatively regulate autophagy, we speculated that curcumin could promote autophagy by blocking the mTOR pathway and subsequently could enhance cardiomyocyte survival after I/R.
In this study, we used both in vitro and in vivo models to test whether D942 and curcumin could protect mouse cardiomyocytes from ischemic damage. Our result indicates that either reagent is capable of enhancing cell survival after ischemia. Combination application of them has the most protective efficacy. Their protective effect is the consequence of autophagy induction via activation of AMPK pathway and inhibition of mTOR signaling. Taken together, our research could provide a novel approach for treating myocardial ischemia.
Materials and Methods
Mouse Primary Cardiomyocyte Culture and OGD/R
The protocol was approved by the Institutional Animal Care and Use Committee of Wuhan University. All animals received treatment in compliance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Research, National Research Council; National Academy Press, 1996).
Primary cardiomyocyte cultures were prepared from ventricles of 1-day-old C57BL/6 mice as described elsewhere with some modifications. Briefly, neonatal mice were anesthetized with pentobarbital sodium (50 mg/kg intraperitoneally). The hearts were removed aseptically, and the ventricles from 3 to 6 hearts were pooled on ice in Hanks’ balanced salt solution (HBSS) without Ca2+ and Mg2+ (137 mmol/L NaCl, 5.4 mmol/L KCl, 0.25 mmol/L Na2HPO4, 0.44 mmol/L KH2PO4, 4.2 mmol/L NaHCO3 and 10 mmol/L 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid [HEPES], pH 7.4). The ventricles were then washed 3 times with HBSS and minced into small fragments. Cells were dissociated at 25°C for 15 minutes with 0.5% trypsin in HBSS without Ca2+ and Mg2+. Cells released after the first digestion were discarded. Cells from subsequent digestions were added to an equal volume of cold HBSS with Ca2+ and Mg2+ until all cardiac cells were isolated. The mixture was centrifuged at 200g for 10 minutes, and the cells were resuspended in Dulbecco modified Eagle medium (DMEM)/Ham F-12 Nutrient Mixture (Sigma-Aldrich, St Louis, Missouri) supplemented with estrogen-free 5% fetal bovine serum (Hyclone, Rockford, Illinois), 100 U/mL penicillin, 100 μg/mL streptomycin, and 25 μmol/L cytosine arabinoside. To exclude nonmuscle cells, the isolated cells were first plated in tissue culture dishes at 37°C for 1 hour in an incubator with 5% CO2. Suspended cardiomyocytes were then plated at a density of 1.0 × 105 cells/cm2 and incubated under the same conditions as mentioned earlier for 24 hours.
For simulated ischemia, OGD medium, that is HBSS with Ca2 + and Mg2 + (137 mmol/L NaCl, 5.4 mmol/L KCl, 1.0 mmol/L MgSO4, 0.25 mmol/L Na2HPO4, 0.44 mmol/L KH2PO4, 1.3 mmol/L CaCl2, 4.2 mmol/L NaHCO3 and 10 mmol/L HEPES, pH 7.4) free of serum and glucose, was preequilibrated in 100% N2 at 37°C for 2 hours. Oxygenated medium was removed from the cardiomyocyte cultures, and the N2-preequilibrated glucose-free medium was immediately added. Cultures were promptly placed in a hypoxia chamber and exposed to 100% N2 for 1.5 hours at 37°C. For reoxygenation, the glucose-free medium was replaced with supplemented DMEM/Ham F-12, and the cells were reoxygenated in 21% O2–5% CO2–74% room air for 24 hours at 37°C.
D942 and Curcumin Treatment
The D942 was purchased from Millipore (Millipore, Billerica, Massachusetts). Stock solution was prepared by dissolving D942 in dimethyl sulfoxide (DMSO) at a concentration of 10 mmol/L and was stored at 4°C before use. For testing its cytotoxic or effective doses, the stock solution was diluted with cardiomyocyte culture medium to reach indicated concentrations immediately before treatment. Twenty-four hours after seeding cells, the culture medium was discarded, and the cells were gently rinsed once with phosphate-buffered saline (PBS). The D942-containing culture medium was then added into each well and incubated for 24 hours.
Curcumin was purchased from Sigma-Aldrich. Stock solution was prepared by dissolving curcumin in DMSO at a concentration of 10 mmol/L and was stored in a dark-colored bottle at −20°C. For testing its cytotoxic or effective dose, the stock solution was diluted with cardiomyocyte culture medium to required concentrations immediately before treatment. Twenty-four hours after seeding cells, the culture medium was discarded, the and cells were gently rinsed once with PBS. The curcumin-containing culture medium was then added into each well and incubated for 24 hours.
For treating cells after OGD, the cells were first subjected to OGD as described previously. The D942 and curcumin were diluted in supplemented DMEM/Ham F-12 before use. Then, OGD-treated cells were incubated in D942- and/or curcumin-containing medium in 21% O2–5% CO2–74% room air for another 24 hours at 37°C.
In some experiments, the AMPK inhibitor compound C (Sigma-Aldrich) or phosphatidylinositol-3-kinase (PI3K) inhibitor pervanadate was used at indicated concentrations in the presence of D942 or curcumin to treat cells.
For in vivo induction of autophagy, 200 mg/kg D942 and 100 mg/kg curcumin were injected into 8 to 12-week-old male C57BL/6 mice via tail vein once a day for 3 consecutive days. Then, the mice were sacrificed by CO2 inhalation, and hearts were collected and homogenized in radio immunoprecipitation assay (RIPA) buffer with homogenizers. Tissue was lysed in RIPA buffer on ice for 30 minutes, and lysates were stored in −80°C until analysis.
Cell Viability Assay
Cell viability assay was performed using calcein-acetoxymethyl ester (calcein-AM) (R&D Systems) following the manufacture’s instruction. Briefly, cells were grown in 96-well plates before the assay. The OGD/R and treatment with D942 and/or curcumin were performed as described previously. The culture medium was carefully discarded, and the cells were gently washed with PBS twice. Calcein-AM at 1 µmol/L was prepared by diluting calcein-AM in prewarmed PBS and was added into each well and incubated for 30 minutes at 37°C in an incubator before recording fluorescence on a Tecan Infinite 200 PRO (Tecan Systems, San Jose, California) plate reader.
Flow Cytometry
Oxygen-glucose deprivation and reoxygenationand treatment with D942 and/or curcumin were performed as described previously. Floating cells were collected after treatment. Adherent cells were incubated in 2 mmol/L EDTA–PBS at 37°C for 10 minutes. The cells were gently flushed with a pipette to dissociate cells from the bottom of wells and pool flushed cells with floating cells. Cells were then centrifuged and stained with propidium iodide (BD Biosciences, San Jose, California) and annexin V (BD Biosciences) according to the manufacture’s instruction. Briefly, the cells were washed with PBS twice and were incubated in 100 µL of binding buffer (10 mmol/L HEPES, pH 7.4; 140 mmol/L NaCl; 2.5 mmol/L CaCl2) containing annexin V-biotin (1:20) for 15 minutes at room temperature (RT) in the dark. Cells were then washed with PBS once followed by incubation in 100 µL of binding buffer containing 5 µg/mL propidium iodide and 5 µg/mL streptavidin–fluorescein isothiocyanate (BD Biosciences) for 15 minutes at RT in the dark. The 1× binding buffer of 400µL was added to each sample. Apoptosis was analyzed by flow cytometry within 1 hour. Apoptosis can be phenotypically divided into early apoptosis and late apoptosis based on cell surface expression of phosphatidylserine (PS) and cell membrane intactness. Early apoptotic cells have PS translocation but cell membrane is still intact, thus showing positive annexin V staining and negative propidium iodide staining. Plasma membrane intactness is compromised in late apoptotic cells, so they are both annexin V and propidium iodide positive.
Murine Myocardial I/R model
Male C57BL/6J mice at 8 to 10 weeks of age were used for the surgical procedure, which was performed according to the Guiding Principles in the Care and Use of Animals of the American Physiological Society and in accordance with the Guide for the Care and Use of Laboratory Animals. Mice were anesthetized with 2% halothane (Sigma-Aldrich) and 40% oxygen and maintained with 0.5% halothane and 40% oxygen. Tracheotomy was performed to provide artificial ventilation (0.3 mL tidal volume, 120 breaths/min), and the left coronary artery was ligated with an 8-0 nylon surgical suture 1.0 mm distal from tip of the left auricle. Sham-operated animals underwent the same surgical procedure except that the suture was not tied around the left anterior descending artery. After 30-minute ligation and 3-day reperfusion, hearts were removed for protein extraction.
Western Blot
Cell or tissue lysate was prepared by homogenization in RIPA buffer (20 mmol/L Tris–HCl, pH 7.5, 150 mmol/L NaCl, 1 mmol/L Na2EDTA, 1 mmol/L ethylene glycol tetraacetic acid, 1% nonidet P40, 1% sodium deoxycholate) and lysed on ice for 30 minutes. Protein amount was quantified using Pierce 660 nm Protein Assay (Thermo Scientific, Rockford, Illinois) following the manufacture’s instruction. Total protein from each sample was loaded onto 8% (for acetyl-CoA carboxylase [ACC]) or 10% (for extracellular signal-regulated kinase (Erk), AMPKs, B cell lymphoma 2 [Bcl-2], and β-actin) sodium dodecyl sulfate polyacrylamide gel electrophoresis gel, and electrophoresis was performed. For LC3B, proteins were loaded onto 4% to 20% Mini-PROTEAN TGX Precast Gels (Bio-Rad, Hercules, California). Proteins were then transferred onto nitrocellulose membranes and blocked with 5% nonfat dry milk-PBS for 1 hour at RT. After 3 washes with 0.05% Tween-20-PBS (PBST), the membranes were incubated with the following primary antibody reagents overnight at 4°C: (1) rabbit monoclonal anti-AMPKα, anti-phospho-AMPKα, anti-ACC and antiphospho-ACC, anti-Erk1/2 antiphospho-Erk1/2 (cell signaling) and (2) mouse monoclonal anti-Bcl-2 and anti-β-actin (Santa Cruz, Dallas, Texas). Membranes were washed 3 times with PBST and then incubated with horseradish peroxidase-conjugated secondary antibodies for 1 hour at RT. Membranes were developed with SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific), and the optical density of the identified protein bands on membranes was gained using a Biospectrum 500 imaging system (UVP, LLC, St Upland, California). To quantify the protein levels, the optical density of target proteins was normalized to that of β-actin of the corresponding groups. Normalized optical density of each group was then normalized to that of vehicle control group. To quantify the phosphorylation of signaling molecules, the optical density of phosphorylated proteins was normalized to that of corresponding native proteins in each group.
Statistical Analysis
Data were analyzed, and results were presented as mean ± standard deviation. Student t test or 1-way analysis of variance was used for comparison of mean between the groups, and P < .05 was considered significant.
Results
D942 and Curcumin Protect Cardiomyocytes From OGD/R-Induced Apoptosis
Cardiomyocytes are sensitive to I/R and undergo apoptosis due to insufficient blood flow or oxygen supply. Before exploring their potential protective effects, we first tested whether D942 and curcumin are cytotoxic to cells under normal culture conditions. Treatment of mouse cardiac myocytes with D942 and curcumin for 24 hours at indicated dose range did not increase either early or late apoptosis, suggesting that D942 or curcumin had little proapoptotic effect (Figure 1). In addition, as compared with untreated cells, D942 and curcumin did not reduce HT22 spontaneous apoptosis, indicating that D942 or curcumin was not effective in inhibiting spontaneous apoptosis (Figure 1). Then, we studied the effects of D942 or curcumin on OGD/R-induced apoptosis in cardiac myocytes. As predicted, OGD/R robustly increased both early and late apoptosis, while D942 and curcumin significantly inhibited cell death as well as promoted cell survival. Notably, combination treatment with D942 and curcumin was the most effective in inhibiting OGD/R-induced apoptosis and maintaining cell survival (Figure 2A and 2B). The increase in early apoptosis in the treated groups is probably not due to enhanced induction of early apoptosis but instead reflects blocked entry into late apoptosis and necrosis, since late apoptosis and necrosis were decreased and living cells were increased. Cell viability assay also confirmed the protective effects of D942 and curcumin (Figure 2C).

D942 and curcumin were not cytotoxic to cardiomyocytes. A, Cardiomyocytes were treated with D942 and curcumin at indicated concentrations for 24 hours. Apoptosis was evaluated by flow cytometry. This is the representative of 3 independent experiments. B, Statistical analysis of cardiac myocyte apoptosis. N = 3 in each group. D indicates D942; C, curcumin.

D942 and curcumin protected cardiomyocytes from OGD/R. A, Cardiomyocytes were subjected to OGD/R before treatment with D942 (10 µmol/L) and/or curcumin (10 µmol/L). Cell apoptosis was evaluated by flow cytometry. This is the representative of 3 independent experiments. B, Statistical analysis of cardiac myocyte apoptosis. C, Cell viability assay with Calcein-AM showed increased cell viability after D942 and/or curcumin treatment. Intensity of Calcein-AM of each group was normalized to that of control group. N = 3 in each group. **P < .01; ***P < .001 compared with OGD/R group. D indicates D942; C, curcumin; OGD/R, oxygen-glucose deprivation and reoxygenation; Calcein-AM, calcein-acetoxymethyl ester.
D942 and Curcumin Promote Autophagy in Cardiomyocytes
The D942 is a well-defined AMPK activator and has been shown to be able to induce autophagy in various cell types. Curcumin is a selective mTOR inhibitor and its ability to activate autophagy in cardiomyocytes has been determined elsewhere. Since autophagy has been previously shown to be protective for several cell types, including cardiomyocytes under nutrient-insufficiency conditions, we speculated that autophagy induced by D942 and curcumin could be a mechanism of protection after OGD/R. To confirm this hypothesis, we first asked whether D942 and curcumin can induce autophagy in our experimental settings. Cardiomyocytes were treated with D942 and/or curcumin under normal culture condition for 24 hours. The OGD/R was performed as a positive control, since it has been well documented as an autophagy inducer. After treatment, autophagic markers were detected by Western blot (Figure 3A). The D942 or curcumin significantly induced conversion of LC3B-I to LC3B-II under normal culture conditions, which is a marker for autophagosome formation. Beclin-1, which is also referred to as Atg6, is one component of a large molecular complex required for the initiation of autophagic vesicle formation. The D942 or curcumin significantly upregulated beclin-1 expression in cardiomyocytes. The p62, which is degraded upon stimulation of autophagy due to its role of targeting ubiquitinated proteins to the autophagic vacuoles, was decreased by D942 or curcumin. The OGD/R exposure also induced similar but weaker changes in the above-mentioned markers in comparison with D942 or curcumin treatment, suggesting either agent is more potent in evoking autophagy. It is noteworthy that combined treatment with D942 and curcumin after OGD/R induced the most significant changes in autophagic markers among all treated groups, suggesting combination of D942 and curcumin is the most potent autophagy activator under our conditions. Taken together, these data indicate that both D942 and curcumin are able to activate autophagy.

D942 and curcumin activated autophagy in cardiomyocytes. A, Cardiomyocytes were treated with D942 (10 µmol/L) and/or curcumin (10 µmol/L) for 24 hours. Conversion of LC3B, protein levels of beclin-1, and p62 were detected by immunoblotting. β-actin was used as internal control. B, Chloroquine inhibits autophagy induced by D942 or curcumin. Note that p62 level was decreased by D942 with curcumin, while chloroquine stabilized p62 level by inhibiting autophagy. C, Combination of D942 with curcumin induced higher autophagy after OGD/R. N = 4 to 5 in each group. *P < .05; **P < .01; ***P < .001. # P < .05; ## P < .01. V indicates vehicle; D, D942; C, curcumin; Ch, chloroquine; N, normal culture condition.
D942 and Curcumin Protect Cardiomyocytes after OGD/R Through Induction of Autophagy
To confirm the role of autophagy in D942 and curcumin-induced cardioprotection, we treated cardiomyocytes with D942 and curcumin in the presence or absence of chloroquine. Chloroquine has been shown to effectively inhibit autophagy by imparing lysosomal acidification and subsequent protein degradation. Chloroquine itself increased the levels of LC3B-II and p62 in comparison with vehicle control, due to inhibited degradation of these autophagic proteins. However, the beclin-1 level was not altered by chloroquine alone. Chloroquine effectively inhibited D942 and curcumin-induced reduction of p62, suggesting autophagy was indeed blocked by chloroquine (Figure 3B). To test whether D942 and curcumin can also induce autophagy after OGD/R, the cells were subjected to OGD/R followed by combination treatment with D942 and curcumin. The OGD/R itself is an autophagy inducer, demonstrated by higher LC3B-II and lower p62 levels compared with cells under normal culture conditions. Combination treatment induced an even higher level of LC3B-II and lower level of p62, in comparison with vehicle control, suggesting that this treatment triggered more autophagy in addition to OGD/R-induced autophagy (Figure 3C). Cell death evaluation after treatment showed that the protective effect of D942 and curcumin was significantly inhibited when chloroquine was applied (Figure 4). Taken together, these data suggest that the protective efficacy of D942 and curcumin is indeed mediated by induction of autophagy.
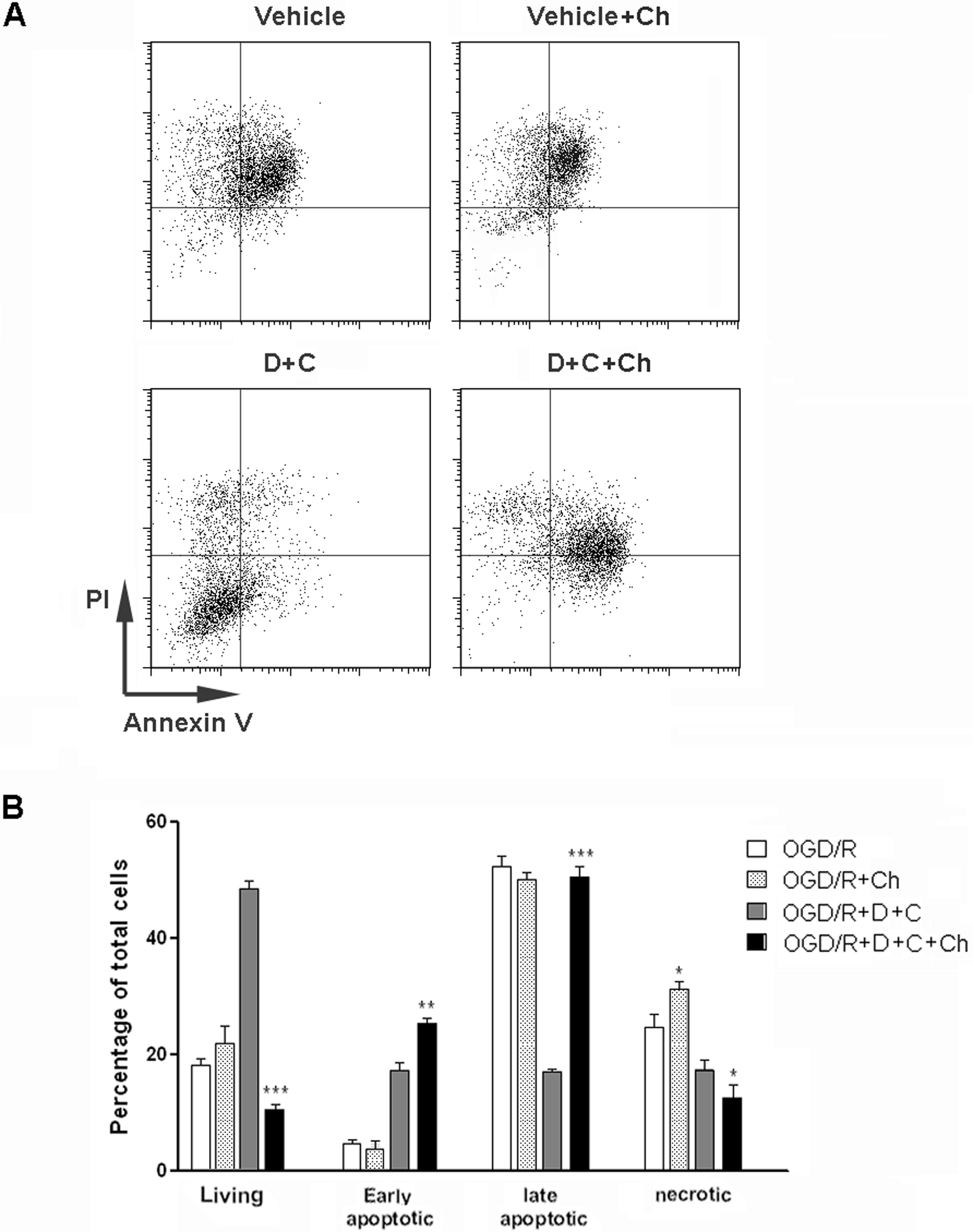
Chloroquine abolished cardioprotective effects of combination of D942 and curcumin. A, Cardiomyocytes were exposed to OGD/R before treatment with D942 (10 µmol/L) and/or curcumin (10 µmol/L). Cell apoptosis was evaluated by flow cytometry. This is the representative of 3 independent experiments. B, Statistical analysis of cardiac myocyte apoptosis. N = 3 in each group. *P < .05; **P < .01; ***P < .001 compared with OGD/R + D + C group. D indicates D942; C, curcumin; Ch, chloroquine; OGD/R, oxygen-glucose deprivation and reoxygenation.
D942 and Curcumin Activate AMPK and Inhibit mTOR Signaling in Cardiomyocytes
To confirm that AMPK and mTOR signaling are indeed influenced by D942 and curcumin, we then detected activation status of these signal pathways in treated cardiomyocytes by Western blot. Activating phosphorylation level of AMPKα was significantly higher in the presence of D942 than those in nontreated group and curcumin alone group. The ACC phosphorylation was also enhanced in the presence of D942, indicating promoted AMPK activity (Figure 5A). Meanwhile, both D942 and curcumin decreased phosphorylation of p70S6 kinase and eukaryotic translation initiation factor 4E-binding protein 1(4EBP1) at their activating sites, suggesting mTOR signaling is substantially inhibited after treatment. It is noteworthy that combination of D942 with curcumin induced a more robust decrease in activating phosphorylation of p70S6K and 4EBP1, suggesting the most potent inhibitory effect of combination treatment (Figure 5B).
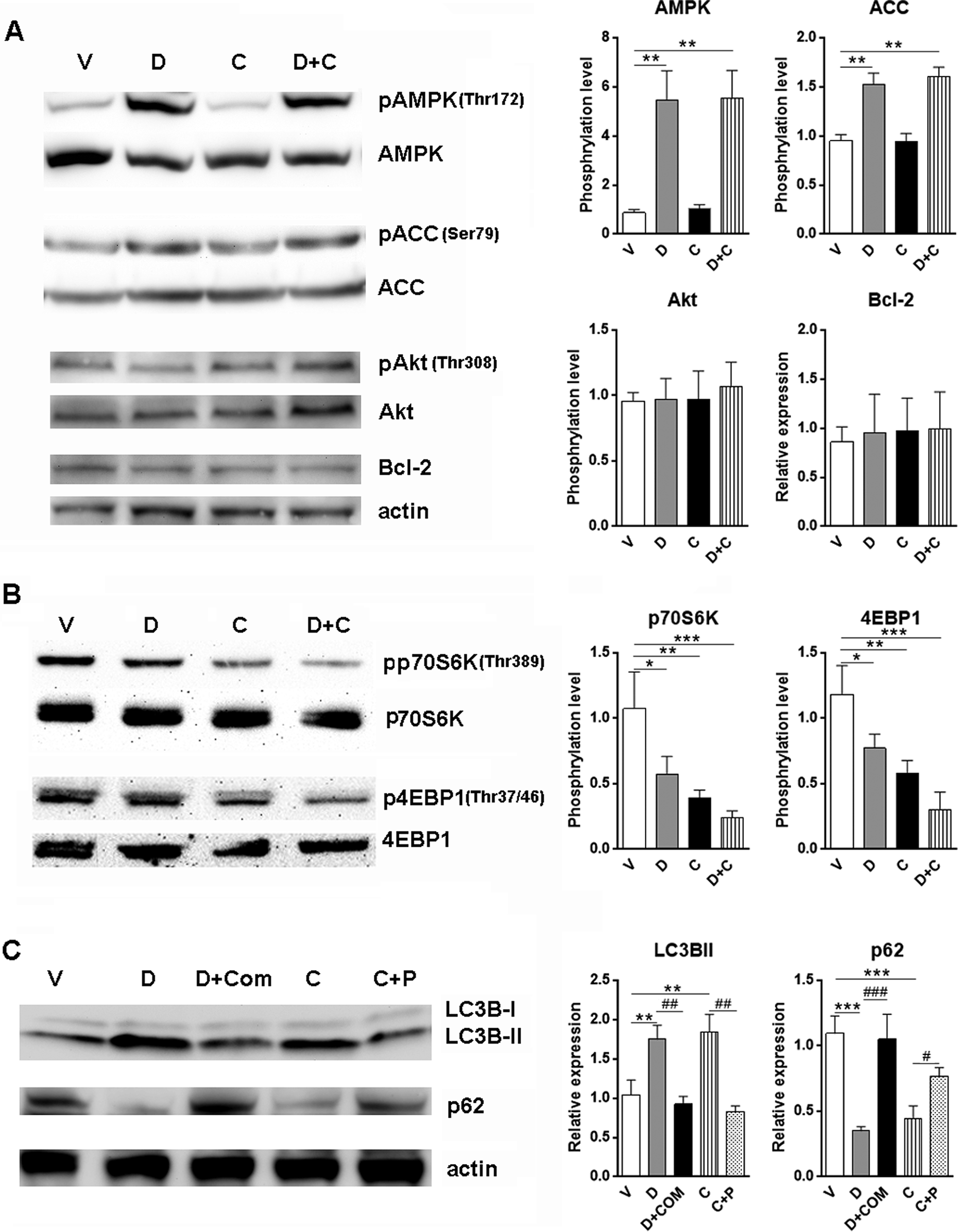
D942 and curcumin-induced autophagy in cardiomyocytes via AMPK and mTOR signal pathway. A, Cardiomyocytes were treated with D942 (10 µmol/L) and/or curcumin (10 µmol/L) for 24 hours. AMPK signaling, Akt signaling, and Bcl-2 expression were determined by immunoblotting. B, mTOR signaling was detected in cardiomyocytes treated with D942 and/or curcumin. C, Compound C and pervanadate were used to determine the roles of AMPK and mTOR signaling in inducing autophagy. N = 4 in each group. *P < .05; **P < .01; ***P < .001. # P < .05; ## P < .01. D indicates D942; C, curcumin; Com C, compound C; P, pervanadate; AMPK, adenosine monophosphate-activated protein kinase; mTOR, mammalian target of rapamycin; Bcl-2, B cell lymphoma 2; Akt, α-serine/threonine kinases.
To test whether other autophagy-related signal pathways are involved in D942 and curcumin-induced autophagy, we tested for activation of Akt and expression of Bcl-2 in cardiomyocytes. None of them was altered after treatment with D942 or curcumin, suggesting neither agent has impacts on these signal pathways (Figure 5A).
D942 and Curcumin Induce Autophagy Through AMPK and mTOR Signal Pathways
Both AMPK and mTOR signaling have been proven to be closely related to autophagy in multiple cell types including cardiomyocytes. To directly link AMPK and mTOR signaling to D942 and curcumin-induced autophagy, we used compound C, a specific AMPK inhibitor, to block AMPK signaling. We also used pervanadate, a protein tyrosine phosphatase inhibitor, to activate Akt-mTOR signal pathways. In the presence of compound C, LC3B-II conversion induced by D942 was significantly inhibited, along with stabilization of p62 protein level (Figure 5C). These data suggest AMPK signaling is indeed involved in autophagy induction by D942. Meanwhile, similar but less significant change was observed after curcumin treatment in the presence of pervanadate, suggesting mTOR signaling could be a key player in autophagy induction by curcumin (Figure 5C).
D942 and Curcumin Induce Autophagy in Murine Hearts in vivo
We speculated that D942 and curcumin can also induce autophagy in cardiomyocytes in vivo. To test this hypothesis, we intravenously (iv) injected D942 and/or curcumin into mice. Then autophagic markers in hearts were determined by Western blot (Figure 6). No significant abnormalities in the shape, size, and structure of the hearts were observed. Consistent with in vitro result, both D942 and curcumin increased LC3B-II conversion, while combination treatment induced the most robust conversion. The D942 promoted AMPKα phosphorylation whereas curcumin reduced 4EBP1 phosphorylation, suggesting their capability of activating AMPK signaling and inhibiting mTOR signaling, respectively. Taken together, these data indicate that D942 and curcumin are able to induce autophagy in murine hearts through AMPK and mTOR pathways.

In vivo induction of autophagy in murine hearts by D942 and/or curcumin. D942 and/or curcumin were intravenously injected into mice and AMPK signaling, mTOR signaling, and LC3B conversion were determined by immunoblotting. N = 5 in each group. *P < .05; **P < .01; ***P < .001 compared with the vehicle group. D indicates D942; C, curcumin; AMPK, adenosine monophosphate-activated protein kinase; mTOR, mammalian target of rapamycin.
D942 and Curcumin Promote Autophagy in Murine Hearts After I/R
To explore the potential use of D942 and curcumin to protect hearts from I/R-induced myocardial damage, we applied a mouse model of myocardial I/R and injected D942 together with curcumin from the time point of reperfusion. We found that D942 and curcumin induced a higher level of autophagy in comparison with vehicle control, demonstrated by higher level of LC3B-II and lower level of p62 (Figure 7). Taken together, our data suggest that combination treatment with D942 and curcumin could be therapeutical for myocardial I/R.

Combination treatment with D942 and curcumin reduced infarct size and induced autophagy in a murine myocardial ischemia/reperfusion model. Lysates of heart tissue were used in Western blot to evaluate the induction of autophagy after ischemia/reperfusion. N = 6 in each group. *P < .05; **P < .01 compared with vehicle group. V, vehicle; D + C, D942 + curcumin.
Discussion
Autophagy is a major catabolic pathway by which mammalian cells degrade and recycle macromolecules and organelles. It plays a critical role in removing protein aggregates, as well as damaged or excess organelles, to maintain intracellular homeostasis and to keep the cell healthy. 20 In the heart, autophagy occurs at low levels under normal conditions. However, this pathway is rapidly upregulated under environmental stress conditions. 21–23 Many studies have reported that the number of autophagosomes increases in the heart during ischemia/perfusion (I/R) in animal models. 24,25 Enhanced autophagy has also been observed in isolated cardiac cells, where exposure to hypoxia/reoxygenation caused an increase in the number of autophagic vesicles. Signals that induce autophagy in I/R include ATP levels and AMPK, hypoxia and Bnip3, calcium, reactive oxygen species and reactive nitrogen species, mitochondrial permeability transition pore opening, ER stress, and the unfolded protein response. The function of autophagy during I/R is still under debate, since in some studies autophagy has been demonstrated to play a cardioprotective role in the heart following myocardial infarction, while other studies suggest autophagy is detrimental as a cause or a contributor to cell death. 26,27 Some speculate that the beneficial or detrimental role of autophagy is a consequence of balance depending on the extent of autophagy. The mechanisms underlying the cardioprotection by autophagy include ATP maintenance, mitophagy, protein clearance, and nutrient recycling. Thus, many studies are focusing on looking for agents that can properly enhance autophagy during or after cardiac infarction.
Our research demonstrates that D942 and curcumin can effectively protect in vitro cultured cardiomyocytes after OGD/R. The D942 is a cell-permeable furancarboxylic acid compound that activates cellular AMPK activity by inhibiting mitochondrial complex 1 and thus elevating intracellular AMP level. The AMPK plays a key role as a master regulator of cellular energy homeostasis. The kinase is activated in response to stresses that deplete cellular ATP supplies such as low glucose, hypoxia, ischemia, and heat shock. The AMPK activation positively regulates signaling pathways that replenish cellular ATP supplies. It also stimulates catabolic processes such as fatty acid oxidation and glycolysis via inhibition of ACC and activation of PFK2. 28 In addition, it has been shown that AMPK signaling positively regulates autophagy by activating Ulk1 through phosphorylation of Ser 317 and Ser 777 or indirectly by inhibiting mTOR signaling. 29,30 Our data indicate that D942 enhances AMPK signaling and subsequently augments autophagy in cardiomyocytes. However, it is still not clear whether other mechanisms, such as AMPK-induced replenishment of cellular ATP or downregulation of gluconeogenesis are involved in the protective effects. Further investigation will be needed to thoroughly elucidate the effects of D942 on cardiomyocytes.
Curcumin is a major active component of turmeric C longa. It inhibits mTOR signaling and activates autophagy. The mTOR signal transduction is the key regulator of autophagy. High mTOR activity prevents Ulk1 activation by phosphorylating Ulk1 Ser 757. 31,32 The Ulk1 is one of the mammalian autophagy-initiating kinases. The Ulk1 activation and association with other molecules such as Atg13 and FIP200 initiate autophagic membrane nucleation. Some other signal pathways, for example PI3K/Akt signaling and MAPK/Erk1/2 signaling, influence autophagy indirectly through regulating mTOR pathway. 33 Our results prove curcumin is protective for cardiomyocytes after OGD/R. Furthermore, D942 can also inhibit mTOR signaling, probably due to the activation of AMPK. When D942 and curcumin are administered together, mTOR signaling is robustly inhibited, and autophagy is highly promoted, compared with any agent alone. It is noteworthy that OGD/R itself also induces autophagy in cardiomyocytes, but the extent of induction is much less than that by D942 and curcumin. Thus, we concluded that enhanced autophagy by D942 and curcumin is beneficial for cardiomyocyte survival. However, we cannot exclude the possibility that excessive induction of autophagy by abuse of either agent would incur severe cell damage. Proper dosages of D942 and curcumin have to be taken into account.
Our data also demonstrated the similar autophagy-inducing and cardioprotective effects of combination of D942 and curcumin in vivo. The I/R itself is a strong inducer of autophagy while combination of D942 and curcumin induces even higher level of autophagy, which could be beneficial for vital nutrient supply and cardiomyocyte survival after stroke. However, further discrete testing for proper dosage and duration of treatment is still indispensable, since excessive autophagy could also be detrimental to cardiomyocytes. Unfortunately, there seems to be no clear border of “good autophagy” and “bad autophagy” in the complicated intracellular and extracellular microenvironment. And currently there is no available method to accurately measure the extent of autophagy. Hence, careful preliminary study to evaluate the effect of D942 and curcumin will be in high demand.
For potential clinical application of D942 and curcumin, another important concern is their side effects. Curcumin seems to be a quite safe agent for patients, since previous clinical studies showed little toxicity except mild nausea and diarrhea. 34 However, this conclusion was based on a study using oral administration. We used iv injection, because it is a direct way to distribute curcumin into blood circulation to access cardiomyocytes. However, whether curcumin in blood could cause damage to other tissues and organs is still unknown. Although we did not observe any abnormalities in the appearance and behavior of the mice tested, we cannot exclude the possibility that high concentration of curcumin in blood could cause damage in other tissues, especially in blood cells, kidney, and lung. The side effects of D942 are unknown due to the lack of study on D942 as a potential therapeutic agent. It might influence the metabolism because of its potency to activate AMPK pathway. Hence, further research on the side effects of these 2 agents will give us more information on their safe use.
Taken together, our research demonstrated cardioprotective effects of D942 and curcumin after OGD/R exposure. Based on our discovery, a potentially novel therapeutic approach could be proposed for myocardial I/R.
Footnotes
Authors’ Note
Hong Jiang conceived and designed the experiments, Keping Yang performed the experiments, Xin Li analyzed the data, Chenhong Xu contributed reagents/materials/analysis tools, and Keping Yang wrote the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
