Abstract
Background and Purpose:
To investigate the effect of thiamine and thiamine pyrophosphate on doxorubicin-induced cardiotoxicity biochemically and histopathologically and to examine whether doxorubicin cardiotoxicity is related to the conversion of thiamine into thiamine pyrophosphate and inhibition of thiamine pyrophosphokinase (TPK) enzyme.
Experimental Approach:
A total of 48 Albino Wistar male rats were used. Rats were divided into groups as thiamine + doxorubicin (TIA + DOX), thiamine pyrophosphate + doxorubicin (TPP + DOX), DOX, and healthy (HEA) groups. One hour after the administration of thiamine and TPP in 25 mg/kg doses, 5 mg/kg doxorubicin were injected to all groups except HEA group during 7 days. Then, the samples were collected for biochemical (glutathione [GSH], malondialdehyde [MDA], DNA damage, creatine kinase (CK), CK-MB, and troponine I [TP-I]), molecular (TPK), and histopathological examinations.
Key Results:
Oxidant parameters (MDA and DNA damage) decreased and antioxidant parameter (GSH) increased in TPP + DOX group. In addition, levels of CK, CK-MB, and TP-I were low in the TPP + DOX group and high in the TIA + DOX and DOX groups. Cardiac tissue was protected in TPP + DOX group, and no protective effect was observed in TIA + DOX and DOX groups. Messenger RNA expression of TPK was decreased in DOX and TIA + DOX groups.
Conclusion and Implications:
The cardiotoxic effect of doxorubicin originated from the inhibition of TPK enzyme resulting in insufficient production of thiamine pyrophosphate.
Introduction
Thiamine, vitamin B1, is a water-soluble essential vitamin that is found in many foods and is one of the major compounds of cellular metabolism. Thiamine deficiency is not seen in humans who have a balanced diet, 1 but underfed and/or chronic alcoholic people have thiamine deficiency. 2 Biventricular heart failures, 3 beriberi, and peripheral neuropathy can be seen in thiamine deficiency. 4
In the human body, thiamine is found in different forms, such as thiamine triphosphate, thiamine diphosphate, and thiamine monophosphate. Among these forms, thiamine diphosphate, also called thiamine pyrophosphate (TPP), is the active form of thiamine. 5 For this reason, TPP is the best indicator of the efficiency level of thiamine. 5,6 In liver, thiamine is phosphorylated by thiamine pyrophosphokinase (TPK) and converted into the active form (TPP), 7 which is an important coenzyme for mitochondrial oxidative decarboxylation. 8 Thiamine pyrophosphate is also a cofactor of the pyruvate-2 oxoglutarate dehydrogenase complex, which is essential for the mitochondrial synthesis of adenosine triphosphate (ATP), and a cofactor of transketolase enzyme, which has an important role in the maintenance of cell redox by synthesizing nicotinamide adenine dinucleotide phosphate (NADPH) and glutathione (GSH). 6,9 -11 Therefore, TPP plays an important role, particularly in the energy production of visual, cardiovascular, muscular, and central nervous systems, and the deficiency of this component may be life threatening. In addition, thiamine may be deficient in tissues even when it is found in normal levels in the blood. 12
B vitamins are essential in the maintenance of normal myocardial functions, and the deficiency of thiamine, in particular, is provided as a reason for cardiomyopathy. 13 In cardiac failures, levels of urine thiamine and erythrosine TPP have been used to identify thiamine deficiency, 14 -16 and thiamine deficiency was found in 30% of hospitalized patients with acute heart failure. 14 Insufficient TPP production has been suggested as a leading cause to cardiovascular problems. 17
Some substances can cause vitamin deficiency by affecting absorption, metabolism, and/or activation of vitamins. Alcohol and diuretics decrease the efficiency of thiamine, and some anticancer drugs prevent the conversion of thiamine into TPP. 2,14 Diuretics have been shown to increase urinary excretion of thiamine, 14 and chronic alcohol decreases the expression of TPK in renal epithelial cells. 2 Gastaldi et al showed that an anthelmintic levamisole and an amino acid phenyl alanine inhibited TPK 18 ; for this reason, the toxic effects of some drugs have been thought to be linked to the inhibition of TPP production.
Doxorubicin is an anthracycline derivative and antineoplastic agent and has many side effects, of which the most important, cardiotoxicity, was first noticed in children who took doxorubicin therapy. 19,20 To date, reactive oxygen species (ROS) have been blamed for the cardiotoxicity of doxorubicin, but the mechanism for this cardiotoxic effect has not been clarified. 21 To our knowledge, there is no existing information on whether the cardiotoxic effect of doxorubicin is linked to the inhibition of TPP production.
The aim of this study was to investigate the biochemical and histopathological effects of thiamine and TPP on doxorubicin-induced cardiotoxicity and to examine whether doxorubicin cardiotoxicity is related to the conversion of thiamine into TPP and inhibition of the TPK enzyme.
Materials and Methods
Animals
We obtained 48 male albino Wistar rats each weighing between 200 and 220 g from the Ataturk University Medicinal and Experimental Application and Research Center. The animals were divided into treatment groups before the experimental procedures were initiated. The animals were housed and fed under standard conditions in a laboratory where the temperature was kept at 22°C. Animal experiments were performed in accordance with the national guidelines for the use and care of laboratory animals and were approved by the local animal care committee of Ataturk University.
Chemicals
Thiamine, TPP, and other chemicals used in assays were obtained from Sigma Co (Munich, Germany), doxorubicin from Med Drug (Istanbul, Turkey), thiopental from IE ULAGAY (Istanbul, Turkey), troponin I (TP-I) kit from VIDAS, bioMérieux SA (Marcy l’Etoile, France), creatine kinase (CK) and CK-MB kit from Roche Diagnostics Ltd (Rotkreuz, Switzerland), Ambion PureLink RNA Mini Kit from Life Tech (California), High Capacity cDNA Reverse Transcription Kit 200 reactions, Housekeeping Assay: ACTB, and Taq Man Gene Expression Master Mix kit from Applied Biosystems (California), and TPK enzyme assay from Primer Design (Southampton, United Kingdom).
Doxorubicin-Induced Cardiotoxicity Experiment
Rats were divided into the following groups of 12 rats each: thiamine + doxorubicin (TIA + DOX), thiamine pyrophosphate + doxorubicin (TPP + DOX), doxorubicin (DOX) control, and healthy (HEA). Thiamine of 25 mg/kg was administered to the TIA + DOX group and 25 mg/kg TPP was injected intraperitoneal (ip) in the TPP + DOX group. 22 Distilled water was injected ip in the DOX and HEA groups. One hour after the injections, 5 mg/kg doxorubicin was administered to all groups except the HEA group. 23 This procedure was repeated for 7 days, after which blood samples were collected from the rats under thiopental anesthesia, and their hearts were removed. Collected samples were used for biochemical, histopathological, and molecular studies. Results were assessed by comparing groups with the DOX and HEA groups.
Biochemical Analyses
Total GSH analysis
The amount of total GSH (tGSH) in the total homogenate was measured according to the method of Sedlak and Lindsay with some modifications. 24 The tissue was weighed and homogenized in 2 mL of 50 mmol/L Tris-HCl buffer containing 20 mmol/L EDTA and 0.2 mmol/L sucrose at pH 7.5. The homogenate was immediately precipitated with 0.1 mL of 25% trichloroacetic acid, and the precipitate was removed after centrifugation at 4200 rpm for 40 minutes at 4°C. The supernatant was used to determine GSH, using 5,5-dithiobis(2-nitrobenzoic acid). Absorbance was measured at 412 nm using a spectrophotometer.
Malondialdehyde analysis
The concentrations of tissue lipid peroxidation were determined by estimating malondialdehyde (MDA), using the thiobarbituric acid test. 25 Tissue was weighed and then homogenized in 10 mL of 100 g/L KCl. The homogenate (0.5 mL) was added to a solution containing 0.2 mL of 80 g/L sodium lauryl sulfate, 1.5 mL of 200 g/L acetic acid, 1.5 mL of 8 g/L 2-thiobarbiturate, and 0.3 mL distilled water. The mixture was incubated at 98°C for 1 hour. Upon cooling, 5 mL of n-butanol–pyridine (15:1) was added. The mixture was vortexed for 1 minute and centrifuged for 30 minutes at 4000 rpm. Absorbance of the supernatant was measured at 532 nm. A standard curve was obtained by using 1,1,3,3-tetramethoxypropane.
Isolation of DNA From Cardiac Tissue
Tissue was drawn and DNA isolated using the modified method of Shigenaga et al. 26 Samples (50–200 mg) were homogenized at 4°C in 1 mL of homogenization buffer (0.1 mol/L NaCl, 30 mmol/L Tris, pH 8.0, 10 mmol/L EDTA, 10 mmol/L 2-mercaptoethanol, 0.5%, v/v, Triton X-100) with 6 passes of a Teflon-glass homogenizer (Thomas Scientific, New Jersey) at 200 rpm. The samples were centrifuged at 4°C for 10 minutes at 1000g to pellet nuclei. The supernatant was discarded, and the crude nuclear pellet was resuspended and rehomogenized in 1 mL of extraction buffer (0.1 mol/L Tris, pH 8.0, 0.1 mol/L NaCl, 20 mmol/L EDTA) and recentrifuged as described earlier for 2 minutes. The washed pellet was resuspended in 300 µL of extraction buffer with a wide-orifice 200-µL Pipetman tip (Eppendorf AG, Germany). The resuspended pellet was subsequently incubated at 65°C for 1 hour with the presence of 0.1 mL of 10% sodium dodecyl sulfate, 40 μL of proteinase K, and 1.9 mL of leukocyte lysis buffer. Then, ammonium acetate was added to the crude DNA sample to give a final concentration of 2.5 pmol/L and centrifuged in a microcentrifuge for 5 minutes. The supernatant was removed and mixed with 2 volumes of ethanol to precipitate the DNA fraction. After centrifugation, the pellet was dried under reduced pressure and dissolved in sterile water. The absorbance of this fraction was measured at 260 and 280 nm. Purification of DNA was determined as A260/280 ratio 1.8.
DNA Hydrolysis With Formic Acid
Approximately 50 mg of DNA was hydrolyzed with 0.5 mL of formic acid (60%, v/v) for 45 minutes at 150°C. 27 The tubes were allowed to cool. The contents were then transferred to Pierce microvials (Sigma Co., Germany), covered with Kleenex tissues (Kimberly-Clark, Wisconsin) cut to size (secured in place using a rubber band), and cooled in liquid nitrogen. Formic acid was the removed by freeze drying. Before analysis by high-performance liquid chromatography (HPLC), the contents in the vials were redissolved in the eluent (final volume 200 µL).
Measurement of 8-Hydroxy-2 Deoxyguanine With HPLC
The amount of 8-hydroxy-2 deoxyguanine (8-OHdG) and deoxyguanine (dG) was measured using an HPLC system equipped with an electrochemical detector (HP Agilent 1100 module series, E.C.D. HP 1049 A), as described previously. 27,28 The amounts of 8-OHdG and dG were analyzed on a 250 × 4.6-mm Supelco LC-18-S reverse-phase column (Phenomenex, California). The mobile phase was 50 mmol/L potassium phosphate, pH 5.5, with acetonitrile (97:3, v/v), and the flow rate was 1.0 mL/min. The detector potential was set at –0.80 V for measuring the oxidized base. The dG and 8-OHdG (25 pmol) were used as standards. The DNA damage was expressed as the number of 8-OHdG/105 dG molecules. 29
Creatine Kinase Analysis
Plasma CK measurement was performed photometrically in a Roche/Hitachi cobas c 701 system (Roche Diagnostics, Indiana). All steps were conducted in accordance with the test procedures. Equimolar NADPH and ATP occurred at the same speed. The NADPH occurrence speed was directly proportional to the CK activity at 340 nm photometrically.
Creatine kinase-MB analysis
Plasma CK-MB measurement was photometrically performed in a Roche/Hitachi cobas c 701 system. All steps were conducted in accordance with the test procedures, using an immunological ultraviolet test.
Troponin I analysis
Plasma TP-I levels were measured with a VIDAS Troponin I Ultra kit via an enzyme-linked fluorescent assay technique. All steps were automatically conducted in accordance with the test procedures, using the VIDAS equipment.
Histopathological analyses
Hearts of rats were fixed in 10% formaldehyde. Cardiac tissue from all groups was placed in bottles containing 4% formaldehyde. For routine tissue follow-up, tissue was bathed during the day. Then, tissue was placed in 70% ethanol for 1 night and placed in 80%, 96%, and 100% ethanol for 1 hour each. Tissues that were being placed in a xylene series were incubated for 4 hours at 60°C in xylene + bead paraffin. Tissues were embedded into paraffin blocks and prepared for sectioning. Sections of 5 µm were obtained, and these were stained with hematoxylin and eosin (H&E) for general observation and Masson trichrome for interstitial fibrosis. Stained sections were examined under a light microscope, and photographs were obtained for all groups.
Measurement of Relative TPK Messenger RNA Expression in Cardiac Tissue With Real-Time Polymerase Chain Reaction
Each cardiac tissue sample was weighed, and total RNA isolation was performed according to the manufacturer guidelines (Pure Link RNA mini kit; Ambion). Then reverse transcriptase reaction and complementary DNA (cDNA) synthesis were performed using the high-capacity cDNA Reverse Transcription kit (Applied Biosystems; serial no: 4368813). Complementary DNA synthesis composition was determined in a Veriti 96-well Thermal Cycler (Applied Biosystems) in steps as follows: 10 minutes at 25°C, 120 minutes at 37°C, 5 minutes at 85°C, and ∞ at 4°C. Level of cDNA was determined in a nano drop spectrophotometer (EPOCH Take3 Plate; Biotek, Biotek Biomedical Devices, Turkey), and cDNA was stored at –20°C. Messenger RNA (mRNA) expression of TPK was measured via the Taq Man Gene Expression Master Mix kit. Amplification and quantification steps were performed in the StepOne Plus Real-Time polymerase chain reaction (PCR) System (Applied Biosystems). For 100 ng cDNA and for a weel, TPK gene (Primer Design Rattus norvegicus TPK1) plus mastermix (total 10 µL) was used for TPK analyses and ACTB gen (Rn00667869_m1; Applied Biosystems) plus mastermix (total 10 µL) was used for housekeeping gene analyses. Primer sequences for TPK1 and ACTB were as follows: TPK1 forward, 5′-AAA CTG ACC ATC CGC TCC T-3′; reverse, 5′-GAA GGC ACA GGC ACA AGA T-3′; ACTB forward, 5′-TGG TGG GTA TGG GTC AGA AG-3′; reverse, 5′-GAC AAT GCC GTG TTC AAT GG-3′. They were diluted to 20 µL with RNAase-free water. The plate was placed at 50°C for 2 minutes and at 95°C for 10 minutes. After these steps, the plate was cycled at 95°C for 15 seconds and 60°C for 1 minute for 40 cycles. The Ct values were changed into ΔΔCt, and relative TPK mRNA expression results were obtained. The HEA group was used as the reference value.
Statistical Analyses
Results were expressed as mean ± standard error of the mean. Data were subjected to 1-way analysis of variance (ANOVA) using the SPSS (version 18.0) software. Differences among groups were obtained using the Tukey test option and were considered significant at P < .05.
Results
Biochemical Results
Total GSH and MDA analysis
As seen in Table 1, there was a very significant difference between HEA and DOX groups (P < .0001) in tGSH levels. Administration of TPP caused a very significant increase in TPP + DOX group in the tGSH levels when compared to DOX group (P < .0001). In contrast, no significant decrease was observed with TIA administration. (P > .05).
Levels of Total Glutathione (tGSH), Malondialdehyhde (MDA), and Oxidative DNA Damage in Cardiac Tissue of Experimental Groups.a

Abbreviations: TIA, thiamine; DOX, doxorubicin; TPP, thiamine pyrophosphate; HEA, healthy; 8-OHdG, 8-hydroxy-2 deoxyguanine; ANOVA, analysis of variance; dG, deoxyguanine.
aN = 6. Results were expressed as mean ± standart error. P < .05 was evaluated as significant in 1-way ANOVA test, Tukey option.
A significant difference was observed between HEA and DOX groups (P < .0001) in MDA levels as well. When compared to DOX group, TPP administration in TPP + DOX group caused a very significant decrease (P < .0001) but not TIA in TIA + DOX group in this level (P > .05).
Oxidative DNA damage (8-OHdG/105 dG) analysis
The 8-OHdG/105 dG level in cardiac tissue of the DOX group increased significantly versus the HEA group (P < .0001). Oxidative DNA damage was reduced in the TPP + DOX group significantly (P < .0001) but not in TIA + DOX group compared to the intact rat group (P > .05; Table 1).
Creatine kinase, CK-MB, and TP-I analysis
As shown in Figure 1, levels of CK, CK-MB, and TP-I were lower in the HEA group when compared to the DOX group (P < .0001). These levels were significantly lower in the TPP + DOX group and nearly same as in the HEA group versus the DOX group. In contrast, these levels were very high in TIA + DOX group similar to DOX group (P > .05).

Measurements of creatine Kinase (CK; A), CK-MB (B), and troponin I (TP-I; C) in serum of doxorubicin-induced cardiotoxicity experiment’s rat groups. Thiamine of 25 mg/kg was administered to the TIA + DOX group and 25 mg/kg thiamine pyrophosphate was injected to the TPP + DOX group. One hour after the injections, 5 mg/kg doxorubicin was administered to all groups except the HEA group. N = 6. Results were expressed as mean ± standart error. P < .05 was evaluated as significant in 1-way analysis of variance (ANOVA) test, Tukey option (* means P < .05). DOX indicates doxorubicin; HEA, healthy; TIA, thiamine; TPP, thiamine pyrophosphate.
Histopathological Results
Healthy rat group. Cardiac tissue
As seen in Figure 2, histopathological evaluation was normal in the HEA rat group.
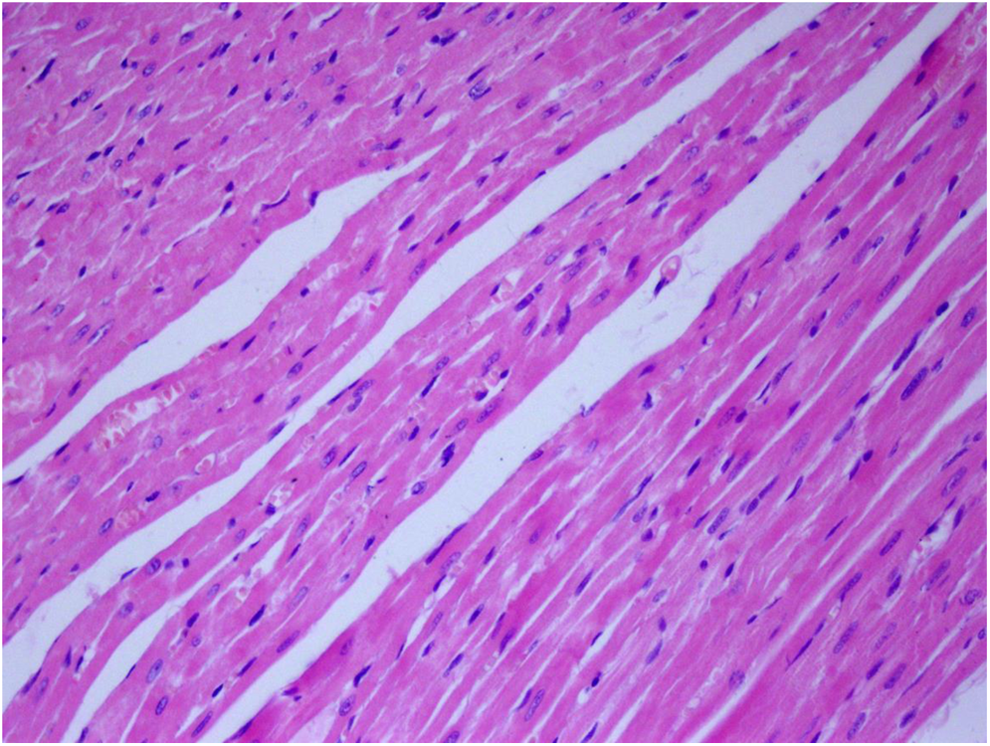
Histopathological evaluation of cardiac tissue of healthy rat group (stain: hematoxylin and eosin, magnification: ×200).
Doxorubicin rat group. Cardiac tissue
Vacuoles were present within the cytosol of Doxorubicin treated rats. The cardiac damage was also manifest by intracellular edema, myofibrillar loss, and interstitial fibrosis.
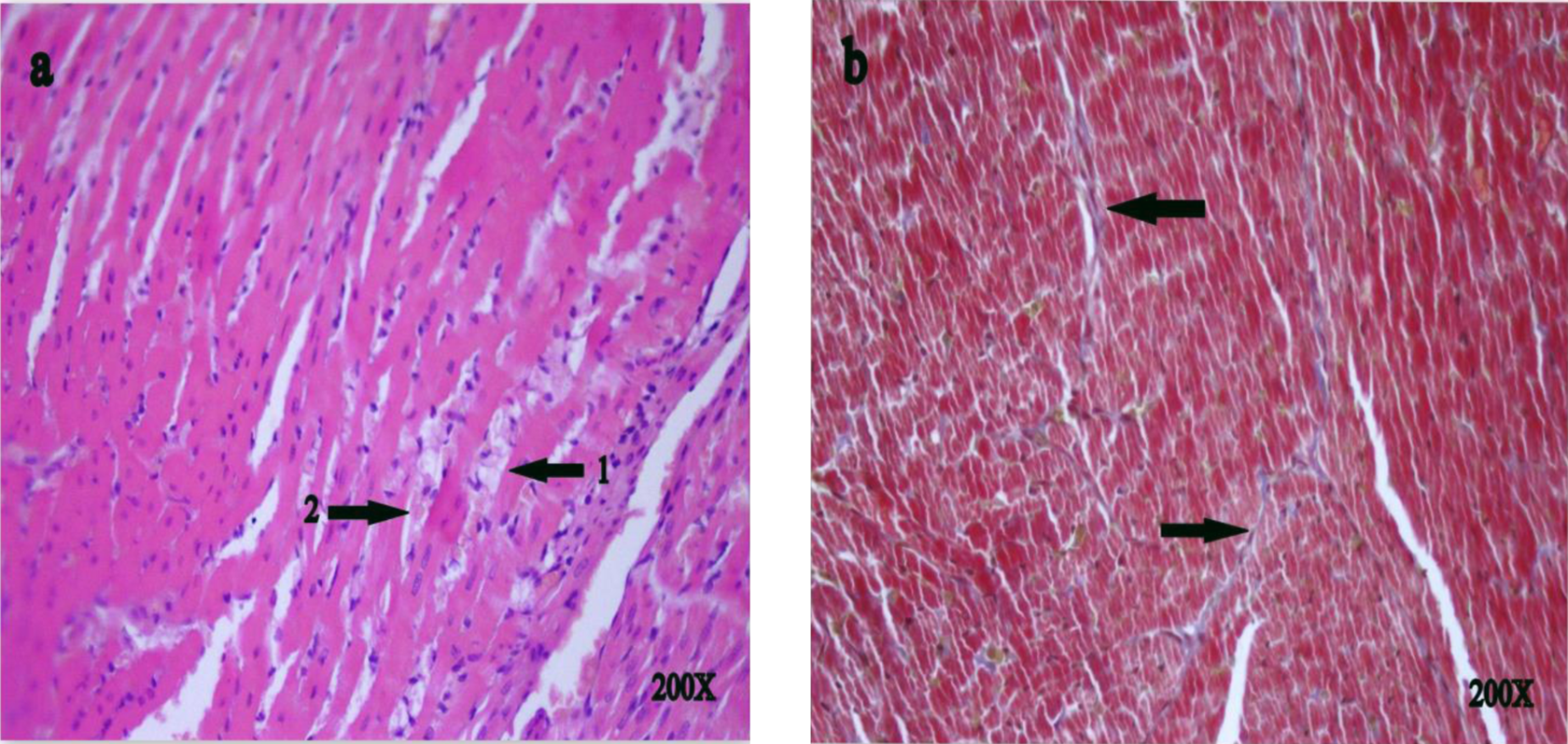
Histopathological evaluation of cardiac tissue of doxorubicin rat group. Cytoplasmic vacuolization increase (A, arrow 1, H&E, magnification: ×200), cardiac tissue damage such as edema and myofibrillar loss (A, ok-2, hematoxylin and eosin [H&E], magnification: ×200), and interstitial fibrosis (B, Masson Trichrome) were shown in doxorubicin (DOX) group.
The TPP + DOX rat group cardiac tissue
In this group, cytoplasmic vacuolization, edema, myofibrillar loss (Figure 4A), and interstitial fibrosis (Figure 4B) were not seen.
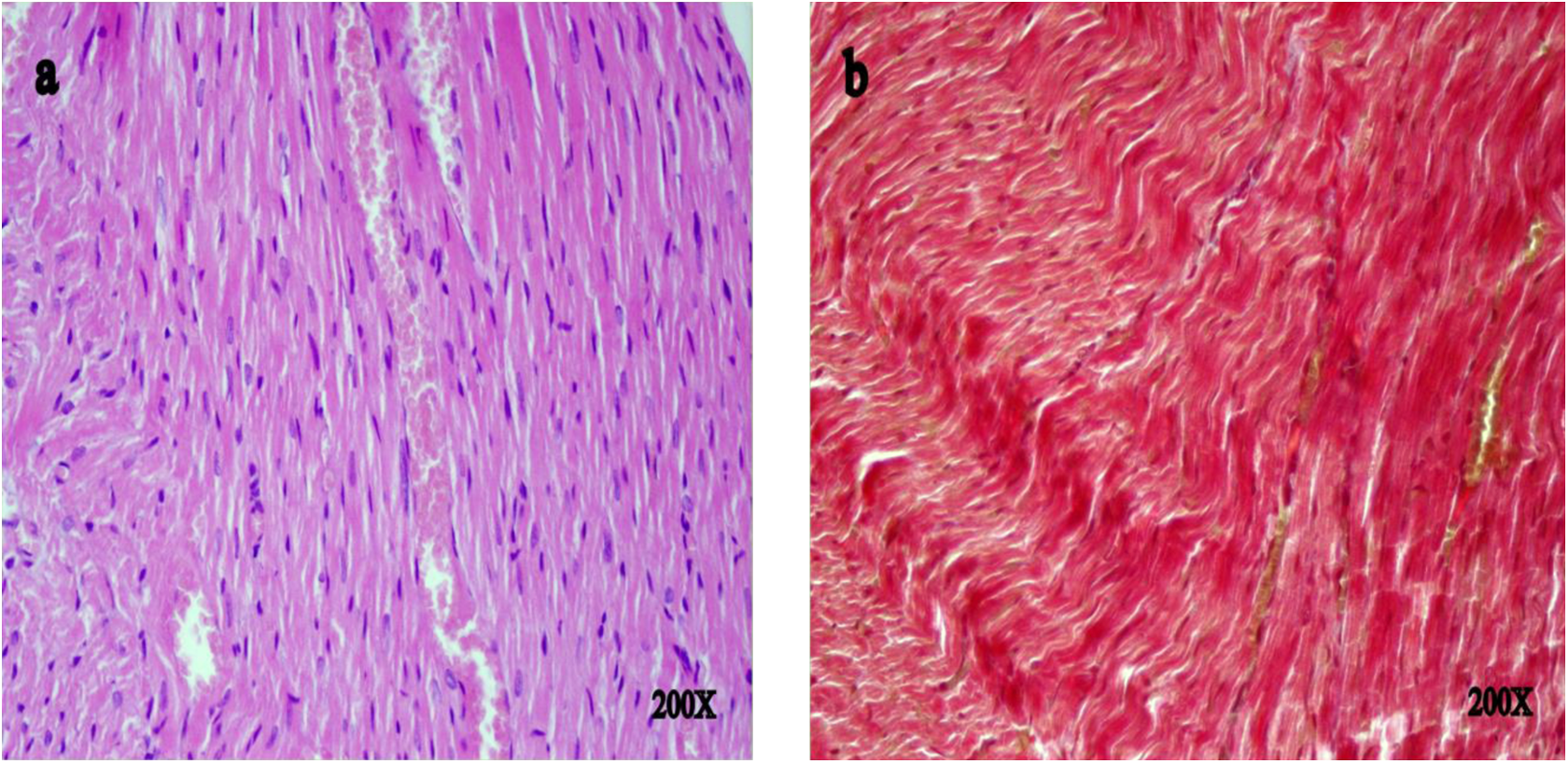
Histopathological evaluation of cardiac tissue of thiamine pyrophosphate (TPP) + doxorubicin (DOX) rat group. Cytoplasmic vacuolization, edema, myofibrillar loss (A, hematoxylin and eosin [H&E], magnification: ×200), and interstitial fibrosis were not seen (B, Masson’s Trichrome, magnification: ×200).
The TIA + DOX rat group cardiac tissue
Edema (Figure 5A, arrow 1), myofibrillar loss (Figure 5A, arrow 2), and interstitial fibrosis (Figure 5B, Masson Trichrome) were observed in the TIA + DOX group.

Histopathological evaluation of cardiac tissue of thiamine + doxorubicin (TIA + DOX) rat group. Edema (A, arrow 1, hematoxylin and eosin [H&E], magnification: ×200), myofibrillar loss (arrow 2), and interstitial fibrosis were observed (B, arrow, Masson’s Trichrome, magnification: ×400).
Relative TPK mRNA expression analysis in rat cardiac tissue
As seen in Figure 6, when the HEA group was used as the reference group (1.0-fold), expression of TPK mRNA decreased by 0.44-fold in the DOX group and 0.50-fold in the TIA + DOX group. These 2 decreases were significant when compared to the HEA group.

Relative thiamine pyrophosphokinase (TPK) messenger RNA (mRNA) expression fold in rat cardiac tissue in quantitative polymerase chain reaction (PCR). Thiamine of 25 mg/kg was administered to the TIA + DOX group. One hour after the injections, 5 mg/kg doxorubicin was administered to all groups except the HEA group. N = 6. Results were expressed as mean ± standart deviation. P < .05 was evaluated as significant in 1-way ANOVA test, Tukey option (*means P < .05). DOX indicates doxorubicin; HEA, healthy; TIA, thiamine.
Discussion
Reactive oxygen species are responsible for doxorubicin cardiotoxicity as well as the pathogenesis of other diseases. 30 Physiologically, ROS and antioxidant defence mechanisms are in balance in the body. When this balance is impaired by ROS, detrimental oxidative stress occurs. Damage starts with the formation of lipid radicals; this radical first converts to lipid hydroperoxides and then to toxic radicals, such as aldehydes, alkenes, and MDA. 31 Consequently, MDA can be used as a damage indicator. Reactive oxygen species also have many detrimental effects on the DNA molecule, similar to the lipid radicals. 32 Oxidative damage to approximately 100 DNA bases have been identified. 33 In recent studies, base damage has been analyzed as an indicator of DNA damage. Because Cu++ ions have been found in G–C-rich zones in DNA, guanine is the base most susceptible to oxidative damage. 34 Consequently, 8-OHdG is the most analyzed base and is accepted as a biomarker of base damage. 35
Our results showed that levels of MDA and 8-OHdG/105 dG in cardiac tissue of the DOX group were significantly higher when compared to the HEA group. No significant difference was observed in the cardiac tissues of the TIA + DOX group for MDA and 8-OHdG/105 dG levels. These levels were significantly lower in the TPP + DOX group and nearly the same as the HEA group. These results showed that oxidant/antioxidant balance was impaired in favor of oxidants in the DOX group and an oxidative stress occurred in their hearts. It was also observed that thiamine administration did not fix this imbalance but TPP injection reversed this oxidative damage by decreasing levels of MDA and 8-OHdG/105 dG. Increased doxorubicin-induced cardiotoxicity from MDA has been documented. 30
One of the basic antioxidant substances that protect cardiomyocytes from ROS damage is GSH. Glutathione and other antioxidants, such as melatonin and vitamin, prevent tissue damage by modulating ROS in the cells. 36 If the GSH level decreases, cardiac tissue cannot protect itself against ROS damage and cardiotoxicity occurs. Doxorubicin has been reported to cause cardiotoxicity by decreasing the GSH level. 37 In this study, significant difference was shown in the HEA group when compared to the DOX group. There was no significant difference in GSH level between the TIA + DOX and the DOX groups. However, in the TPP + DOX group, a significant increase occurred in the GSH level and this was similar to the level of the HEA group. Administration of TPP increased the GSH level and protected against doxorubicin damage. In light of this finding, we consider that TPP but not thiamine has an antioxidant property, and therefore thiamine and TPP have different effects on oxidative stress. Thiamine and TPP were compared in another clinical and experimental study and no difference was found. 16
To understand whether there was a difference between thiamine and TPP for cardioprotection, the clinical markers of cardiac damage, including CK, CK-MB activities, and TP-I levels, were measured. We found that CK and CK-MB activities showed a statistically significant increase in the DOX group. This increase was in accordance with the current studies. 37,38 When compared to the DOX group, a significant decrease was shown in the TPP + DOX group and no significant difference was seen in the TIA + DOX group for CK and CK-MB activities. Creatine kinase is a dimeric enzyme and has subtypes, such as CK-MM (skeletal muscle type), CK-BB (brain type), and CK-MB (myocardial type). 39 As a marker of cardiac damage, CK and especially CK-MB activities were used in the clinical diagnosis and follow-up of myocardial infarction and myopathies. 40 The MB type is found in appreciable values (15%-20%) in myocardial tissues, meaning that CK-MB is shown in serum by reflecting its own structure. 39 Another cardiac marker, TP-I, was also measured. Administration of DOX increased TP-I level in serum. 30 Our TP-I results changed in parallel to changes seen in CK and CK-MB levels. Like CK and CK-MB, TP-I is also sensitive and unique to myocardial damage, so it is used in the diagnosis of myocardial infarction. 41 If we need to talk about measured cardiac markers for this study, doxorubicin increased them because of its cardiotoxic effect. Thiamine administration had no effect on this increase, but TPP administration inhibited this increase in cardiac markers and resulted in similar results as the HEA group.
In the histopathological analysis of rat cardiac tissue, myocardial morphology was seen as normal in the HEA group. But in DOX group, severe damage was seen in cardiac tissues. In these tissues, in addition to edema, myofibrillar loss and interstitial fibrosis, increase in cytoplasmic vacuolization was also shown. In the TPP + DOX group, it was shown that cytoplasmic vacuolization, edema, myofibrillar loss, and interstitial fibrosis were not seen and myocardial damage decreased. Histopathology results were in accordance with biochemical results. The TPP protected cardiac tissue against doxorubicin damage, but thiamine did not show any protective effect. Studies have been conducted to identify whether thiamine has a protective effect on cardiac tissue, but results were controversial. In one study, a silent healing was observed in the left ventricle by thiamine administration, and in another study no positive progress was exhibited. 42 -44 In order to address this controversial situation, TPK mRNA expression was determined. Previous studies have also been indefinite about which substance should be used in the determination of micronutrient deficiency, such as thiamine. For example, in heart failure and thiamine deficiency studies, both urinary thiamine levels and erythrocyte TPP levels were used to evaluate the deficiency. 14 -16
Thiamine has an important role in carbohydrate metabolism and it is converted into active TPP by the TPK enzyme. 45 So we decided to investigate whether there might be a problem in the conversion of thiamine to its active form TPP. We therefore consider that doxorubicin could result in TPK enzyme blockage. So TPK mRNA expression was studied in rat cardiac tissue by real-time PCR. Doxorubicin administration decreased TPK mRNA expression and DOX + THI administration did not stop this decrease. Therefore, doxorubicin decreased TPK activity but thiamine did not change this decrease. As a result of TPK blockage, thiamine could not turn into the active form, TPP. Exogenous thiamine did not show a protective effect against doxorubicin cardiotoxicity. Under this condition, TPP administration can be both effective and sensitive.
In conclusion, along with ROS, TPP deficiency has a role in the cardiotoxic effect mechanism of doxorubicin. Our results showed that doxorubicin inhibited TPP production by decreasing TPK enzyme expression and as a result exogenous thiamine was deficient. Our results indicated that TPP but not thiamine had a protective effect.
Footnotes
Author Contributions
B. Polat, H. Suleyman, E. Sener, E, and F. Akcay contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
