Abstract
Direct experimental safety comparisons of Xa coagulation factor direct inhibitors, apixaban and rivaroxaban, on their approved therapeutic indications have not been identified. Due to recently raised safety concerns, a meta-analysis was carried out pooling data from studies identified on a Medline and Cochrane Library search in order to better evaluate the safety profile of both drugs. Abstracts from scientific meetings were also searched from 2003 to 2011. Primary and secondary outcome measures were major bleeding and total bleeding, respectively. Relative risks (RRs) were estimated using random effects models and statistical heterogeneity was estimated with I2 statistics. Of the 160 screened publications, 12 clinical trials were included in which enoxaparin was the active control. For knee arthroplasty, apixaban was associated with significantly fewer major bleeding events (6496 patients, RR 0.56, 95% confidence interval [CI] 0.32-0.96) and fewer total bleeding events (6496 patients, RR 0.81, 95% CI 0.67-0.97). There were no significant differences in the incidence of major bleeding events (5699 patients, RR 1.40, 95% CI 0.56-3.52) or in the incidence of total bleeding events for rivaroxaban (5699 patients, RR 1.09, 95% CI 0.91-1.30). No differences were found when thromboprophylaxis after hip replacement was the case. Apixaban seems to be associated with a lower risk of the incidence of hemorrhagic events after total knee arthroplasty. For hip arthroplasty, no differences were found between the studied drugs.
Introduction
Patients submitted to major orthopedic surgery, such as elective total knee or hip arthroplasty, represent a group at high risk of venous thromboembolism (VTE). 1 Almost half of the patients who underwent arthroplasty are affected by asymptomatic deep venous thrombosis (DVT), although most of these thrombi resolve without long-term complications. 2 –4 For some patients, propagation of the existing thrombus can cause symptoms as a result of venous occlusion. 5 Symptomatic VTE is common after discharge from hospital. 6,7 The most frequent cause for readmission to the hospital following total knee arthroplasty is VTE. 8
The provision of thromboprophylaxis during hospitalization has been established as standard of care for the last 2 decades. 9 With anticoagulant therapy, incidence of fatal pulmonary embolism (PE) decreased to 0.2%, although symptomatic VTE continues to be reported in 1.3% to 10% of patients within 3 months after surgery. 2,10 Low molecular weight heparins (LMWHs), fondaparinux, and vitamin K antagonists have been used as pharmacological agents for VTE prophylaxis. Although these therapies have shown to be effective, they are associated with some practical limitations. 11 Low molecular weight heparin and fondaparinux have the inconvenience of subcutaneous administration, which also can increase the risk of injection site hematomas. 12,13 Furthermore, subcutaneous administration of anticoagulants is difficult to provide after hospital discharge. Vitamin K antagonists are being abandoned in Europe due to concerns about their delayed onset of action, unpredictable pharmacokinetic and pharmacodynamic effects, and need for frequent monitoring. 14,15 Mechanical VTE prophylaxis is known to be cumbersome, and its efficacy is found to be lower when compared with anticoagulant therapy, especially after hip arthroplasty. 2
The specific limitations of the currently available anticoagulant agents led to the development of new therapies for preventing VTE. 16 –18 The direct thrombin inhibitor, dabigatran (Pradaxa, Boehringer Ingelheim, Ingelheim am Rheim, Germany), and the oral direct inhibitors of coagulation factor Xa, rivaroxaban (Xarelto, Bayer, Berlin, Germany) and apixaban (Eliquis, Bristol-Myers Squibb/Pfizer EEIG, Uxbridge, UK), were recently approved by the European Medicines Agency (EMA) for thromboprophylaxis after total knee and hip arthroplasty. 19,20 Both oral coagulation factor Xa direct inhibitors were approved based on evidence provided by phase III randomized clinical trials (RCTs) using once daily 40 mg enoxaparin (European regimen) or twice daily 30 mg enoxaparin (North American regimen) as the active control. 21 –24 These studies found that oral coagulation factor Xa direct inhibitors are effective in VTE prevention. However, safety concerns have been raised regarding thromboprophylaxis with rivaroxaban. 25 –28 One study has shown an increased risk of wound complications associated with rivaroxaban. 25 Since the prevalence of fatal PE after total knee arthroplasty is 0.1% and the risk of major and clinically relevant bleeding events with thromboprophylaxis is 3.0%, risk and benefits should be balanced before starting anticoagulation therapies. 29 The safety of Xa coagulation factor direct inhibitors holding a European market authorization, apixaban and rivaroxaban, have never been evaluated by direct comparisons in randomized controlled trials.
Objective
The aim of this study was to carry out a meta-analysis in order to comparatively evaluate the safety profile of the direct inhibitors of coagulation factor Xa, apixaban and rivaroxaban, as thromboprophylaxis agents after total knee or hip arthroplasty.
Methods
We searched EMA Web site for direct inhibitors of coagulation factor Xa which had been already approved. 30,31 Data search and safety analysis were performed according to the therapeutic indication approved by EMA.
Search Strategy
Medline and the Cochrane Library were searched from its inception until June 27, 2011 in order to identify relevant studies comparing direct factor Xa inhibitors with enoxaparin. Search terms related with knee and hip arthroplasty (eg “knee operation,” “knee surgery,” “knee arthroplasty,” “hip arthroplasty,” “hip replacement,” and “hip surgery”) were combined with thromboembolism prophylaxis terms, such as “thrombosis prophylaxis,” “deep venous thrombosis prophylaxis,” and “PE prophylaxis.” Text words, brand names, and manufacturer's coded designations were used to identify both factor Xa inhibitors. All languages were considered in the search strategy. The bibliographic list of all relevant clinical trials was hand searched in order to identify additional eligible studies. Study lists from systematic reviews and meta-analysis identified during the search process were also considered. The databases of the American Society of Haematology (starting on the 2004 issue) and the International Society on Thrombosis and Haemostasis (starting on the 2003 issue) were searched in order to identify relevant studies published as abstracts. Food and Drug Administration (FDA) and EMA publicly available records were searched in order to identify studies that met our inclusion criteria. Articles that were not available were requested to the authors. The electronic databases search strategy is available in the Table A1.
Study Selection
Literature was searched and relevant studies were examined for further assessment. The studies inclusion criteria were (1) randomized, controlled clinical trials, (2) patients of all ages undergoing total hip or knee arthroplasty, (3) comparison of safety of factor Xa direct inhibitors with enoxaparin for thromboprophylaxis. Only the oral direct inhibitors of factor Xa holding marketing authorizations in the EU were evaluated. Both trials with blinded and unblinded design were included. Studies focusing on pharmacokinetic or pharmacodynamic variables were excluded.
Qualitative Assessment
Quality assessment of studies was based on the recommendations of the Cochrane Handbook of Systematic Review of Interventions on assessing adverse effects. 32 The value of trial data on adverse effects relies on 2 major characteristics: the rigor of monitoring for the adverse effects during the study and the completeness of reporting. Allocation concealment and the withdrawal rates were also evaluated.
Data Extraction
Two reviewers independently extracted data from the included studies. Any disagreements were resolved by consensus or arbitration by a third reviewer. We contacted the authors of studies for missing data when necessary. Data on study characteristics (methodology, included population, study design, and drugs evaluated) and outcomes (bleeding and adverse events) during treatment were extracted.
Outcomes Assessed
The primary outcome of this meta-analysis was the incidence of major bleeding beginning after the first dose of the study drug and up to 2 days after the last dose of the study drug (on-treatment period). Major bleeding was defined as bleeding that was fatal, involved a critical organ (eg, retroperitoneal, intracranial, intraocular, and intraspinal), required reoperation or that was clinically overt, extra-surgical site bleeding associated with a fall in hemoglobin level of at least 2 g/dL, calculated from the day 1 postoperative baseline value, or requiring infusion of 2 or more units of whole blood or packed cells. Other safety outcomes included any on-treatment bleeding, any on-treatment clinically relevant nonmajor bleeding, drug-related adverse events, drug-related serious adverse events, and wound complications. Wound complications outcome is the composite of major and/or nonmajor wound bleedings. Clinically relevant nonmajor bleeding was defined according to the RCTs that evaluated apixaban and rivaroxaban. 21
Statistical Analysis
Review Manager (RevMan) version 5.1.2 (Cochrane Collaboration, Oxford, UK) was used to calculate relative risk (RR) and 95% confidence intervals (CIs) for all the primary and secondary outcomes throughout the meta-analysis. All reported P values are 2-sided with significance set at <.05. Statistical heterogeneity was assessed by calculating a chi-square test and the I 2 measure of inconsistency. 33 Statistical heterogeneity was considered low when 0% < I 2 < 25%, moderate when 25% < I 2 < 50%, and high when I 2 > 50%. We planned to pool data across studies using the DerSimonian and Laird random effects model. 34 The publication bias was assessed by examining the funnel plot. 35 We performed a sensitivity analysis to explore the influence on effect size of blinding of outcome assessment and the methodological quality of included trials. For each Xa coagulation factor direct inhibitor, the results of total hip or knee arthroplasty subgroups were compared with the overall meta-analysis. For the sensitivity analysis, RCTs using twice daily enoxaparin 30 mg (North American regimen) as active control were excluded.
Results
Study Selection
Figure 1 presents the flow of the search strategy criteria. The electronic databases searches returned 165 potentially relevant articles. After review of the titles and abstracts, 142 citations were refused; 16 articles were duplicates; 23 articles were selected for further evaluation. After application of inclusion criteria, 12 studies were eligible for inclusion. 21 –24,36 –42 The review of reference lists scientific society’s databases did not find any other relevant studies. No further studies were identified in the FDA and EMA publicly available records.
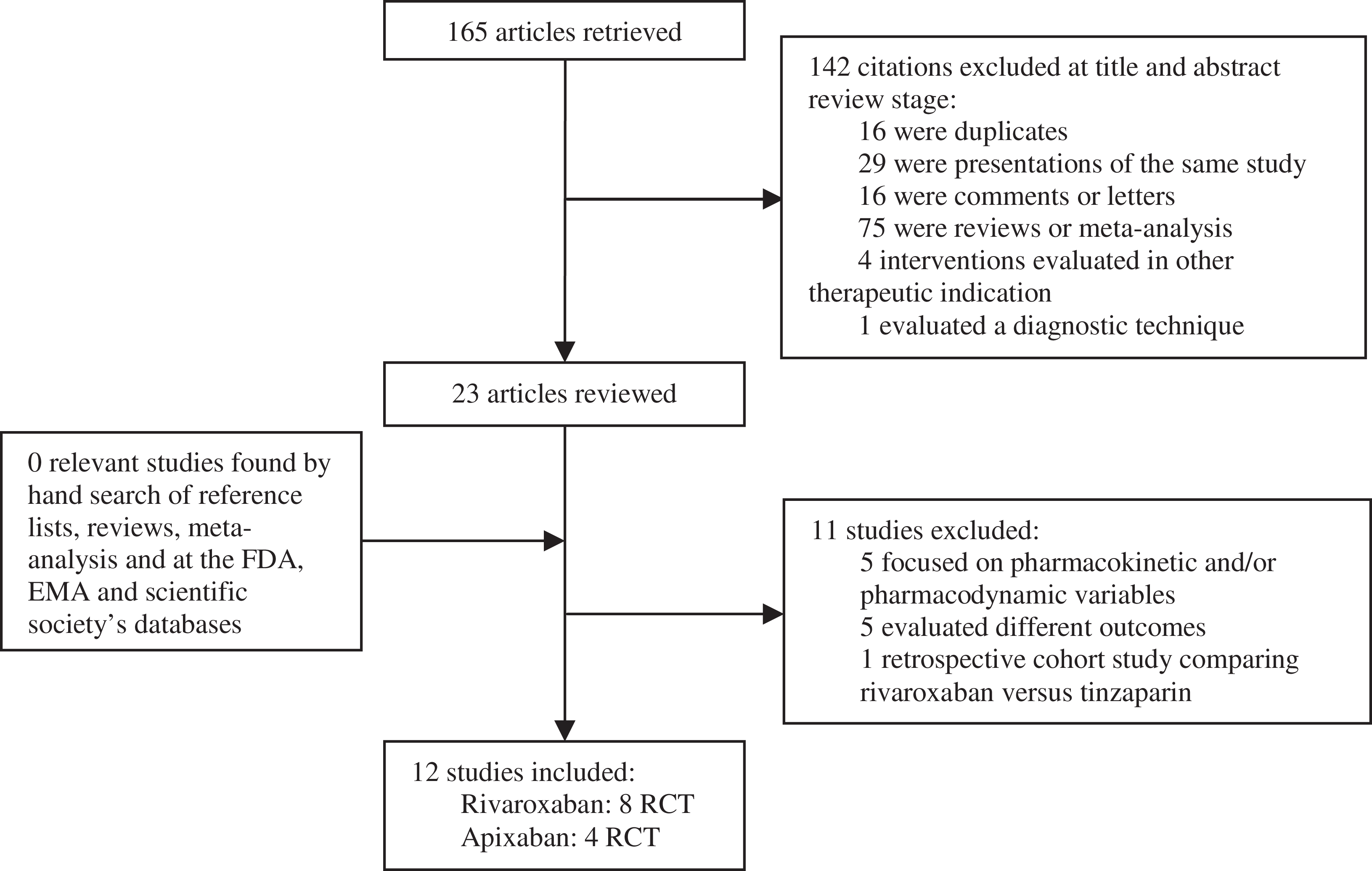
Flow diagram of identification of studies for inclusion.
Characteristics of the Included Studies
The main characteristics of the studies are presented in Table 1. Study design, duration of treatment, demographic characteristics of patients, drugs under evaluation, and number of participants are described. A total of 12 RCTs were found, of which 8 evaluated the efficacy and safety of rivaroxaban versus enoxaparin and 4 evaluated the efficacy and safety of apixaban versus enoxaparin. These clinical trials included 28 483 patients, in which 15 586 were randomized to receive rivaroxaban or enoxaparin, and 12 897 were randomized to receive apixaban or enoxaparin. All the RCTs were performed in adult patients undergoing total knee or hip arthroplasty. Five RCTs evaluated rivaroxaban for thromboprophylaxis in patients undergoing total hip replacement and 3 RCTs evaluated rivaroxaban for thromboprophylaxis in patients undergoing total knee replacement. Apixaban was evaluated as thromboprophylaxis agent after total knee arthroplasty in 3 RCTs and 1 RCT evaluated apixaban as thromboprophylaxis agent in patients undergoing total hip replacement.
Characteristics of the Studies Included in the Meta-analysis
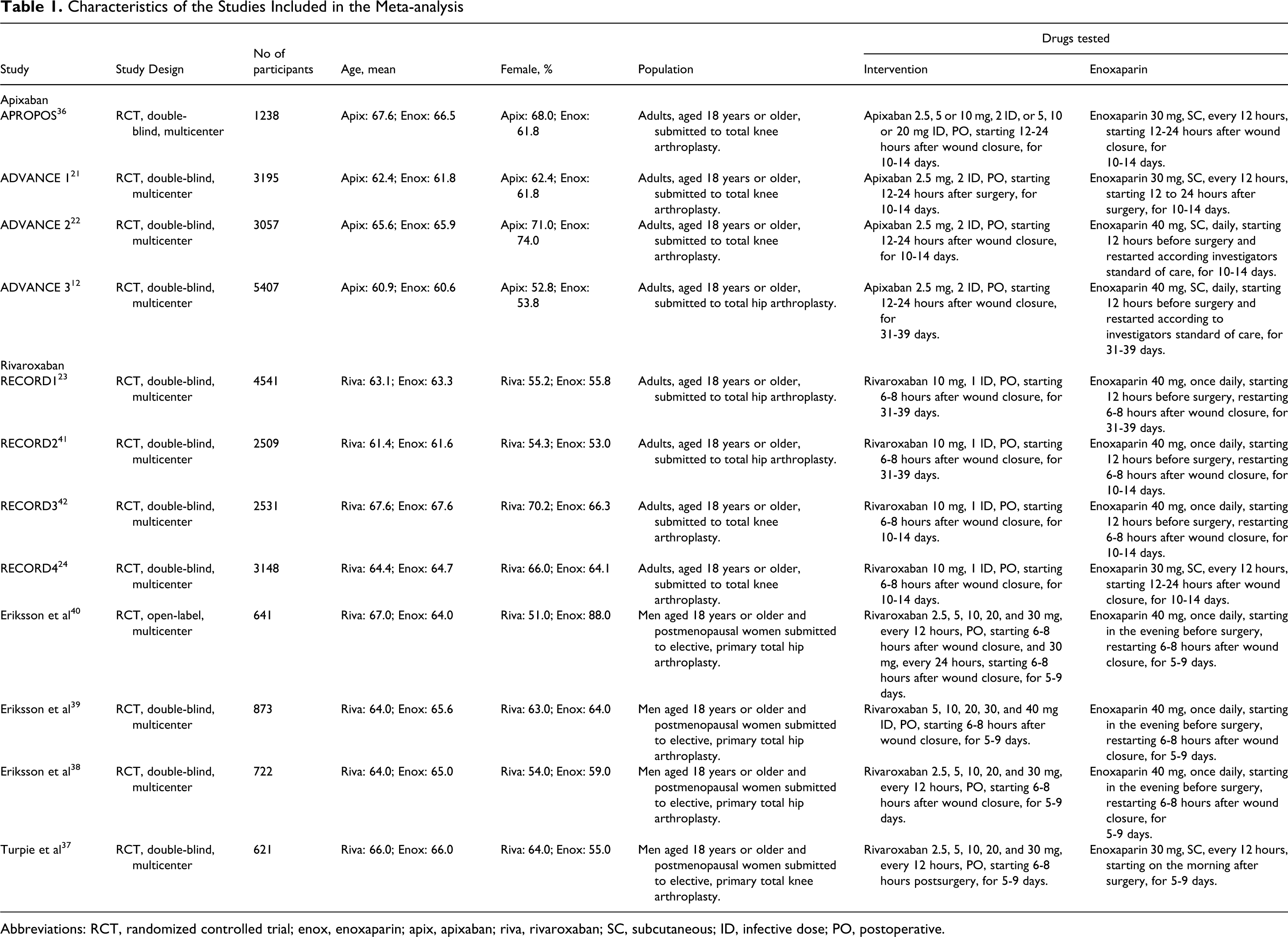
Abbreviations: RCT, randomized controlled trial; enox, enoxaparin; apix, apixaban; riva, rivaroxaban; SC, subcutaneous; ID, infective dose; PO, postoperative.
Rivaroxaban treatment schedules for thromboprophylaxis were comparable among the included RCTs. The patients of the rivaroxaban group received the first dose 6 to 8 hours after skin wound closure. To avoid clinical heterogeneity, for the dose-ranging studies, only the patients that received a total daily dose of 10 mg were included in the analysis. In our study, all the patients received 10 mg/d of rivaroxaban (single dose or 5 mg twice day).
In the 4 apixaban RCT, the apixaban treatment group received the first dose 12 to 24 hours after wound closure. In the apixaban dose-ranging study, only the patients that received 2.5 mg twice daily were included in the analysis. All the patients included in our study treated with apixaban received 2.5 mg twice a day.
The included treatment arms of apixaban and rivaroxaban correspond to the approved total daily dosage for thromboprophylaxis after total hip or knee arthroplasty, which is 5 mg for apixaban, given 2.5 mg twice daily, and 10 mg for rivaroxaban, given once daily or 5 mg twice daily.
Clinical trials included in the meta-analysis used either the enoxaparin dose and regimen approved for use in Europe (40 mg once daily, first dose received 12 hours or the evening before the surgery and medication resumed 6-8 hours after wound closure) or the regimen approved for use in North America (30 mg twice daily, first dose received on the morning after surgery or 12 or 24 hours after wound closure). The enoxaparin European regimen was administrated in 6 RCTs that evaluated rivaroxaban and in 2 RCTs that evaluated apixaban. The enoxaparin (North American regimen) was administrated in 2 RCTs that evaluated rivaroxaban and in 2 RCTs that evaluated apixaban.
Quality Assessment of the Included Studies
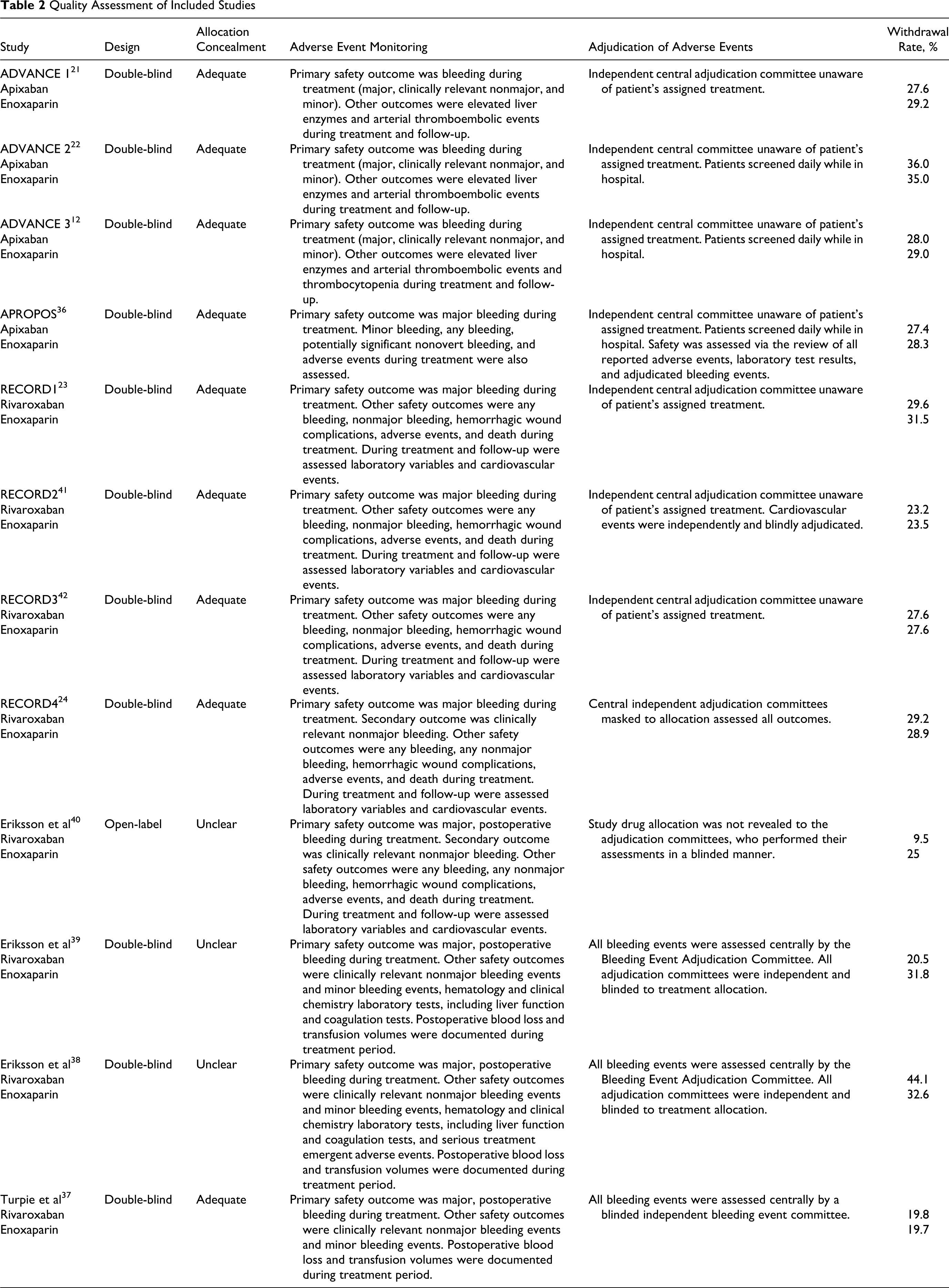
The quality assessment of the included trials is presented in Table 2. All but 1 RCT were double blind. Allocation concealment was adequate in 9 trials. In 3 trials, allocation concealment was unclear. Information on withdrawal rates was available for all trials and ranged from 9.5% to 44.1%. The monitoring of bleeding events was performed in all trials. Besides bleeding events, few adverse effects were specifically monitored. All trials used an independent adjudication committee masked to allocation to assess the outcomes.
Quality Assessment of Included Studies
Publication Bias
We examined the funnel plot (standard error [SE] of log RR plotted against RR) and did not find evidence of publication bias.
Safety Evaluation Results
Results of the meta-analysis comparing apixaban and rivaroxaban with enoxaparin are shown in Tables 3 and 4.
Outcomes of Meta-analysis Comparing Apixaban and Enoxaparin
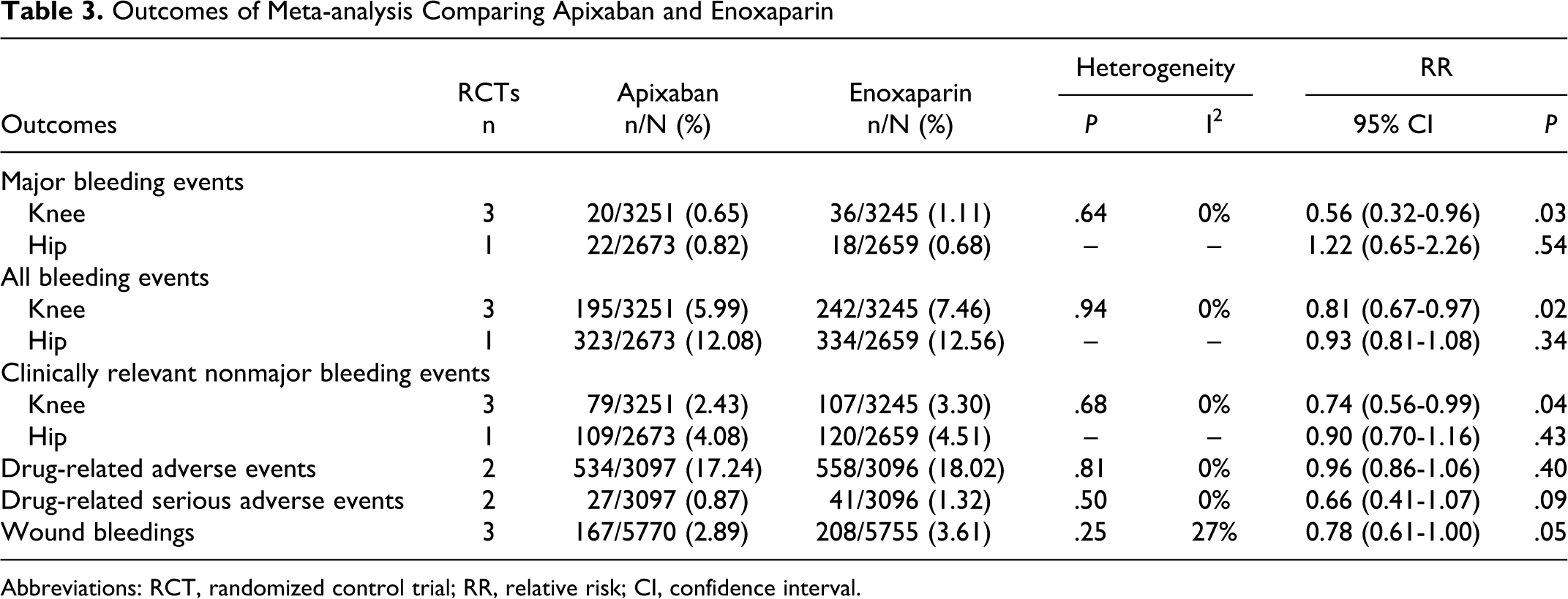
Abbreviations: RCT, randomized control trial; RR, relative risk; CI, confidence interval.
Outcomes of Meta-analysis Comparing Rivaroxaban and Enoxaparin
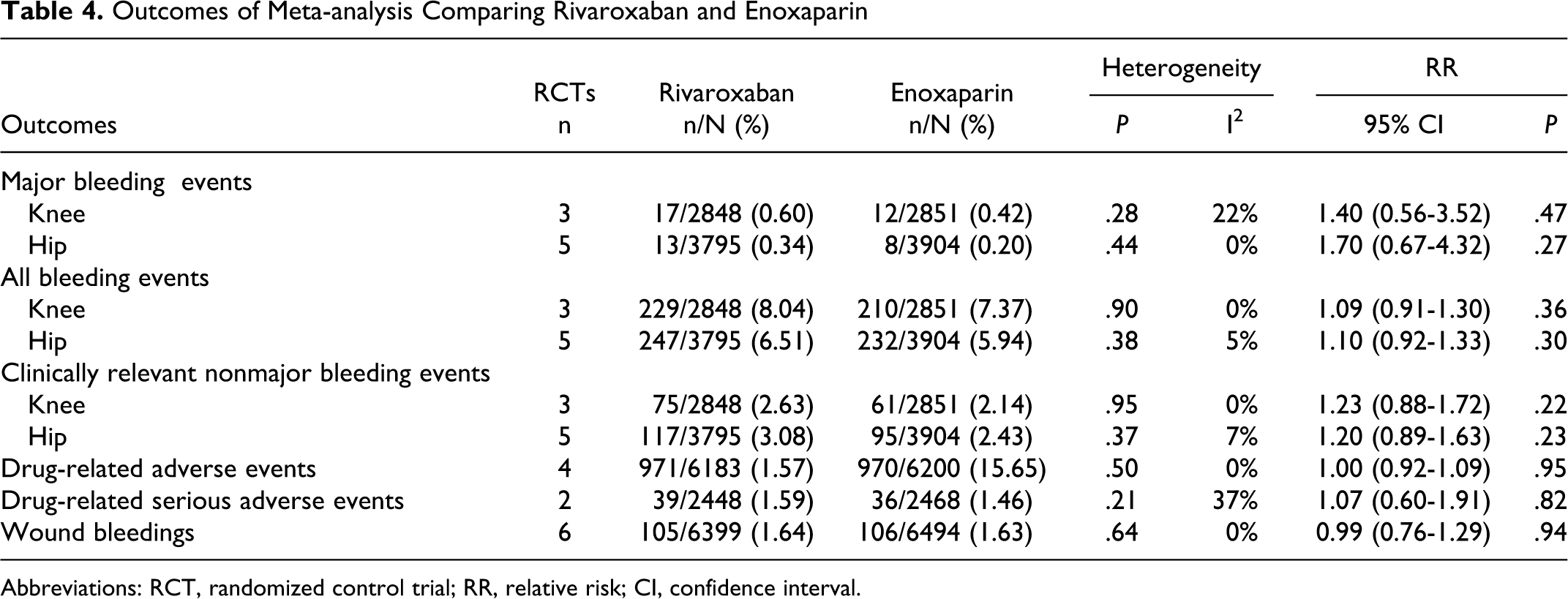
Abbreviations: RCT, randomized control trial; RR, relative risk; CI, confidence interval.
According to the results, apixaban presented a more favorable safety profile compared with enoxaparin in the following outcomes: major bleeding events, all bleeding events, and clinically relevant nonmajor bleeding events for thromboprophylaxis after total knee arthroplasty. No significant differences were found when thromboprophylaxis after total hip arthroplasty was the case. For rivaroxaban, no statistically significant differences in safety profile were found when compared with enoxaparin.
Sensitivity Analysis
The sensitivity analysis limited to double-blind RCTs did not change the results. After removing a phase II, dose-ranging RCT, there were no significant differences for the incidence of clinically relevant nonmajor bleeding events between apixaban and enoxaparin thromboprophylaxis after total knee arthroplasty (6193 patients, RR: 0.75, 95% CI: 0.56-1.00, P = .05). For all other outcomes in knee or hip arthroplasty thromboprophylaxis, the removing of any other phase II, dose-ranging, study did not change the results.
For the sensitivity analysis, studies that compared direct inhibitor of coagulation factor Xa with twice daily enoxaparin 30 mg were excluded (Table A2). The removal of RCTs using enoxaparin on the North American regimen as active control did not significantly affect any outcome with rivaroxaban after total hip arthroplasty. One RCT compared rivaroxaban and enoxaparin on European regimen for thromboprophylaxis after total knee arthroplasty. 42 The results of that RCT were similar to those obtained in this meta-analysis, for all outcomes. One RCT compared apixaban with enoxaparin European regimen for thromboprophylaxis after total knee arthroplasty. 22 The results of this RCT were different from those obtained in this meta-analysis. According to the results of that RCT, there were no significant differences in the incidence of bleeding outcomes when apixaban was compared with once daily enoxaparin 40 mg (major bleeding events: 3009 patients, RR: 0.65, 95% CI: 0.28-1.49, P = .30; all bleeding events: 3009 patients, RR: 0.83, 95% CI: 0.65-1.06, P = .14; clinically relevant nonmajor bleeding events: 3009 patients, RR: 0.76, 95% CI: 0.52-1.12, P = .17). After removing the RCTs that used enoxaparin on North American regimen as active control, no statistically significant differences were observed for wound bleeding rates, drug-related adverse event rates, and drug-related serious adverse event rates for both apixaban and rivaroxaban.
Discussion
Clinical guidelines recommend pharmacological prophylaxis for patients undergoing total knee and hip arthroplasty for at least 10 days after the surgery. 2,3,29 New oral anticoagulants, such as apixaban and rivaroxaban, may provide a more suitable antithrombotic therapy and increase patient compliance, when compared with available alternatives (LMWH and vitamin K antagonists).
Several RCTs compared rivaroxaban versus enoxaparin for thromboprophylaxis after total knee or hip arthroplasty, reporting higher efficacy of rivaroxaban when compared with both European and North American enoxaparin regimens. 23,24,41,42 A previous meta-analysis confirmed that rivaroxaban was superior to enoxaparin as a thromboprophylaxis agent after total hip and knee surgery. 27 However, a higher number of bleeding events associated with rivaroxaban was identified. As a consequence, the authors do not recommend the use of rivaroxaban in patients susceptible to hemorrhage.
Apixaban was proven to be at least as effective as enoxaparin for thromboprophylaxis after total knee or hip arthroplasty. 12,21,22 Although in ADVANCE 1 study apixaban was not shown to be superior to enoxaparin (North American regimen) after knee arthroplasty, it was associated with a lower bleeding risk. 21 When compared with enoxaparin European regimen, apixaban was superior in preventing thromboembolism after knee and hip arthroplasty without increased bleeding risk. 12,22 Another published meta-analysis points out apixaban as effective as enoxaparin for thromboprophylaxis after total knee arthroplasty. 43 In this study, apixaban is associated with significantly fewer major bleeding events. These findings raised the need to comparatively evaluate the safety profiles of apixaban and rivaroxaban, once both the drugs were proven to be efficacious in preventing VTE events.
Our meta-analysis included enoxaparin controlled trials, in the absence of studies comparing directly both drugs. The results suggest that thromboprophylaxis with apixaban after total knee arthroplasty is associated with a lower risk of major, clinically relevant nonmajor, and total bleeding events, when compared with rivaroxaban. No differences were observed when apixaban and rivaroxaban were compared for thromboprophylaxis after total hip arthroplasty.
In order to avoid clinical heterogeneity of the studies, 2 subgroup meta-analyses were carried out according to the approved therapeutic indications (hip and knee surgery). A sensitivity analysis was also performed aiming at preventing erroneous interpretations of the results. The removing of phase II, dose-ranging, RCTs from the meta-analysis did not significantly altered the results. Eight RCTs (2 with apixaban and 6 with rivaroxaban) used enoxaparin according to the European regimen (40 mg, once daily) and 4 RCTs (2 with apixaban and 2 with rivaroxaban) used enoxaparin according to the North American regimen (30 mg, twice daily). The indirect comparison of apixaban with rivaroxaban based on the enoxaparin European regimen significantly altered the results. These findings could suggest that twice daily enoxaparin 30 mg is associated with a higher risk of bleeding. Although both the enoxaparin regimens have never been directly compared, this risk was also observed in a previous meta-analysis. 43 Nonetheless, indirect comparisons between the 2 inhibitors of coagulation factor Xa based on both enoxaparin regimens (European and North American) consistently present the trend for lower bleeding risk in patients treated with apixaban.
The overall incidence of drug-related adverse reactions of any cause was similar for both drugs. Apixaban was found to be less associated with wound hemorrhages, although this difference did not reach statistical significance. However, when major and nonmajor wound hemorrhages are observed separately, such difference was no longer observed.
Preclinical studies have shown that both rivaroxaban and apixaban are highly selective for factor Xa. 17 Although it is suggested that rivaroxaban could be associated with an increased factor Xa inhibitory potential, 44 this may cause differences in the efficacy and safety profile of both drugs. Therefore, there is a rationale to compare both rivaroxaban and apixaban safety profiles, since differences in the incidence of PE and major bleeding events can change benefit–risk balance that supports therapeutic decisions.
For the phase II dose-ranging studies, both apixaban and rivaroxaban were compared with enoxaparin regimens. 36 –40 Based on their phase II programs, rivaroxaban 10 mg once daily and apixaban 2.5 mg twice daily have proven to have similar efficacy and safety to enoxaparin. Therefore, an indirect comparison can be established between rivaroxaban and apixaban safety profile based on their approved daily doses.
The findings of the present study are based on a pooled analysis of 28 483 patients. Safety outcomes considered in this meta-analysis were those defined in the included RCTs. To our knowledge, this is the first study comparing the safety of the 2 marketed direct inhibitors of coagulation factor Xa. The findings of this meta-analysis may be useful to more accurately establish benefit–risk ratios of both drugs and in the individualization of drug therapy.
The present study has limitations. First, although it includes the results of well-designed RCTs with a large number of patients, some relevant clinical outcomes such as rates of wound infection and wound healing were not assessed in all the included RCTs. 25 Second, only 1 RCT evaluated the safety of apixaban in the thromboprophylaxis after total hip arthroplasty. 12 Although the RCT included a large number of patients, the availability of more studies would strength the analysis. Third, some heterogeneity between RCTs was found. Such differences could be due to differences in prophylactic treatment duration but also to different enoxaparin regimens (North American and European) used as active control. However, heterogeneity should always be taken into account since each RCT included different populations. Fourth, the number of eligible studies to perform a sensitivity analysis is few. Although an indirect comparison between apixaban and rivaroxaban had been done based on the enoxaparin European regimen, the sample of both included trials could not be powered enough to detect significant differences.
Bleeding is a major concern in patients submitted to thromboprophylaxis after hip or knee arthroplasty after hospital discharge. 2,3,29 New direct inhibitors of the coagulation factor Xa have proved to be effective in reducing the risk of TVE in a single, unmonitored dose, given orally, which can lead to a more effective anticoagulant therapy. The results of this study suggest that apixaban may be a safer alternative than rivaroxaban for thromboprophylaxis, when total knee arthroplasty is the case. However, more studies are needed, in particular with direct comparisons, in order to better establish the risk profile of these 2 therapeutic agents.
Footnotes
Appendix
Results of the Sensitivity Analysis Comparing Apixaban and Enoxaparin (40 mg Once Daily) for Thromboprophylaxis After Total Knee Arthroplasty (ADVANCE 2 Study Results)
| RCTs | Apixaban | Enoxaparin | Heterogeneity |
RR |
|||
|---|---|---|---|---|---|---|---|
| Outcomes | n | n/N (%) | n/N (%) | P | I2 | 95% CI | P |
| Major bleeding events | 1 | 9/1501 (0.60) | 14/1508 (0.93) | – | – | 0.65 (0.28-1.49) | .30 |
| All bleeding events | 1 | 104/1501 (6.93) | 126/1508 (8.36) | – | – | 0.83 (0.65-1.06) | .14 |
| Clinically relevant nonmajor bleeding events | 1 | 44/1501 (2.93) | 58/1508 (3.85) | – | – | 0.76 (0.52-1.12) | .17 |
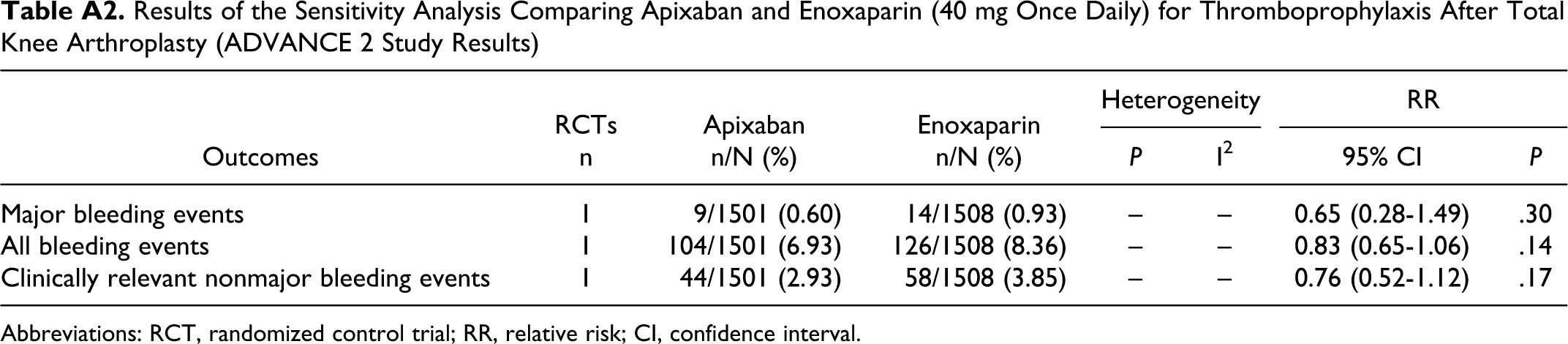
Abbreviations: RCT, randomized control trial; RR, relative risk; CI, confidence interval.
Acknowledgments
The authors would like to acknowledge the support of Dr Zilda Mendes, statistician at CEFAR–Portuguese National Association of Pharmacies, Lisbon, Portugal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Carlos Alves is supported by a research grant from Foundation for Science and Technology (FCT), Portugal, reference: SFRH/BD/64957/2009. Francisco Batel-Marques and Ana Filipa Macedo did not receive any financial support for the research, authorship, and/or publication of this article.
