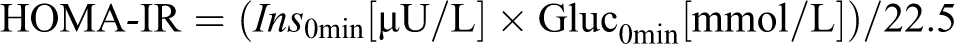Abstract
This study examined the effect of acute and sustained transdermal glyceryl trinitrate (GTN) therapy on insulin and glucose regulation. Totally, 12 males (18-30 years) underwent a glucose tolerance test at baseline (visit 1), 90 minutes after acute transdermal GTN 0.6 mg/h (visit 2), following 7 days of continuous GTN (visit 3), and 2 to 3 days after stopping GTN (visit 4). At each visit, plasma glucose and insulin concentrations were measured before and 30, 60, 90, and 120 minutes after a 75-g oral glucose load. Indices of glucose metabolism that were examined included the insulin sensitivity index, the homeostasis model assessment of insulin resistance (HOMA-IR), and the insulinogenic index. The acute administration of GTN had no effect on glucose and insulin responses (visit 2). However, after 7 days of GTN exposure (visit 3) there was an increase in the mean glucose concentration measured after the oral glucose load. On visit 1, the mean glucose concentration (± standard deviation) following the 75 g oral glucose challenge was 5.7 ± 0.5 µmol/L. On visit 3, after 7 days of transdermal GTN therapy, the mean glucose concentration after the oral glucose was significantly higher; 6.2 ± 0.5 µmol/L (P < .015; 95% confidence intervals 0.25-0.77). There was also an increase in the HOMA-IR index; on visit 1, the median HOMA-IR (interquartile range) was 5.2 (3.9) versus 6.9 (6.8) on visit 3 (P < .015). Other indices of glucose metabolism did not change. These observations document that GTN therapy modifies glucose metabolism causing evidence of increased insulin resistance during sustained therapy in normal humans.
Introduction
The pharmacodynamic effects of glyceryl trinitrate (GTN) and other organic nitrates are believed to involve the release of nitric oxide (NO) or some NO-containing moiety. 1,2
Although NO has many cardiovascular effects, it also has an important role in the control of several metabolic pathways. Importantly, little is known about the impact of organic nitrate therapy on glucose and insulin metabolism, particularly during sustained therapy. Therefore, the purpose of the study presented here was to explore the impact of acute and sustained transdermal GTN therapy on glucose metabolism and indices of insulin resistance in normal humans.
There is evidence that endogenous NO plays a role in the regulation of β-cell function (BCF), insulin release, and insulin sensitivity. 3,4 Lautt has documented that the postprandial rise in insulin activates hepatic parasympathetic nerves, leading to generation of NO, which via activation of guanylate cyclase increases insulin sensitivity. 5 –7 Furthermore, insulin sensitivity is impaired with intraportal administration of NO synthase inhibitors and restored with NO donors. 6 The NO bioavailability has also been shown to be an important regulator of peripheral glucose uptake in humans. 8 There is a close correlation between insulin sensitivity and endothelial NO bioavailability in healthy patients. 9 In both animal and human models, acute GTN therapy is associated with an increase in peripheral insulin sensitivity. 10,11 Similarly, the NO donor nitroprusside, when given by intra-arterial infusion, increases arteriovenous glucose extraction. 12 In contrast, the administration of intravenous nitroprusside and the oral nitrates, isosorbide-5-mononitrate and pentaerythritol tetranitrate, had no effect on glucose metabolism in normal volunteers. 13
Sustained therapy with GTN and other organic nitrates has been shown to be associated with increased vascular production of radicals. 14 –17 Although studies on the redox response to nitrates have focused on their apparent negative impact on vascular function, it is plausible that chronic nitrate therapy may have an impact on insulin and glucose interactions that is different from that which has been observed following acute exposure. Indeed, in a rabbit model Bajza et al reported that sustained, 7-day exposure to GTN therapy (which induced hemodynamic tolerance) was associated with a decrease in insulin sensitivity. 10 To date, no study has reported the impact of sustained GTN therapy on whole-body estimates of insulin sensitivity and BCF in humans. The objective of the current study was to quantify the effect of acute and sustained GTN therapy on insulin sensitivity and BCF as assessed by the oral glucose tolerance test (OGTT). We hypothesized that the acute administration of GTN would increase insulin sensitivity, but that insulin resistance would be increased during sustained therapy. Since long-acting nitrates continue to have a role in the therapy on both chronic angina and congestive heart failure, it is important to better understand their potential impact on glucose and insulin metabolism.
Methods
The Mount Sinai Research Ethics Board approved the study and all patients gave written informed consent according to the Declaration of Helsinki.
Study Population
Totally, 12 healthy nonsmoking, male volunteers (18-30 years old) were enrolled after providing written informed consent. Participants enrolled were on no medications and completed a medical questionnaire to confirm that they had no history of any significant illness. Participants were required to consume >200 g/d of carbohydrates in their diet for 3 days prior to each visit and to fast for 12 hours before each OGTT. All OGTT studies were carried out at approximately 0930 hours.
Study Design
Screening visit
The screening visit included a general medical history and physical examination by a clinician. Standing blood pressure (BP) and heart rate measurements were determined as an average of 3 measurements using an automatic, calibrated sphygmomanometer (Critikon Company LLC, Tampa, Florida). Fasting venous blood samples were drawn to ensure glucose, cholesterol, and triglyceride levels were within the normal range. 18,19
Visit 1
Participants were studied in the fasting state. A forearm venous catheter was inserted for repeated blood sampling. Baseline glucose and specific insulin samples were obtained. Subsequently, participants underwent a standard 75 g OGTT involving the ingestion of a 75-g glucose load (time = 0) and venous blood samples were drawn at 30, 60, 90, and 120 minutes with glucose as well as specific insulin measured at each time point. Insulin concentrations at each time point were determined from a single blood sample. Blood samples, collected in tubes containing sodium fluoride preservative, were kept on ice until processing in the hospital core laboratory. The results of this baseline OGTT served as the data for visit 1. This and all OGTT at subsequent visits were performed beginning at approximately 0930 hours.
Visit 2
All participants returned to the laboratory at 0800 hours after a 12-hour overnight fast and blood samples were drawn for serum glucose and insulin. A transdermal GTN 0.6 mg/h preparation was applied (Transderm Nitro, Novartis, Dorval, QC, Canada). Participants remained fasting and an OGTT was performed 90 minutes later, as described above. The BP and heart rate measurements were determined and the participants were instructed to wear the 0.6 mg/h GTN preparation continuously, changing it daily at 0900 hours, until their return visit in 7 days.
Visit 3
Participants returned to the laboratory after 7 days of continuous therapy with GTN and fasting blood drawn for glucose and insulin. Following another OGTT, patients were instructed to remove their GTN patch and to return to the laboratory in 2 to 3 days for their final visit.
Visit 4
Participants returned to the laboratory 2 to 3 days after discontinuation of GTN therapy. Fasting blood glucose and insulin levels were drawn, as described above. All measures, including an OGTT, were repeated as described in visit 1. At the end of this final OGTT the participants were discharged from the laboratory.
Analytical Methods
Serum glucose concentrations were measured by the glucose oxidase technique on an automated analyzer, with samples processed by the Mount Sinai Hospital central laboratory. Specific insulin was measured using the Roche Elecsys 1010 immunoassay analyzer and the electrochemiluminescence immunoassay kit (Roche Diagnostics, Laval, QC, Canada). This assay shows 0.05% cross-reactivity to intact human proinsulin and the primary circulating split form des-31,32 proinsulin.
Calculations
Whole-body insulin sensitivity during the OGTT was estimated using the composite insulin sensitivity index (ISIcomp) equation as described by Matsuda and DeFronzo.
20
This equation describes the:
The incremental glucose (G30) and insulin (I30) responses were calculated as the difference between the values 30 minutes after glucose intake and those before glucose intake.
21
From this the early phase insulin secretion, in response the glucose ingestion, can be calculated as the insulinogenic index (IGI):
To evaluate pancreatic BCF, insulin secretion must be assessed with respect to the current insulin sensitivity. The measure of BCF used in this study is a well-established OGTT-based measure
22
–24
:
An estimate of insulin resistance, the homeostasis model assessment-insulin resistance (HOMA-IR index),
25
was derived using fasting insulin and glucose values:
Statistical Analysis
Based on the results of a previous study 11 examining the effect of transdermal GTN on ISI, sample size calculations revealed that approximately 10 patients would be required to detect a 30% change in ISI with 90% power (2-sided α of 0.05). The effects of acute and sustained GTN therapy were evaluated by comparing values on visit 1 with subsequent visits (2, 3, and 4). All values were tested for normality using the Kolmogorov-Smirnov test. Repeated measures analysis of variance was used to analyze normally distributed values. A 2-sided P value of less than .05 was considered to indicate statistical significance. Scheffé's method was used to correct for multiple comparisons. Nonparametric data were analyzed using the Wilcoxin signed rank test with Benferroni correction for multiple comparisons. Parametric data presented are expressed as mean ± standard deviation; for some data the 95 % confidence intervals are also presented. Nonparametric data are presented as the mean with interquartile range. Statistical analyses were performed with Statview 5.01 (SAS Institute Inc, Cary, North Carolina).
Results
The baseline clinical characteristics of all participants are outlined in Table 1. The BP and heart rate were measured prior to the OGTT at each visit and did not vary across visits (data not shown).
Baseline Clinical Characteristics of Patients.a

Abbreviations: BMI, body mass index; BP, blood pressure.
aData are presented as mean ± standard deviation.
Effect of GTN on Insulin Concentrations and ISI
Neither acute nor sustained therapy with GTN had a significant effect on plasma insulin concentrations either at individual time points or when averaged on each visit (Figure 1, Table 2). There was no significant change in the ISIcomp, IGI, or BCF with acute or sustained GTN therapy (Table 2). The HOMA-IR index of insulin resistance after the oral glucose load on visit 2 (after acute exposure to GTN) was not significantly different than that observed on visit 1. On day 3, after 7 days of GTN exposure, the HOMA-IR increased (P < .015; Table 2).

Plasma insulin concentrations during the oral glucose tolerance test on visit 1 (control), on visit 2 (before and after acute therapy with 0.6 mg/h transdermal glyceryl trinitrate), visit 3 (after 7 days of sustained therapy with transdermal glyceryl trinitrate 0.6 mg/h), and visit 4 (2-3 days after discontinuation of glyceryl trinitrate therapy).
Metabolic Values, Estimates of Insulin Resistance, and β-Cell Function.a

Abbreviations: HOMA-IR, homeostasis model assessment of insulin resistance index; ISI
aResults are presented as mean ± standard deviation (95% confidence interval) unless otherwise indicated. Nonparametric data is presented as the median with interquartile range. Visit 1, baseline values prior to therapy with glyceryl trinitrate; visit 2, after 90 minutes of transdermal glyceryl trinitrate therapy; visit 3, after 7 days of transdermal glyceryl trinitrate therapy; and visit 4, 1 to 2 days after discontinuation of glyceryl trinitrate.
b P < .015 versus visit 1.
c P < .015 versus visit 1.
Effect of GTN on Plasma Glucose Concentrations
The change in glucose concentrations during each OGTT on each of the study days is shown in Figure 2. Plasma glucose concentrations remained within the normal range at each sampling time point on all study days. The average glucose concentration after the oral glucose load on visit 2 (after acute exposure to GTN) was not significantly different than that observed on visit 1 (Figure 2, Table 2). Of note, on visit 3, after 7 days of GTN exposure, the average glucose concentration was significantly increased when compared to that observed on visit 1 (P < .015; Figure 2, Table 2).

Plasma glucose concentrations during the oral glucose tolerance test on visit 1 (control), on visit 2 (before and after acute therapy with 0.6 mg/h transdermal glyceryl trinitrate), visit 3 (after 7 days of sustained therapy with transdermal glyceryl trinitrate 0.6 mg/h), and visit 4 (2-3 days after discontinuation of glyceryl trinitrate therapy). * indicates P < .015; difference in average plasma glucose post oral glucose tolerance test on visit 3 versus visit 1.
Discussion
Importantly, the impact of sustained exposure to GTN on insulin and glucose metabolism in humans has not been examined. The results of the current study illustrate that, in healthy participants, sustained GTN therapy causes small but significant increases in plasma glucose concentrations after an oral glucose load without significant changes in plasma insulin, suggesting an increase in peripheral insulin resistance. Consistent with this, there was an increase in the HOMA-IR measure of insulin resistance on visit 3 although there were no other changes in measures of insulin sensitivity or BCF. Our investigation does not address the mechanism, but it is possible that a decrease in NO bioavailability associated with a net increase in free radical bioavailability, known to occur during sustained GTN therapy, may lead to these changes in glucose metabolism. 16,17
The current findings do not confirm the findings of Kovacs et al who found that the acute transdermal GTN caused a decrease in plasma insulin responses during an OGTT without a parallel change in plasma glucose. 11 This difference may be the result of the larger sample size and differences in both the dose of transdermal GTN and the timing of the OGTT after GTN administration. 11 Furthermore, other studies have failed to observe an acute effect of NO donors on the measures of glucose and insulin metabolism. For example, Henstridge et al also did not observe an effect of either nitroprusside or oral organic nitrates (isosorbide-5-mononitrate and pentaerythritol tetranitrate) on measures of plasma glucose and insulin before and after an OGTT.
In the current study sustained (7 days) continuous transdermal therapy was employed. This is a dosing regimen that has been documented on multiple occasions to cause tolerance with complete loss of hemodynamic effects within 24 hours of the initiation of therapy. It is now recognized that loss of hemodynamic effect does not equate with a loss of the biochemical action of GTN and other organic nitrates. It has been demonstrated in both animal and human models that sustained GTN therapy is associated with an increase bioavailability of free radical species and a decrease in the bioavailability of NO. 16,17 These effects have important physiologic consequence, as they lead to the development of endothelial dysfunction, 14,26 heightened sensitivity of the vasculature to vasoconstrictors, 27 evidence of free radical-induced lipid injury 28 along will changes in autonomic nervous system regulation. 29 It is these “nonhemodynamic” effects of organic nitrates that led to the hypothesis that continuous GTN might modify glucose homeostasis. Sustained exposure to the organic nitrates, including GTN, has been repeatedly shown to cause plasma volume expansion. 30,31 The mechanism of this effect is unclear although a change in membrane permeability or of transmembrane oncotic forces have been hypothesized. Whatever the mechanism, an increase in plasma volume should not be associated with an increase in plasma glucose concentrations, but it is possible that the observed changes in glucose concentrations during GTN therapy could result from an effect not involving insulin secretion or glucose uptake.
Limitations
It is recognized that the current observations were made in lean, normal volunteers. Future studies should assess the effect of GTN use on insulin sensitivity in patients with obesity, glucose intolerance, or overt diabetes, particularly in the setting of coronary artery disease where increased free radical bioavailability and abnormalities of NO metabolism may amplify the impact of GTN on measures of glucose metabolism. We used a continuous dosing regimen of GTN, an approach that is generally not used in clinical practice. It is recognized that the observed results might well be different if an intermittent GTN dosing regimen had been used. However, our purpose was to induce the abnormal biochemical responses and changes in free radical associated with continuous exposure to GTN, since we hypothesized that these changes might impair glucose homeostasis.
Conclusion
In summary, the current study demonstrates that sustained transdermal GTN causes small, but significant increases in plasma glucose concentrations in response to a glucose load along with an increase in an estimate of insulin resistance. The possibility that sustained organic nitrate therapy could worsen abnormalities of glucose metabolism in patients with insulin resistance and/or frank diabetes is a question that warrants future investigation.
Footnotes
Authors’ Note
John D. Parker is the recipient of a Career Investigator award of the Heart and Stroke Foundation of Ontario.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by an operating grant of the Heart and Stroke Foundation of Canada.