Abstract
We investigated the effect of erythropoietin (EPO) posttreatment on survival time and vascular functions in a mouse model of sepsis. Sepsis was induced by cecal ligation and puncture. After 20 ± 2 hours of sepsis, thoracic aorta was isolated for assessing its reactivity to norepinephrine (NE) and acetylcholine (ACh). We also measured the tissue nitric oxide (NO) level, inducible nitric oxide synthase (iNOS), endothelial nitric oxide synthase (eNOS), G protein-coupled receptor kinase 2 (GRK2), and α1D adrenoceptor messenger RNA (mRNA)/protein expression. In septic mice, EPO moderately improved the survival time from 19.68 ± 0.75 to 34.7 ± 3.2 hours. Sepsis significantly decreased the aortic contractile response to NE along with reduced α1D mRNA and protein expression. Erythropoietin significantly preserved the α1D receptor expression and restored NE-induced contractions to control levels in septic mice. Further, it attenuated the aortic α1D receptor desensitization in sepsis which was evident from reduced GRK2 mRNA expression. Accordingly, a selective GRK2 inhibitor markedly restored the contractile responses to NE in sepsis. Erythropoietin treatment attenuated iNOS mRNA expression and iNOS-induced overproduction of NO, but improved endothelium-dependent relaxation to ACh associated with increased eNOS mRNA expression. In conclusion, EPO seems to reverse sepsis-induced vasoplegia to NE through the preservation of α1D adrenoceptor mRNA/protein expression, inhibition of GRK2-mediated desensitization, and attenuation of NO overproduction in the mouse aorta.
Introduction
Sepsis, the most common cause of death among patients in intensive care unit, occurs due to multiple organ dysfunctions, 1 and the incidence and mortality rates remain high around the world. 2,3 Recent data suggest that 18 million new cases occur each year with a mortality of 30%. 4 The only Food and Drug Administration-approved drug, human recombinant activated protein C, for the treatment of sepsis has recently been withdrawn from the market. 5,6 Thus, finding any new treatment strategy becomes a challenge to the researchers and the clinicians.
Erythropoietin (EPO) is commonly used for the treatment of anemia in humans. 7 However, there is increasing evidence from animal studies that EPO is beneficial in sepsis. The protective effect of the drug in sepsis is mainly attributed to the improvement in cardiac function, 8,9 prevention of apoptosis, and decrease in tissue hypoxia. 10 Hyporesponsiveness to vasoconstrictors (vasoplegia) is one of the characteristic features of sepsis in its later stage, which poses a serious problem for the clinicians to treat hypotension in patients with sepsis. However, it is not clear whether EPO treatment can reverse vasoplegia in sepsis and, if so, what are the mechanisms involved. Hypertension is the main side effect of patients with anemia receiving EPO. 11 Therefore, it is logical to believe that it might improve reactivity of vascular smooth muscles to vasoconstrictor drugs in sepsis. Although, EPO lacks direct vasoconstrictor effect, it has been reported to augment the contractile potency of norepinephrine (NE) through the release of endothelin 1. 12 Therefore, the present study was undertaken to evaluate the effect of EPO on sepsis-induced vasoplegia in mice.
We have recently reported that vasoplegia due to sepsis was associated with decreased α1D adrenergic receptor (AR) messenger RNA (mRNA) expression and overproduction of inducible nitric oxide synthase (iNOS)-derived nitric oxide (NO) in the mouse aorta. 13 Further, it has been known that plasma catecholamines levels are elevated due to increased sympathetic activity in sepsis. 14 Garcia-Sainz and coworkers 15 have suggested that constant stimulation with NE rapidly phosphorylates α1D ARs, resulting in receptor desensitization. This desensitization is initiated following phosphorylation of agonist-occupied receptors by G protein-coupled receptor kinases (GRKs). A previous study demonstrates that in vascular smooth muscle, α1D ARs are desensitized by GRK-2. 16 Therefore, we evaluated the effect of EPO treatment on α1D AR desensitization in sepsis.
Endothelial dysfunction plays an important role in the pathophysiology of sepsis. 17,18 Nitric oxide is the primary endothelium-derived vasodilator in aorta, 19 and reduction in the expression and activity of endothelial nitric oxide synthase (eNOS) is associated with caecal ligation and puncture (CLP) model of sepsis. 20,21 Therefore, the second objective of the present study was to evaluate the effect of EPO on endothelial dysfunction in sepsis.
We hypothesized that improvement in vascular functions might improve the survival in mouse model of sepsis. Therefore, in addition to assessing vascular functions, we conducted experiments to examine the effect of EPO on survival in CLP-induced sepsis in mice.
Materials and Methods
Animals
Healthy adult male Swiss Albino mice (30-35 g) were procured from the Laboratory Animal Resource Section, Indian Veterinary Research Institute, Izatnagar, Uttar Pradesh, India. Mice were housed in polypropylene cages with free access to feed and water in the Divisional Animal House. All the experimental protocols were approved by Institutional Animal Ethics Committee of the Indian Veterinary Research Institute, Izatnagar.
Induction of Sepsis in Mice
Sepsis was induced by cecal ligation and puncture described by Wicherman and coworkers. 22 It is characterized by initial hyperdynamic phase followed by late hypodynamic phase and simulates the natural sepsis in clinical conditions observed in humans. 23 Mice were fasted overnight before the induction of sepsis but allowed water ad libitum. The animals were anesthetized by injection of xylazine (10 µg/g body weight, intraperitoneally) and ketamine (80 µg/g body weight, intraperitoneally), and a 2-cm ventral midline incision was made. The cecum was then exposed and ligated with 3-0 silk just distal to the ileocecal valve to avoid the intestinal obstruction, punctured twice with a 21-gauge needle, and returned to the abdomen. The abdominal incision was closed in layers. To prevent dehydration, isotonic sodium chloride solution (1 mL/mouse) was given subcutaneously to all mice. Sham-operated (SO) mice had undergone the same surgical procedure, except CLP. All the surgical maneuvers were carried out according to the procedures laid down by the Institutional Animal Ethics Committee.
Dose and Treatment Schedule
After 7 days of acclimatization period, the mice were divided into 4 groups: (1) SO + vehicle, (2) sepsis (CLP) + vehicle, (3) SO + EPO, and (4) sepsis + EPO (CLP + EPO). Erythropoietin was procured from Zuventus Healthcare Ltd, a joint Venture of Emcure, Mumbai, India. The SO/CLP mice were injected subcutaneously with normal saline (0.1 mL), and SO + EPO/CLP + EPO mice were injected EPO (1000 IU/kg) subcutaneously. The dose and time of administration of EPO were based on previous reports. 8,10
Survival Study
After the induction of sepsis, the animals were treated with either vehicle or EPO per the dosage schedule mentioned earlier. All the animals from different groups were kept under observation for 72 hours. The degree of sepsis was assessed by the presence of conjunctivitis, absence of grooming activities with resulting ruffled fur, reduced intake of feed and water, and lethargy. The survival curves and mean survival time for all the groups were obtained by Kaplan-Meier survival curve and analyzed using log-rank test.
Determination of Serum Lactate Level
Blood samples were collected by cardiac puncture from mice of different groups at 20 ± 2 hours postsurgery. Serum was separated by centrifuging at 1717g for 10 minutes and kept at −80°C until further use. Serum lactate was estimated by enzyme-linked immunosorbent assay kit (Sigma-Aldrich, St Louis, Missouri) per the manufacturer’s instruction. The principle of the test was based on the formation of a product due to oxidation of lactate by lactate dehydrogenase, which interacts with a probe to produce red dye. The intensity of the dye was determined at 570 nm, and results were expressed as mmol/L.
Tissue Collection
Twenty hours after surgery, the mice were killed by bleeding from vena cava under xylazine–ketamine anesthesia. Lung and the heart were removed en bloc after opening the chest and abdomen, and the thoracic aorta were immediately placed into cold (4°C) modified Krebs-Henseleit solution (MKHS) with the following composition (mmol/L): 118.0 NaCl, 4.7 KCl, 2.5 CaCl2·2H2O, 1.2 MgSO4·7H2O, 1.2 KH2PO4, 11.9 NaHCO3, and 11.1 glucose. Thoracic aorta was cleaned of adhering tissue and cut into 3- to 4-mm long rings.
Recording of Isometric Tension and Experimental Protocol
The arterial rings were mounted between 2 “L”-shaped hooks made of 37-gauge stainless steel wire and kept under a resting tension of 1.0g in a thermostatically controlled (37.0°C ± 0.1°C) organ bath (Ugo Basile, Italy) of 10-mL capacity containing MKHS and were continuously bubbled with medical gas (74% N2 + 21% O2 + 5% carbon dioxide). The arterial rings were equilibrated for 60 to 80 minutes in organ bath filled with MKHS before recording tension. During equilibration period, the bath fluid was repeatedly changed once in every 15 minutes. The change in tension was measured by a high-sensitivity isometric force transducer and recorded in a PC using LabChart V6.1.3 Pro software program (Powerlab; AD Instruments, Australia). After equilibration, the arterial rings were contracted with high K+ (80 mmol/L) depolarizing solution to assess the viability of the tissue. The preparations were then washed with normal MKHS to restore the baseline resting tension. After a lapse of 30 minutes and 2 to 3 washes with normal MKHS, concentration-dependent contractile response to NE (0.1 nmol/L-1 µmol/L) was elicited in the arterial rings from different groups at an increment of 0.5 log unit. To evaluate the influence of iNOS-derived NO, GRK2, and Gβγ subunit in mediating vascular hyporeactivity to NE in sepsis, aortic rings from different groups were preincubated with iNOS inhibitor N-[[3-(aminomethyl)phenyl]methyl]-ethanimidamide dihydrochloride (1400 W), a selective inhibitor of GRK2 methyl 5-[2-(5-nitro-2-furyl)vinyl]-2-furoate, and a selective inhibitor of Gβγ subunit gallein, respectively, for 30 minutes before recording the concentration-dependent responses to NE. To rule out the nonspecific action of GRK2 inhibitor, calcium chloride (CaCl2)-induced contractile response was elicited in the absence and presence of GRK2 inhibitor. Calcium chloride-induced contraction in K+-depolarized vascular tissue preparations involves Ca2+ influx through voltage-gated L-type calcium channels and is independent of G-protein coupled receptor (GPCR) activation. Thus, receptor phosphorylation and desensitization by GRK2 are not involved in CaCl2-induced response in the aorta. In this set of experiment, after equilibration period, aortic rings were contracted with K+-depolarizing solution followed by wash with Ca2+-free MKHS with ethylene glycol tetraacetic acid (EGTA) solution to remove any extracellular calcium. Then, the tissue was incubated with Ca2+-free K+-depolarizing solution without EGTA for 30 minutes. Then, concentration-dependent contractions to CaCl2 (10 μmol/L-10 mmol/L) were elicited in the absence and presence of 1 µmol/L GRK2 inhibitor. In a separate set of experiment, to assess the endothelium-dependent and independent relaxation of aortic rings, dose-dependent response to acetylcholine (ACh; 1nmol/L-10 µmol/L) and sodium nitroprusside (SNP; 0.1 nmol/L-0.1 µmol/L) was recorded, respectively, in phenylephrine (PE)-precontracted aortic rings from different groups.
Measurement of Aortic NO Level
Aortic total tissue NO production in terms of nitrate was estimated per the method described earlier, 24,25 with some modifications. Briefly, around 10 mg (wet weight) of tissue was homogenized in MKHS (10%) in a 1.5-mL centrifuge tube and incubated for 30 minutes. The tubes contained MKHS (control) or MKHS + 1400 W (10 µmol/L), an iNOS-specific inhibitor. The second set of tubes (MKHS + 1400 W) was kept to estimate the eNOS-derived NO production. Deproteinization of the samples was carried out by adding 1 mL ZnSO4 (75 mmol/L) and 12.5 mL NaOH (55 mmol/L), mixed thoroughly, and allowed to stand for 10 minutes. The tubes were then centrifuged at 5724g for 10 minutes, and 500 µL of supernatant was added with Griess reagents (SRL, Maharashtra, INDIA) containing 450 µL of sulfanilamide (1%) and 50 µL of N-(1-naphthyl) ethylenediamine (0.2%) and were incubated at 37°C for 20 minutes. After incubation, absorbance of the samples was measured at 545 nm and compared with standard curve. To construct a standard curve, the same procedure was followed except that individual tubes contained 1, 2.5, 5, 7.5, or 10 µmol/L of nitrite diluted from a stock solution of NaNO2 (10 mmol/L). Absorbance was recorded and converted to a straight line with regression analysis (Y = ax + b; R 2 > .99). Values for nitrite were computed from the linear regression formula.
Messenger RNA Expression Study
Total RNA isolation
After 20 ± 2 hours postsurgery, thoracic aorta from mice of different groups were isolated in 0.1% diethyl pyrocarbonate–treated autoclaved phosphate-buffered saline. After removing the adjacent adipose tissues under a microscope, tissues were stored in RNA later and kept at −80°C until further use. Total RNA was isolated with Mini RNA isolation kit (IBI Scientific, Peosta, Iowa, USA) per the manufacturer’s instructions. The samples were treated with Rnase-free Dnase, and the DNase was subsequently inactivated by heating at 56°C for 10 minutes and immediately chilled to 4°C. The purity of the RNA was quantified by NanoDrop spectrophotometer (Thermo Scientific, Waltham, MA USA), and only RNA samples with an A260–A280 ratio of 1.8 to 2.0 were used for reverse transcription. Synthesis of complementary DNA (cDNA) was carried out using Revertaid first strand cDNA synthesis kit (Thermo Scientific, Waltham, MA USA) using moloney murine leukemia virus reverse transcriptase enzyme by following manufacturer’s instructions.
Quantitative real-time polymerase chain reaction
Real-time polymerase chain reaction (PCR) was performed using SYBR Green I master mix (Maxima SYBR Green/ROX quantitative PCR master mix [2×]; Thermo Scientific, Waltham, MA USA). Each sample was run in duplicate in a 25-µL reaction mixture. The 25 µL reaction mixture consisted of 12.5 µL SYBR Green/ROX master mix, 1.0 µL from 10 pmol/µL stock solution of each of the gene-specific forward and reverse primers, and 1 µL of cDNA, and the volume was made up to 25 µL with RNAse-free water. The real-time PCR reaction was started with initial incubation at 95°C for 10 minutes, followed by 40 cycles of amplification with denaturation at 95°C for 30 seconds, annealing (temperature varied depending on the primer pairs) for 30 seconds, and extension at 72°C for 30 seconds each. The optimum annealing temperatures determined by PCR for the respective gene using the specific primers were as follows: the α1D adrenoceptor genes were F 5′-GCCTCTGAGGTGGTTCTGAG-3′, R 5′-GGACGAAGAAAAAGGGGAAC-3′ (208 bp) at 57°C; for GRK2 gene F 5′-GGCGATACTTCTACTTGTTCCC-3′, R 5′-CGTTCCTTGATCTGTGTCTCTT-3′ (118 base pairs) at 57°C; for the eNOS gene: F 5′-GGCTGGGTTTAGGGCTGT-3′, R 5′-GCTGTGGTCTGGTGCTGGT-3′ (107 base pairs) at 57°C; for iNOS gene F 5′-ACATCGACCCGTCCACAGTAT-3′, R 5′-CAGAGGGGTAGGCTTGTCTC-3′ (177 base pairs) at 60°C; and for the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene: F 5′-AACTTTGGCATTGTGGA AGG-3′, R 5′-ACACATTGGGGGTAGGAACA-3′ (223 base pairs) at 58°C. To assess the specificity of the amplified product, a dissociation curve was generated at temperatures of 55°C through 95°C. The results were expressed as threshold cycle values (CT). This value is the cycle number when the fluorescence of the reporter dye is appreciably higher than the background fluorescence. The threshold, automatically adjusted by the instrument, was used for the generation of CT values.
Immunohistochemistry of α1D Adrenoceptor
Transverse cross sections (5 µm) from neutral-buffered, formalin-fixed, paraffin-embedded aorta were incubated overnight with rabbit anti-mouse polyclonal antibody against α1D AR (1:50, Abcam, Cambridge, MA, USA) at 4°C in a moist chamber. After that sections were incubated with goat anti-rabbit polyclonal immunoglobulin G tagged with horseradish peroxidase (1:500; Enzo Life Sciences, Farmingdale, New York, USA) for 30 minutes at room temperature in a moist chamber. Counterstaining was done with Mayer hematoxylin stain for 30 to 40 seconds. Negative control slides were treated similarly with normal anti-rabbit serum. The sections were visualized for α1D AR protein under light microscope (OLYMPUS, IX51 Tokyo, Japan). The density of α1D AR was counted in 5 different areas located in 4 sections from each aorta under 20× magnification. The receptor density in different groups was measured semiquantitatively by ImageJ software (ImageJ, NIH, Maryland, USA) and expressed as arbitrary unit (AU) in relation to SO control (ie, considering the density in SO control = 1).
Estimation of Plasma NE Level
At 20 ± 2 hours postsurgery, blood samples from different groups were collected in EDTA-containing tubes, and plasma was separated by centrifuging the blood at 4000 rpm for 10 minutes at 4°C. The samples were then immediately aliquoted and stored at −80°C for NE measurement using commercially available mouse catecholamine EIA kit (Cloud-Clone Corp, Texas, USA). Concentration of plasma NE was expressed in pg/mL.
Drugs and Chemicals
Norepinephrine, ACh, and SNP were purchased from Sigma Chemical (St Louis, Missouri). 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one and N-([3(aminomethyl)phenyl]methyl) ethanimidamide dihydrochloride (1400 W), methyl 5-[2-(5-nitro-2-furyl)vinyl]-2-furoate, and gallein were procured from Cayman Chemicals (AnnArbor, Michigan), Calbiochem (MA, USA), and Tocris (Bristol, United Kingdom), respectively. Norepinephrine was dissolved in 0.1 N HCl and 0.01% ascorbic acid, whereas methyl 5-[2-(5-nitro-2-furyl) vinyl]-2-furoate and gallein were dissolved in dimethylsulfoxide. All other chemicals were of analytical grade. All other drugs were dissolved in distilled water.
Statistical Analysis
Results are expressed as mean ± standard error of the meanwith “n” equal to number of animals. The overall difference in survival time was determined by the Kaplan-Meier test followed by the log-rank test. The maximal response (Emax) and the concentration producing 50% of the maximal response (EC50) were determined by nonlinear regression analysis (sigmoidal dose response with variable slope), using GraphPad Prism version 4.00 (San Diego, California). Sensitivity/potency was expressed as pD2 = −log EC50. Concentration-dependent agonist response data were analyzed by 2-way analysis of variance (ANOVA) followed by Bonferroni post hoc test. Differences in repeated measurements were analyzed by 2-way ANOVA followed by multiple comparisons with Bonferroni post hoc test; otherwise, 1-way ANOVA followed by Newman-Keuls post hoc analysis was applied to determine statistical significance using GraphPad Prism software. To study the relative change in gene expression, the 2−ΔΔCT method was used as described previously by Livak and Schmittgen. 26 The formula used to calculate the fold change in gene expression was “fold change = 2−ΔΔCT” (where ΔΔCT = [CT, target gene − CT, GAPDH] treatment – [CT, target gene − CT, GAPDH] control). The gene-specific amplification was corrected for the difference in input of RNA by taking housekeeping gene GAPDH to account. For CLP + vehicle, SO + EPO, CLP + EPO groups, evaluation of 2−ΔΔCT indicates the fold change in gene expression relative to SO control (ie, fold change in sham control = 1). The results were analyzed in comparison with the CT (minimum threshold of amplification) value of the target gene and the reference gene (GAPDH). Differences in values were considered statistically significant at P < .05.
Results
Effect of EPO Posttreatment in Survival Time in Septic Mice
Figure 1 shows survival curves and mean survival time for mice treated with EPO after onset of sepsis. The mean survival time in sepsis (vehicle treated) was 19.68 ± 0.75 hours (n = 26), while EPO posttreatment (1000 IU/kg body weight subcutaneously) significantly (P < .001) improved the survival time to 34.7 ± 3.2 hours, n = 21. All the SO (n = 7) and SO + EPO (n = 12) mice survived during the 72-hour observation period.
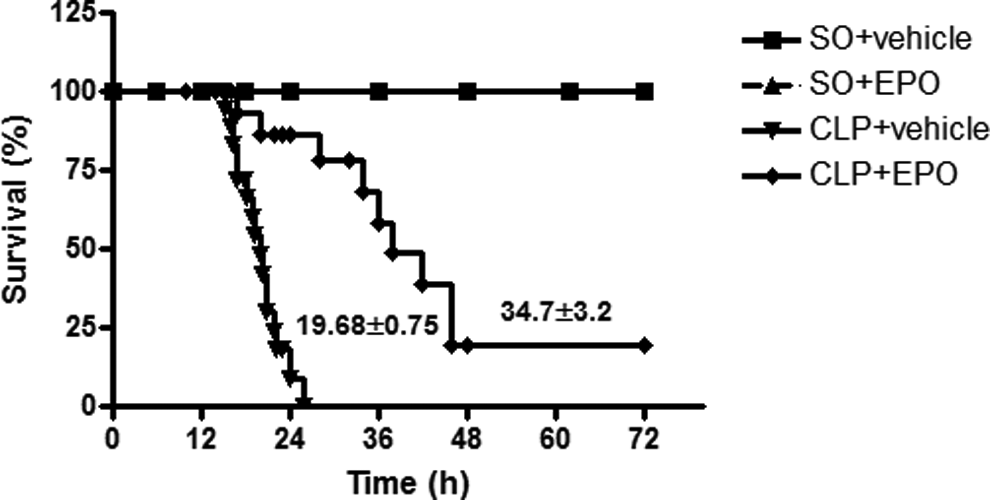
Effect of erythropoietin (EPO) posttreatment on survival time in septic mice. Treatment with EPO at 1 hour after surgery (CLP + EPO) significantly (P < .001) improved the mean survival time in comparison to the untreated sepsis (CLP + vehicle) group. All the sham operated (SO) + vehicle treated and SO + EPO mice survived during the 72-hour observation period. The overall difference in survival time was determined by the Kaplan-Meier test followed by the log-rank test. *P < .001in comparison to CLP + vehicle.
Effect on Serum Lactate Level
An elevated lactate concentration in sepsis is a marker of disease severity. As a marker of sepsis, the lactate level was estimated in the serum of mice from different groups after 20 ± 2 hours of surgery. In CLP mice, the serum lactate level (4.64 ± 0.05 mmol/L, n = 6) was significantly (P < .001) higher than that of the SO mice (1.61 ± 0.37 mmol/L; n = 6). Treatment with EPO significantly (P < .001) reduced the serum lactate level (2.85 ± 0.09 mmol/L; n = 6) in septic mice. However, serum lactate level of EPO-treated SO mice (2.80 ± 0.09 mmol/L; n = 6) was significantly (P < .01) higher than the vehicle-treated SO mice.
Effect on Hyporeactivity of Aortic Smooth Muscle to NE in Septic Mice
Vasoplegia or vascular hyporeactivity is one of the clinical characteristics of sepsis. We used NE to assess the reactivity of mouse aorta ex vivo. Figure 2 illustrates the reactivity of mouse aorta to NE in different groups. The raw tracing in Figure 2A shows that aortic rings from different groups of mice exhibited concentration-dependent contractions to NE (0.1 nmol/L-10 µmol/L). In SO animals, the pD2 and Emax values were 7.85 ± 0.09 and 0.45 ± 0.03 g (n = 9), respectively. Sepsis significantly (P < 0.05) decreased the Emax (0.11 ± 0.01 g; n = 13) as well as the potency of the agonist NE (pD2 7.30 ± 0.14; n = 13). Treatment of CLP mice with EPO (P < .05) preserved the contractions to NE (Emax 0.37 ± 0.03 g; pD2 7.732 ± 0.11; n = 10), almost to the control level. The SO mice treated with EPO showed no significant change in the contraction of aortic rings to NE (Emax 0.49 ± 0.04 g and pD2 7.427 ± 0.11; n = 8) in comparison to vehicle-treated SO mice.

Effect of erythropoietin (EPO) posttreatment on hyporeactivity of aortic smooth muscle to norepinephrine (NE) in septic mice. Representative tracings show concentration-dependent contractions to NE (0.1 nmol/L-1 µmol/L) in the aorta taken from sham operated (SO) + vehicle (A), CLP + vehicle (B), SO + EPO (C), and CLP + EPO (D) treated septic mice. The line diagram (E) depicts the mean concentration–response curves elicited with cumulatively added NE on the aortic rings obtained from different treatment groups. Vertical bars represent standard error of the mean (SEM). Data were analyzed by 2-way analysis of variance (ANOVA) followed by Bonferroni post hoc tests. *P < .05 in comparison to SO + vehicle; # P < .05 in comparison to CLP + vehicle.
Effect on α1D mRNA Expression in Septic Mouse Aorta
In order to examine the hypothesis that hyporeactivity to NE in septic mouse aorta may relate to an alteration in the transcriptional regulation of receptor expression, the effect of different treatments on α1D mRNA expression was studied. As presented in Figure 3, sepsis significantly (P < .001) decreased the mRNA expression of α1D in the aorta to 0.41 ± 0.09 (n = 6) in comparison to SO animals (1.0; n = 6). Treatment with EPO (P < .001) preserved the α1D mRNA expression (0.81 ± 0.09-fold; n = 6). However, EPO treatment of SO mice had no significant effect on aortic α1D mRNA expression (0.85 ± 0.06; n = 6) compared to vehicle-treated SO animals.

Effect of erythropoietin (EPO) posttreatment on sepsis-induced decrease in α1D adrenoceptor mRNA expression in the mouse aorta. Sepsis significantly (P < .001) attenuated the α1D-adrenoceptor mRNA expression compared to sham operated (SO) + vehicle-treated controls. Erythropoietin posttreatment significantly (P < .001) preserved the α1D mRNA expression in the aortic segments from septic mouse. Data were analyzed by 1-way analysis of variance (ANOVA) followed by Newman-Keuls multiple comparison test. Vertical bars represent standard error of the mean (SEM). *P < .001 compared with SO +vehicle; # P < .001 compared with CLP + vehicle. mRNA indicates messenger RNA.
Effect on α1D Protein Expression in Septic Mouse Aorta
To relate the improved vascular reactivity to NE in treatment groups to receptor protein expression, the α1D AR protein expression in mouse aorta was studied in different groups. The representative immunohistochemical images of α1D AR protein in different groups are presented in Figure 4. Sepsis significantly (P < .001) reduced the protein density to 0.38 ± 0.04 AU (n = 4) when compared to SO mice (1.00 ± 0.00; n = 4). Treatment of septic mice with EPO significantly (P < .05) increased the protein density to 0.59 ± 0.08 AU (n = 4). However, treatment with EPO in SO mice did not produce any significant change in the protein density (0.92 ± 0.06 AU; n = 4) compared to SO animals.

Representative immunohistochemical images and bar diagram depict the α1D adrenoceptor protein density in the aortic section obtained from different groups. Data were analyzed by 1-way analysis of variance (ANOVA) followed by Newman-Keuls multiple comparison test. Vertical bars represent standard error of the mean (SEM). *P < .001 compared with sham operated (SO) + vehicle; # P < .05 compared with CLP + vehicle.
Effect on Plasma NE Levels
One of the mechanisms of receptor desensitization relates to an increased level of the agonist at the receptor site. With a view to examining this concept, we measured plasma NE level in different groups. Compared to the NE levels in SO + vehicle-treated animals (80.52 ± 1.71 pg/mL; n = 6), there was a significant (P < .001) rise in the plasma NE level in CLP + vehicle-treated mice (144.90 ± 12.76 pg/mL; n = 8). Treatment with EPO significantly (P < 0.001) reduced NE level (73.14 ± 5.47 pg/mL; n = 6) in septic mice. However, plasma NE level was not significantly altered in the SO mice treated with EPO (94.72 ± 7.72 pg/mL; n = 6) compared to vehicle-treated SO mice.
Effect of GRK2 Inhibitor (1 µmol/L) on Hyporeactivity of Mouse Aorta to NE in Sepsis
G protein receptor kinase 2 plays a significant role in the desensitization of GPCRs. To study the role of α1D adrenoceptor desensitization mechanism in impairing NE-induced contraction in sepsis, we used methyl 5-[2-(5-nitro-2-furyl)vinyl]-2-furoate, a selective inhibitor of GRK2. The comparative line diagrams in Figure 5 show the mean concentration–response curves of NE in the absence and presence of GRK2 inhibitor in aortic rings from different groups. As shown in Figure 5B, GRK2 inhibitor significantly (P < .05) restored the contractile response of aortic rings to NE (0.35 ± 0.03 g; n = 8 vs 0.11 ± 0.01 g; n = 13) in septic mice. However, it did not produce any additional improvement in NE-induced contractile response in any of the 2 treatment groups compared to their respective controls. For example, the Emax values of NE in the absence and presence of GRK2 inhibitor were 0.50 ± 0.03 g versus 0.53 ± 0.03 g (n =6-8) in EPO-treated SO mice and 0.37 ± 0.03 g versus 0.40 ± 0.04 g (n = 7-10) in EPO-treated septic mice.

Role of G protein receptor kinase-2 (GRK2) and Gβγ subunit signaling on hyporeactivity of aortic rings to norepinephrine (NE) in septic mouse aorta. Comparative line diagrams showing the mean concentration–response curves of NE elicited in the absence and presence of GRK2 inhibitor (1 µmol/L) or Gβγ inhibitor gallein (100 µmol/L) in aortic rings from mice of sham operated (SO) + vehicle (A), CLP + vehicle (B), SO + erythropoietin (EPO; C), and CLP + EPO (D) groups. Bar diagram (E) showing the effect of EPO posttreatment on sepsis-induced enhanced GRK2 messenger RNA (mRNA) expression in the mouse aorta. Note that the GRK2 inhibitor and gallein partially restored the NE-induced contractile response only in the aortic rings obtained from CLP + vehicle group. Vertical bars represent standard error of the mean (SEM). Data were analyzed by 2-way analysis of variance (ANOVA) followed by Bonferroni post hoc tests. *P < .05 in comparison to respective control, # P < .05 in comparison to CLP + vehicle.
To eliminate the off-target effect of the GRK2 inhibitor, methyl 5-[2-(5-nitro-2-furyl)vinyl]-2-furoate, we assessed its effect on CaCl2-induced contractions (unrelated to GPCR) in Ca2+-free high K+ (80 mmol/L) solution in the aorta from SO and CLP mice. The SO mice exhibited concentration-dependent contractions to CaCl2 (10 μmol/L-10 mmol/L) with pD2 and Emax values of 3.37 ± 0.21 and 0.51 ± 0.05 g (n = 5), respectively. Sepsis significantly (P < .05) decreased the maximal response (Emax 0.28 ± 0.03 g; n = 6) without affecting the potency (pD2 3.48 ± 0.17; n = 6). However, GRK2 inhibitor did not influence the response of CaCl2 either in SO mice (Emax 0.46 ± 0.03 g; pD2 3.70 ± 0.19; n = 4) or in CLP mice (Emax 0.26 ± 0.01 g; pD2 3.25 ± 0.13; n = 4) compared to their respective controls. This indicates that GRK2 inhibitor specifically reverses NE-induced hyporeactivity in CLP mice.
Effect of Gallein (100 µmol/L) on Hyporeactivity of Mouse Aorta to NE in Sepsis
Gβγ subunit of GPCRs recruits GRK2 for phosphorylation and subsequent desensitization of the receptors. To assess the role of Gβγ subunit in α1 AR desensitization, we used gallein, a selective inhibitor of Gβγ. The comparative line diagram in Figure 5 shows the mean concentration–response curves of NE in the absence and presence of gallein in aortic rings from different groups. As shown in Figure 5B, gallein partially (P < .05) restored the contractile response of aortic rings to NE (0.28 ± 0.02 g; n = 8 vs 0.11 ± 0.01 g; n = 13) only in septic mice. However, it did not produce any additional improvement in NE-induced contractile response in any of the 2 treatment groups compared to their respective controls. For example, Emax values of NE in the absence and presence of gallein were 0.49 ± 0.03 g versus 0.38 ± 0.02 g (n = 6) in SO + EPO and 0.36 ± 0.03 g versus 0.41 ± 0.05 g (n = 7-8) in EPO-treated septic mice, respectively.
Effect on GRK2 mRNA Expression in Mouse Aorta
Figure 5E illustrates the effect of different treatments in sepsis on GRK2 mRNA expression in the mouse aorta. Sepsis significantly (P < .05) increased the mRNA expression of GRK2 in the aorta to 2.03 ± 0.30-fold (n = 4). Erythropoietin treatment of septic mice restored the mRNA expression (0.88 ± 0.21-fold; n = 4) to SO control level. However, the mRNA expression of GRK2 in EPO-treated SO mice was 0.76 ± 0.31-fold, n = 4.
Effect on iNOS mRNA Expression in Mouse Aorta
To examine whether the improved vasoreactvity to NA in sepsis relates to reduced iNOS-derived NO, we studied iNOS gene expression in the mouse aorta from different groups. As illustrated in Figure 6A, sepsis significantly (P < .001) increased the iNOS mRNA expression in aorta to 4.32 ± 0.35-fold (n = 4) in comparison to SO controls (1.00; n = 4). This increased mRNA expression was significantly (P < .001) attenuated in septic mice treated with EPO (1.73 ± 0.54; n = 4). However, treatment with EPO in SO mice (1.22 ± 0.27; n = 4) produced no significant change in the iNOS mRNA expression compared to SO + vehicle-treated mice.
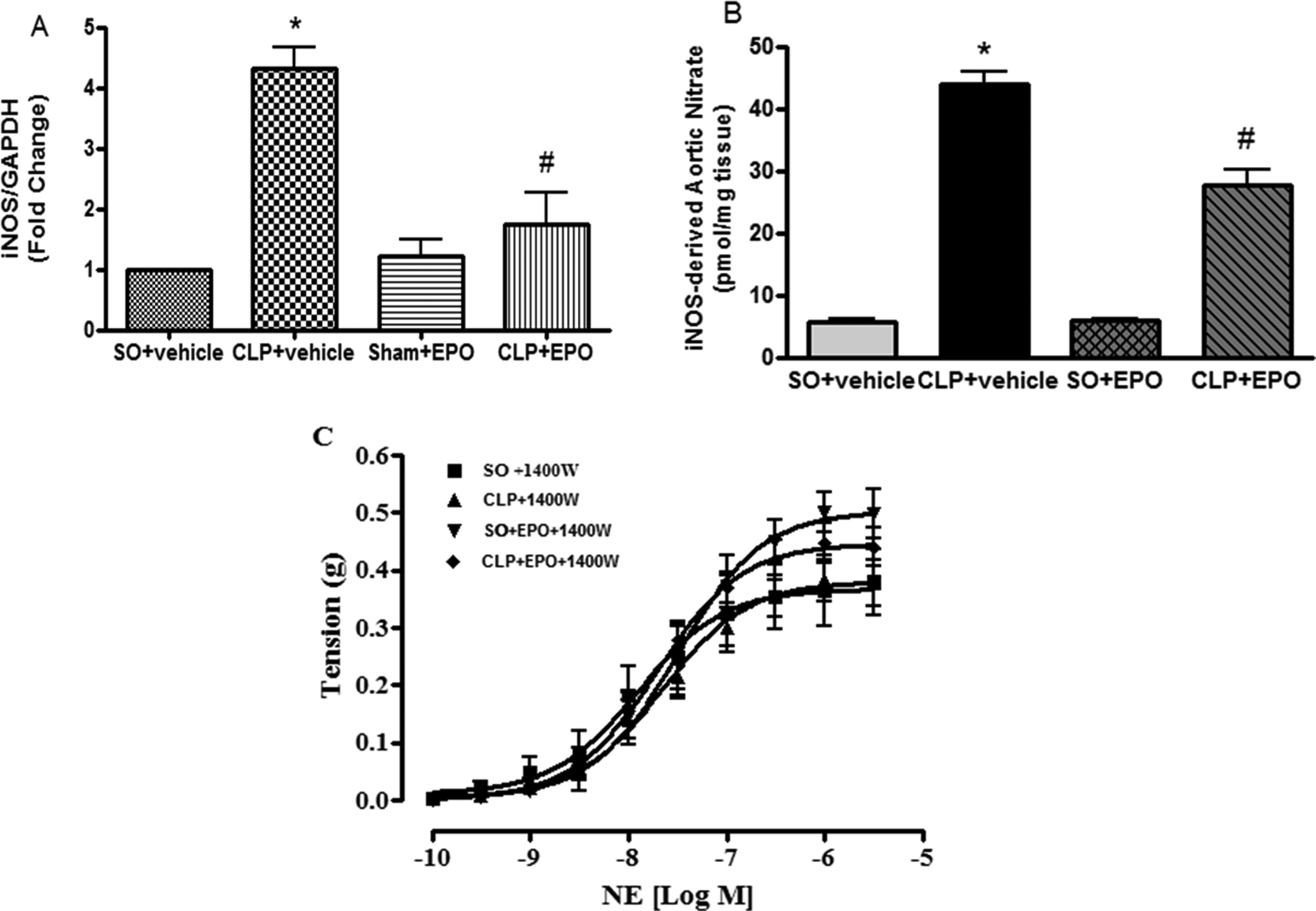
Effect of erythropoietin (EPO) posttreatment on inducible nitric oxide synthase (iNOS)-derived nitric oxide on hyporeactivity of aortic rings to norepinephrine (NE) in sepsis. Note that sepsis significantly elevated iNOS mRNA expression (A) and iNOS-derived nitrate level (B) in the mouse aorta. Treatment with EPO at 1 hour after surgery significantly attenuated aortic iNOS mRNA expression and iNOS-derived aortic nitrate level. Data were analyzed by 1-way analysis of variance (ANOVA) followed by Newman-Keuls multiple comparison test. The line diagram (C) depicts the mean concentration–response curves elicited with cumulatively added NA in the presence of selective iNOS inhibitor, 1400 W, on the aortic rings obtained from different treatment groups. Data were analyzed by 2-way ANOVA followed by Bonferroni post hoc tests. Vertical bars represent standard error of the mean (SEM). *P < .05 in comparison with sham operated (SO) + vehicle; # P < .05 in comparison with CLP + vehicle.
Effects on iNOS-Derived Nitrate Level in Aortic Homogenate
To establish whether the change in iNOS gene expression correlates with the product of the iNOS gene, we have measured the nitrate level in mouse aortic homogenates from different groups. Figure 6B illustrates the effect of different treatments on inducible nitrate content in mouse aorta. Inducible nitric oxide synthase-derived nitrate level in aorta from SO + vehicle-treated animals was 5.68 ± 0.60 pmol/mg wet tissue (n = 6). Sepsis significantly (P < .001) increased the aortic nitrate level to 43.89 ± 2.06 pmol/mg wet tissue (n = 6). Treatment with EPO in septic mice significantly (P < .001) reduced the nitrate level (27.62 ± 2.60 pmol/mg wet tissue; n = 6). Erythropoietin in SO mice (6.03 ± 0.15 pmol/mg wet tissue; n = 6) did not significantly alter the iNOS-derived nitrate level in aorta compared to SO + vehicle-treated mice.
Effect of iNOS Inhibitor 1400 W (10 µmol/L) on Hyporeactivity of Mouse Aorta to NE in Sepsis
To assess the role of iNOS-derived NO in sepsis-induced vascular hyporeactivity to NE, 1400 W was used to selectively inhibit iNOS. Figure 6C illustrates the effect of 1400 W (10 µmol/L) on NE-induced contractile responses in mouse aorta. In the presence of 1400 W (10 µmol/L), no significant difference was observed in either the maximal response or the potency of NE among the different groups. For example, the pD2 and Emax values were 7.89 ± 0.20 and 0.38 ± 0.05 g (n = 7) in SO, 7.65 ± 0.11 and 0.37 ± 0.04 g (n = 7) in sepsis, 7.50 ± 0.11 and 0.50 ± 0.04 g (n = 7) in SO + EPO, and 7.71 ± 0.08 and 0.44 ± 0.03 g (n = 6) in CLP + EPO-treated septic mice, respectively.
Effect on Endothelium-dependent Relaxation in Septic Mice
Figure 7A illustrates the endothelium-dependent relaxation to ACh (1 nmol/L-10 µmol/L) in PE (0.1-10 µmol/L)-precontracted mouse aorta from different groups. Acetylcholine-induced relaxation was significantly (P < .05) attenuated in septic mice (58.66% ± 5.89%; pD2 6.87 ± 0.12; n = 6) compared to SO + vehicle-treated mice (100.8% ± 2.7% and pD2 7.46 ± 0.10; n = 6). Concentration-dependent relaxation to ACh was significantly (P < .05) improved in the septic mice treated with EPO (Emax 83.4% ± 3.2% and pD2 7.47 ± 0.09; n = 7). Erythropoietin treatment had no significant effect on ACh-induced, endothelium-dependent relaxation in SO mice (SO + EPO, 83.51% ± 3.41%, and pD2 7.23 ± 0.07, n = 6 vs SO, 100.8% ± 2.7%; pD2 7.46 ± 0.10; n = 6).

Effect of erythropoietin (EPO) posttreatment on sepsis-induced endothelial dysfunction in mouse aortic rings. Line diagrams depict the mean concentration–response curves of acetylcholine (ACh) (A) and sodium nitroprusside (SNP; B) on the aortic rings obtained from different groups. Note that sepsis significantly attenuated endothelium-dependent, ACh-induced relaxation while it had no effect on nitric oxide (NO) signaling in vascular smooth muscle (SNP-induced relaxation). Treatment with EPO at 1 hour after surgery significantly preserved the ACh-induced relaxation. Vertical bars represent standard error of the mean (SEM). Data were analyzed by 2-way analysis of variance (ANOVA) followed by Bonferroni posttests. Bar diagrams showing the effect of EPO posttreatment on sepsis-induced reduced aortic endothelial NO synthase (eNOS) mRNA expression (C) and eNOS-derived nitrate level in aorta (D). Treatment with EPO at 1 hour after surgery significantly reversed sepsis-induced reduced aortic eNOS mRNA expression and eNOS-derived aortic nitrate level. Data were analyzed by 1-way ANOVA followed by Newman-Keuls multiple comparison test. *P < .05 in comparison with sham-operated (SO) +vehicle; # P < .05 in comparison with CLP + vehicle. mRNA indicates messenger RNA.
Effect on Endothelium-Independent Relaxation in Septic Mice
In order to evaluate the contribution of endothelial NO signaling in vascular smooth muscle to impaired endothelium-dependent relaxation in sepsis, we studied the relaxant effect of endothelium-independent nitrovasodilator SNP in aortic rings from different groups. Figure 7B shows the cumulative dose–response curves of SNP (0.1 nmol/L-0.1 µmol/L) in PE (0.1-10 µmol/L)-precontracted aortic rings. The pD2 and Emax values of SNP in SO + vehicle-treated mice were 8.07 ± 0.05 and 104.95% ± 4.36% (n = 9), respectively. Sepsis had no significant effect on SNP-induced smooth muscle relaxation (pD2 7.96 ± 0.10 and Emax 115.88% ± 6.05%; n = 9). Smooth muscle relaxation to SNP was not altered in septic mice treated with EPO (Emax 112.25% ± 1.34% and pD2 7.86 ± 0.08; n = 9) and SO + EPO (Emax 119.01% ± 5.77%; pD2 7.82 ± 0.04; n = 9) when compared to SO + vehicle-treated mice.
Effect on eNOS mRNA Expression in Mouse Aorta
In order to find out whether the reduced endothelium-dependent relaxation in sepsis is due to its effect on eNOS gene, we studied the expression of eNOS gene in mouse aorta from different groups. As illustrated in Figure 7C, sepsis significantly (P < .001) reduced the eNOS mRNA expression in aorta to 0.19 ± 0.08-fold (n = 4) in comparison to SO controls. Treatment with EPO significantly (P < .05) restored the eNOS mRNA expression (0.61 ± 0.17; n = 4) in septic mice. However, treatment with EPO (0.86±0.14; n = 4) in SO mice produced no significant change in eNOS mRNA expression when compared to SO mice.
Effects on eNOS-Derived Nitrate Level in Aortic Homogenate
To establish whether the increase in eNOS gene expression correlates with the product of the eNOS gene, we measured the eNOS-derived nitrate level in mouse aorta from different groups. Figure 7D depicts the effect of different treatments in sepsis on constitutive total nitrate level in mouse aorta. The eNOS-derived nitrate content in aorta from SO + vehicle-treated animals was 31.22 ± 1.75 pmol/mg wet tissue (n = 6). Sepsis significantly (P < .001) decreased the constitutive nitrate content to 13.68 ± 1.41 pmol/mg wet tissue (n = 6). Treatment with EPO significantly (P < .001) increased the nitrate level to 29.13 ± 1.00 pmol/mg wet tissue (n = 6). In SO mice, EPO did not cause any change in constitutive nitrate content (30.06 ± 2.25 pmol/mg wet tissue; n = 6) when compared to SO + vehicle-treated mice.
Discussion
The major findings of the present study are as follows: (1) EPO treatment after onset of sepsis in mice significantly increased the survival time in the 72-hour observation period, (2) preserved the vasoconstrictor response to NE in aorta impaired by sepsis, (3) increased the aortic α1D mRNA as well as protein expression and attenuated GRK-2 expression in sepsis, (4) decreased the upregulation of iNOS as well as iNOS-derived basal NO release and tissue content of NO in aorta caused by sepsis, and (5) restored endothelial dysfunction which was evident from the preservation of ACh-induced relaxation, eNOS-derived NO release/nitrate, and rise in eNOS mRNA expression.
In a randomized study involving critically ill patients, EPO treatment was associated with lower mortality, 27 and it was attributed to cytoprotective, antiapoptotic, and anti-inflammatory effects. 28 Roberta and coworkers 29 demonstrated the beneficial effects of a low dose of recombinant EPO in rodent models of endotoxemia. Additionally, the administration of a large dose of EPO in lipopolysaccharide (LPS) model of sepsis improved survival, and the beneficial effects were associated with inhibition of apoptosis, NO production, and tissue hypoxia. 10 As observed in the present study, EPO posttreatment significantly improved the survival time in CLP-induced sepsis in mice, which is in agreement with survival benefit observed with EPO posttreatment in CLP model of sepsis in an earlier study. 10
An increased blood lactate level is widely believed to be a marker of inadequate oxygen delivery and anaerobic metabolism. 30 Furthermore, the rate of decline in lactate concentration (lactate clearance) has been recommended as an end point of early goal-directed therapy in critically ill patients with sepsis. As evidenced in the present study, an elevated serum lactate level in sepsis indicates severity of disease, and EPO posttreatment reverses the severity by reducing the serum lactate level.
Septic shock is associated with profound refractory hypotension that necessitates administration of vasopressor agents to maintain arterial pressure. Drugs such as NE, dopamine, and vasopressin are commonly used in clinical conditions of sepsis in humans to maintain hemodynamic functions. 31 –33 In the present study, we investigated whether EPO treatment can reverse the hyporeactivity of mouse aorta to NE and if so what are the mechanisms involved. According to our findings, sepsis significantly inhibited the maximal response to NE with a significant change in the agonist affinity for the α-adrenoceptors. We postulated that the suppression of the NE-induced contraction in the aorta may involve (1) the lower expression of the α1D-adrenoceptors and/or (2) desensitization of these receptors due to an increased plasma concentration of NE. Hosoda and coworkers 34 demonstrated the presence of mRNA for α1A, α1B, α1D adrenoceptors in the mouse aorta. According to their study, the rank order of each subtype in terms of its vasoconstrictor action was in good agreement with the level of mRNA expression, that is, α1D > α1B > α1A adrenoceptor. As α1D AR subtype is most abundantly present 34,35 and contributes to the vasoconstrictor response 16,36 in mouse aorta, we evaluated the influence of EPO treatment on this receptor subtype. Previous studies from our laboratory demonstrated downregulation of α1D adrenoceptor in septic mice. 13 In the present study, we observed that posttreatment with EPO reversed the downregulation of α1D mRNA and protein expression, which is consistent with the restoration of the aortic contractile action of NE in sepsis.
G protein-coupled receptors are modulated by a family of specific GRKs. Receptor phosphorylation by GRKs is followed by binding of uncoupling proteins called β-arrestins leading to loss of receptor responsiveness, a process called desensitization. 37,38 G protein receptor kinase-2 is a ubiquitous member of the GRK family that is able to phosphorylate a variety of GPCRs in an agonist-dependent manner, suggesting its participation in the modulation of multiple signaling pathways. The key role of GRK2 in GPCR desensitization mechanisms suggests that changes in the expression or function of this protein may affect the efficacy of signal transduction. Reports have shown that GRK2 function is of major relevance for cardiovascular physiology. 39–40 Recently, Thangamalai and coworkers 41 demonstrated the downregulation of GRK2 mRNA as well as protein in septic heart. As evidenced in the present study, sepsis caused downregulation of GRK2 mRNA expression in the mouse aorta. To establish a link between GRK2 mRNA expression and the desensitization mechanism, we used GRK2 inhibitor ex vivo to examine its effect on the aortic contractile response to NE in sepsis. In accordance with our hypothesis, GRK2 inhibitor restored the vascular reactivity to NE in sepsis. However, it had no significant effect on NE responses in tissues obtained from SO, EPO-treated SO, and EPO-treated septic mice. The lack of effect of the GRK2 inhibitor in these groups unlike that of the CLP group suggest that there is a definite link between expression of GRK2 and modulation of NE responses by the inhibitor. For example, we found a profound increase in GRK2 mRNA expression only in the CLP mice, whereas the gene expression in EPO-treated groups was comparable to SO animals. These observations clearly demonstrate that the α1D adrenoceptor present in the mouse aorta is a substrate for GRK2-induced phosphorylation and desensitization in sepsis. Our hypothesis is supported by a previous study in mouse aorta, which demonstrated that α1D AR is desensitized by GRK2. 16 Garcia-Sainz and coworkers 15 have also reported that phosphorylation and desensitization of α1D ARs are increased following stimulation with NE in rat fibroblast in both whole cell and membrane preparations. An interaction between GRK2 and Gβγ subunit of GPCRs is an established mechanism of receptor desensitization. 42 Gallein inhibits Gβγ subunit-dependant signaling and has been used to interrupt the interaction between GRK and Gβγ subunit, thereby reversing the mechanisms of GPCR desensitization. 43 In the present study, we documented that gallein partially reversed the hyporeactivity to NE in the aorta taken from CLP mice. This further strengthens our hypothesis that GRK2 and Gβγ subunit pathways play a significant role in the hyporeactivity of mouse aorta to NE in sepsis.
Nitric oxide overproduction due to iNOS induction has been proposed as an important pathway leading to vascular hyporeactivity to vasopressor drugs and resultant hypotension in LPS 44 and peritonitis models of shock. 45 However, the involvement of iNOS-derived NO pathway in vascular hyporeactivity and hypotension is controversial. For instance, in the rat model of peritonitis, low level of expression and limited role for the iNOS in the vascular hyporeactivity have been demonstrated. 46 We observed that in the mouse model of sepsis, 1400 W, a selective inhibitor of iNOS, completely preserved the contractile responses to NE in sepsis. Thus, there was no significant difference in the NE responses between SO and septic animals. The functional responses assessed in the presence of 1400 W corroborate with the overexpression of iNOS mRNA and overproduction of iNOS-derived NO in sepsis. Further, the lack of effect of 1400 W in augmenting aortic contractions in tissues taken from SO, SO + EPO, and CLP + EPO corresponds to identical mRNA expression/NO production in these groups. Our observations with EPO are supported by a previous study from our laboratory looking at the effects of 1400 W on NE responses in CLP model of mice. 13
The endothelium is known to play an important role in regulating vascular tone through the generation of several potent vasoactive substances such as NO and/or prostaglandin I2. Nitric oxide is the primary endothelium-derived vasodilator that mediates the relaxation response to ACh in mouse aorta. 19 Kandasamy and coworkers 13 observed impaired endothelium-dependent vasodilation in mouse model of sepsis. As observed in the present study, the inhibition of ACh-induced endothelium-dependent relaxation was related to decreased eNOS mRNA expression and reduced endothelial NO production in the aorta from septic mice. Then the question arises if sepsis affected endothelial NO-mediated relaxation by influencing nitric oxide/soluble guanylyl cyclase/cyclic guanine monophosphate (NO/sGC/cGMP) signaling at the aortic smooth muscle level. We observed that sepsis did not influence SNP-induced endothelium-independent relaxation in the aorta. Therefore, NO signaling at the vascular cell level is not influenced by sepsis in the mouse model. This observation is supported by a previous report from our laboratory in the rat sepsis model. 21 Results of the present study showed that EPO markedly preserved ACh-induced relaxation in the aorta from septic mice. This is consistent with the restoration of eNOS mRNA expression and reversal of decreased eNOS-derived NO release and tissue NO content in sepsis. Our findings are in agreement with improved ACh-induced relaxation and NO release in the aorta from LPS-treated rats receiving EPO. 29
Clinical Relevance
In conclusion, EPO increased the survival time, preserved the vasoconstrictor response to NE, and endothelial function. Improvement in vascular functions may partly explain the survival benefit of EPO in sepsis. The present investigation in the mouse model of sepsis is of sufficient clinical relevance for further studies on EPO as an adjunct therapy in the management of sepsis.
Footnotes
Author Contribution
K. Kandasamy and S. K. Mishra contributed to conception and design; acquisition, analysis, and interpretation; drafted the article; critically revised the article; gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. S. Choudhury, V. Singh, S. A. Darzi, K. K. Jaya, T. Kumar, and F. Sultan contributed to analysis. M. P. Addison contributed to acquisition and analysis. R. Thangamalai and J. R. Dash contributed to acquisition and interpretation. T. U. Singh and S. Parida contributed to conception and design; acquisition, analysis, and interpretation; drafted the article, critically revised the article, and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Author(s) received financial support from Indian Veterinary Research Institute, Izatnagar, Uttar Pradesh, India for this research.
