Abstract
Objective:
To determine the efficacy and safety of erythropoiesis-stimulating agents (ESAs) for the treatment of anemia in patients with systolic heart failure.
Data Sources:
A search of MEDLINE (1946-January 2014) and EMBASE (1947-January 2014) was conducted using the search terms erythropoietin and systolic heart failure. In addition, bibliographies of relevant articles were reviewed for additional citations.
Study Selection and Data Extraction:
All English language randomized controlled trials evaluating clinical outcomes or adverse events when using ESAs in the setting of systolic heart failure were included.
Data Synthesis:
A total of 9 studies were reviewed. All studies examining hematological parameters found a statistically significant increase in hemoglobin levels with active treatment versus placebo. Of the 7 trials evaluating exercise tolerance or capacity, only 4 demonstrated statistically significant improvement in these measures in patients receiving ESAs, whereas the remainder showed no clinical benefit. Four studies examined quality-of-life measures. Although numerical improvements were observed in most trials, statistical significance was reached in only 2 trials. A nonsignificant trend for decreased mortality in patients treated with darbepoetin with a similar adverse event profile compared to placebo was shown in one study; however, the largest trial to date showed no benefit in all-cause mortality or heart failure-related hospitalizations with the use of ESAs. Additionally, a statistically significant increase in the number of cerebrovascular events and thrombotic events was found.
Conclusion:
There is inconclusive evidence to suggest that the use of ESAs in treating anemia in patients with heart failure is beneficial. Although ESAs demonstrated a clear ability for increasing hemoglobin levels, the data regarding clinical outcomes such as exercise parameters, quality of life, and hospitalizations are conflicting. In addition, a mortality benefit has not been shown; therefore, the potential for improved symptomatology must be weighed against the potential for adverse events.
Background
Patients with concomitant anemia and heart failure have a significantly increased risk of mortality. 1 Unfortunately, the prevalence of anemia in patients with heart failure is difficult to establish due to varying definitions of anemia used. The World Health Organization defines anemia as a hemoglobin value less than 13 g/dL in men and less than 12 g/dL in women, while the National Kidney Foundation defines anemia as less than 12 g/dL in both men and postmenopausal women. 2 This lack of consensus in the definition of anemia makes isolating the true number of patients with anemia difficult.
The exact pathophysiology of anemia in heart failure is unknown. It is most likely multifactorial and involves both natural disease progression and effects from the standard of care medications these patients are receiving. 2 Patients who have left ventricular dysfunction usually develop decreased cardiac output, leading to decreased renal perfusion. In normal physiology, decreased renal perfusion causes the renin–angiotensin–aldosterone system to become activated, leading to salt and water retention and increases in cardiac output. In patients with heart failure, extra fluid can cause hemodilution leading to anemia. Another proposed mechanism for this type of anemia is due to adverse effects of the medications used in the treatment of heart failure. 2 According to the American College of Cardiology Foundation/American Heart Association 2013 heart failure guidelines, patients diagnosed with systolic heart failure should be placed on an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB). 3 These drug classes have shown morbidity and mortality benefits in patients with heart failure. 2,4,5 In a healthy patient, angiotensin II increases erythropoietin (EPO) secretion by decreasing renal blood flow due to the vasoconstrictor effect of angiotensin II. When an ACEi or ARB is used, this action is prevented. Other proposed causes of anemia in patients with heart failure include proinflammatory cytokines and concomitant chronic kidney disease. 2
Therapies that have been evaluated for the treatment of heart failure include blood transfusions, iron therapy, and erythropoiesis-stimulating agents (ESAs). 2 Erythropoiesis-stimulating agents have shown benefits in multiple types of anemia, including anemia due to chronic kidney disease, chemotherapy treatment, and zidovudine use in human immunodeficiency virus infection; however, they are not currently approved for anemia due to heart failure. 6 Known side effects of ESAs are hypertension, iron deficiency, and increased risk of venous thromboembolism, stroke, and death. 6,7 The objective of this literature review was to determine the efficacy and safety of ESAs for the treatment of anemia in patients with systolic heart failure.
Literature Search
A search of MEDLINE (1946-January 2014) and EMBASE (1947-January 2014) was conducted using the medical subject heading terms erythropoietin and systolic heart failure. The search was limited to articles written in English that were randomized controlled trials whose patient population was treated for anemia with ESAs. Trials were required to include either clinical outcomes such as exercise capacity, hospitalization for heart failure, or mortality or provide information on adverse events when using ESAs. Bibliographies of relevant articles were reviewed for additional citations. A total of 9 studies met inclusion criteria. The reviewed articles are discussed based on their contributions to either efficacy or safety. A summary of the trials is listed in Table 1. 8 –16
Summary of Studies Using Erythropoiesis-Stimulating Agents for the Treatment of Anemia in Patients With Systolic Heart Failure.
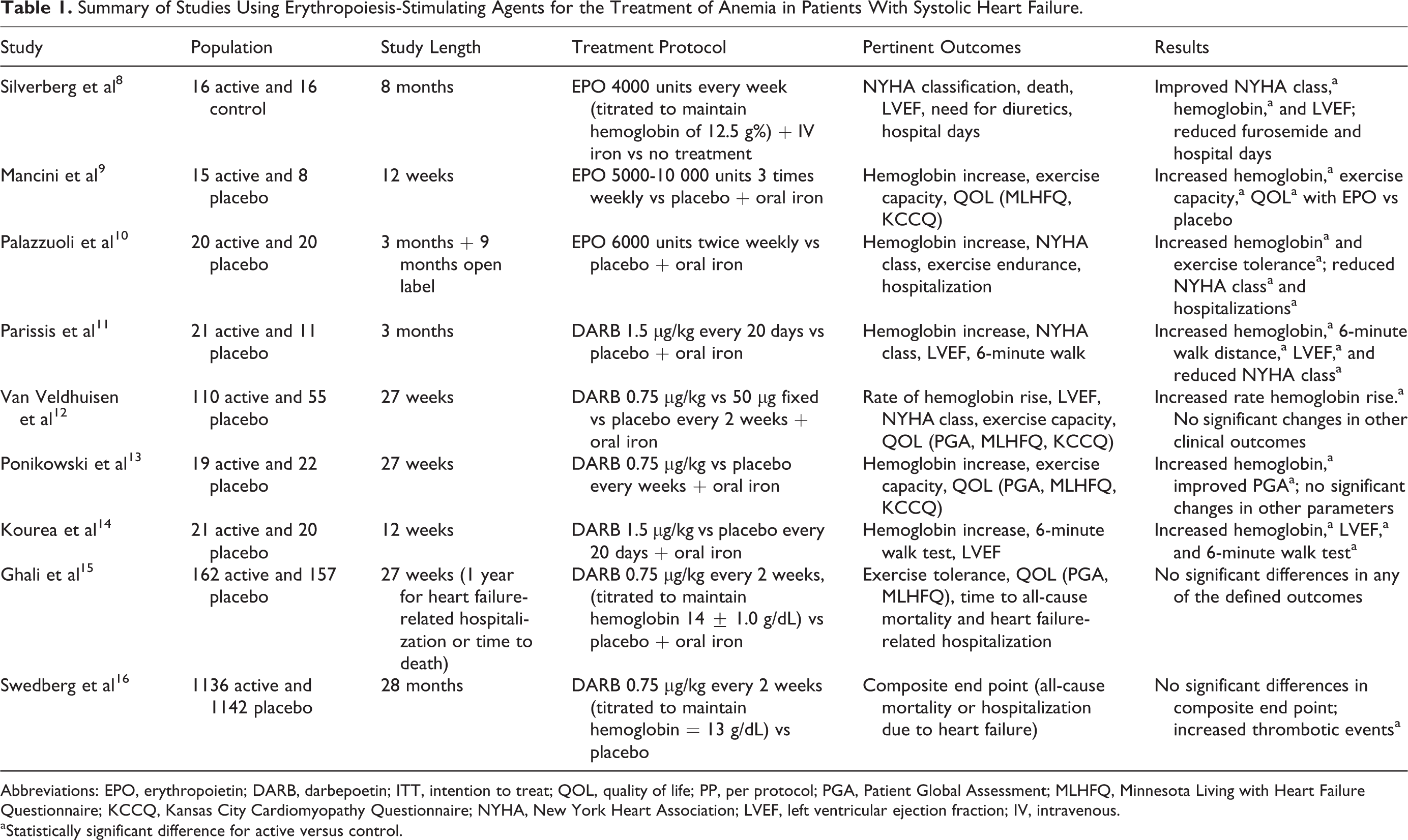
Abbreviations: EPO, erythropoietin; DARB, darbepoetin; ITT, intention to treat; QOL, quality of life; PP, per protocol; PGA, Patient Global Assessment; MLHFQ, Minnesota Living with Heart Failure Questionnaire; KCCQ, Kansas City Cardiomyopathy Questionnaire; NYHA, New York Heart Association; LVEF, left ventricular ejection fraction; IV, intravenous.
aStatistically significant difference for active versus control.
Literature Review
Silverberg et al conducted a randomized controlled trial to evaluate the effect of correcting mild anemia in patients with severe congestive heart failure (CHF). 8 This single-center trial enrolled 32 patients with New York Heart Association (NYHA) class III to IV disease. Patients were randomized in a consecutive fashion into the active (n = 16) or control group (n = 16). Erythropoietin (EPO) was administered for active treatment at an initial dose of 4000 units/wk and titrated to maintain a hemoglobin of 12.5 g/%. In addition, intravenous iron was administered to this group at a dose of 200 mg every 2 weeks until serum ferritin reached 400 μg/L, %Fe saturation was 40%, or the hemoglobin target was met. Patients were followed for a mean 8.2 ± 2.7 months. The groups were well matched at baseline in terms of CHF medications, ejection fraction, renal function, and hemoglobin values. A statistically significant improvement from baseline in NYHA classification was demonstrated in the active group compared to the control group (3.8 ± 0.4 to 2.2 ± 0.7 vs 3.5 ± 0.7 to 3.9 ± 0.3, P < .0001). During the follow-up period, 4 deaths occurred in the control group compared to 0 in the active group. Other positive findings of this study were an improvement in ejection fraction, a reduction in CHF-related hospitalization days, and a lower furosemide dose requirement in the patients treated with EPO. The limitations of this study included a small sample size, as well as a lack of blinding and use of a placebo control. Additionally, this study excluded patients with less severe CHF (NYHA class II) and had a narrow entry criteria for hemoglobin values (10-11.5 g/dL).
Mancini et al conducted a small, single-blind, randomized, and placebo-controlled trial to investigate the effect of EPO therapy on exercise capacity in patients with chronic heart failure after a 3-month trial period.
9
Patients were randomized in a 2:1 ratio to 5000 units of EPO dosed 3 times a week (increased to 10 000 units after 4 weeks if hemoglobin increase was below 1 g/dL) or placebo (0.9% saline) dosed once a week. A total of 26 patients were randomized and 23 completed the study. There was a statistically significant change in hemoglobin levels in response to active treatment (11± 0.6 to 14.3± 1.2 g/dL, P < .0001), while the hemoglobin in the placebo group remained unchanged (10.9 ± 1.1 to 11.5 ± 1.3 g/dL). Peak oxygen consumption (V
Palazzuoli et al conducted a randomized, double-blind, and single-center study examining the impact of anemia treatment on exercise tolerance and other clinical markers in patients with heart failure. 10 Forty patients with hemoglobin values <11 g/dL and NYHA classification III to IV were randomized to receive EPO 6000 units twice weekly for 3 months (n = 20) versus normal saline placebo (n = 20). Both groups were given daily oral iron therapy. After the intervention phase, patients were followed for an additional 9 months. At baseline, the groups were similar in terms of NYHA class and exercise duration. Hemoglobin values increased significantly from baseline in the active treatment group compared to placebo after the intervention period (10.4 ± 0.6 to 12.4 ± 0.8 vs 10.6 ± 0.7 to 10.5 ± 0.6 g/dL, P < .1). Exercise endurance, as measured by distance (278 ± 55 to 356 ± 88 m, P < .01) and duration (5.8 ± 2.2 to 7.8 ± 2.5 min, P < .01), was markedly improved in the EPO-treated group. After 1 year, fewer hospitalizations were observed in the patients receiving EPO compared to placebo. In all, 20% of patients treated with EPO were admitted to the hospital versus 44.4% of those receiving placebo (P < .01). Death occurred in 1 patient in the EPO group and 2 in the placebo group. This trial was limited by its small sample size and narrow patient population, as it only included those with NYHA class III to IV from a single center.
Parissis et al enrolled patients with NYHA class II to III heart failure and a hemoglobin <12.5 g/dL in a randomized, placebo-controlled, single-center study evaluating the effect of darbepoetin on cardiac function. 11 Patients were randomized in a 2:1 manner to receive darbepoetin 1.5 μg/kg every 20 days (n = 21) or placebo (n = 11), in addition to oral iron. Treatment was held if hemoglobin values reached 14 g/dL. The groups were well matched at baseline. After the 3-month treatment period, statistically significant differences between the darbepoetin-treated group and the placebo group were found for hemoglobin level (12.8 ± 1.3 vs 11.8 ± 1.3 g/dL, P = .009), NYHA class (2.1 ± 0.5 vs 3.2 ± 0.6, P = .001), 6-minute walk distance (296 ± 87 vs 167 ± 99 m, P < .001), and left ventricular ejection fraction (LVEF; 31% ± 6% vs 25% ± 5%, P < .001). Adverse effects included 1 patient in the placebo group with transient ischemic attack and 1 patient in the active group who developed arterial hypertension. Limitations of this study included a small sample size, unblinded investigators, and lack of patients with NYHA class IV heart failure.
Van Veldhuisen et al conducted a multinational, double-blind study to evaluate the effect of 2 dosing regimens of darbepoetin in patients with heart failure and anemia with hemoglobin between 9.0 and 12.5 g/dL. 12 A total of 160 patients were randomized to either a weight-based regimen (0.75 μg/kg), a fixed-dose regimen (50 μg), or placebo given every 2 weeks and supplemented with oral iron. The target hemoglobin level was 14.0 ± 1.0 g/dL. Baseline hemoglobin values were similar between groups. At week 27, a significant difference was observed between the weight-based dosing group and placebo for rise in hemoglobin concentration (0.2 g/dL/wk, 95% confidence interval [CI]: 0.16-0.24; P < .001). The mean hemoglobin increase for the weight-based, fixed-dose, and placebo groups was 1.87 ± 1.36, 1.64 ± 0.98, and 0.07 ± 1.08 g/dL, respectively. The 2 darbepoetin regimens were considered equivalent, based on the prespecified range. Equivalence was declared if the difference in the CIs for the average rate of increase in hemoglobin fell within −0.5 to 0.5 g/dL/wk for each regimen. When looking at secondary outcomes, there was no difference found in the 6-minute walk distance, LVEF, or NYHA classification. Regarding the Patient Global Assessment (PGA), 49% of patients in the placebo group versus 65% in the active treatment group reported improvement in symptoms, although this difference was not statistically significant. For the other quality-of-life measures, the MLHFQ, and the Kansas City Cardiomyopathy Questionnaire (KCCQ), there were also numerical improvements in the scores despite not reaching statistical significance. The incidence of any adverse event was similar between the 3 groups. An increase in the number of deaths in the darbepoetin groups (6 vs 0 in placebo) was reported; however, these were not determined to be treatment related. A limitation of this evaluation is that it was only powered to detect a difference in laboratory markers rather than clinical outcomes. In addition, this was a small sample size and may be difficult to extrapolate to a larger, more varied patient population.
Ponikowski et al investigated whether darbepoetin improved exercise tolerance in patients with symptomatic chronic heart failure and anemia.
13
In this phase 2, double-blind, placebo-controlled trial, 41 patients were randomized in a 1:1 fashion to either subcutaneous placebo or subcutaneous darbepoetin 0.75 μg/kg given every 2 weeks. Patients had to have symptomatic CHF with exercise limitation on the treadmill and a hemoglobin level between 9.0 and 12.0 g/dL. The mean change from baseline to week 27 in absolute peak V
Kourea et al performed a study to observe the effects of darbepoetin on proinflammatory cytokines in patients with anemia and chronic heart failure. 14 To be included in this trial, patients had to be diagnosed with NYHA class II to III heart failure, have been treated with guideline-recommended therapy, have a LVEF of less than 40%, a hemoglobin of less than 12.5 g/dL, and a serum creatinine of less than 2.5 g/dL. Forty-one patients were randomized to darbepoetin (1.5 μg/kg every 20 days) plus oral iron twice daily or placebo with oral iron. At the end of the 3-month evaluation period, there was a significant increase in hemoglobin levels from baseline in the darbepoetin group (10.9 ± 1.0 to 12.8 ± 1.4 g/dL; P < .001) but not in the placebo group (11.4 ± 0.8 to 11.7 ± 1.5 g/dL). The LVEF also significantly increased in the active treatment group (26 ± 6 to 32 ± 6, P < .001). Significance was also seen in the 6-minute walk test for patients treated with darbepoetin (201 ± 113 to 274 ± 97 m) versus the placebo group (237 ± 101 to 204 ± 103 m, P < .01). The limitations of this trial included a single-blind design, small sample size, and lack of representation of the sickest patients due to exclusion of those with NYHA class IV heart failure.
Ghali et al conducted one of the largest randomized, double-blind, placebo-controlled, multinational trials evaluating the treatment of anemia in patients with heart failure. 15 To be included in the analysis, patients had to have a LVEF less than 40%, 2 hemoglobin screenings between 9.0 and 12.5 g/dL, and no other causes of anemia. There were 319 patients enrolled from 65 centers randomized in a 1:1 fashion to either darbepoetin (0.75 μg/kg) or placebo given every 2 weeks for up to 1 year. The target hemoglobin value was 14.0 ± 1.0 g/dL. At week 27, the primary end point, which was the change in exercise duration, was not significant between placebo and darbepoetin (45.6 ± 126.4 vs 58.4 ± 111.1 s, P = .46), respectively. Of note, this study successfully achieved a power of greater than 80% to detect a difference ≥60 seconds in this parameter. No statistical difference was identified at week 27 on the MLHFQ or PGA measures. The study was not designed to find a difference between the 2 groups in terms of mortality and first heart failure-related hospitalization; however, a nonsignificant trend of lower mortality in those patients treated with darbepoetin was seen. In the placebo group, 18 patients died from any cause while 11 patients died in the darbepoetin group (hazard ratio [HR] 0.68; 95% CI: 0.43-1.08, P = .10). The adverse event profile including serious, treatment-related, and specific-interest adverse effects was similar between the darbepoetin- and placebo-treated patients A limitation of this study was that patients with severe anemia (ie, hemoglobin less than 9 g/dL) were excluded.
Swedberg et al conducted the largest randomized, double-blind, placebo-controlled trial to date involving patients with systolic heart failure (NYHA classes II-IV) and anemia, defined as a hemoglobin value between 9 and 12 g/dL. 16 There were 1136 patients randomized to receive darbepoetin 0.75 μg/kg every 2 weeks, titrated to target a hemoglobin of 13.0 g/dL, not to exceed 14.5g/dL. The placebo group (n = 1142) also received dose adjustments to mimic the active group. After 28 months of follow-up, there was no statistical difference in the number of deaths or first hospitalization due to worsening heart failure (HR 1.01; 95% CI: 0.90-1.13, P = .87). In terms of adverse events, more patients treated with darbepoetin experienced an embolic or thrombotic event (risk difference 3.5%; 95% CI: 0.9-6.1, P = .009) as well as cerebrovascular ischemic events (risk difference 1.7%; 95% CI: 0.2-3.2, P = .03). Limitations to the study were that patients with more severe anemia (hemoglobin less than 9 g/dL), whom might have the most benefits to gain from treatment, were excluded. The investigators targeted a higher hemoglobin level that is typically the case when using ESAs for chronic kidney disease, which could account for the lack of benefits in terms of the primary outcome as well as the increase in cerebrovascular and thromboembolic events.
Discussion
Efficacy
Current 2013 ACCF/AHA heart failure guidelines do not specify any preferred treatment for anemia in patients with heart failure. 3 Erythropoiesis-stimulating agents such as EPO (4000-10 000 units 1-3 times/wk) and darbepoetin (0.75-1.5 μg/kg every 14-20 days) have been shown to be effective in increasing hemoglobin levels by roughly 2 g/dL in these patients; however, the actual benefits in terms of exercise capacity and quality of life are varying. 8 –16
Exercise capacity has been considered an outcome of interest in patients with heart failure. By increasing exercise capacity in these patients, it is plausible that this may increase quality of life, as well as decrease the number of possible negative outcomes.
17,18
Cardiac function is measured in a variety of ways, several of which were used in the studies. A 6-minute walk test and peak V
Although an improvement in a patient’s exercise capacity is desirable, a better measurement of patient-specific efficacy outcomes could be quality of life. The KCCQ and the MLHFQ have been used in trials to measure health-related quality of life in patients with heart failure. 9,12,13,15 Although these questionnaires may be subjective, they have been used to quantify quality of life in patients with heart failure. Of the trials reviewed, several showed a numerical improvement in these quality-of-life scores; however, statistical significance was not always reached. In the studies that did demonstrate a significant difference, the clinical relevance has yet to be determined. 9,13 Some of the trials had a short duration that leaves a gap in information on long-term efficacy when using ESAs for anemia in patients with heart failure. 9,11,14
Most of these studies were small clinical trials and may not be representative of the entire heart failure population. There is also very limited data on the patients with NYHA class IV, so extrapolation of benefit to this sicker population is difficult.
It should be noted that the larger and more robust trials did not show the same beneficial outcomes as had previously been demonstrated in smaller studies. It is possible that although even mild levels of anemia have been associated with increased mortality and worse NYHA functional class, perhaps it is not the hemoglobin value itself that is the causative factor, thus patients with mild to moderate anemia may not have yet sustained the level of tissue hypoxia required to elicit symptoms and trigger a compensatory response. 19,20 Based on the currently available evidence, treating mild to moderate anemia as a strategy for improving clinical outcomes in heart failure may not be the most optimal approach.
Since most studies excluded patients with severe anemia, further research is warranted in this underrepresented patient population. In addition, further clarification of the true pathophysiologic relationship between anemia, functional capacity, and mortality could help direct other targets and potential treatment approaches for minimizing the morbidity and mortality of heart failure. Finally, additional research examining the utility of targeting less intense hemoglobin values, similar to what is used in other types of anemia, may be of use to evaluate clinical benefit without the additional risks.
Safety
Despite showing clinical efficacy in many studies, ESAs have potential safety concerns, especially when targeting higher hemoglobin levels. The black box warning on all ESAs states that when hemoglobin levels greater than 11 g/dL are targeted, there is an associated increased risk of cardiovascular and thromboembolic adverse events, including increased mortality. 7 Every trial included in this review targeted a goal hemoglobin level above 12.5 g/dL, which places patients at an increased risk of developing an adverse event when being treated with either EPO or darbepoetin. The trial by Swedberg et al is the largest published trial to date examining mortality outcomes and adverse events in more than 2000 patients. It revealed an increase in adverse events with no mortality benefit. More specifically, there was no benefit in using the ESAs in terms of the number of all-cause deaths or heart failure-related hospitalizations. There was also a statistically significant increase in the number of cerebrovascular events as well as embolic or thrombotic events. This trend could be due to the high hemoglobin target that was preselected (greater than 13.0 g/dL). 16
The study conducted by Ghali et al showed a nonsignificant trend for decreased mortality in patients treated with darbepoetin with a similar adverse event profile compared to placebo. 15 Although this became the basis for future studies, this trial was limited in power to detect a meaningful difference in mortality.
Prior to more recent studies, there were no significant differences in adverse events with the use of ESAs in patients with anemia due to heart failure; this was most likely due to small sample sizes since the study conducted by Swedberg et al demonstrated statistical significance in terms of adverse events. This is important to note, since it was the only randomized controlled trial that was statistically powered to detect differences in adverse events. 16
Conclusion
This review demonstrates inconclusive evidence for the use of ESAs in treating anemia in patients with heart failure. Both EPO and darbepoetin showed a clear ability for increasing hemoglobin levels; however, the available evidence is conflicting regarding clinical outcomes such as exercise parameters, quality-of-life markers, and hospitalizations. In addition, the impact on mortality does not appear beneficial. Thus, the potential for improved symptomatology must be weighed against the increase in adverse events seen in the larger trials. Furthermore, randomized controlled trials are needed to evaluate whether a lower hemoglobin target would provide clinical benefits without the increased risk of adverse events.
Footnotes
Author Contribution
Lindquist contributed to conception and design of the study, with substantial contribution to data acquisition, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content. Cruz contributed substantially to data acquisition, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content. Brown contributed to analysis and interpretation of the data and critical revision of the article for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
