Abstract
Background:
Current data indicate a persisting concern about possible cataractogenecity of statins.
Objective:
To perform a meta-analysis of studies pertaining to statins and cataract.
Methods:
We identified 363 records by a systematic search of the MedLine, Web of Knowledge, Cochrane database, and ClinicalTrials.gov. After exclusion of duplicates, studies without cataract as an outcome, reviews, and animal or basic science studies, we analyzed 14 studies. Two end points were examined: clinical cataract (requiring extraction or reported by the patient) and lens opacities discovered by slit-lamp examination.
Results:
Using random effects meta-analysis, a statistically significant decrease in cataracts with statins was observed among studies examining clinical cataract (odds ratio [OR] 0.81, 95% confidence interval [CI] 0.71-0.93,
Conclusion:
This meta-analysis indicates a clinically relevant protective effect of statins in preventing cataracts. The effect is more pronounced in younger patients and with longer duration of follow-up, while there is no difference by gender.
Introduction
Cataract is the leading cause of visual impairment other than uncorrected refractive errors worldwide. Statins decrease morbid and mortal cardiovascular events in primary and secondary prevention, in both genders, different ages, and patient subsets. 1 –3 In addition, statins have been found beneficial in conditions not directly related to the cardiovascular system such as infections. 4 However, statins have been associated with adverse effects including effects on liver enzymes, myositis, rhabdomyolysis, diabetes, and with ambiguous findings regarding cancer, and dementia. 5,6 Cataracts were considered a side effect of statins in early studies. High doses of lovastatin, the first Food and Drug Administration (FDA)-approved statin, caused cataracts in beagle dogs. 7 In 1987, lovastatin was approved for human use with the precaution “that patients placed on lovastatin therapy be examined with a slit-lamp before and shortly after initiation of treatment, and annually thereafter.” 8,9 This recommendation was removed from the labeling by the FDA in 1991. Prior publications have reported inconsistent findings on the effect of statins on cataract. Recently, Machan et al reported that in the Waterloo Eye Study, statin use was substantially higher in patients with type 2 diabetes and was associated with cataracts. 10 Also, recent data on statins indicate continuing concern among patients, pharmacists, and other health care providers as reported in electronic drug information sites. Drug Facts and Comparisons 11 state that in the package inserts, cataract is associated with the statin class. On the other hand, the opposite may also be true, that is, statins may prevent cataracts through their antioxidant and other pleiotropic effects. 12 –15
In order to address this issue, that is, concern regarding cataractogenecity versus possible prevention of cataracts by statins, we performed a meta-analysis of published studies on the association of statins with cataracts and examined the influence of age, gender, and duration of therapy on the observed effects.
Methods
Studies Included in the Meta-analysis and Data Extraction
We performed a meta-analysis according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. A systematic search of the MedLine, Web of Knowledge, Cochrane Database, and ClinicalTrials.gov was performed for the intersection of the terms “statins,” each of the marketed statins individually, and “cholesterol lowering medications” with “cataract” through the end of October 2012 (Supplemental Table 1; Figure 1). The searches were performed by the 2 investigators together while evaluation of the citations was done independently by each of the 2 investigators, and disagreements were reconciled at scheduled meetings. Studies were included if they were carried out on humans, had comparison groups, and had cataract as an outcome. Placebo or usual care was used as comparison group. Basic science studies, nutrition or animal studies, reviews, editorials/letters, case reports, and studies without comparison groups were excluded. Using the search strategy described in Supplemental Table 1, we identified 363 potentially appropriate titles for possible inclusion in the analysis (Figure 1). Of the 363 records retrieved (359 by the literature searches and 4 identified from other sources, that is, reading the references of reviews and the articles retrieved), we excluded 185 duplicate titles and screened 178 abstracts relevant to the analysis. Of the remaining 178 records,110 were excluded by reading the full-text articles. Of the remaining 68 records, 55 were excluded because they did not include cataract as an outcome, were basic science, animal or nutrition studies, duplicate reports from the same trial, reviews, editorials, letters, case reports, or they did not have a comparison group leaving 13 records for inclusion in the meta-analysis. 16 –28
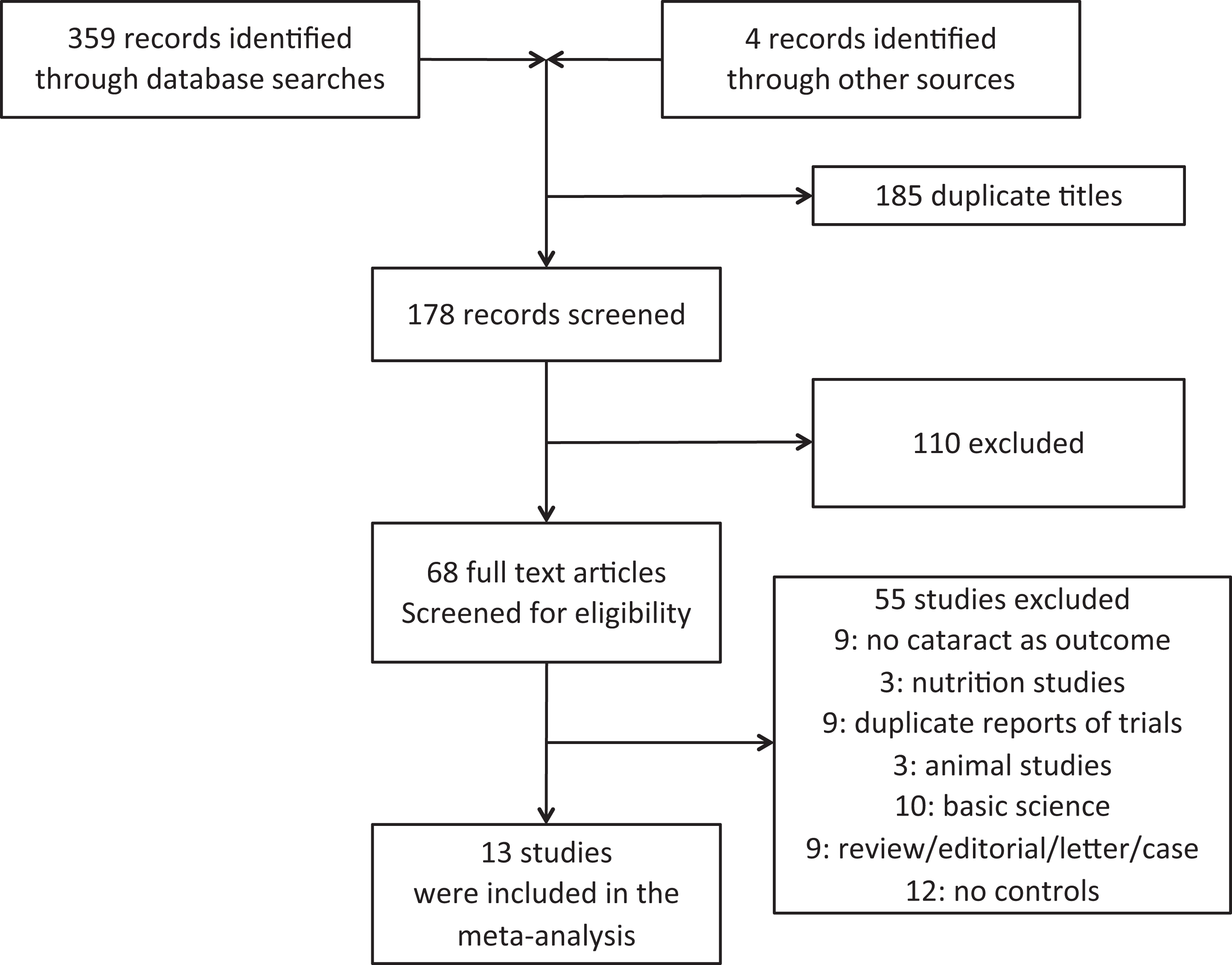
Search strategy.
Each of the 2 authors independently reviewed and tabulated data individually by reading the reports of all studies included in the meta-analysis and adjudicated disagreements by discussion. Two end points were analyzed: clinical cataract (those requiring extraction or those reported by the patient) and lens opacities (discovered by slit-lamp examination). Since age-matched reports of the number of cataracts were not available in the reports by Chodick et al, 16 Schlienger et al, 24 Smeeth et al, 25 Collins and Altman, 18 and Tan et al, 26 we used the weighted average of age-adjusted hazard ratios and the number of cataracts in the comparison groups to calculate the number of cataracts in the active groups. In the study by Havel et al, where no opacities were observed in either the placebo or lovastatin group, we entered 1 cataract in each group in order to avoid division by 0 to obtain an odds ratio (OR). 27 The specific statin, type of study (randomized vs observational), duration of follow-up in months, percentage of patients who were men, and average age were recorded. Lovastatin was used in 3 studies, 17,22,27 and simvastatin in 4 studies. 19,23,28 In 7 studies, more than 1 statin was used, and the data were presented in the aggregate rather than by individual statin. The following statins were used in these 7 studies: in the study by Tan et al, simvastatin, fluvastatin, lovastatin, atorvastatin, and pravastatin were used. 26 In the study by Hermans et al, simvastatin, atorvastatin, rosuvastatin, and pravastatin were used. 20 In the study by Klein et al, simvastatin, fluvastatin, lovastatin, atorvastatin, and pravastatin were used. 21 In the study by Schlienger et al, simvastatin (72%), pravastatin (18%), and other (10%) were used. 24 In the study by Smeeth et al, simvastatin, atorvastatin, pravastatin, fluvastatin, and cerivastatin were used. 25 In the study by Chodick et al, lovastatin, pravastatin, simvastatin, fluvastatin, cerivastatin, and rosuvastatin were used. 16 In the study by Collins and Altman, pravastatin, simvastatin, fluvastatin, rosuvastatin, and atorvastatin were used. 18 In the study by Pedersen et al, cataract was not the primary end point, and the data were derived by the authors in a post hoc analysis. 23 In studies where all pertinent information was not included in the primary publication of the trial, we used other publications from the same study in order to obtain the data. When specific data on the rate of the occurrence of cataract were not included in the publications, we contacted the senior authors to obtain the requisite information. When the dose of the individual statins used was known, the relative dose was calculated by multiplying the dose used times the relative potency (1 for lovastatin, fluvastatin, and pravastatin; 2 for simvastatin; 4 for atorvastatin; and 8 for rosuvastatin). 29 –31 Metaregression of studies with known relative potency was performed. There were no studies using either atorvastatin or rosuvastatin alone.
In addition to examining studies identified by the search strategy, the authors performed an additional search of the primary publications of the important randomized trials of statin therapy in cardiovascular disease. This was performed in order to identify additional studies where cataract was not an end point but was mentioned in the text. Mention of cataract was not detected in any of the 27 trials examined (Supplemental Table 2).
Statistical Methods
The primary outcome examined was cataract. Specific subanalyses were performed for clinical cataracts and opacities separately. For clinical cataracts, metaregression was performed by the specific type of study outcome, that is, cataract extraction, mixed outcome (including some extractions and some cataracts reported by patients), and studies that included only cataracts reported by patients. The number of primary and secondary end points and the number of patients in the statin and comparison groups were recorded. The types of all analyses were prespecified.
Data Synthesis and Analysis
The rates of cataract were calculated for the intervention and comparison groups of each study. JMP version 9.0.2 (2010 SAS Institute Inc, Cary, North Carolina) was used for univariate analysis and distributions, and Comprehensive Meta-Analysis 2.2 (Biostat, Englewood, New Jersey) was used to perform the meta-analyses and metaregressions. Odds ratios and 95% confidence intervals (CIs) were calculated for each study and weighted pooled effects were computed using random effects models. Heterogeneity was evaluated by the use of the Q statistic, and sensitivity analysis was performed by repeating the analyses 14 times while sequentially removing 1 study at a time. Additional sensitivity analyses were performed after exclusion of observational studies with very large numbers of patients, that is, Chylack et al
17
and Collins and Altman
18
studies together. Also, sensitivity analysis was done after the exclusion of the Havel study, where no cataracts occurred in either of the randomized groups. Randomized and observational studies were analyzed separately. In addition, we performed sensitivity analysis by excluding the study by Havel where cataract was not observed in either the statin or the comparison group. We examined publication bias by performing cumulative meta-analysis by the Duval and Tweedie’s Trim and Fill method and the fail-safe N models of Rosenthal and Orwin.
32
–34
Metaregressions of the log ORs of the effects over the duration of the studies and over the mean age of the participants of each study were performed. To investigate whether the effect of statins on cataract was influenced by the gender of the participants, we performed 2 additional sensitivity analyses: comparing the studies with percentage of men <55% to all other studies and comparing the studies with percentage of men
Results
Description of the studies
The characteristics of the studies included are shown in Table 1, and the quality of the studies are shown in Supplemental Table 3. The following items were assessed for each study: randomization, method of diagnosis of cataract, baseline similarity, defined eligibility, placebo, adjustment for confounders, and blinding. In addition, possible bias in each study is tabulated using the guidelines laid down by the Cochrane library (Supplemental Table 4).
Listing of Studies Included in the Meta-Analysis.


Continued.

Abbreviations: Avg, average; AR, absolute risk; FU, follow-up; N, number RR, relative risk; MSD, Merck Sharp Dohme; MRC, Medical Research Council.
The meta-analysis included 13 clinical trials. 16 –28 One of the studies had separate data for clinical cataract and for lens opacities detected by slit-lamp examination. 23 Overall, the meta-analysis included 2 403 644 patients (2 399 200 if data from Pedersen et al 23 with separate analyses for clinical cataract and lens opacities were counted only once) and 25 618 cataracts. The average number of patients in the studies analyzed was 171 689, standard deviation (SD) 586 097, median 2746. When the 2 largest observational studies 16,18 were not included, the total number of patients was 92 577 (average 7715, SD 12 573, median 1 046), and the total number of cataracts was 2465 (average 205, SD 227, median 125). The average duration of treatment was 54 months, SD 43, median 60; average age was 61 years, SD 8, median 61; and 58% were men, SD 16, median 55.
Clinical Cataracts and Opacities
Overall, statin use was associated with a 19% decrease in the risk of cataract (OR 0.81, 95% CI 0.72-0.92,

Forest plot of the effect of statins on cataract by type of cataract (clinical, ie, reported by the patient vs opacities determined by slit-lamp examination). Solid squares represent the odds ratios in individual trials and have a size proportional to the inverse of the variance. Horizontal lines, diamonds, and squares denote the 95% confidence intervals for individual trials and summary statistics. Pooled estimates were computed from a random effects model. For the clinical trials, Q was 6.079 with 9 degrees of freedom,

Forest plot of the effect of statins on cataract in observational and randomized trials. The effect observed in the 8 observational studies was statistically significant (OR 0.81, 95% CI 0.70-0.93,

Metaregression of duration of statin therapy in months versus the log odds ratio for cataract.
There was no significant heterogeneity among any of these analyses with

Forest plot of the effect of statins on cataract by whether cataract extraction or no extraction was the dependent variable. The benefit is significantly more pronounced for studies reporting cataract extraction than in those that used softer end points (overall
Sensitivity Analysis
To investigate whether 1 study biased the results, we performed the analysis 14 times removing 1 study at a time sequentially. The ORs ranged from 0.78, 95% CI 0.69-0.88,
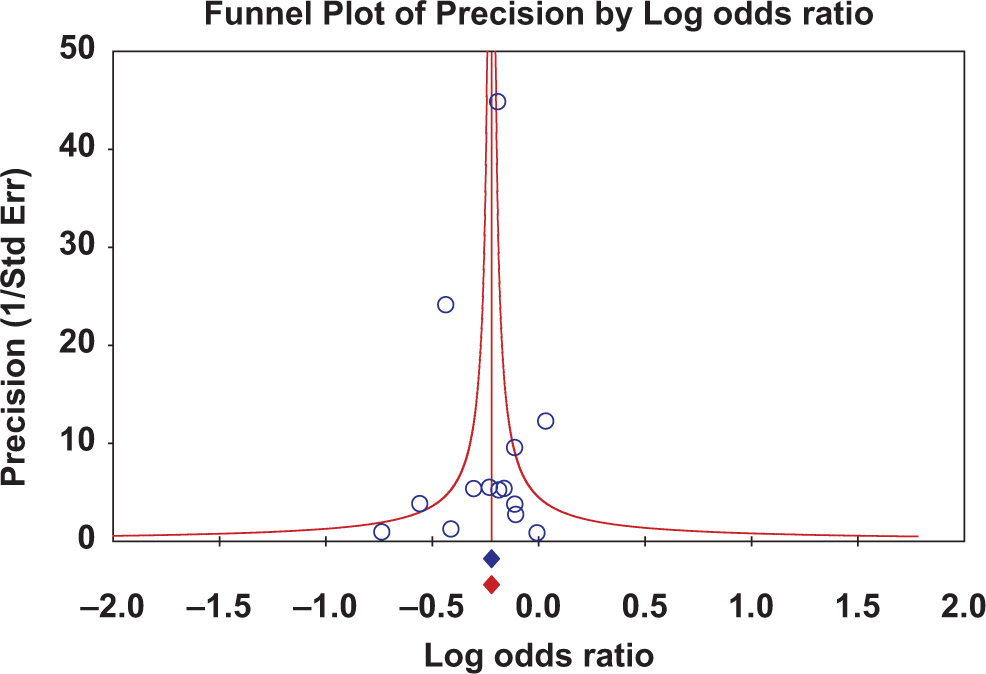
Funnel plot of precision (1/standard error) versus log odds ratio is symmetrical.
When the analysis was restricted to the 13 smaller studies by excluding the study by Collins, the benefit of statins remained significant and nearly identical to the one described previously (OR 0.81 95% CI 0.69-0.95,
When the 2 larger studies (Chylack
17
and Collins and Altman
18
) were excluded, the OR was 0.91, 95% CI 0.82-1.0,
Discussion
This study demonstrated a clinically relevant and statistically significant protective effect on statins (19% lower rate of occurrence). This effect remained significant after several types of sensitivity analysis, was present in both studies reporting on cataract extraction as well as those with softer end points, and there was no significant heterogeneity among studies included in the analysis. The protective effect of statins in preventing cataracts was more pronounced in younger patients and with longer duration of follow-up. The bigger benefit in younger persons and the related decrease in the benefit of advanced age may be due to the slow time course of the development of cataract and the increased baseline risk of this condition in older persons. We did not observe a difference with respect to gender. Individual studies have not shown a statistically significant benefit of statins in women especially in primary prevention. 3 This may be due to the lower representation of women in these studies since a meta-analysis of randomized trials with gender-specific outcomes demonstrated that statin therapy was associated with significant decreases in cardiovascular events and in all-cause mortality in women as well as in men. In agreement with this meta-analysis, we observed similar benefits with respect to cataract in men and women. 3 New onset diabetes occurs in some patients with statin therapy, and the risk is higher with high-intensity dose statin therapy usually with rosuvastatin or atorvastatin. 35 In this meta-analysis, there were no studies where either atorvastatin or rosuvastatin were used alone, and the dose was available only in 6 studies. Examining these 6 studies, we did not find a significant relationship between relative dose of statins and cataract.
Although the benefit of statins in preventing cardiovascular events has been established by controlled clinical trials, reviews, and meta-analyses and has been vetted by clinical guidelines throughout the globe, there have been concerns about a possible association of statins with adverse events such as cancer, cognitive dysfunction, and diabetes as well as with cataracts. 5,6 The concern that statins might cause cataracts when these agents were first marketed persists among some physicians, patients, pharmacists, and other health care providers as reported in electronic drug information sites. 11 –13
The mechanism of the effect of statins in preventing cataracts is not known. It may be related to low-density lipoprotein lowering although our literature search did not identify a citation relating cataracts with familial hypercholesterolemia. The benefit may be related to an antioxidant pleiotropic effect of statins. 36 Patients with cataract had lower plasma antioxidant levels and higher levels of oxidative stress than those of healthy controls. 37
This study has limitations inherent to meta-analyses including studies of different designs, randomized clinical trials where cataract was not a predefined end point, imprecise ascertainment of exposure in several observational studies, incomplete adjustment for confounders, possible reporting bias, and incomplete follow-up in some studies. It is possible that the combination of observational studies and randomized studies in the same meta-analysis has increased the introduction of bias and confounding and may have magnified the effect. To address this question, we performed the meta-analysis both separately for observational and randomized trials and with both types of trials together. The effect observed in the 8 observational studies is statistically significant while the effect in the 6 randomized trials was similar (OR 0.84 vs OR 0.81) in magnitude but not statistically significant. The absence of a statistically significant effect in the randomized trials may imply that the effect, if it exists, is small. Observational studies have inherent biases and confounding whose identity is difficult, if not impossible, to discern and they may introduce bias and confounding while decreasing the role of chance. Combined analysis of randomized and observational studies is not uncommon and as Concato and associates reported that the results of well-designed observational studies do not systematically overestimate the magnitude of the effects. 38
However, our study has significant strengths in that it includes all published reports on the topic and that the effect is consistent when analyzed from various aspects. The effect of statins in preventing cataract was significantly more pronounced for the hard end point of cataract extraction than patient reported or slit lamp detected cataract where there may be detection bias. This point makes our observations more reliable, since there is no detection bias with respect to cataract extraction. Also, there was no publication bias as determined by a symmetrical funnel plot.
Conclusion
In conclusion, this meta-analysis indicates a clinically relevant protective effect of statins in preventing cataracts, and this effect is more pronounced in younger patients and with longer duration of follow-up while there is no effect by gender.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Kotis has served on the speakers bureau and as consultant for Merek, Sanofi, Pfizer, related to the treatment of hypercholesterolemia.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
