Abstract
Acute heart failure, advanced cardiac failure, cardiac surgery, and sepsis are conditions that require simultaneous treatment to stimulate contractility and/or reduce systemic vascular resistance, with levosimendan and milrinone being treatment options. This research's aim is to review the current indications and evidence for these medications across various scenarios. Evidence suggests that levosimendan is a non-inferior alternative to dobutamine and superior to milrinone in treating low cardiac output syndrome following cardiac surgery. In cases of septic shock, levosimendan has been linked to lower mortality rates compared to placebo, while milrinone's efficacy remains inconclusive. Furthermore, postoperative patients undergoing correction for congenital heart disease have shown reduced mechanical ventilation time and intensive care unit stays when treated with levosimendan, although differences exist between the populations assigned to each intervention. In conclusion, levosimendan, compared to milrinone, appears to offer better hemodynamic favorability in patients undergoing cardiac surgery. However, additional research is necessary to further understand its impact on hemodynamic outcomes, mortality, intensive care unit, and hospital stays in patients with cardiogenic shock of both ischemic and non-ischemic etiologies, as well as septic shock.
Keywords
Background
For patients with cardiovascular disease, finding a drug that simultaneously boosts heart contractility and reduces systemic vascular resistance (SVR) can be a critical decision.1,2 While other options like combining vasoactive drugs exist, the increasing prominence of milrinone and levosimendan has made them first-line choices.2,3 The use of inodilators is especially important in patients with chronic heart failure or pulmonary hypertension associated with right heart failure. However, there is increasing evidence regarding the use of these medications in different scenarios such as septic cardiomyopathy, postoperative cardiovascular surgery, and even ventilator weaning in patients at high risk of extubation failure.3,4 Although evidence in these scenarios is still limited and few clinical practice guidelines consider them as first-line, these medications could have a beneficial effect based on the pathophysiology, pharmacokinetic and pharmacodynamic (PK/PD) characteristics of each population.1‐5
Unlike dobutamine, an inotrope widely used in clinical practice, milrinone and levosimendan do not influence adrenergic receptors, which could decrease the risk of adverse events associated with adrenergic stimulation.4,6 Additionally, they are not affected by the decreased expression of adrenergic receptors in patients with chronic use of beta-blockers.2,3 Furthermore, the pharmacokinetics of these medications and their interaction with potassium channels and their effect on phosphodiesterase 3 confer beneficial properties such as improving myocardial energy efficiency and decreasing ventricular afterload, thereby enhancing ventricular–arterial coupling.3,5‐15
But the question remains: which drug offers greater benefit? This review tackles this question by first examining in detail the pharmacological properties, pros, and cons of both drugs, then delving into the existing research comparing them in different critical settings.
Materials and Methods
Study Design and Search Strategy
An exploratory literature search was conducted using electronic databases including PubMed and Scopus, covering the period from their inception until December 2023. This scoping systematic review followed the methodology proposed by Arksey and O'Malley, 16 later refined by Levac et al. 17 This methodology consists of 5 fundamental steps: (1) identification of the research question, (2) search for relevant documents, (3) selection of studies, (4) data extraction, and (5) summary and reporting of results. This article follows the PRISMA extension to report scoping systematic reviews (PRISMA-ScR). 18
The search employed:
PubMed: ((“Milrinone/administration and dosage”[Mesh] OR “Milrinone/pharmacokinetics”[Mesh] OR “Milrinone/pharmacology”[Mesh] OR “Simendan/administration and dosage”[Mesh] OR “Simendan/pharmacokinetics”[Mesh] OR “Simendan/pharmacology”[Mesh] OR “Cardiac Surgical Procedures”[Mesh] OR “Critical Care”[Mesh] OR “Heart Failure/drug therapy”[Mesh] OR “Heart Failure/physiopathology”[Mesh] OR “Heart Failure/prevention and control”[Mesh]) AND (“Anesthesia, Cardiac Procedures”[Mesh] OR “Anesthesia, Cardiac Procedures/adverse effects”[Mesh] OR “Anesthesia, Cardiac Procedures/psychology”[Mesh])) Filters: from 1000/1/1 - 2023/12/1 Scopus: (TITLE-ABS-KEY (milrinone) OR TITLE-ABS-KEY (simendan AND levosimendan) AND TITLE-ABS-KEY (cardiac AND surgical) AND TITLE-ABS-KEY (critical AND care) AND TITLE-ABS-KEY (heart AND failure) OR TITLE-ABS-KEY (anesthesia, AND cardiac)) AND PUBYEAR > 1990 AND PUBYEAR < 2024
Inclusion Criteria
The inclusion criteria were structured around the PICO (Population, Intervention, Comparison, Outcome) framework.
Population: Patients with acute heart failure, cardiogenic shock, cardiovascular surgery, and septic shock. Intervention: Pharmacological treatment with levosimendan and/or milrinone. Comparison: Levosimendan compared to milrinone. Outcomes: The review aimed to summarize the clinical pharmacology and PK/PD of levosimendan and milrinone, as well as evidence related to hemodynamic outcomes, mortality rates, intensive care unit (ICU) and hospital stay durations, and days of mechanical ventilation.
The review included research articles, literature reviews, randomized clinical trials (RCTs), systematic reviews, and meta-analyses. Figure 1 illustrates the PRISMA-ScR flow diagram of the articles included in the review.
18
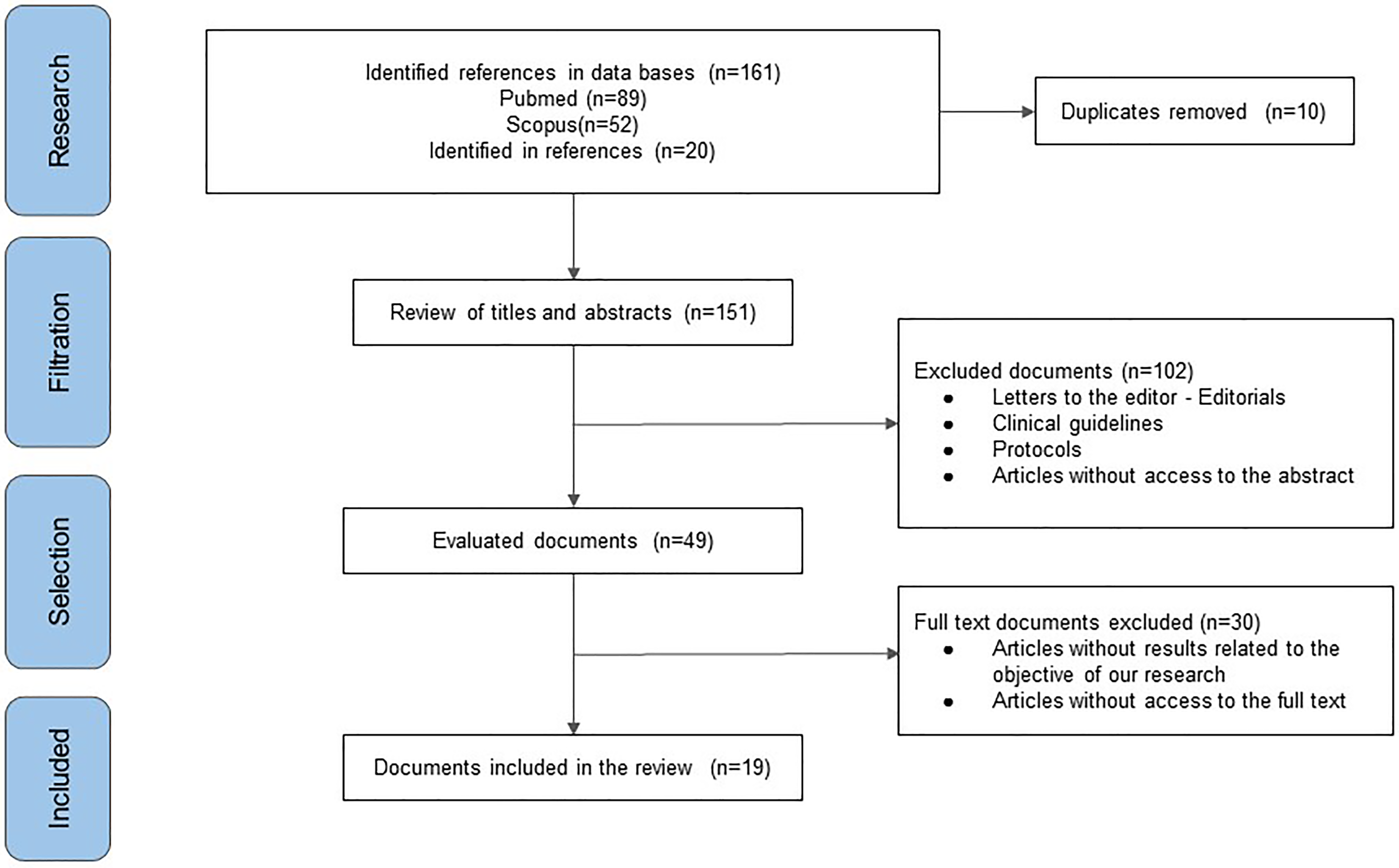
PRISMA-ScR flowchart of the included publications.
Study Selection and Data Extraction
Study titles and abstracts retrieved from the search were independently screened by 4 authors (AQA, HRA, ETQ, AB). Relevant data, including study characteristics (authors, year of publication), population details (number of cases), evaluated pathology (acute heart failure, cardiogenic shock, cardiovascular surgery, septic shock), and hemodynamic characteristics, diagnostic features, and treatment types were extracted. The accuracy of data extraction was verified by another author (HRA, JOCB) who also contributed clinical and scientific insights.
Data Synthesis
Data from the selected studies were extracted using a standardized template. The synthesis focused on evaluating mortality, ICU and hospital stay durations, and hemodynamic outcomes.
Levosimendan: Pharmacokinetics and Pharmacodynamics
Levosimendan is the active enantiomer of simendan, a compound derived from dinitrile-pyridazinone. Three mechanisms have been described as the main physiologic effects of levosimendan and of its active metabolite OR-1896: Inotropism, vasodilation, and cardiac cytoprotection.1,2 The first of the 3 is thought to be mediated through a link between the drug and cardiac troponin C (cTnC). This interaction increases sensitivity towards calcium and prolongs the interaction of cTnC and cardiac troponin I (cTnI). As a result, cardiac contractility is augmented without affecting serum calcium levels. 3 The latter aspect differentiates levosimendan with other inotropic agents known to increase calcium levels, an effect that has been associated with cardiac remodeling, arrhythmias, and increased oxygen consumption.3,4 Secondly, both venous and arterial vasodilations are processes mediated through the opening of ATP-dependent potassium channels in smooth muscle cells, inducing hyperpolarization and thus vasodilation. 5 Lastly, a cardioprotective mechanism of levosimendan has been proposed through the observation of diminished reactive oxygen species (ROS) following the aperture of mitochondrial ATP-dependent potassium channels. 6
In spite of the aforementioned mechanisms, the use of levosimendan increases cardiac output (CO) and stroke volume (SV), reduces mean arterial pressure (MAP), pulmonary wedge pressure (PWP), and peripheral vascular resistance (PVR). 7
The pharmacokinetics of levosimendan show a short half-life elimination of approximately 1 h; however, its most active metabolite, OR-1896, reaches a peak half-life elimination between 70 and 80 h after administration. 8
Finally, the most important side-effects reported from levosimendan use include hypotension, headache, and dizziness. Although controversial, some studies suggest that levosimendan increases the incidence of atrial fibrillation (AF) in comparison to dobutamine.9,10 The differences in pharmacokinetics between levosimendan and milrinone are described in Table 1.
Pharmacokinetics Differences Between Levosimendan and Milrinone.
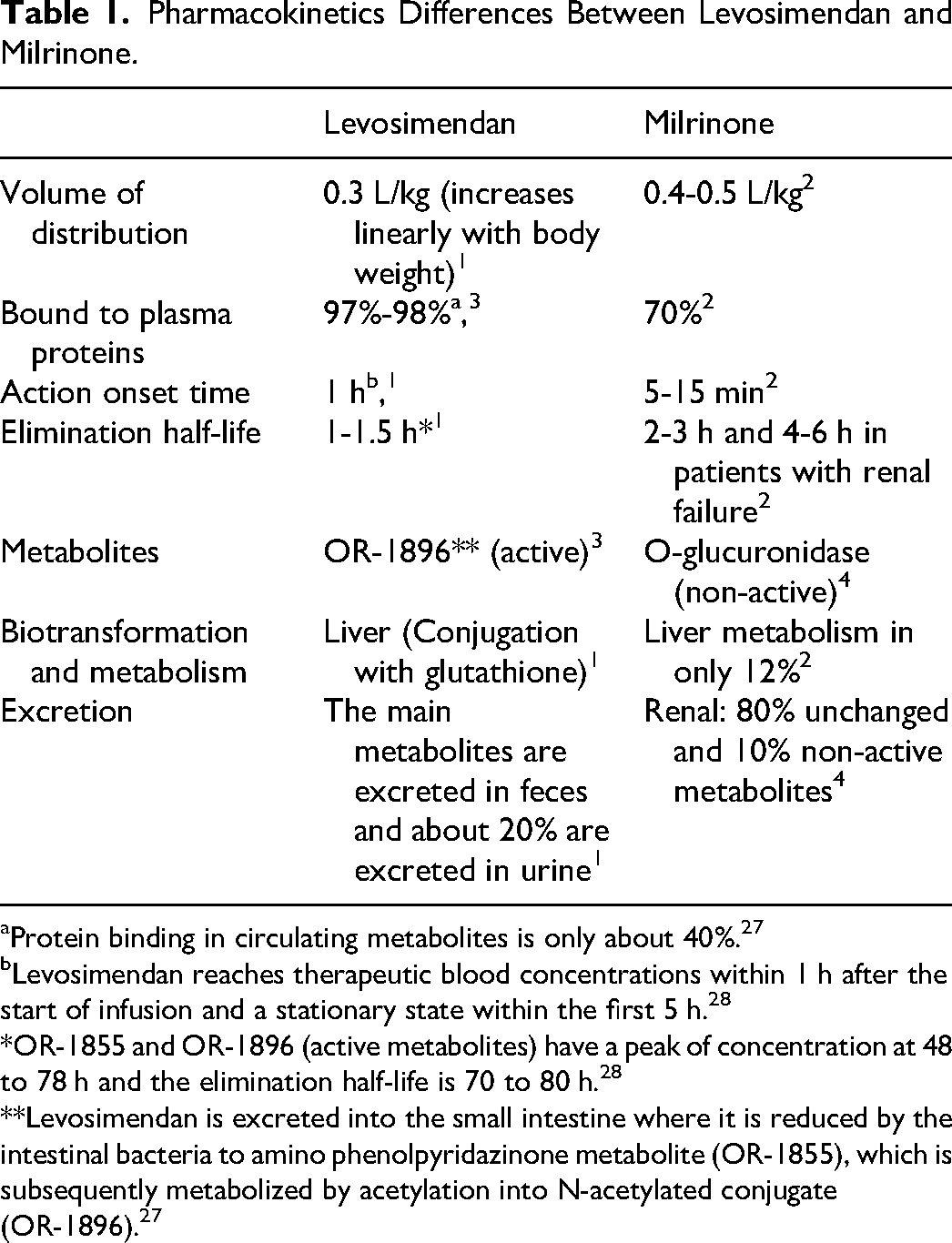
Protein binding in circulating metabolites is only about 40%. 27
Levosimendan reaches therapeutic blood concentrations within 1 h after the start of infusion and a stationary state within the first 5 h. 28
*OR-1855 and OR-1896 (active metabolites) have a peak of concentration at 48 to 78 h and the elimination half-life is 70 to 80 h. 28
**Levosimendan is excreted into the small intestine where it is reduced by the intestinal bacteria to amino phenolpyridazinone metabolite (OR-1855), which is subsequently metabolized by acetylation into N-acetylated conjugate (OR-1896). 27
Current Uses of Levosimendan
Acute Heart Failure
Levosimendan has been compared with placebo and with other vasoactive agents such as dobutamine in the context of acute heart failure (HF). The LIDO study compared short-term outcomes comparing a 24-h infusion of levosimendan and dobutamine in a randomized clinical trial with over 200 patients presenting with acute HF. The primary outcome was hemodynamic improvement defined as at least a 30% increase in CO and a decrease of 25% or more in pulmonary capillary wedge pressure. While dobutamine was infused in a constant fashion of 5 mcg/kg/min, levosimendan was infused at a rate of 0.1 mcg/kg/min with a previous loading dose of 24 mcg/kg, both for 24 h. Results showed a superior outcome for levosimendan which both reduced pulmonary capillary pressure compared with dobutamine and decreased mortality at 180-day follow-up (levosimendan 26% vs dobutamine 38%, P = .002). 11
The REVIVE-II study randomized 600 patients with HF with a left ventricular ejection fraction (LVEF) < 23% and resting dyspnea to receive either standard therapy or standard therapy plus levosimendan. The primary outcome was patient-referred symptom relief and total hospital stance. Both primary outcomes were superior for the levosimendan group compared with standard therapy alone. Additionally, there was a decrease in the need for rescue therapy in the levosimendan group (15% vs 26%). Nevertheless, adverse effects were higher in the intervention group with levosimendan, particularly regarding hypotension (49.2% vs 14.6%), headache (29.4% vs 14.6), ventricular tachycardias (VT) (24.1% vs 16.9%), and AF (8.4% vs 0.2%). 12
The RUSSLAN study (n = 504) analyzed the efficacy and safety of a 6-h levosimendan infusion in patients with ventricular dysfunction secondary to myocardial infarction (MI). It was found that, compared with placebo, a 0.1 to 0.2 mcg/kg/min 6-h infusion of levosimendan significantly reduced progression to overt HF and reduced mortality in all pre-stablished time measures: 24 h, 14 days, and 180 days (4.0% vs 8.8%. P = .04; 11.7% vs 19.6% P = .03; and 22.6% vs 31.4% P = .05 respectively). In this case it is important to mention that there was no statistically significant difference regarding ischemia and hypotension as adverse reactions in both groups (P = .319). 13
Lastly, the SURVIVE study (n = 1327) compared the effects of levosimendan and dobutamine in hospitals from 9 countries who had acute HF with inotropic support requirements. As main result of the trial, levosimendan and dobutamine had no statistically significant differences in mortality at 180 days (P = .40). Additionally, there was a greater reduction in natriuretic atrial peptide (BNP) in patients who received levosimendan. 14
While this information might not have been encompassed within the original literature review, the European Society of Cardiology advises that, owing to its non-adrenergic mechanism, levosimendan could be a preferred option over dobutamine for patients with acute heart failure undergoing beta-blocker therapy.11‐15
Levosimendan in Advanced Heart Failure
The LION-HEART study, a multicentric double-blind, randomized clinical trial that included patients with advanced HF being treated in outpatient facilities, compared the efficacy and safety of intermittent levosimendan every 2 weeks for 12 weeks. The main outcome was the concentrations of NT-proBNP, which was significantly lower in the levosimendan group when compared with placebo (P = .003). Additionally, the mean hospitalization stance was lower in the levosimendan group (P = .001). 15
Garcia et al published the LAICA study in 2021 with the objective of evaluating the long-term effectiveness and safety of intermittent infusion in patients with advanced HF. They administered 0.1 µg/kg/min of levosimendan as a continuous 24-h intravenous infusion once monthly for 1 year. It was evidenced that patients in the levosimendan group had a lower cumulative incidence of HF decompensation and/or death at 1 and 3 months than the placebo group (5.7% vs 25.9% [P = .004] and 17.1% vs 48.1% [P = .001], respectively), but not at 6 and 12 months. Survival probability was significantly higher in patients who received levosimendan compared with those who received placebo (log rank: 4.06; P = .044). 19
In 2023, the Pözl group conducted the LeoDOR trial evaluating the efficacy of intermittent levosimendan therapy in the vulnerable phase following hospitalization for acute HF. In patients with LVEF ≤30%, levosimendan was administered either at 0.2 µg/kg/min for 6 h every 2 weeks or as a continuous infusion for 24 h at a rate of 0.1 µg/kg/min every 3 weeks. Among patients recently hospitalized with HF and reduced LVEF, intermittent levosimendan therapy did not improve post-hospitalization clinical stability. 20
The varied outcomes observed within this demographic underscore the necessity for further studies aimed at elucidating the impact of levosimendan usage in patients presenting with advanced HF.
Levosimendan in Cardiac Surgery
In a meta-analysis by Maharaj et al involving 729 patients requiring inotropic support after coronary artery bypass grafting (CABG) or percutaneous coronary intervention (PCI), levosimendan demonstrated a mortality benefit compared to placebo, dobutamine, and milrinone. Additionally, it reduced inpatient stay and the incidence of postoperative AF. 21
Another meta-analysis, including over 3100 patients with left ventricular dysfunction undergoing cardiac surgery, found that prophylactic levosimendan significantly reduced mortality (OR: 0.67; 95% CI: 0.49-0.91; P = .0087) compared to the control group. Levosimendan also led to a lower incidence of low cardiac output syndrome (LCOS) and reduced rates of acute kidney injury (AKI) and renal replacement therapy (RRT). 22
The clinical trials CHEETAH 1, 23 LICORN, 24 and LEVO-CTS 25 investigated the use of levosimendan in patients undergoing cardiac surgery requiring hemodynamic support. They found that, in patients with low ejection fraction, adding levosimendan to standard care did not reduce 30-day mortality compared to placebo. There were also no significant differences in the need for prolonged catecholamines, mechanical left ventricular assist devices, or renal replacement therapy between patients treated with levosimendan and those who received placebo. Additionally, levosimendan prophylaxis did not decrease the rate of adverse events compared to placebo in patients with reduced ejection fraction undergoing cardiac surgery with cardiopulmonary bypass.
Levosimendan in Sepsis
Cardiac dysfunction is a frequent complication among patients with sepsis. This condition usually develops in the first 3 days from the onset of shock and resolves near the 10th day when the septic state has ceased. 26
In a randomized control trial conducted by Morelli et al, patients in septic shock who, after 48 h, had a LVEF < 45% despite adequate support therapy, were assigned to receive dobutamine or levosimendan. Levosimendan showed an increase in systolic volume (SV), an increased systolic index (SI), cardiac index (CI), and left ventricle work index. Additionally, urinary output, creatinine clearance, and lactate concentrations decreased. 27
Zengzheng et al conducted a meta-analysis of 11 studies (n = 1044) with the aim of assessing the efficacy of levosimendan in patients with sepsis or septic shock. Following the administration of levosimendan, there were no significant differences between the groups in 28-day mortality (34.9% and 36.2%; OR: 0.93; 95% CI: 0.72-1.2). However, in the levosimendan group, patients exhibited a significant improvement in CI, LVEF, and serum lactate levels. 28
Zangrillo et al in 2015 conducted a meta-analysis including 7 randomized trials on the use of levosimendan in patients with severe sepsis and septic shock, comprising a total of 246 patients. The use of levosimendan was associated with a significantly reduced mortality compared to standard inotropic therapy. Specifically, in the levosimendan group, 59 out of 125 patients (47%) died, whereas in the control group, 74 out of 121 patients (61%) died (risk difference = −0.14, risk ratio = 0.79 [0.63-0.98], P = .03, numbers needed to treat = 7). Furthermore, it was also found that in the levosimendan group, lactate levels were lower, and patients had a higher CI. 29
The LeoPARDS RCT, a multicenter, randomized, double-blind, parallel-group, placebo-controlled study conducted in UK intensive care units, sought to evaluate the efficacy and safety of levosimendan in adult patients with septic shock.30,31 Despite its potential as a calcium-sensitizing drug with inotropic properties, the addition of levosimendan to standard care did not demonstrate a reduction in organ dysfunction or mortality compared to placebo. Moreover, patients receiving levosimendan showed a decreased likelihood of successful extubation and an increased risk of supraventricular tachyarrhythmias. This trial highlights the need for further research, including comparative studies against alternative inotropes, such as dobutamine, to determine the optimal management of septic shock in patients with low cardiac output.
Levosimendan and Renal Considerations
Forty patients diagnosed with end-stage HF and awaiting heart transplant were randomly assigned to receive either levosimendan (a 12 mcg/kg loading dose followed by a 0.1 mcg/kg/min infusion for 24 h) or placebo in a study by Zemljic et al. After a 3-month follow-up, the levosimendan group displayed lower blood creatinine levels compared to placebo (1.6 +/− 0.26 mg/dL vs 1.90 +/− 0.14 mg/dL; P = .05). 32 This finding aligns with the LIDO study (n = 203), a separate study that also showed a reduction in serum creatinine as a secondary outcome when levosimendan was compared with dobutamine (P = .03). 11
While further research is needed to confirm the long-term implications, these results suggest that levosimendan may offer potential benefits for kidney function in patients with severe HF.
Levosimendan and Respiratory Support Weaning
In 2021, Chao Luo et al conducted a meta-analysis investigating the efficacy of levosimendan in facilitating weaning from mechanical ventilation and extracorporeal membrane oxygenation (ECMO) using the Newcastle-Ottawa Scale, with 60% of the included studies classified as moderate quality. Levosimendan effectively improved weaning rates in both ventilated patients with a low LVEF (OR: 2.04; 95% CI: 1.25-3.34) and patients with ECMO for cardiogenic shock (OR: 1.98; 95% CI: 1.34-2.91), as defined by successful discontinuation of mechanical ventilation within a specified timeframe. These findings suggest potential benefits of levosimendan for weaning in patients with these critical conditions, although further research with higher-quality studies is needed to confirm long-term efficacy and safety. 33
Milrinone: Pharmacokinetics and Pharmacodynamics
Milrinone is an inotropic agent with a dipyridinic structure and without catecholaminergic activity. It acts through the inhibition of phosphodiesterase 3 (PDE-3) which in turn increases cyclic adenosine diphosphate (cAMP). Cyclic AMP facilitates cellular influx of calcium ions through activation of protein kinase A (PKA) and thus increases inotropism. In vascular smooth muscle, cAMP inhibits the myosin light chain-kinase from adding a phosphate group to myosin, therefore, promoting the relaxed state of smooth muscle cells in peripheral vasculature. 34
Administration of milrinone is commonly done through a 0.3 to 0.7 mcg/kg/min continuous infusion. Its onset of action rounds the 10-min mark, and peak action is usually reached between 30 and 60 min. It presents a high binding rate with plasmatic proteins (77%-89%) with a volume of distribution ranging from 0.4 to 0.45 L/kg. Its half-life is of 2 to 2.5 h, and its clearance is 0.13 to 0.14 L/kg/h. Most of milrinone and its secondary metabolites are excreted through urine via a 0.3 L/min rate, and even 90% of the infused dose can be measured after 8 h of administration. 35 In the myocardium, milrinone increases systolic function through its inotropic effect and has a reflex chronotropic increase due to its vasodilating activity. Pulmonary vascular resistance decreases, diminishing work and afterload on the right ventricle, and SVR also decreases, increasing CO and SV, as well as lowering the myocardial oxygen consumption (VO2). Additionally, a positive lusitropic effect has been observed with milrinone, and thus it has been hypothesized that it might improve diastolic dysfunction. 36
In the myocardium, milrinone increases systolic function through its inotropic effect and has a reflex chronotropic increase due to its vasodilating activity. Finally, the most common adverse effects that follow the use of milrinone are risk of hypotension, sinus bradycardia, rebound hypertension, oliguria, hemorrhage, and myocardial dysfunction in patients with a LVEF < 40% 35 (Table 1).
Milrinone and Acute Heart Failure
A retrospective study conducted by Kelly et al included 135 patients from a single center in whom milrinone and dobutamine were used for the treatment of acute HF. Dobutamine was associated with a 24% increase on mortality compared with milrinone, which on the other hand, was associated with an increased ICU stance (P < .01). 1
A double-blind randomized study included 23 patients with acute HF either NYHA III and LVEF > 50% who at least had been hospitalized once in the past 12 months compared milrinone with placebo. The primary outcome was safety of the intervention measured by changes in hemodynamic parameters, mainly HR (P = .94), SBP (P = .57), and DBP (P = .29). As a secondary outcome, symptom evaluation questionnaires were assessed, particularly the Kansas Cardiomyopathy Questionnaire, showing an improvement (+10 +/−1 3 vs −3 +/−15; P = .046). Additionally, the 6-min walk test did not show a statistically significant difference (+22 [−8 to 49] vs −47 [−97 to 12]; P = .092). Finally, no significant echocardiographic changes nor pro-arrhythmic effect was seen with the use of milrinone.37,38
A systematic review and meta-analysis, comprising 3 randomized controlled trials (RCTs) (I2 = 0%) involving over 300 patients with myocardial infarction (MI) and acute heart failure showed significant increases in LVEF (P < .001) and cardiac output (CO) (P = .002). However, no differences were found in mortality (P = .16), end-diastolic left ventricle (EDLV) dimensions (P = .06), heart rate (HR), or blood pressure (BP) with the use of milrinone. Nevertheless, the study is limited by its sample size and overall low study quality. 39
The OPTIME-CHF, a double-blind, multicentric RCT included over 900 patients with diastolic dysfunction and HF. In a 60-day follow-up, milrinone use was associated with an increase in length of stay or death (P = .055), death and rehospitalization (P = .01), and in-hospital mortality (P = .04) in patients with an ischemic etiology. On the other hand, milrinone decreased length of stay (P = .055), death or rehospitalization (P = .01), and in-hospital mortality (P = .04) in patients with non-ischemic etiology, Finally, the study suggests that there are no benefits in the routine use of milrinone in acute heart failure; however, benefits were demonstrated in those with non-ischemic cardiomyopathy, emphasizing the importance of defining the etiology of heart failure. 40
Milrinone in Advanced Heart Failure
The PROMISE study, a double-blind, multicenter randomized controlled trial involving 1088 patients with advanced left ventricular dysfunction compared the effects of 40 mg of oral milrinone daily versus placebo. Milrinone therapy, compared to placebo, was associated with a 28% increase in all-cause mortality and a 34% increase in cardiovascular mortality. These findings suggest that despite the favorable hemodynamic effects of milrinone, long-term therapy may lead to higher morbidity and mortality 41
Zewail et al 42 evaluated the hemodynamic and clinical outcomes of long-term combination therapy with intravenous milrinone and oral beta-blockers in patients with severe congestive heart failure. The mean duration of milrinone treatment in this combination treatment group was 269 days (range, 14-1026 days). Functional class improved from IV to II-III with milrinone therapy. The total number of hospital days decreased from 450 to 380 (a 15.6% reduction), and the mean length of hospital stay decreased by 1.4 days (a 14.7% reduction). Treatment-related sudden death is relatively infrequent during the combination regimen.
Milrinone and Cardiac Surgery
A double-blind RCT including 40 patients with severe pulmonary hypertension (PSAP >= 60 mm Hg) caused by mitral stenosis who underwent valvular replacement compared dobutamine + nitroglycerine versus milrinone after weaning of CPB. Patients with a LVEF < 40% were excluded. There was no difference in mortality risk using EUROscore II. The group using dobutamine + nitroglycerine had a greater MAP reduction (P < .05), a greater increase in CO (P < .05), and a greater increase in venous oxygen partial pressure (P < .05), when compared with milrinone. On the other hand, the group allotted to milrinone had a greater reduction in central venous pressure (CVP) (P < .05), a greater reduction in mean pulmonary arterial pressure (MPAP) (P < .05), a decrease in pulmonary capillary wedge pressure (PWCP) (P < .05), and a lower reduction in SVR (P < .05). 36
A systematic review and meta-analysis included 16 RCTs including almost 700 patients undergoing CABG on and off-pump and who were allotted to receive either milrinone or placebo. Mortality was similar between both groups, and no statistically significant difference was found in the on-pump (OR: 1.17, CI: 0.37-3.72; P = .649; I2: 0%) and off-pump (OR: 1.00, CI: 0.14-7.30; P = 1.00; I2: 0%) subgroups. Additionally, the use of milrinone was associated with a lower incidence myocardial ischemia (RR: 0.29, CI: 0.16-0.52; P < .0001; I2: 0%), and arrhythmias (OR: 0.53, CI: 0.31-0.91; P = .02; I2: 0%). There was no association between milrinone and stroke (P = .33), or AKI (P = .80). 43
Ushio et al conducted another meta-analysis including 12 RCTs with over 500 patients undergoing cardiac surgery (CABG, CABG + valve replacement, valve replacement alone, and of-pump CABG). Milrinone was evaluated pre-CPB, during CPB, and off-pump. No difference was found on mortality in short-term (P = .67). Additionally, the use of milrinone was associated with a decrease in ventricular arrhythmias (P = .01) and had no association with development of AF (P = .84). Heterogeneity, however, was considered an important limitation of the meta-analysis, so conclusions must be met with caution. 34
A 2015 meta-analysis including 4 RCTs evaluated the use of milrinone in 252 patients undergoing CABG surgery; no statistically significant difference was found regarding the requirement of intra-aortic balloon pump (IABP) (P = .06), or inotropic support (P = .62). However, there was a significant decrease of inotropic support duration (P = .010; RR: −2.84, CI: −5-0.69), mechanical ventilation duration (P = .0009; RR: 14.19%, CI: −6.52 to −1.68), and incidence of post-CABG ischemia and infarction (P < .00001; and P = .002, respectively). Finally, no difference was found regarding ICU length of stay or mortality. 44
Lastly, in a meta-analysis evaluating the outcomes of 20 RCTs compared the effect of milrinone versus the drug upon mortality in 1037 patients undergoing cardiac surgery. Majure et al found that there was no statistically significant difference in mortality either in adult (RR: 1.17, CI: 0.54-2.53; P = .7) or in pediatric population (RR: 1, CI: 0.07-14.79; P = .9). Nevertheless, in 4 of the studies, mortality was twice that of the patients treated with levosimendan without it being statistically significant (P = .3), a phenomenon perhaps explained by the studies being underpowered. 45
Milrinone in Sepsis
A 2021 big-data real-world study of MIMIC III public database included 235 patients with sepsis. Zhu et al used a propensity score matched analysis to adjust for confounding. For the primary outcome of hospital mortality there was no significant difference (OR: 0.84, 95% CI: 0.45-1.56; P = .574). More patients in the milrinone group received RRT (46.2% vs 22.4%, P = .001) and had longer length of ICU stay (20.97 ± 22.84 days vs 11.10 ± 11.54 days, P = .004) 46
Milrinone and Respiratory Pathologies
Few studies have explored the use of milrinone in respiratory pathologies. Sobhy et al conducted an RCT including 50 patients with life-threatening asthma, as defined by the 2008 NICE guidelines, to compare milrinone in addition to standard therapy (oxygen, nebulized short-acting beta agonists, short-acting muscarinic agents, intravenous corticosteroids, and magnesium sulfate) versus standard therapy alone Table 2. 47 Peak end-expiratory volume (PEEV) was assessed at both 10 min and 1 h post-treatment. The milrinone group exhibited significantly improved PEEV compared to the standard therapy group at both time points (P < .001 at 10 min; P = .002 at 1 h). Furthermore, respiratory rate and partial pressure of arterial oxygen (PaO2) significantly improved in the milrinone group (P < .001). However, there were no significant changes observed in peripheral oxygen saturation (SpO2), partial pressure of carbon dioxide (PaCO2), or pH. The milrinone group also showed a lower requirement for invasive mechanical ventilation (P = .047). While mean arterial pressure (MAP) was higher in the milrinone group at 10 min (P < .001), the difference was not statistically significant at 1 h (P < .654). 47 These findings suggest that milrinone may offer promise as an adjunctive therapy for acute life-threatening asthma exacerbations.
Available Evidence of the Uses of Milrinone and Levosimendan.

Levosimendan and Milrinone: Head-to-Head
Even though levosimendan and milrinone have not been directly compared in a thorough manner, some evidence has already been published in order to shed light upon potential benefits in different relevant contexts. These section aims to summarize the scenarios in which either levosimendan or milrinone might be preferred in spite of head-to-head comparisons as well as to show in which scenarios evidence is yet to determine if a clear benefit exists when using one over the other.
One of the main scenarios in which both drugs have been compared is cardiac surgery. In a single-center RCT, 40 patients were allotted to either receive milrinone or levosimendan after undergoing mitral or aortic valve repair and in whom pulmonary hypertension (MPAP > 40 mm Hg) and LVEF < 50% in stenosis or <60% in regurgitation) was also present. Levosimendan was associated with a statistically significant increase in heart rate (P < .016) as well as with a decreased MAP (P < .001) and a greater postoperative norepinephrine base (NEb) requirement (P < .039).48‐50
Another condition highly associated with high morbidity and mortality in patients in whom cardiac surgery was performed is LCOS. Sunny et al, compared milrinone, levosimendan, and dobutamine titrated up to a CI of >2.5 Lmin/m2 in patients who underwent elective valve replacement. Dobutamine was found superior when hemodynamic parameters were considered compared with levosimendan and milrinone when titrated up to an adequate CI 20 min after weaning of CPB (P < .05). Likewise, levosimendan and dobutamine were superior to milrinone for achieving an optimal LVEF 10 min after CPB (P < .05). Thus, authors concluded that levosimendan might be a non-inferior alternative compared with dobutamine and superior to milrinone regarding the treatment of LCOS in patients who underwent elective valve replacement surgery. 51
Jaky et al compared the effectiveness of levosimendan with milrinone for weaning patients of CPB after undergoing cardiotomy for advanced HF. In the study, the authors found that hemodynamic support with NEb was similar with levosimendan and milrinone both after weaning of CPB (0.06 vs 0.07 mcg/kg/min) as well as 24 h after (0.06 vs 0.04 mcg/kg/min). On the other hand, mortality was similar in both groups at the 28-day (35% vs 40%) and 180-day mark (50% vs 44%); supporting both effectiveness and safety of levosimendan for the weaning after CPB when compared to milrinone. 52
Both levosimendan and milrinone have been compared in myocardial revascularization surgery where hemodynamic parameters have a key role in morbidity and mortality. Amin et al randomized 60 patients who underwent CABG surgery and who had a LVEF < 40% to receive either levosimendan or milrinone after anesthesia induction. Results showed a greater LVEF after ICU discharge with levosimendan when compared with milrinone (P < .05), as well as a shorter ICU length of stay (P < .01), and a shorter requirement of mechanical ventilation in the postoperative period (POP) (P < .01), providing insight into the safety and efficacy of levosimendan in the POP of CABG surgery. 53
Controversy exists regarding the use of levosimendan and milrinone in acute HF and cardiogenic shock since some old RCTs showed an increase in mortality associated with the use of this pharmacological agent. Nevertheless, a network meta-analysis of RCTs including over 5300 patients showed no statistically significant difference in mortality with the use of levosimendan and milrinone in acute HF (OR: 1.97; IP 95%: 0.53-7.29). Additionally, no differences were found when comparing dobutamine with levosimendan (OR: 0.66; IP 95%: 0.30-1.48) or milrinone (OR: 1.30; IP 95%: 0.34-4.96). 54 Another network meta-analysis including 1800 patients presenting with cardiogenic shock, showed that among 7 possible interventions with vasoactive agents (epinephrine, NEb, dopamine, dobutamine, levosimendan, milrinone, and recombinant atrial natriuretic peptide), milrinone had the highest probability to decrease mortality (44%) followed by levosimendan (26%). 53 Likewise, in acute cardiogenic shock following acute MI, a 2020 meta-analysis conducted by Karami et al including over 2450 patients from 19 RCTs evaluated the impact of inotropic agents and vasopressors in mortality associated with the mentioned presentation. Levosimendan was associated with a lower mortality when compared with placebo and enoximone (RR: 0.69; 95% CI: 0.47-1.00) and was further confirmed by a sensitivity analysis and bias risk assessment (RR: 0.61; 95% CI: 0.41-0.90). On the other hand, milrinone had non-conclusive results when compared to dobutamine (RR: 0.20; 95% CI: 0.02-1.65). 55
Lastly, levosimendan has been compared with milrinone after correction of adult congenital heart disease. Mauriat et al compared both drugs in the said context base on a propensity scaly analysis. Results showed that levosimendan was associated with a lower duration of mechanical ventilation (P < .01), a shorter total ICU length of stay (P < .009), and with lower scores in the Vasoactive-Inotropic Score scale (P < .001). 56 It should be noted that patients randomized to receive levosimendan had a greater mean score in preoperative risk assessment scales, greater rates of left ventricular dysfunction, right ventricular dysfunction, and had required epinephrine, renal replacement therapy, mechanical ventilation, and ICU care, with a greater frequency than those randomized to receive milrinone. 56
Discussion
As extensively discussed, the use of levosimendan and milrinone has been questioned in multiple scenarios.2,3,10‐13 The first of these is the use of inodilators in acute heart failure, where these drugs compared to other adrenergic inotropes have not shown a decrease in hard outcomes such as mortality.18,19,26‐29,32
The SURVIVE study, 14 a randomized clinical trial comparing levosimendan versus dobutamine, did not demonstrate an impact on overall mortality. However, in the subgroup of patients with a history of heart failure, the decrease in mortality seemed to favor the levosimendan group, especially in the days following the suspension of the inotropic infusion. This finding corresponds to the pharmacokinetic characteristics of levosimendan, which, thanks to its active metabolite, has a half-life of around 80 h. This characteristic is especially useful in patients with decompensated heart failure and represents a significant advantage compared to adrenergic medications. Additionally, the levosimendan group significantly decreased BNP levels, which could be related to improvement in cardiac chamber filling pressures. 14
Regarding adverse events, the REVIVE study showed an increase in adverse events with the use of levosimendan. 12 However, this study combined inotropic and vasodilator treatment regimens, which could have potentiated adverse events and not necessarily be directly linked to the use of levosimendan.14,15,19‐22,29
The most recent study comparing the use of milrinone with dobutamine, published by Rebecca Mathew et al, 57 did not show differences in mortality between both groups, nor was a statistically significant difference demonstrated in hospital stay, development of acute kidney injury, or arrhythmias. However, in the OPTIME HF study, 40 an increase in hospital stay, death, and rehospitalization was found in patients with ischemic etiology heart failure who were administered milrinone versus placebo.
Due to the available evidence, it is possible to conclude that in the setting of acute heart failure, there is still no ideal inotropic agent to fit all.40,51‐58 Therefore, the decision should be based on previous comorbidities, etiology of acute heart failure, risk of arrhythmias, and prior use of beta-blocker therapy. In the setting of chronic heart failure, the evidence is clearer; thanks to the LION-HEART study, 15 the use of levosimendan chronically demonstrated a decrease in hospitalizations due to decompensation of heart failure and therefore an improvement in the quality of life of the patients.
In cardiac surgery, the use of levosimendan has demonstrated a reduction in mortality as preoperative conditioning in patients with ventricular dysfunction. Additionally, a meta-analysis published by Maharaj et al 21 demonstrated a reduction in mortality in the POP of CABG or PCI compared to placebo, dopamine, or milrinone. Similarly, the use of milrinone has shown improvement in hemodynamic variables and mortality in CABG, CABG + valve replacement, and mitral valve replacement. The decision-making algorithm proposed for the choice of inotropic agents in cardiac surgery can be found in Figure 2.

Proposed algorithm for the use of levosimendan and milrinone in heart failure. Abbreviations: AKI, acute kidney injury; RRT, renal replacement therapy; CABG, coronary artery bypass grafting; PCI, percutaneous coronary intervention; AF, atrial fibrillation; CVP, central venous pressure; PAWP, pulmonary artery wedge pressure; SVR, systemic vascular resistance; SV, stroke volume; CI, cardiac index.
The most recent guidelines from the Surviving Sepsis Campaign recommend the use of dobutamine plus NEb or adrenaline alone in cases of septic cardiomyopathy with persistent hypoperfusion despite the use of vasopressors. Ge et al in 2023, 28 which included 11 randomized clinical trials, did not demonstrate a difference in mortality in patients with sepsis and ventricular dysfunction with the use of levosimendan versus dobutamine or placebo. However, it is expected that medical evidence supporting the use of levosimendan in sepsis will be described in the near future. Regarding the use of milrinone, retrospective studies have not shown a difference in mortality; however, there appears to be a higher association with the requirement for renal replacement therapy and hospital stay when milrinone is used. It is worth noting that the available studies in septic cardiomyopathy are small and mostly retrospective, limiting the quality of evidence and recommendations. 28
The main pathophysiological mechanisms of septic cardiomyopathy include mitochondrial dysfunction, oxidative stress, abnormalities in calcium handling, and sensitivity to myofilament. 19 For this reason, the use of levosimendan could be beneficial based on its pharmacokinetic characteristics. However, it is clear that there is currently little evidence to recommend the use of levosimendan by clinical practice guidelines, and dobutamine remains the first-line inotropic agent in this scenario.15,19,49
The use of inodilators has been described in other scenarios, such as cardiovascular optimization for ventilatory weaning in patients on ECMO, where it has shown improvement in the rate of successful weaning with the use of levosimendan or even the use of milrinone as a potent pulmonary vasodilator in the treatment of life-threatening asthma crises.59‐61 Although the evidence is scarce, the results are interesting and could provide the basis for further research on the role of inodilators outside the cardiovascular context.
Considering the paramount significance attributed to the utilization of milrinone and levosimendan, particularly within the realm of acute heart failure, we posit the development and implementation of a meticulously devised algorithm. The primary objective of this algorithm is to facilitate the judicious selection of the most optimal inodilator therapy on an individualized basis. This proposed algorithm is underpinned by a comprehensive consideration of the antecedently elucidated evidentiary framework, thereby ensuring a discerning and tailored approach to therapeutic decision-making in the context of acute heart failure, Figure 2. Through the assimilation of pertinent evidence, our proposed algorithm strives to enhance the precision and efficacy of the therapeutic interventions employed, thereby contributing to an advanced and nuanced management paradigm for acute cardiac dysfunction.
Limitation
The review article presents several limitations that compromise its rigor and reliability. Firstly, the adopted analysis is predominantly narrative rather than following a systematic approach, which could result in a subjective and biased interpretation of the collected data. The absence of grouped data analysis raises questions about the validity of the presented conclusions and leaves unanswered why this methodology was not performed. Additionally, the omission of a meta-analysis or network meta-analysis limits the study's ability to synthesize and generalize results robustly and quantitatively. There is a lack of information regarding the design of included studies which could impact the quality and consistency of the presented evidence. Furthermore, the potential heterogeneity among included studies has not been addressed, and this could significantly affect the interpretation of results and the applicability of conclusions to various clinical contexts. Overall, these limitations highlight the need for a more rigorous and transparent methodology in future research.
Conclusions
Overall, the evidence suggests that levosimendan and milrinone offer similar hemodynamic support in specific post-surgical scenarios, while their roles in critical care settings like acute HF and cardiogenic shock necessitate further investigation. The choice between these agents should be carefully considered based on individual patient characteristics, underlying pathology, and available evidence.
Footnotes
List of abbreviations
Acknowledgment
The authors like to acknowledge the support from Universidad de La Sabana and Fundación Clínica Shaio.
Author Contributions
Conceptualization: AQA, HRA, ISP, ETQ, AB, LMD, MRA, CFN, JCB, CPH, RB. Data curation: AQA, HRA, CFN, JCB, CPH, RB. Formal analysis: AQA HRA, ISP, ETQ, AB, CFN. Methodology: AQA, HRA, ISP, ETQ, AB. Supervision: AQA, HRA, JCB, CPH, RB. Validation: AQA, HRA, ISP, ETQ, AB. Writing—original draft: AQA, HRA, ISP, ETQ, AB. Writing—Final manuscript: AQA, HRA, ISP, ETQ, AB, LMD, MRA, CFN, JCB, CPH, RB.
Consent for Publication
The study was approved by the local ethics committee from Fundación Clínica Shaio.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
We declare that this manuscript collects honestly, accurately, and transparently the information relative to the study that reports there are no financial or personal relationships with other people or organizations that could inappropriately bias our work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Universidad de La Sabana (Grant: MEDESP-3-2024) and Fundación Clínica Shaio.
