Abstract
Background and aim:
Beta-blockers have unfavorable effects on metabolic parameters in hypertensive treatment. New generation beta-blockers with vasodilatory capabilities are superior to traditional beta-blockers, but studies examining their effects on metabolic parameters are still lacking. This study aimed to compare the effects of 2 new generation beta-blockers, carvedilol and nebivolol, on insulin resistance (IR) and lipid profiles in patients with essential hypertension.
Methods:
This was a prospective, randomized, open-label, single-center clinical trial. A total of 80 patients were randomized into 2 groups: the carvedilol group (n = 40, 25 mg of carvedilol daily) and the nebivolol group (n = 40, 5 mg of nebivolol daily). Follow-up was performed for 4 months. Fasting plasma glucose, insulin levels, and the lipid profile (high-density lipoprotein [HDL], low-density lipoprotein [LDL], total cholesterol, triglyceride, apolipoprotein AI, and apolipoprotein B levels) were measured and IR was calculated by the homeostasis model assessment (HOMA) index. These variables were compared before and 4 months after treatment.
Results:
Blood pressure and heart rate were significantly and similarly reduced in the carvedilol and nebivolol groups after treatment compared to those before treatment (both
Conclusion:
New generation beta-blockers, carvedilol and nebivolol, efficiently and similarly decrease blood pressure. They have similar favorable effects on glucose, insulin, IR, and the lipid profile.
Introduction
Beta-blocker drugs have been used to treat hypertension for 4 decades. Recently, use of beta-blockers in antihypertensive treatment has been debated because of the lack of hemodynamic and metabolic benefits compared to placebo and other antihypertensive agents. 1
However, beta-blockers are a group of drugs that show heterogeneity with physiological and pharmacological properties. Effects of new generation vasodilating beta-blockers on hemodynamic and metabolic parameters differ from traditional beta-blockers. Traditional beta-blockers decrease cardiac output and increase peripheral resistance. Therefore, they may cause an increase in insulin resistance (IR), susceptibility to diabetes, and impairment of the lipid profile by reducing the transport of glucose into peripheral tissues. In contrast, vasodilating beta-blockers remove negative effects on glucose and lipid metabolism by decreasing peripheral vascular resistance, without adverse effects on cardiac output. 2,3 Along with this peripheral vasodilatation, new generation beta-blockers, such as carvedilol and nebivolol, have antioxidant effects and nitric oxide–induced benefits, such as reversal of endothelial dysfunction. 4 Despite these differences of traditional and vasodilating beta-blockers, almost all of the studies that led beta-blockers to the background were on traditional agents (eg, propranolol, atenolol, and metoprolol).
New generation beta-blockers have been shown to be superior to the traditional beta-blockers regarding hemodynamic and metabolic parameters. 1 -4 However, direct comparison of the effect of 2 new generation beta-blockers, carvedilol and nebivolol, on metabolic parameters is not well known. In this study, we aimed to compare the effects of 2 new generation beta-blockers, carvedilol and nebivolol, on IR and lipid profiles in patients with essential hypertension.
Materials and Methods
Study Population
A total of 107 newly diagnosed patients with hypertension, who were admitted to the Department of Cardiology at Baskent University Ankara Hospital, were enrolled in this study, between August 2013 and April 2014. Systolic blood pressure and diastolic blood pressure were measured in both arms by the auscultatory method, using a standard mercury sphygmomanometer, after at least 10 minutes of rest in the sitting position. The mean of 2 recordings of blood pressure was taken by the same physician, and 140 mm Hg systolic and/or 90 mm Hg diastolic pressure or higher was considered as hypertension. 5
Patients with secondary hypertension, diabetes mellitus, heart failure, sick sinus syndrome, sinus bradycardia, second- or third-degree heart block, coronary artery disease, severe valvular disease, hypothyroidism or hyperthyroidism, pregnancy or lactation, cerebrovascular disease, liver or kidney failure, or antihypertensive or antihyperlipidemic drug use were excluded from this study.
Study Protocol
The present study was a randomized, open-label, single-center trial. Among the 107 patients who were initially included, 27 patients were excluded after the screening phase. These included 15 patients who declined to participate, 8 whose antihypertensive treatment was changed by another physician, 1 who discontinued taking antihypertensive drugs, and 3 patients were lost to follow-up. Eighty patients were randomly assigned to a 25-mg daily dose of carvedilol (n = 40) or 5-mg daily dose of nebivolol (n = 40). The dose was titrated at 1-week intervals. At the third week of treatment, if blood pressure was not controlled (<140/90 mm Hg), amlodipine (5 mg), which has neutral metabolic effects, was added to the therapy. After providing effective blood pressure control, we planned 4 months of follow-up period to assess the effects of the drugs on IR and the lipid profile.
Lifestyle changes were proposed to all the patients in line with the European Society of Cardiology 2013 guidelines for the management of arterial hypertension. 5
Biochemical Measurements
Fasting blood samples were withdrawn from patients after 12 hours of fasting for measurements of biochemical parameters. Insulin was measured by the chemiluminescence method. Fasting plasma glucose (FPG), high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, and triglyceride levels were measured with enzymatic methods. Apolipoprotein AI (apoAI) and apolipoprotein B (apoB) were measured with immunoturbidimetric methods.
Insulin resistance was determined by homeostasis model assessment of IR (HOMA-IR). HOMA-IR was calculated using the following formula: (fasting insulin [μU/mL] × FPG [mg/dL]/405). An HOMA-IR ≥ 2.7 was accepted as IR. 6 Total cholesterol (TC) was calculated by the Friedewald formula: TC = LDL + HDL + triglycerides/5
Waist circumference was measured. Body mass index (BMI) was calculated using the Quetelet index (weight/height2, kg/m2).
Four months after therapy initiation, the patients’ blood pressure, heart rate, BMI, waist circumference, and biochemical parameters (FPG, insulin, HDL, LDL, triglyceride, apoAI, and apoB levels) were measured. We compared changes in blood pressure, IR, and the lipid profile between the 2 drugs, before and 4 months after treatment.
All of the participants provided written informed consent. This study was approved by the Baskent University institutional review board and ethics committee and Turkey Pharmaceuticals and Medical Devices Agency.
Statistical Analysis
The sample size was determined by power analysis done by “repeated measures analysis of variance for factorial design” (80% power). At the end of the study, we performed statistical analysis of patients who received 4 months of treatment. When the assumptions of parametric tests were fulfilled, the Student
Results
Baseline demographic, clinical, and laboratory characteristics of the carvedilol and nebivolol treatment groups are shown in Table 1. Blood pressure and heart rate reduced significantly and similarly (no significant difference between the groups) in the carvedilol and nebivolol groups after 4 months of treatment compared to before treatment (both
Baseline Demographic, Clinical, and Laboratory Characteristics of Carvedilol and Nebivolol Treatment Groups.a

Abbreviations: apoA-I, apolipoprotein AI; apoB, apolipoprotein B; BMI, body mass index; CCB, calcium channel blocker; DBP, diastolic blood pressure; FPG, fasting plasma glucose; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment of insulin resistance; LDL, low-density lipoprotein SBP, systolic blood pressure; SD, standard deviation.
aValues are mean ± SD or n (%).
bStudent
cChi-square test.
dMann-Whitney
Comparison of Hemodynamic and Metabolic Parameters Between Carvedilol and Nebivolol Treatment Before and After Treatment.
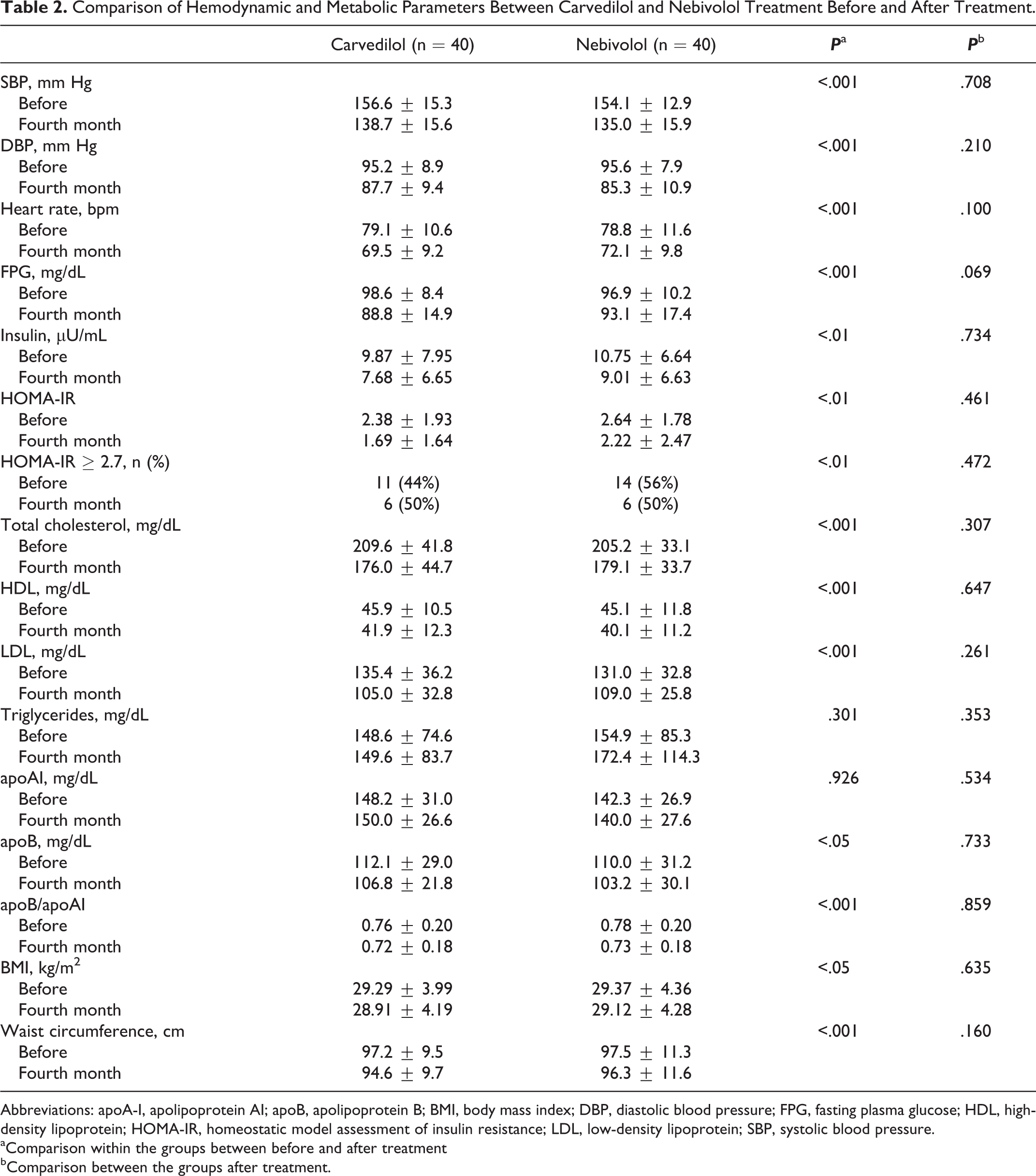
Abbreviations: apoA-I, apolipoprotein AI; apoB, apolipoprotein B; BMI, body mass index; DBP, diastolic blood pressure; FPG, fasting plasma glucose; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment of insulin resistance; LDL, low-density lipoprotein; SBP, systolic blood pressure.
aComparison within the groups between before and after treatment
bComparison between the groups after treatment.
Although 25 patients had IR (11 patients in the carvedilol group and 14 patients in the nebivolol group) before treatment, only 12 patients had IR after 4 months of therapy (6 patients in the carvedilol group and 6 patients in the nebivolol group). This decline in IR was significant (
The ApoB/ApoAI ratio, a predictor of coronary artery disease, was compared between the groups before and after treatment. After treatment, the apoB/apoAI ratio decreased significantly in a similar manner (no significant difference between the groups) in both the groups compared to before treatment (
We observed that in the carvedilol and nebivolol groups, BMI (
Discussion
The current study demonstrated that new generation beta-blockers, carvedilol and nebivolol, efficiently decreased blood pressure, and they had similar favorable effects on glucose levels, insulin levels, IR, and the lipid profile.
Beta-blockers are the most diverse of the antihypertensives. Conventional beta-blockers have negative effects on the lipid profile, leading to a reduction in cardiac output and increase in peripheral resistance. However, vasodilating beta-blockers reduce peripheral resistance without affecting cardiac output. Additionally, they do not have unfavorable effects on IR, glycemic control, or the lipid profile. 7 The current study is important because it directly compared the effect of 2 new generation beta-blockers, carvedilol and nebivolol, on the lipid profile and IR.
The new generation vasodilating beta-blockers carvedilol and nebivolol lower blood pressure as effectively as other antihypertensive drugs. Although effective blood pressure decreases, adverse hemodynamic and metabolic effects are less with new generation beta-blockers than with traditional beta-blockers. 8 After 4 months of therapy, we found that carvedilol and nebivolol treatment significantly decreased systolic blood pressure and diastolic blood pressure and heart rate compared to baseline, and this decline was similar for both the drugs. These findings are similar to those of a study by Erdogan et al. 2 They determined that carvedilol and nebivolol treatment led to a significant decrease in blood pressure and heart rate compared with placebo, but none of the drugs was superior.
Several mechanisms have been suggested for vasodilating beta-blockers and their useful effects on insulin and lipid metabolism. Alpha-1 adrenergic receptor blockade and vasodilation, anti-inflammatory effects, decreased oxidative stress, and decreased weight gain are the main proposed mechanisms. 7
In our study, we found similar decrease in FPG with carvedilol and nebivolol after 4 months of therapy. This finding can be explained by positive effects of new generation vasodilating beta-blockers on glucose metabolism. Furthermore, lifestyle changes, which were started by treating hypertension, might have contributed to this result. In the Glycemic Effects in Diabetes Mellitus: Carvedilol – Metoprolol Comparison in Hypertensives (GEMINI) study, the vasodilating beta-blocker carvedilol and traditional beta-blocker metoprolol were compared in diabetic patients with hypertension. 9 Carvedilol provided better results than metoprolol in glycemic control.
We also found that the decrease in insulin and HOMA-IR values was similar between the carvedilol and the nebivolol groups. This supports the results of a previous study by Yildiz et al who compared the effect of carvedilol and nebivolol on IR in diabetic patients. 10 They found that there was also no superiority of the drug groups.
Dyslipidemia (ie, IR) is a strong and independent predictor of hypertension for cardiovascular morbidity and mortality. 11 Previous studies have shown that traditional beta-blockers may have harmful effects (eg, increase the triglyceride levels and decrease the HDL levels) on the lipid profile. 1 The activity of lipoprotein lipase is reduced in cases of hyperinsulinemia. Therefore, by increasing the activity of lipoprotein lipase, vasodilating beta-blockers can make a positive contribution to the lipid profile. 12 Additionally, nitric oxide release and antioxidant properties of new generation beta-blockers may contribute to this positive effect. 13
In our study, both the new generation vasodilating beta-blockers (carvedilol and nebivolol) significantly and similarly decreased HDL, LDL, and TC values after treatment compared to those before treatment. Triglyceride levels did not change in both the groups. There was no significant difference in apoAI levels (main lipoprotein of HDL cholesterol) between the groups. This finding does not explain the decrease in HDL levels but at least could explain the lack of expected recovery. Levels of apoB (main lipoprotein of LDL cholesterol) were significantly reduced after treatment in both the groups. This finding could explain the decrease in LDL cholesterol levels in our study. Although we expected a decrease in triglyceride levels according to the decline in plasma glucose levels and IR, there was no significant change in triglyceride levels with treatment in both the groups. This finding is consistent with a lack of an increase in triglyceride levels with carvedilol in spite of metoprolol as observed in the GEMINI and Extended-Release Carvedilol Trial Lipid studies. 9,11
However, in another randomized clinical trial, Studinger et al compared carvedilol, nebivolol, and metoprolol on central arterial pressure. After 3 months of therapy, they found no differences in glucose and lipid parameters. 14 The differences between this study and ours might have resulted from the different treatment periods, therapeutic doses, but most likely the lifestyle changes.
The apoB/apoAI ratio is an important indicator for assessing the risk of vascular disease and treatment of atherosclerotic vascular disease with statins. Some studies have suggested that this ratio is superior to other lipoprotein and apolipoprotein parameters for assessment of cardiovascular risk. 15 In our study, there was a significant reduction in apoB and the apoB/apoAI ratio after treatment compared to those before treatment, but no differences were observed between the groups. These findings support that carvedilol and nebivolol could be used in patients with hypertension and atherosclerotic vascular disease.
In the GEMINI study, there was significant weight gain with metoprolol use, but this negative effect was not observed with carvedilol. 9 Although the reason for this disadvantage is not clear with the use of traditional beta-blockers, it has been proposed to result from insulin-evoked endothelial dysfunction. 16 In our study, weight, BMI, and waist measurement values were significantly decreased after treatment compared to that before treatment, and these decreases were similar between the groups. These findings are probably due to lifestyle changes, activation of lipolysis, an increase in insulin sensitivity, and reduction in weight, as expected from vasodilatory beta-blocker therapy. Decrease in BMI (a strong predictor of IR) shows a correlation with improvement in IR, which could be another reason for choosing vasodilating beta-blockers.
Although renin–angiotensin–aldosterone system inhibitors are recommended in patients with hypertension having metabolic syndrome, an insufficient amount of studies have compared these agents with carvedilol or nebivolol. 1 In our study, positive and similar results were obtained with 2 vasodilating beta-blockers.
Study Limitations
Because of ethical concerns, when antihypertensive treatment began in all patients, an untreated control group could not be used. The lack of blinding could pose potential weakness. Study follow-up examinations of blood pressure and heart rate of patients were performed with office and home measurements. More information on blood pressure and heart rate could be obtained with ambulatory measurements. Four months of treatment was planned to evaluate effects of drugs on IR and the lipid profile. A longer follow-up time could provide more accurate metabolic values. Patients were recommended to perform lifestyle changes along with the treatment of hypertension. Lifestyle changes and their positive effects on metabolic parameters may have contributed in part to the useful effects of the drugs. Because of difficulties in standardizing lifestyle changes in patients, the extent of this contribution is unknown. The present study focused specifically on the comparison of the effects of carvedilol and nebivolol on metabolic and lipid parameters. However, it is also recognized that carvedilol and nebiviolol are known to provide benefit in terms of renal protection, reduction in left ventricular hypertrophy, reduction in neointimal hyperplasia, and atherosclerosis regression, parameters that merit further evaluation in additional comparative trials. 1
Conclusion
New generation beta-blockers, carvedilol and nebivolol, efficiently and similarly decrease blood pressure. These beta-blockers also have similar favorable effects on glucose, insulin, IR, and the lipid profile. Carvedilol and nebivolol could be considered in the treatment of hypertension, with no superiority with each other on hemodynamic and metabolic effects. Favorable effects on controlled blood pressure and metabolic parameters need to be investigated in large, randomized, placebo-controlled studies.
Footnotes
Author Contributions
Ozyildiz, A.G. contributed to conception or design, acquisition, analysis, or interpretation; drafted the manuscript; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Eroglu, S. contributed to conception or design, acquisition, analysis, or interpretation; critically revised the manuscript; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Bal, U.A. agreed to be accountable for all aspects of work ensuring integrity and accuracy. Atar, İ. contributed to acquisition, analysis, or interpretation and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Okyay, K. agreed to be accountable for all aspects of work ensuring integrity and accuracy. Muderrisoglu, H. gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was supported by the Baskent University Research Fund.
