Abstract
This study investigated the effect of 6-week supplementation of a probiotic strain Lactobacillus salivarius UBL S22 with or without prebiotic fructo-oligosaccharide (FOS) on serum lipid profiles, immune responses, insulin sensitivity, and gut lactobacilli in 45 healthy young individuals. The patients were divided into 3 groups (15/group), that is, placebo, probiotic, and synbiotic. After 6 weeks, a significant reduction (P < .05) in total cholesterol, low-density lipoprotein (LDL) cholesterol, and triglycerides and increase in high-density lipoprotein cholesterol was observed in the probiotic as well as in the synbiotic group when compared to placebo; however, the results of total cholesterol and LDL-cholesterol were more pronounced in the synbiotic group. Similarly, when compared to the placebo group, the serum concentrations of inflammatory markers such as high sensitivity C-reactive protein, interleukin (IL) 6, IL-1β, and tumor necrosis factor α were significantly (P < .05) reduced in both the experimental groups, but again the reduction in the synbiotic group was more pronounced. Also, an increase (P < .05) in the fecal counts of total lactobacilli and a decrease (P < .05) in coliforms and Escherichia coli was observed in both the experimental groups after 6 weeks of ingestion. Overall, the combination of L salivarius with FOS was observed to be more beneficial than L salivarius alone, thereby advocating that such synbiotic combinations could be therapeutically exploited for improved health and quality of life.
Introduction
Probiotics are live microorganisms that confer health benefits to the host when consumed in sufficient quantities. 1 Selected strains of probiotic bacteria, especially lactobacilli and bifidobacteria, have been found to confer numerous health benefits including antipathogenic, antidiarrheal, bowel improving, hypocholesterolemic, immune potentiating, antiallergic, anti-inflammatory, diabetes delaying, and cancer prevention, and so on.1–9 In addition, prebiotic substrates such as inulin or fructo-oligosaccarides (FOS) that stimulates the growth and metabolism of probiotics in the gut have also been found to exhibit beneficial role against several gut-related complications. 10 Furthermore, blends of pre- and probiotics, collectively known as “synbiotics,” also confer collaborative beneficial effects against several ailments such as allergy, inflammatory bowel disease, irritable bowel syndrome, and acute diarrhea.11–13 The human body is habitat to innumerous and extremely diverse microbial communities, with the densest population residing in the distal gut (the gut microbiota). Although bacteria have long been known to activate inflammatory pathways, recent studies have evidenced that the gut microbiota can affect the lipid metabolism and function and also influences the development of obesity and related metabolic diseases.14,15 Studies have revealed a close relationship between metabolic and inflammatory pathways, with inflammation playing an important role in obesity and metabolic diseases such as insulin resistance (IR), type 2 diabetes (T2DM), nonalcoholic fatty liver diseases, atherosclerosis, metabolic syndrome (MetS), and so on. 16 Obesity and IR are frequently associated with increased inflammation in adipose tissues, indicating that this low-grade “metabolic” inflammation constitutes a causative link between obesity and IR. 17 The gut lumen also serves as a reservoir of lipopolysaccharides (LPS), the major component of the outer membrane of gram-negative bacteria. 18 Detection of low levels of LPS in the blood of healthy humans has suggested that LPS may be absorbed continuously from the gut at a low rate.19,20 Circulating LPS negatively affects insulin sensitivity as evident by its higher levels in the serum of patients with T2DM 20 ; and this LPS-induced, low-grade inflammation has been implicated in the onset or progression of diabetes and cardiovascular diseases (CVD). 21 A positive association also exists between serum LPS and markers of MetS (eg, triglycerides [TGs], IR, chronic inflammation, etc) in type 1 diabetic patients. 22 Plasma levels of LPS-binding protein are also observed to be higher in high-fat diet fed and obese-prone mice. 23 Recently, the noteworthy involvement of intestinal microbes in obesogenesis has been reported24,25,14 and an altered intestinal environment (ie, microbial composition, intestinal permeability, and LPS-detoxifying ability) has been found to contribute to adiposity and IR.24,26,27 In this milieu, various nutraceutical approaches are being explored to combat the rising global epidemic of obesity and diabetes.28–30 Among these approaches, probiotics (eg, lactobacilli, bifidobacteria) and prebiotics (eg, inulin, FOS) have received a special attention and interest in context to their safe and effective therapeutic potential. 31
Lactobacillus salivarius is an indigenous member of human gut and oral microbiota, and its probiotic utility has recently been emphasized.32–38 In addition, FOS (a commonly used prebiotic) has also been shown to exhibit beneficial effects on gut health, diabetes, MetS, adiposity, and immunity.39,40 In this context, the present investigation was carried out with the following 3 main objectives: (a) to observe the functional effects of supplementation of L salivarius UBL S22 with or without prebiotic FOS on overall metabolic health parameters, (b) to explore the effects of probiotic/synbiotic supplementation on proinflammatory cytokine markers, lipid profiles, and insulin sensitivity, and (c) to assess the influence of probiotic/synbiotic intake on specific gut microbial groups in healthy young individuals.
Materials and Methods
Study Design
The study protocol was approved by the Institutional Ethics Committee of National Institute of Nutrition, Indian Council of Medical Research (ICMR), Hyderabad, India. The trial was registered under Clinical Trials Registry India (REF/2012/08/003972), ICMR, India.
Before entering the study, the paarticipants gave their written informed consent. A total of 45 human volunteers (age: 20-25 years) with normal body mass index (BMI; 18.5-24.9 kg/m2) were assigned for the study. participants were apparently free from hypertension, diabetes, or any metabolic, gastrointestinal, and degenerative diseases and had no habit of smoking, tobacco, or alcohol consumption. The study was a randomized, placebo-controlled trial. Participants were randomly divided into 3 groups (15 per group), that is, placebo, probiotic, and synbiotic. Each group had a male–female ratio of 6:7. Participants were asked not to consume any other probiotic-containing products (from a supplied list) during the study period. Participants were given an identification number and were assigned a treatment code by a scientist blind to the treatment codes. All the 3 groups received 2 capsules per person per day. The probiotic and placebo groups were blinded but it was not possible to blind participants or field staff to the FOS supplementations, since the capsules looked different. Nevertheless, all the investigators, including medical doctors (who collected clinical data and anthropometric measurements), laboratory technicians (who carried out the biochemical and microbial analyses), and the statisticians were blind to the treatment. After completion of the biochemical and statistical analysis, the groups were decoded. Hence, there was minimum possibility of any bias that could affect the study results or interpretation.
Lactobacillus salivarius UBL S22 capsules were generously provided by Unique Biotech Pvt Ltd and Research Centre, Hyderabad, India (http://www.uniquebiotech.com/index.html). A high-density culture system was used to produce freeze-dried powder containing L salivarius at a concentration of over 1011 colony-forming units (CFUs) g−1. Each capsule (500 mg) contained 2 × 109 CFUs of L salivarius. Placebo capsules contained only plain gelatin. All capsules were preserved at 4°C. Korea-made FOS in powdered form was supplied in airtight sachets by Xena Bio Herbals Pvt Ltd (Hyderabad, India). Participants in the placebo group (group 1) received gelatin capsules, the probiotic group (group 2) received 1 capsule containing 2 × 109 CFUs of L salivarius, and the synbiotic group (group 3) received 1 capsule of L salivarius and FOS (10 g/d) per day for 6 weeks.
Biochemical Analyses
Biochemical analyses were carried out as mentioned previously. 9 Briefly, blood (5 mL) was drawn by vein puncture after an overnight fast at the baseline and after 6 weeks of intervention. The samples were drawn into standard EDTA tubes and centrifuged. Serum was separated and stored at −20°C for biochemical analyses. The samples were analyzed for total cholesterol, TGs, and high-density lipoprotein (HDL) cholesterol using enzymatic kits from LabCare Diagnostics Pvt Ltd (India). The Friedewald equation 41 was applied to calculate the following plasma lipid fractions: (1) low-density lipoprotein (LDL) cholesterol = total cholesterol − HDL cholesterol − (TG/5), with all concentrations as mg/L; (2) very low-density lipoprotein cholesterol = (TG/5); and (3) atherogenic index = (total cholesterol − HDL cholesterol)/HDL cholesterol. Serum glucose was measured using a glucose measuring kit from Biosystems (Barcelona, Spain). Homeostasis model assessment (HOMA) index was used to evaluate IR before and after the supplementation period. 42 According to international data, a value of HOMA_IR up to 2.0 is considered normal insulin sensitivity, while a value >2.0 is considered as IR. However, from the experience in Indian patients/diabetic patients, the cutoff value for HOMA_IR is up to 3.6 for normal sensitivity, 3.7 to 4.2 for IR, and >4.2 for impaired glucose tolerance. 43 Serum high-sensitivity C-reactive protein (hs-CRP) was estimated by dbc-hs Krishgen CRP kit (Biosystems, India). Serum cytokines (tumor necrosis factor α [TNF-α], interleukin [IL] 6, and IL-1β) were also analyzed by Krishgen kit (BioSystems, India). All assays were carried out according to the manufacturer’s instructions.
Stool Sampling and Colony Counting
At baseline and after 6 weeks of supplementation, participants collected early morning stool samples in sterile plastic containers which were kept in a thermal box containing ice bags and anaero packs. The samples were brought to the laboratory at earliest possible time (same day). The collected fecal samples were subsequently processed and subjected to microbial analyses, preferably on the same day or within 2 days after collection. Approximately 1 g of each sample was homogenized with sterile 9 mL of phosphate-buffered saline (PBS) in duplicate tubes. The homogenate was serially diluted using PBS, and the serial dilutions were finally inoculated onto the respective agar plates using selective media for enumeration of total lactobacilli (Lactobacillus MRS Agar, HiMedia, India), Escherichia coli (Eosin Methylene Blue Agar, HiMedia, India), and coliforms (Violet Red Bile Agar, HiMedia, India). 9 The plates were incubated (in triplicate) at 37°C aerobically or anaerobically as appropriate, and the colonies were counted after 48 hours.
Statistical Analysis
Assuming that the ingestion of L salivarius and FOS would reduce concentration of hs-CRP, a sample size of 15 in each arm was calculated to detect a 20% reduction in hs-CRP with treatment, with a power of 80% and 5% significance using Power and Sample Size Calculation software, version 3.0.14. Variation between groups at baseline was evaluated by 1-way analysis of variance (ANOVA). Changes from baseline to end point were evaluated by applying 1-way ANOVA. When there was significant change over time, analysis of covariance was used to analyze the difference between groups after adjusting for significant differences at the baseline. Statistical software SAS 9.1 (SAS Institute, Inc Cary, North Carolina) was used throughout, and P < .05 was considered to indicate statistical significance.
Results
Baseline Values
The mean BMI and mean age of the participants in the 3 groups was comparable at the baseline. The fasting blood glucose, total cholesterol, and TG levels were within the normal range in all the volunteers; however, HDL cholesterol was less than 40 mg/dL in 22 participants. Similarly, IIR (HOMA > 3.6) was observed in 28 participants. The level of hs-CRP was also high in 25 volunteers, with hs-CRP ranging from 1 to 3 pg/mL in 15 and above 3 pg/mL in 10 volunteers.
Effect of Supplementation on BMI, Serum Glucose, and Insulin Resistance
After 45 days of supplementation, the BMI did not change in the placebo and the probiotic groups; however, it was found to be decreased significantly (P < .05) in the synbiotic (probiotic plus FOS) group, when compared to the baseline as well as the end point values of control and probiotic group. Fasting serum glucose levels were comparable between the 3 groups after 6 weeks of supplementation, although there was slight numerical decrease observed in serum glucose in all the 3 groups (Table 1). After 6 weeks, the probiotic group had significantly (P ≤ .05) lower insulin when compared to the placebo group. Similarly, the synbiotic group showed a significantly (P ≤ .05) decreased insulin concentration, when compared to the placebo as well as the probiotic group (Table 1). However, the decrease observed in the probiotic and the synbiotic groups cannot be substantiated, since a decrease in serum insulin was also observed in the placebo group after 6 weeks of supplementation. Nevertheless, the decrease observed in the probiotic and the synbiotic groups was clearly more pronounced than that in the placebo group. On the other hand, the mean IR was found to be reduced only in the probiotic and the synbiotic groups. Although, the IR index was reduced in both the experimental groups, the reduction observed in the synbiotic group was more significant (P < .05) than that in the probiotic group (Table 1).
Effect of Ingestion of Lactobacillus salivarius UBL S22 Alone (Probiotic) or in Combination With Prebiotic Fructo-Oligosaccharide (synbiotic) on BMI, Serum Glucose, Fasting Insulin, and Insulin Sensitivity.

Abbreviations: BMI, body mass index; HOMA-IR, homeostasis model assessment-estimated insulin resistance; SE, standard error.
a Significant (P < .05) difference between baseline and end point values within groups (within columns).
b,c Significant (P < .05) difference between placebo and experimental groups (within rows). Data expressed as mean ± SE (n = 15).
Effect of Supplementation on Serum Lipid Profiles
At baseline, the serum total cholesterol and TGs were within normal range for all the volunteers; however, HDL levels were lower than 40 mg/dL among few participants from both the experimental groups (Figure 1). After 6 weeks of supplementation, a significant (P < .05) decrease in total cholesterol, LDL, and TGs accompanied with an increase in HDL was observed in both the experimental groups when compared to their respective baseline values, while there was no such change observed in the control group (Figure 1). Also, the total cholesterol and TGs were observed to be reduced significantly (P < .05) in the probiotic group, when compared to the end point values from the placebo group. Interestingly, the total cholesterol and LDL in the synbiotic group were found to be more reduced (P < .05), when compared to the end point values of the probiotic group. Moreover, the decreases observed in TGs and increase in HDL, although not significant, were more pronounced in the synbiotic group, when compared to those in the probiotic group (Figure 1).
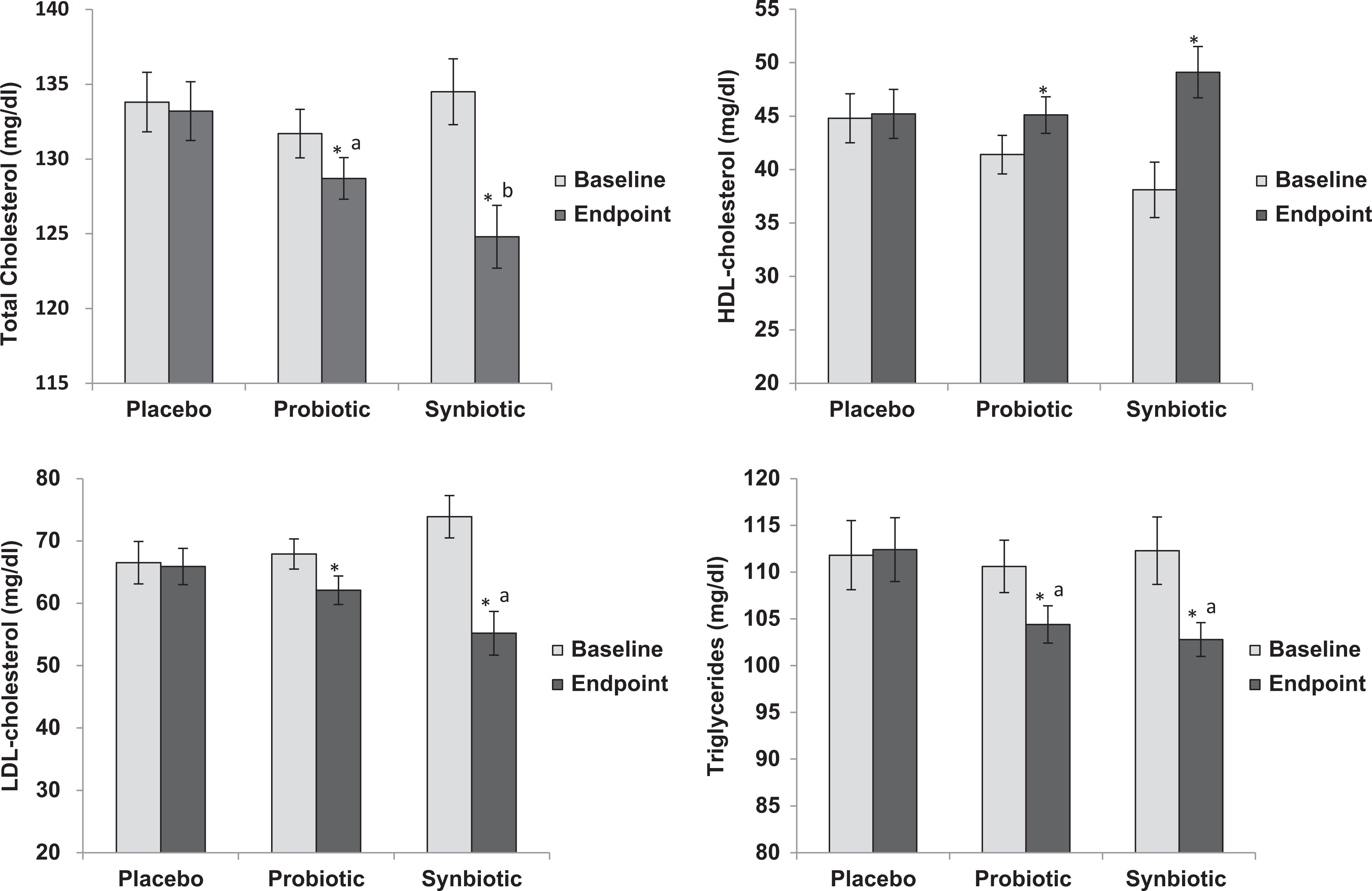
Effect of ingestion of Lactobacillus salivarius UBL S22 alone (probiotic) or in combination with prebiotic fructo-oligosaccharide (synbiotic) on serum lipid profiles. *Significant (P < .05) difference between baseline and end point values within the same groups. a,bSignificant (P < .05) difference between the groups. Data expressed as mean ± standard error (SE; n = 15).
Effect of Supplementation on Inflammatory Markers
At baseline, the serum hs-CRP was high (>1 pg/mL) in 8 probiotic (53.3%), 10 synbiotic (66.7%), and 7 placebo (46.7%) group participants (Figure 2). However, after 6 weeks of supplementation, this percentage reduced to 41.8% and 62.2% in the probiotic and the synbiotic groups, respectively, while no such reduction was observed in the placebo group. Overall, the serum concentrations of CRP, IL-1β, and TNF-α were found to be significantly (P < .05) reduced in the probiotic group as well as in the synbiotic group when compared to their respective baseline values or to the end point values from the control group. Furthermore, the concentrations of CRP, IL-6, and TNF-α were significantly (P < .05) more reduced in the synbiotic group when compared to those in the probiotic group (Figure 2).

Effect of ingestion of Lactobacillus salivarius UBL S22 alone (probiotic) or in combination with prebiotic fructo-oligosaccharide (synbiotic) on serum inflammatory interleukin markers. *Significant (P < .05) difference between baseline and end point values within the same groups. a,bSignificant (P < .05) difference between the groups. Data expressed as mean ± standard error (SE; n = 15).
Effect of Supplementation on Gut Microbial Populations
After 6 weeks of supplementation, the total Lactobacillus count was 6.61 ± 0.12, 7.23 ± 0.11, and 7.96 ± 0.10 log10 CFU/g in the placebo, probiotic, and synbiotic groups, respectively (Figure 3). Although the count of E coli was quite high (6.73 ± 0.17 log10 CFU/g) in the placebo group even after 6-week supplementation, it was found to be much lower, that is, 6.37 ± 0.12 and 6.19 ± 0.05 log10 CFU/g in the probiotic and the synbiotic groups, respectively (Figure 3). Concomitantly, the fecal coliform count was also highest in the placebo group (6.61 ± 0.14 log10 CFU/g), lower in the probiotic group (6.35 ± 0.21 log10 CFU/g), and lowest in the synbiotic group (5.88 ± 0.16 log10 CFU/g). Overall, the total Lactobacillus count was significantly (P < .05) increased in the probiotic group as well as in the synbiotic group when compared to the respective baselines and to the placebo group end point; with a more pronounced increase in the synbiotic group than the probiotic group. Although there was no significant decrease in E coli count in the probiotic group when compared to the baseline, there was a significant (P < .05) decrease when compared to the control group. In the synbiotic group, the E coli count reduced significantly when compared to that at the respective baseline and control group end points but not from the probiotic group end point. Intriguingly, the coliform count was not reduced in the probiotic group; however, it was found to be significantly (P < .05) reduced in the synbiotic group, when compared to the respective baseline and to the end point values from the placebo and the probiotic groups (Figure 3).
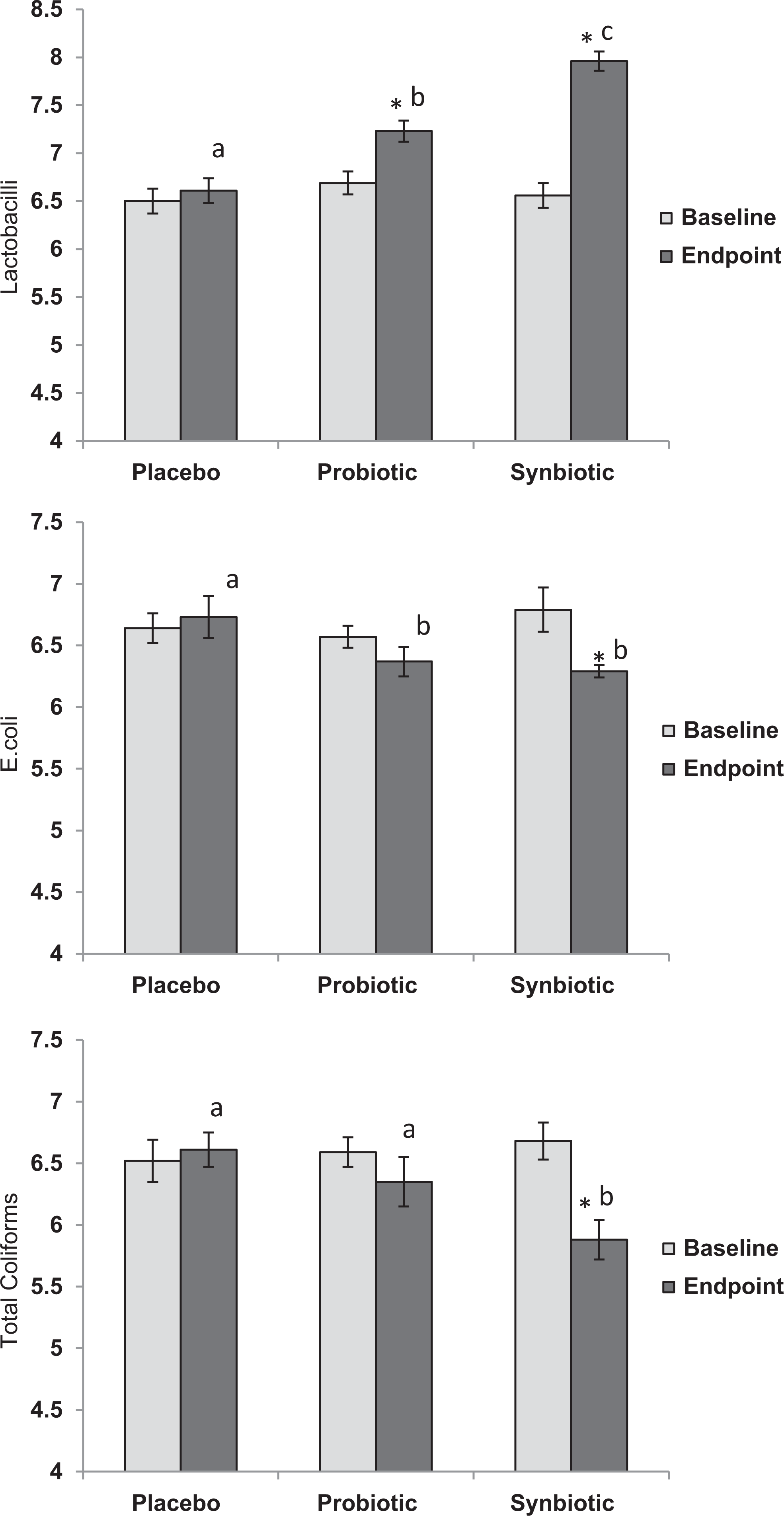
Effect of ingestion of Lactobacillus salivarius UBL S22 alone (probiotic) or in combination with prebiotic fructo-oligosaccharide (synbiotic) on fecal bacterial counts (data expressed as log10 colony-forming unit (CFU) per gram of fecal dry weight). *Significant (P < .01) difference between baseline and end point values within groups. a,b,cSignificant (P < .05) difference between the groups (within rows). Data expressed as mean ± standard error (SE; n = 15).
Discussion
The probiotic strain L salivarius UBL S22 used in the present trial has earlier been shown to exhibit immune-modulating effects in vitro32–34; however, to our knowledge, this appears to be the first study to examine the clinical efficacy of a combination of this strain with prebiotic FOS on lipid profiles, proinflammatory cytokines, and insulin sensitivity in healthy young individuals. The major finding observed is that, in comparison to being used alone, the “symbiotic” blend of putative L salivarius preparation with prebiotic FOS exhibited superior influence on serum lipid profiles, proinflammatory markers, and insulin sensitivity. Furthermore, as anticipated, the synbiotic combination also modulated the gut microflora by increasing the number of beneficial lactic acid bacteria and decreasing the harmful gram-negative bacteria.
Consumption of probiotic-fermented milks has earlier been shown to improve the total–HDL cholesterol and LDL–HDL cholesterol ratio in healthy volunteers. 44 Klein et al 45 also observed a 35%, 32%, and 11.6% decrease in LDL, total cholesterol, and serum triglyceride levels, respectively, after administration of probiotics. Kumar et al 5 also reported the hypocholesterolemic effect of probiotic Lactobacillus plantarum in hypercholesterolemic animal models. Recently, Mohania et al 46 observed an improvement in plasma, hepatic, and aortic lipid profiles, and attenuation of diet-induced hypercholesterolemia in hypercholesterolemic rats following the supplementation of probiotic-fermented milk containing L plantarum. In a randomized, placebo-controlled, double-blind synbiotic study on normocholesterolemic men by Schaafsma et al, 47 a combination of probiotic L acidophilus plus FOS supplementation significantly reduced the LDL, TC, and LDL–HDL ratio. In a similar study on men and women with hypercholesterolemia, a significant reduction in LDL and TC was observed after 12-week supplementation of a combination of L acidophilus plus prebiotic inulin. 48 Interestingly, the results of the synbiotic combination used in this study concur well with these investigations, thereby further advocating the positive influence of probiotics and synbiotics on lipid profiles. Very recently, DiRienzo 49 meta-analyzed 26 clinical studies and concluded that probiotics or synbiotics can be exploited as viable recommendation for therapeutic dietary options for better lipid profiles and cardiovascular health. Furthermore, in this study, although there was a decrease in serum total cholesterol, LDL-cholesterol, and TGs in both the treatment groups, the decrease observed in the synbiotic group was more pronounced. These results further support the previous observations that addition of prebiotics may augment the ameliorating effect of probiotics on serum lipid profiles.
Furthermore, the synbiotic group also exhibited more significant decrease (P ≤ .05) in the serum levels of CRP and proinflammatory cytokines after 6-week supplementation when compared to the probiotic group. Nevertheless, the probiotic group also showed a relative decrease in the serum concentration of CRP and interleukins. Several studies have confirmed that the interaction of probiotics with inflamed intestinal tissues downregulates the production of IL-6 and increases the number of apoptotic T cells in the lamina propria. 50 Several probiotic strains that pass through the gastrointestinal tract can also induce anti-inflammatory cytokine production.51,52 In this context, combining probiotics with prebiotics (that selectively promote the growth and activities of certain beneficial bacterial species and improve nutrient absorption, transit, and fermentation) could be a more effective strategy. 53 Guigoz et al 54 observed decreased phagocytic activity of granulocytes and monocytes and decreased expression of IL-6 messenger RNA in the peripheral blood monocytes in healthy elderly people after 3 weeks of FOS consumption. Roller et al 55 also found an increased local immunity in the ileum of rats after feeding a combination of oligofructose-enriched inulin, L rhamnosus GG, and bifidobacteria. Vulevic et al 56 reported increased phagocytosis and activities of natural killer cells and IL-10 and reduced production of IL-6, IL-1β, and TNF-α in healthy elderly volunteers after 10 weeks of galacto-oligosaccharides intake. Several mechanisms can explain the reduction in proinflammatory cytokines and improved insulin sensitivity after the ingestion of probiotic/ synbiotic products. Several gram-negative gut pathogens stimulate LPS production and its leakage from intestinal epithelial cells. This LPS then binds to the cytokine receptors on hepatocytes and/or adipocytes and triggers a proinflammatory cytokine release and insulin resistance. Probiotics, prebiotics, or both together could improve the intestinal barrier function, encourage the growth of gut beneficial or commensal microbes, and reduce the growth of several gram-negative pathogens, thereby reducing the LPS leakage and eventually leading to the decreased proinflammatory cytokine production in adipose tissues. However, we acknowledge the need for future studies in order to evaluate whether the probiotic/synbiotic treatment used in this study led to a reduced proinflammatory state through gut microbiota modulation, immune modulation, alteration in the intestinal barrier function, weight loss, or through some other mechanisms. In addition, although the decreased IR in probiotic and synbiotic groups may indicate an optimizing effect of probiotic and synbiotic supplementation on insulin sensitivity, however, the mechanisms underlying this phenomenon remain unclear. Nevertheless, the positive effect on glucose homeostasis can be supported by a number of animal studies which have demonstrated the antidiabetic potential of specific probiotic strains.57,58 Because IR is associated with low-grade inflammation, the beneficial effects of probiotic/synbiotic treatment on glucose homeostasis could potentially involve anti-inflammatory mechanisms. It may be noted that, in this study, L salivarius alone also exhibited some positive impact on IR and inflammation, which is in line with the previous studies. Insulin resistance, together with resultant compensatory hyperinsulinaemia, can lead to a cluster of metabolic abnormalities such as glucose intolerance, hyperlipidemia, hepatic steatosis, and hypertension and, hence, is considered as the pathophysiological basis of MetS, a major risk factor for obesity, T2DM, CVD, and several other related ailments. In context to the beneficial effects of prebiotics and probiotics on endotoxemia, inflammation, and IR, the 2 hormones glucagon-like peptide (GLP) 1 (GLP-1) and GLP-2 have also been found to play an important role.58,59,18 Significantly increased levels of circulating GLP-1 and GLP-2 have been observed in mice on a prebiotic diet. 60 Intake of prebiotics may also stimulate the enteroendocrine L-cell differentiation and increases GLP-1 response.59,18 An increased production of GLP-2 following a prebiotic diet is associated with increased number of beneficial gut bacteria, improved integrity of intestinal barrier, and lowered metabolic inflammation and endotoxemia. 60 Maybe, a GLP-mediated mechanism is also involved in the ameliorating effect of L salivarius plus FOS on IR and glucose tolerance observed in this study. Prebiotics are fermented by gut bacteria to produce SCFAs including butyrate, and butyrate supplies energy to the colonic cells thereby improving the gut health. Although, butyrate is not produced by probiotic lactobacilli and bifidobacteria, acetate and lactate produced by these probiotics can be converted into butyrate by other gut bacteria, again justifying a superior effect of synbiotics (as reported in the present study) on colonic health than probiotics or prebiotics alone.
The ability of probiotic lactobacilli and FOS to positively modulate the gut microbiota has been observed in several studies.9,27,61 Positive modulation of gut microecology is acknowledged as one of the key mechanisms underlying the beneficial effects of probiotics and prebiotics on gut functions, immune modulation, and metabolism. Supplementation with probiotics and prebiotics can promote the growth and activities of the host’s endogenous beneficial or commensal bacteria, thereby aiding in the correction of dysbiosis. 62 In this context, the end point counts of lactobacilli, E coli, and coliforms observed in the present study speculate that the intake of L salivarius or its supplementation with FOS could promote the population of Lactobacillus spp in the gut microbiota of healthy individuals (Figure 3). A number of trials testing the effectiveness of probiotics, prebiotics, or synbiotics in atopic eczema, irritable bowel syndrome, or antibiotic-associated diarrhea have also revealed that probiotics could be useful if provided under specific conditions, since the beneficial effect is strain dependent and could differ from organism to organism.63–65 Nevertheless, the positive modulation of gut microflora, that is, the increased number of beneficial/commensal groups and reduced counts of harmful bacteria, is considered as one of the key mechanisms involved in these beneficial effects. 66 There is a close relationship between gut microflora and intestinal inflammation, since the primary sites that are affected by inflammation in most of the metabolically diseased states are those which are generally colonized by the highest number of bacteria, such as the distal ileum and the colon. There are indications that some probiotic strains, particularly Lactobacillus and bifidobacteria, can reduce the incidences of diarrhea and other intestinal disorders by competitively inhibiting the pathogens. 67 Recently, several studies have proposed the gut microbiota as an environmental factor involved in the development of MetS,14,68 thereby indicating that probiotics and prebiotics may ameliorate metabolic abnormalities by modulating the gut microbiota.
Our study has several limitations such as (a) a short intervention period, (b) lack of “FOS-only” group, and (c) inability to study enough molecular parameters to reveal molecular mechanisms involved. However, the study was significantly and adequately powered to investigate the efficacy of probiotic with prebiotics on IR and inflammation. Our aim in this preliminary investigation was to determine whether a synbiotic combination of L salivarius with FOS is more efficacious than L salivarius alone for the management/improvement in normal gut health, insulin sensitivity, and lipid profiles in apparently healthy young individuals. In conclusion, we have demonstrated that the supplementation of L salivarius UBL S22 along with prebiotic FOS could ameliorate the insulin sensitivity, serum lipid profiles, proinflammatory cytokines, and gut microbial populations. The study advocates that this probiotic strain or its synbiotic combination may also be used therapeutically to prevent lifestyle-associated, inflammation-associated, or gut microbiome-associated metabolic disorders through amelioration of IR, serum lipid profiles, inflammatory status, and gut microbial populations. Nevertheless, further molecular as well as clinical investigations are warranted to elucidate the in-depth mechanisms of action and the efficacy of this probiotic/synbiotic supplementation in humans.
Footnotes
Author Contributions
HR and MK conceived and designed the experiment. MK, ND, and HRC performed the biochemical and microbiological analyses. HR, MK, and RN analyzed the data. MK and RN wrote the article. RN and HR edited and improved the article. HR, MK, ND, NKS, HRC, and RN read and approved the final draft.
Authors’ Note
The work was supported by and executed at NIN. This work was carried out at the Department of Microbiology and Immunology, National Institute of Nutrition, Hyderabad (Andhra Pradesh), India.
Acknowledgments
The authors wish to thank the Director, National Institute of Nutrition, India, for providing facilities and support system to carry out this work. The authors also thank all the volunteers for participating in the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RN is post doc fellow at Juntendo; however, this work does not represent any scientific or monetary views of Juntendo.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
