Abstract
Background:
Coumarin anticoagulants (acenocoumarol, phenprocoumon, and warfarin) are generally used for the prevention of stroke in patients with atrial fibrillation or for the therapy and prevention of venous thromboembolism. However, the safe use of coumarin anticoagulants is restricted by a narrow therapeutic window and large interindividual dosing variations. Some studies found that the effectiveness and safety of coumarin anticoagulants therapy were increased by pharmacogenetic-guided dosing algorithms, while others found no significant effect of genotype-guided therapy.
Methods:
Four electronic databases were searched from January 1, 2000, to March 1, 2014, for randomized controlled trials of patients who received coumarin anticoagulants according to genotype-guided dosing algorithms. The primary outcome was the percentage of time that the international normalized ratio (INR) was within the normal range (2.0-3.0). Secondary outcomes included major bleeding events, thromboembolic events, and INR ≥4 events.
Results:
Eight studies satisfied the inclusion and exclusion criteria. Genotype-guided dosing of coumarin anticoagulants improved the percentage of time within the therapeutic INR range (95% confidence interval [CI], 0.02-0.28;
Conclusions:
This meta-analysis showed that genotype-guided dosing increased the effectiveness and safety of coumarin therapy compared with standard dosing but did not have advantages compared with clinical variables-guided dosing.
Introduction
Coumarin anticoagulant agents such as acenocoumarol, phenprocoumon, and warfarin are frequently used for prophylactic and therapeutic reasons, such as in patients diagnosed with atrial fibrillation, pulmonary embolism, or deep vein thrombosis. 1 Warfarin is the most frequently used, but acenocoumarol and phenprocoumon are used in some regions. 2 The safe use of coumarin anticoagulants is restricted by a narrow therapeutic window and large interindividual variations in coumarin anticoagulants dose requirements. 3,4
A narrow therapeutic index means that an increase or decrease in international normalized ratio (INR) may result in major hemorrhagic or thrombotic complications. Many studies have evaluated the genetic and clinical factors associated with coumarin anticoagulant dose requirements and their influence on INR stability, including the effects of age, race, weight and height, target INR, and polymorphisms in the genes encoding the vitamin K epoxide reductase complex subunit 1 (VKORC1) and cytochrome P450 2C9 (CYP2C9). 5 –8 Variants in CYP2C9 and VKORC1 have consistently been associated with dose requirements in populations worldwide and together account for approximately 40% of the variation in dose requirements. 9 –11
Genotype-guided dosing algorithms may improve prediction of the required coumarin dose and thus increase its effectiveness and safety. 12 –16 The CoumaGen-II 15 study showed that patients treated according to pharmacogenetic-guided dosing had a higher percentage of patients who remained within the therapeutic INR range, fewer with INR ≥4, and fewer serious adverse events. However, other recent studies failed to support this conclusion and showed that genotype-guided dosing of coumarin anticoagulants did not improve the percentage of time in the therapeutic INR range. 2,17 –19 We therefore performed a meta-analysis of randomized controlled trials (RCTs) in an attempt to resolve this discrepancy and provide evidence to clinical physicians.
Methods
Study Selection
We searched PubMed, Embase, web of science, and Cochrane Library from January 1, 2000, to March 1, 2014. We limited the key words to (VKORC1) OR (CYP2C9) and (genotype) OR (genotype-guided) OR (pharmacogenetic) and (acenocoumarol) OR (phenprocoumon) OR (coumarin) OR (warfarin). Searches were limited to RCTs of patients who received coumarin anticoagulants according to a genotype-guided dosing algorithm, and trials in which both efficacy and safety outcomes were reported. Eight RCTs were collected (Figure 1).

Flow chart of literature review.
Two reviewers (Tang and Li) independently extracted data using a data abstraction instrument: the number of participants in each arm, study quality, length of follow-up period, intervention and control dosing algorithms, and primary and secondary outcomes. Our primary outcome of interest was time that the INR was within the therapeutic range (INR 2.0-3.0), and secondary outcomes included major bleeding events, thromboembolic events, and INR ≥4 events.
Data Extraction and Quality Assessment
Details of the publication, inclusion and exclusion criteria, demographics of the enrolled patients, interventions used, and outcome definitions and outcomes were collected and collated. Risk of bias in RCTs (including masking of participants, method of sequence generation and allocation concealment, intention-to-treat analysis, early trial stopping for efficacy before the planned enrolment was completed, and loss to follow-up) was assessed. 20 Study quality was assessed using the Modified Jadad scale (Table 1). 21 Any discrepancies between reviewers were resolved by consensus.
Quality Assessment of Randomized Controlled Trials.a

a1 to 3 points = low quality; 4 to 7 points = high quality.
Statistical Analysis
All data synthesis was done using Review Manager (Cochrane Collaboration software, version 5.1). The principal measure of effect size between the intervention and control arms was the standardized mean difference for the percentage time within the therapeutic INR range. 22 We prespecified the use of random-effects models for all analyses; these contained between-trial heterogeneity and give wider and more conservative confidence intervals (CIs) compared with fixed-effects models, which do not incorporate heterogeneity. 23 Statistical heterogeneity was assessed using I2, defined as the percentage of total variability across studies attributable to heterogeneity rather than chance, and using reported guidelines for low (I2 = 25%-49%), moderate (I2 = 50%-74%), and high (I2 ≥ 75%) heterogeneity. 23 We used meta-regression subgroup analyses to explore the sources of heterogeneity. Secondary outcomes, including major bleeding events, thromboembolic events, and INR ≥4 events, were analyzed using risk ratios to pool outcomes with a 2-sided significance level of 5%. 24
Study Characteristics and Quality
Ten RCTs included experimental groups that received coumarin anticoagulants according to a genotype-guided dosing algorithm and control groups that received coumarin anticoagulants according to a nongenotype-guided dosing algorithm. All RCTs were not drug clinical trials, and 2 articles were excluded. Caraco et al 25 and Borgman et al 26 studies were pseudo RCTs, because there was no randomization method and study quality was low (Jadad score is 2 points). Table 2 shows the baseline characteristics for each study. Pirmohamed et al 12 and Verhoef et al 2 examined the primary outcome twice, at 4 weeks and 12 weeks. Hillman et al 27 used a genotype-guided dosing algorithm based on the CYP2C9 gene, while other studies used both CYP2C9 and VKORC1 genes. Four RCTs 12,13,16,27 included control groups of patients treated with standard dosing (coumarin anticoagulants at a standard initial dose, such as 10, 5, or 2.5 mg), while other studies included control groups dosed according to a clinical variables-guided dosing algorithm (coumarin anticoagulant dose calculated according to age, smoker status, body surface area [BSA], race, etc). 28 The trials did not include sufficient information on weight, height, smoker status, BSA, and race to allow them to be analyzed. The primary outcome of percentage of time within the therapeutic INR range is shown in Table 3. The qualities of the trials were assessed using the Modified Jadad score (Table 1), where 1 to 3 points = low quality and 4 to 7 points = high quality.
Baseline Characteristics of Randomized Controlled Trials of Genotype-Guided Coumarin Dosing.
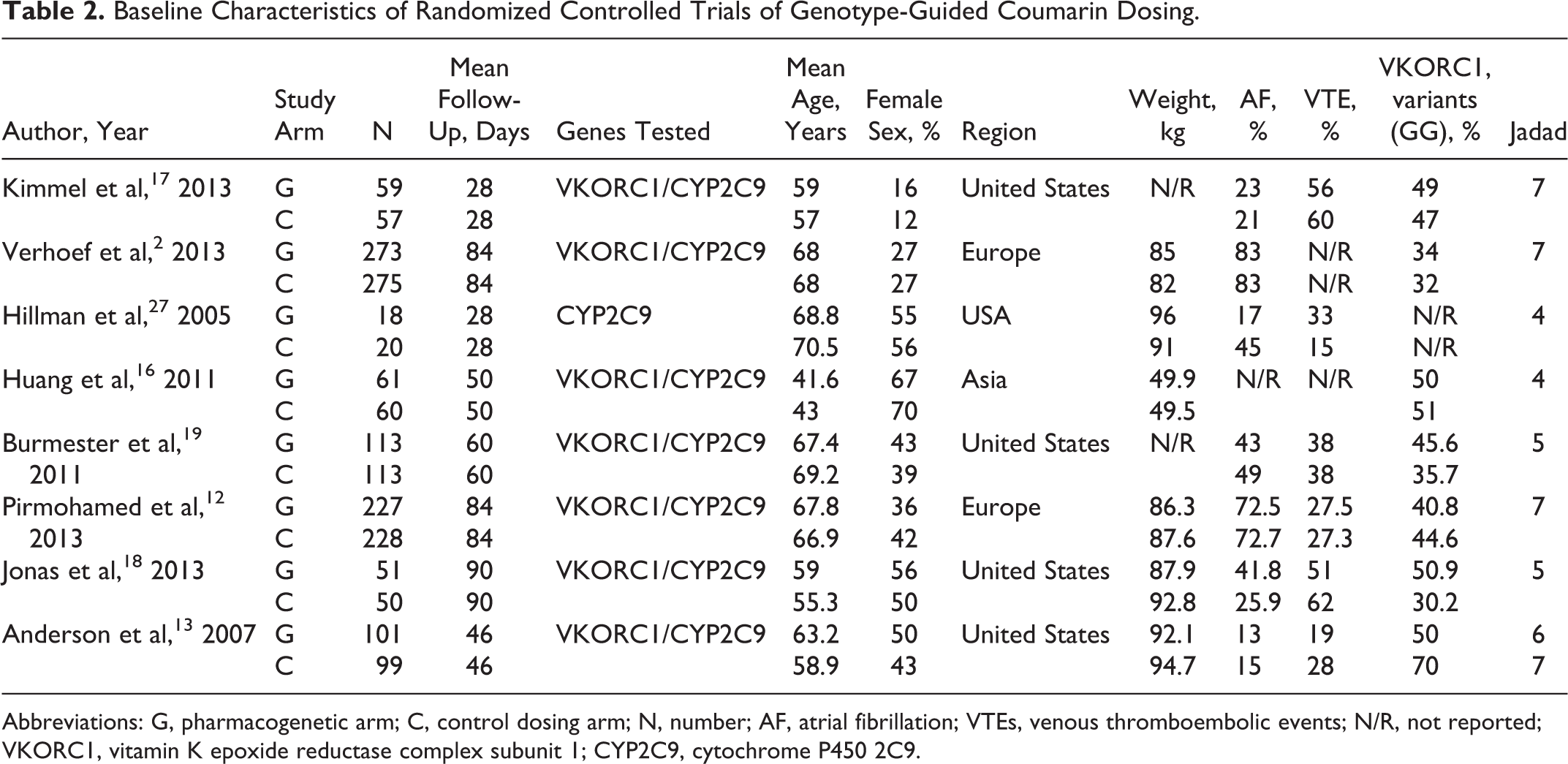
Abbreviations: G, pharmacogenetic arm; C, control dosing arm; N, number; AF, atrial fibrillation; VTEs, venous thromboembolic events; N/R, not reported; VKORC1, vitamin K epoxide reductase complex subunit 1; CYP2C9, cytochrome P450 2C9.
Primary and Secondary Outcomes in Randomized Controlled Trials.

Abbreviations: INR, international normalized ratio; VTE, venous thromboembolic event; N/R, not reported; SD, standard deviation.
aPirmohamed and Verhoef assessed the primary outcome at 4 weeks and 12 weeks.
Results
Primary Outcome
Meta-analysis indicated that coumarin anticoagulants administered according to genotype-guided dosing resulted in a significantly greater percentage of time within the therapeutic INR range, compared with nongenotype-guided dosing (95% CI, 0.02-0.28;
Stata 12.0 Meta-Regression of Randomized Controlled Trials for Heterogeneity.a

Abbreviations: CI, confidence interval; CYP2C9, cytochrome P450 2C9; INR, international normalized ratio; SE, standard error; VKORC1, vitamin K epoxide reductase complex subunit 1.
aType: nongenotype dosing based on standard dosing or clinical variables-guided dosing; time: time of INR%; gene: genotype based on CYP2C9 and VKORC1 or just CYP2C9; age: age ≥65 or <65 years; and sample size: sample ≥50 or <50.
Subgroup analysis demonstrated that the percentage of time within the therapeutic INR range was similar in patients treated according to genotype-guided and clinical variables-guided dosing algorithms (95% CI, −0.07-0.15;
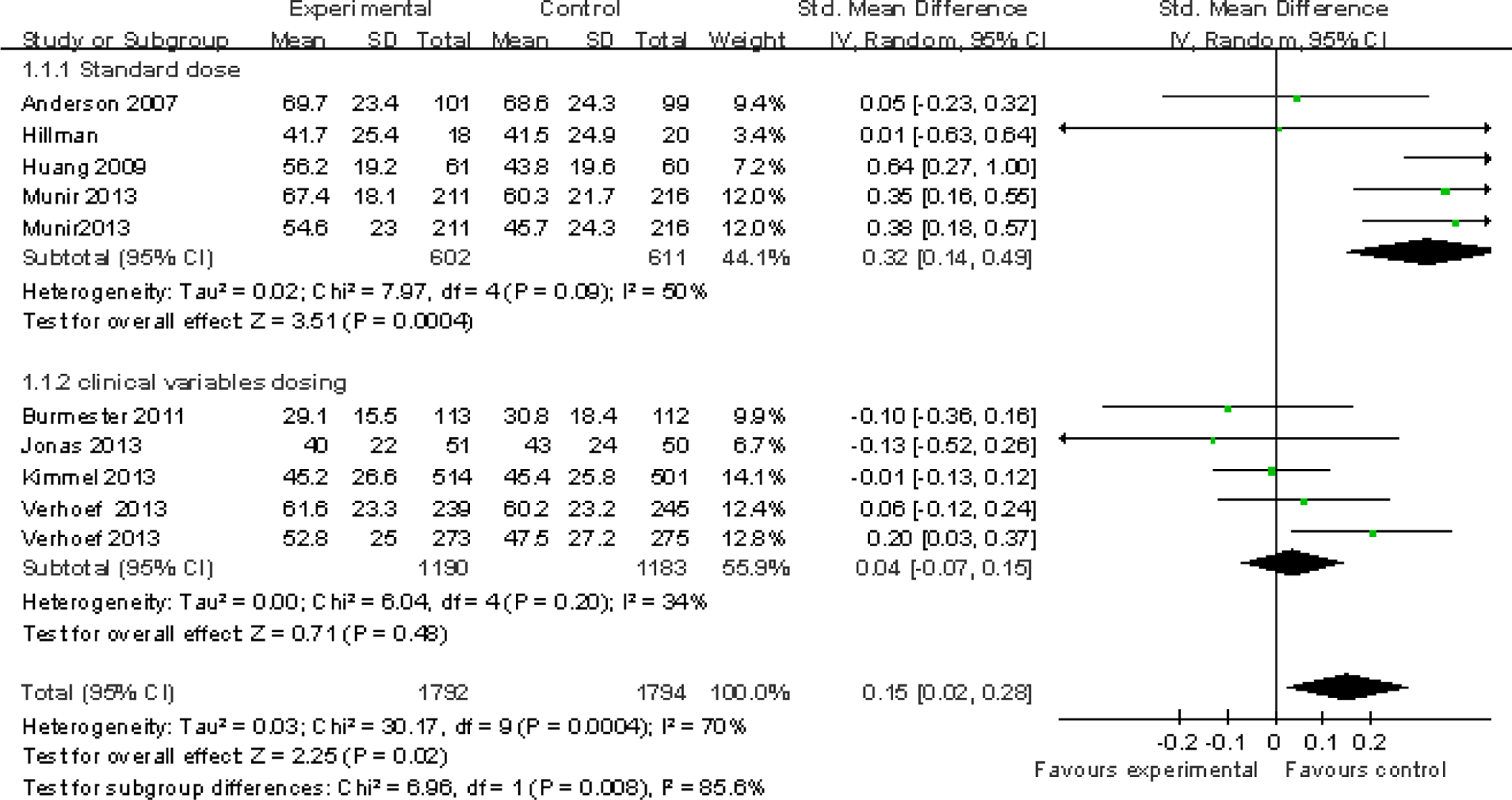
Meta-analysis of effects of pharmacogenetic dosing and control dosing on therapeutic INR range with subgroup analysis. *Pirmohamed et al and Verhoef assessed the primary outcome at 4 and 12 weeks. Percentage of time within the therapeutic INR range (total 95% CI, 0.02-0.28;
Secondary Outcomes
Secondary outcomes included major bleeding events, thromboembolic events, and INR ≥4 events. Kimmel et al
17
study found no significant differences between the groups in terms of the principal secondary outcomes or other adverse events. However, Pirmohamed et al found that patients in the genotype-guided group were less likely to have an INR of ≥4.0 than those in the control group,
12
while Anderson et al
13
had no exact statistical data on major bleeding events and thromboembolic events. No major bleeding or thromboembolic events occurred during the follow-up period in Huang et al study,
16
and Pirmohamed et al had no major bleeding events during follow-up. There is a statistically significant reduction in numbers of secondary outcomes (INR ≥4 events, major bleeding events, and thromboembolic events; 95% CI, 0.79-1.00;

Meta-analysis of effects of pharmacogenetic dosing and control dosing on secondary outcomes with subgroup analysis (total 95% CI, 0.79-1.00;
Discussion
Coumarin anticoagulants continue to be the most commonly used anticoagulants worldwide. 30 However, coumarin anticoagulants such as acenocoumarol, phenprocoumon, and warfarin have a narrow therapeutic index, and the mean daily dose of coumarin varies widely from patient to patient. 31 A subtherapeutic INR is associated with an increased risk of thromboembolism, whereas a supratherapeutic INR is associated with an increased risk of bleeding. 32 The typical therapeutic INR for most indications is 2.0 to 3.0, and clinicians need to define and implement dosing regimens that achieve therapeutic anticoagulation. 33
Variations in coumarin anticoagulant dosing depend on several factors, including age, weight, height, BSA, sex, concomitant medication use, smoking status, and race, and genetic factors also play an important role. 5,28 A large proportion of the variation in coumarin dose requirements between patients is attributable to coding variations in multiple genes. 34 Variants in the genes that encode enzymes responsible for coumarin metabolism (eg, CYP2C9) and the molecular target of coumarin anticoagulants, VKORC1, influence coumarin dosing. 12 VKORC1 produces an integral protein in the endoplasmic reticular membrane that catalyzes one of the steps in the vitamin K cycle, reducing vitamin K 2,3-epoxide and vitamin K to vitamin K hydroquinone. 34 CYP2C9 is a CYP isoform that is involved in pharmaceutical metabolism. Coding variations in CYP2C9 are connected to warfarin dose variability. 35 Many studies have identified significant correlations with 2 genes in particular, the cytochrome-P450 gene CYP2C9 and the VKORC1. 36
Although coumarin anticoagulant dosing algorithms do not avoid the need for continual INR monitoring and dose titration, they can, even in the absence of genotype information, provide a good estimate of the appropriate coumarin anticoagulant dose by taking into account clinical information such as age, BSA, weight, and smoking status. 36,37
We analyzed data from 8 RCTs 2,12,13,16 –19,27 and showed that a genotype-guided dosing algorithm for coumarin anticoagulants was safer and more effective than standard dosing but was not significantly different from a clinical variables-guided dosing algorithm. We identified 8 RCTs in which the experimental group received coumarin anticoagulants according to a genotype-guided dosing algorithm while the control group was treated according to a nongenotype-guided dosing algorithm. Although these 8 RCTs were assessed as high quality, the percentage of time within the therapeutic INR still showed moderate heterogeneity. Given that the INR could be affected by many factors, 28 we performed subgroup analysis and meta-regression 29 to explore the source of the heterogeneity and produce a more credible result.
Different follow-up times may have affected the percentage of time within the therapeutic INR.
2
We therefore performed meta-regression
38
by dividing the studies into those with follow-up periods ≥4 and <4 weeks and found no effect of time on the period of therapeutic INR (
Four RCTs
12,13,16,27
included control groups treated with standard-dose coumarin anticoagulants, while the other studies included control groups dosed according to clinical variables-guided algorithms,
28
which may have contributed to the observed heterogeneity. Subgroup analysis of percentage of time within the INR therapeutic range (total heterogeneity: I2 = 70%;
In the COAG trial, the mean percentage of time within the therapeutic INR range among black patients was less in the genotype-guided group than in the clinically guided group (
Our study had limitations. Although we explored the sources of heterogeneity, we were unable to rule out the involvement of BSA, smoking status, weight, variant alleles, and race. 1,28 Further studies based on unified data are needed to reduce potential differences and thus produce more credible results.
Monitoring the percentage of time spent within an INR range serves as a marker of bleeding and thromboembolic risks. 12 The results of our meta-analysis suggest that using a clinical variables-guided algorithm to calculate the dose of coumarin anticoagulants may be an important factor influencing the effects of anticoagulant therapy.
Conclusions
The results of this meta-analysis indicate that genotype-guided dosing of coumarin anticoagulants could increase their effectiveness and safety compared with standard dosing, but that genotype-guided dosing had no apparent advantage over clinical variables-guided dosing. There was therefore insufficient evidence to support the use of pharmacogenetics to guide coumarin anticoagulants therapy in patients already treated according to a clinical variables-guided dosing.
Footnotes
Author Contributions
Tao Tang contributed to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content. Jie Liu contributed to conception and design of the study, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content. Keqiang Zuo contributed to conception and design of the study, data acquisition, drafting of the article, and critical revision of the article for intellectual content. Jie Cheng contribution to data acquisition, analysis and interpretation of the data, and drafting of the article. Linyin Chen contribution to data acquisition and analysis and interpretation of the data. Chenhui Lu contribution to conception and design of the study and analysis and interpretation of the data. Shilong Han contribution to data acquisition, analysis and interpretation of the data, and drafting of the article. Jichong Xu contribution to analysis and interpretation of the data. Zhongzhi Jia contribution to conception and design of the study and analysis and interpretation of the data. Meng Ye contribution to data acquisition and analysis and interpretation of the data. Erli Pe contribution to conception and design of the study, data acquisition, and drafting of the article. Xiaoping Zhang and Maoquan Li contribution to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content. Tao Tang, Jie Liu, and Keqiang Zuo contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
