Abstract
Although both sevoflurane postconditioning (SPoC) and delayed remote ischemic preconditioning (DRIPC) have been proved effective in various animal and human studies, the combined effect of these 2 strategies remains unclear. Therefore, this study was designed to investigate this effect and elucidate the related signal mechanisms in a Langendorff perfused rat heart model. After 30-minute balanced perfusion, isolated hearts were subjected to 30-minute ischemia followed by 60-minute reperfusion except 90-minute perfusion for control. A synergic cardioprotective effect of SPoC (3% v/v) and DRIPC (4 cycles 5-minute occlusion/5-minute reflow at the unilateral hindlimb once per day for 3 days before heart isolation) was observed with facilitated cardiac functional recovery and decreased cardiac enzyme release. The infarct size-limiting effect was more pronounced in the combined group (6.76% ± 2.18%) than in the SPoC group (16.50% ± 4.55%, P < .001) or in the DRIPC group (10.22% ± 2.57%, P = .047). Subsequent analysis revealed that an enhanced heme oxygenase 1 (HO-1) expression, but not protein kinase B/AKt or extracellular signal-regulated kinase 1 and 2 activation, was involved in the synergic cardioprotective effect, which was further confirmed in the messenger RNA level of HO-1. Such trend was also observed in the nuclear factor erythroid 2-related factor 2 (Nrf2) nuclear translocation, an upstream regulation of HO-1. In addition, correlation analysis showed a significantly positive relationship between HO-1 expression and Nrf2 translocation (r = 0.729, P < .001). Hence, we conclude that DRIPC may produce an additive cardioprotection to SPoC through an enhanced HO-1 expression partly via Nrf2 translocation.
Keywords
Introduction
Acute myocardial injury is one common complication following adult cardiac surgery in association with increased mortality and morbidity. 1 Ischemia–reperfusion (I/R) injury has been recognized as an important contributing factor among the proposed mechanisms. 2 Effectiveness in cardioprotection by different conditioning protocols during myocardial I/R injury has been proven in various young and healthy animal models. 3 However, these beneficial effects have been found in preclinical studies to be confounded with age, 4 –6 gender, 7,8 diabetes mellitus, 9 –11 or concurrent medications, 12,13 all of which are relevant in clinical patients. 14,15 This phenomenon indicates that any single protective strategy may face growing challenge in the clinical translation. Therefore, combination with different conditioning strategies to obtain an enhanced cardioprotective potential is of significant clinical importance. 16 –20
Postconditioning at the onset of reperfusion with sevoflurane, a widely used volatile anesthetic, has been reported to be protective in isolated perfused rat hearts by our 21,22 and other recent publications. 23,24 Similar cardioprotection by remote ischemic preconditioning (RIPC) has also been proven by applying several cycles of transient ischemic stimulus at a remote organ (mostly limb) before myocardial ischemia in various animal models as outlined in several comprehensive reviews. 25,26 Remote ischemic preconditioning presents the protective effect in 2 different phases: early phase (<4 hours) and delayed phase (24-72 hours). 27 Although the precise mechanisms are not fully understood, activation of reperfusion injury salvage kinases (RISK) pathway including phosphatidylinositol-3-kinase (PI3K)-protein kinase B (PKB)/AKt and extracellular signal-regulated kinase 1 and 2 (ERK1/2) has been proposed as the crucial mechanisms for SPoC 23,28 and early RIPC. 29 –31 On the other hand, a few studies have reported that nitric oxide synthase, 32 calcitonin gene-related peptide, 33 or nuclear factor κB 32 may be involved in the cardioprotection elicited by delayed RIPC (DRIPC). Recently, the role of heme oxygenase 1 (HO-1) in delayed preconditioning has been indicated in RIPC-induced hepatic protection 34 and H2S-induced cardioprotection. 35 In an isolated rat heart model from our previous study (data not shown), DRIPC was also found to increase HO-1 protein expression.
It is generally accepted that failure in the enhancement of combined conditioning may be attributed by similar protective mechanism of 2 protocols. 16,17 Both SPoC and RIPC are very applicable for the noninvasive nature and easy for manipulation, indicating a promising perspective in the clinical practice. However, few publications have been concerned about the potential cardiac effect of combined conditioning with these 2 interventions. In this study, we hypothesized that a combination of SPoC and DRIPC confers more pronounced cardioprotection than SPoC or DRIPC alone and tried to elucidate the underlying mechanisms in an isolated heart model with focus on the RISK pathway and HO-1.
Methods and Materials
All procedures were approved by the Institutional Animal Care and Use Committee of Fuwai Hospital, and all animals received appropriate care according to the Guide for the Care and the Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996).
Perfusion and Monitoring of Isolated Rat Hearts
A total of 120 male adult Sprague-Dawley rats (250-350 g) were injected intraperitoneally with ethyl carbamate (1.5 g/kg) and heparin (1000 IU/kg). After the tail-clamp test disappeared and 2% lidocaine infiltrated into the incision, the hearts were rapidly excised, immersed in ice-cold phosphate buffer solution, mounted on a Langendorff apparatus, and perfused without pacing at a constant flow (10 mL/min) with 95% oxygen oxygenated Krebs-Henseleit buffer solution (KHB) containing (in mmol/L) NaCl 118.5, KCl 4.75, MgSO4 1.19, NaHCO3 25.0, KH2PO4 1.2, glucose 11.0, 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid 10.0, and CaCl2 1.4, pH 7.4 at 37°C. A water-filled latex balloon attached to a pressure transducer (Memscap, Norway) was inserted into the left ventricle (LV) via a small incision of the left atrial appendage. The cardiac function was recorded with a Powerlab data-acquisition system (Labchart 7.0, AD Instruments Inc, Colorado Springs, Colorado). The left ventricular end-diastolic pressure (LVEDP) was set to a fixed value that ranged 5 to 10 mm Hg by adjustment of balloon volume. Heart rate (HR), left ventricular developed pressure (LVDP), and maximum LVDP increase rate (+dp/dt) and maximum LVDP decrease rate (−dp/dt) rate were continuously collected.
Experimental Protocols
After 30 minutes at stablized perfusion, isolated hearts were randomized by sealed envelopes into 7 groups (n = 15 per group accounting to a total of 105): (1) time control (TC) group: continuous perfusion for 120 minutes; (2) I/R group: ischemia for 30 minutes followed by 60-minute reperfusion; (3) sevoflurane postconditioning (SPoC) group: perfused with 3% (v/v) sevoflurane-bubbled KHBs (Maruishi Pharmaceutical Co, Japan) oxygenated with 95% oxygen for 10 minutes at the onset of reperfusion and then with normal KHBs for the remaining 50 minutes. The concentration was monitored by an anesthetic gas monitor (Datex Capnomac Ultima, Division of Instrumentarium Corp, Helsinki, Finland) and a gas chromatograph (GC/MSQP2010PLUS, Shimadzu, Kyoto, Japan): 3% (v/v) sevoflurane (0.51 ± 0.04 mmol/L) 22 ; (4) SPoC plus LY294002 (SPoC + LY) group: perfused with 3% (v/v) sevoflurane-bubbled KHBs containing 10 μmol/L LY294002 (a specific PI3K inhibitor, Tocris Bioscience, United Kingdom) for 10 minutes at the onset of reperfusion, and then with normal KHBs for the remaining 50 minutes; (5) DRIPC group: 4 cycles of 5-minute occlusion and 5-minute reflow at unilateral hindlimb once per day for 3 days before heart isolation. Ischemia was confirmed using modified pulse oxymetry for the rats with anesthesia by ethyl carbamate (intraperitoneally, 1.0 g/kg). The time interval between each preconditioning was limited to 23 to 24 hours; (6) DRIPC plus zinc protoporphyrin (ZnPP; DRIPC + ZnPP): ZnPP (50 μg/kg body weight, a specific inhibitor of HO-1, Tocris Bioscience, United Kingdom) 36 was injected intraperitoneally before preconditioning in each day; and (7) combination of SPoC and DRIPC (DRIPC + SPoC): delayed preconditioned hearts were then postconditioned by sevoflurane at the onset of reperfusion for 10 minutes. The detailed grouping is illustrated in a schematic form (Figure 1).
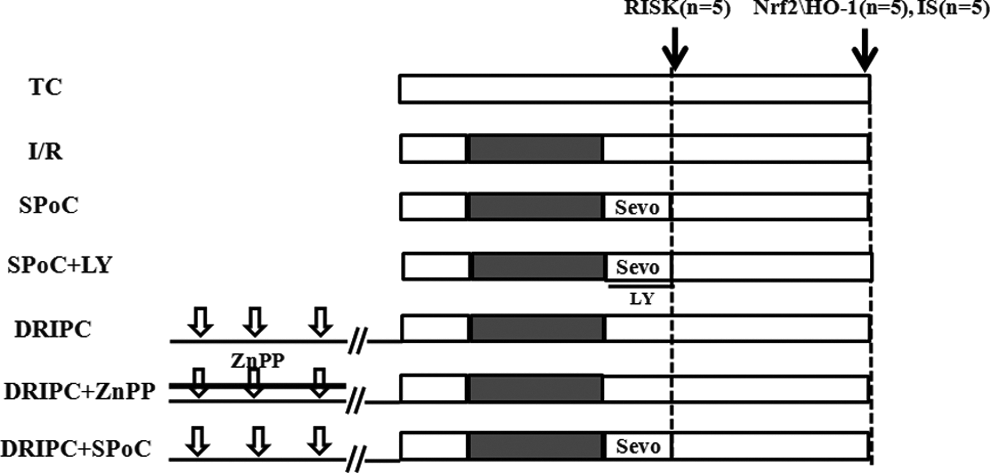
Experiment protocols in seven groups (n=15). TC, time control; I/R, ischemia/reperfusion; SPoC, sevoflurane postconditioning; DRIPC, delayed remote ischemic preconditioning; LY, LY294002, a specific inhibitor of phosphatidylinositol-3-kinase (upstream of protein kinase B/AKt pathway); ZnPP, a specific inhibitor of heme oxygenase 1; DRIPC + SPoC, a combination of DRIPC and SPoC; Sevo, sevoflurane; RISK, reperfusion injury salvage kinase; IS, infarct size.
Infarct Size Measurement
2,3,5-Triphenyltetrazolium chloride (TTC; Sigma-Aldrich, St. Louis, MO) staining was used to determine the myocardial infarct size (IS) at the end of perfusion (n = 5). After being frozen at −20°C for 1 hour, the heart was sliced transversally into 5 sections with about 1-mm thickness and incubated in 0.1% TTC solution for 10 minutes at 37°C. The slices were then fixed in 4% formalin solution overnight. The IS, expressed as a percentage of total ventricular area, was measured by the planimetry with the use of ImageJ 1.43 (Wayne Rasband, Bethesda, MD).
Cardiac Troponin I detection
Coronary effluent (≈1 mL) was collected at the baseline and at the end of 60-minute perfusion for the detection of cardiac troponin I (cTnI) levels using ACS 180 automated chemiluminescence system with commercial kits (Bayer Corp, Whippany, NJ) in a blinded fashion (n = 10).
Western Blotting
The total tissue, cytosolic, and nuclear protein fractions were isolated from the frozen LV samples (n = 5) as described previously. 35,37 Equal amounts of protein (60 μg/lane) were separated on 10% sodium dodecyl sulfate polyacrylamide gel (80 V for 2 hours) by electrophoresis and transferred to nitrocellulose membranes (Pall, Whippany, NJ) using a semi-dry electroblotting apparatus (Bio-Rad, Hercules, CA). After being blocked for 1 hour at room temperature in 5% skim milk, the membranes were incubated overnight at 4°C with primary antibodies. After being washed with Tris-HCl buffer solution containing 0.1% Tween 20 for 3 times, the membranes were incubated for 1 hour at room temperature with horseradish peroxidase-conjugated secondary antibodies. The washed membranes were then used for analysis with a chemiluminescence detection kit (Pierce, Rockford, IL). Target signals and the glyceraldehyde 3-phosphate dehydrogenase (GAPDH; loading control, 1:5000, Zhongshanjinqiao, China) signal were analyzed semiquantitatively using Quantity One system (Bio-Rad, Hercules, CA).
The primary antibodies are as follows: rabbit antibodies against Akt, p-AktSer-473, ERK1/2, p-ERK1/2Thr-202/Tyr-204 (all monoclonal, 1:1000, Cell Signal Technique, Boston, MA), nuclear factor erythroid 2-related factor 2 (Nrf2; polyclonal, 1:400, Santa Cruz, Dallas, TX), and HO-1 (polyclonal, 1:500, Abcam, Cambridge, MA).
Real-Time Polymerase Chain Reaction
The messenger RNA (mRNA) level of HO-1 was analyzed by real-time polymerase chain reaction (RT-PCR). Total RNA was isolated from the LV samples lysed in Trizol solution (Invitrogen, Carlsbad, CA; n = 3). The RNA samples were then reversely transcribed with First Strand complementary DNA (cDNA) synthesis kit (Roche, Indianapolis, IN) in a 20 μL reaction system. The cDNA was then amplified with Universal SYBR Green Master [Rox] in the LightCycle 480 system (Roche, Indianapolis, IN) using the following primers: (1) HO-1 forward, CAAGCACAGGGTGACAGAAG; reverse, CCTCTGGCGAAGAAACTCTG; (2) GAPDH forward, CAACTCCCTCAAGATTGTCAG; reverse, ATGGCATGGACTGTGGTCAT.
Statistical Analysis
All data were expressed as mean ± standard deviation (SD). The sample size was chosen according to the previous studies by our group. 21,22,28,38 A sample size of 3 to 5 was set to obtain a SD in statistical analysis for protein expression and transcription determination. In all, 5 hearts per group (n = 5) were used for IS analysis, 5 per group at 60-minute reperfusion (n = 5) for Western blotting and quantitative RT-PCR, and these 10 per group were also used for heart function measurement and troponin analysis, and another 5 per group at 15-minute reperfusion (n = 5) for Western blotting of RISK pathway. Data with abnormal distribution were transformed before analysis (a log transformation for IS or square-root transformation for phosphorylation of AKt, cytosol-to-nuclear ratio of Nrf2, and HO-1 protein level). Data of cardiac function were analyzed by repeated-measures analysis of variance (ANOVA) followed by least significant difference (LSD) post hoc test. Other data were subjected to 1-way ANOVA followed by LSD post hoc test. P < .05 was considered statistically significant. All analyses were performed in SPSS (version 11.0).
Results
Cardiac Function
The hemodynamic parameters were similar in all groups at the baseline. These variables remained stable in the TC group. During the reperfusion, a markedly elevated LVEDP and dramatic decreases in RPP (the product of HR and LVDP) and ±dp/dt were observed in the I/R group (P < .05). The hearts in the DRIPC + SPoC group exhibited a more significant recovery in LVEDP, RPP, and ±dp/dt in comparison to SPoC or DRIPC group alone (P < .05). The recovery of cardiac function in SPoC or DRIPC group was prevented by the specific inhibitor LY294002 or ZnPP, respectively (P > .05 vs TC group; Table 1).
Hemodynamic Parameters in All the Experimental Groups.a

Abbreviations: TC, time control; I/R, ischemia reperfusion; SPoC, sevoflurane postconditioning; DRIPC, delayed remote ischemic preconditioning; LY, LY294002, a specific inhibitor of phosphatidylinositol-3-kinase (upstream of protein kinase B/AKt pathway); ZnPP, a specific inhibitor of heme oxygenase 1; DRIPC + SPoC, a combination of DRIPC and SPoC; LVEDP, left ventricular end-diastolic pressure; RPP, the product of heart rate and left ventricular developed pressure; ±dp/dt, maximum LVDP increase rate; −dp/dt, maximum LVDP decrease rate; SD, standard deviation.
a Data are presented as mean ± SD (n = 10).
b P < .05 vs baseline.
c P < .05 vs TC group.
d P < .05 vs I/R group.
e P < .05 vs DRIPC + SPoC group.
Infarct Size and Cardiac Enzyme Release
Sevoflurane postconditioning or DRIPC reduced the IS caused by I/R injury (16.50% ± 4.55% in the SPoC group and 10.22% ± 2.57% in the DRIPC group vs 35.00% ± 5.24% in the I/R group, P < .001, respectively). The IS-limiting effect was further enhanced while combining these 2 interventions (6.76% ± 2.18%, P < .001 vs SPoC group, P = .047 vs DRIPC group) whereas it was nearly abolished by LY294002 (29.31% ± 6.31%, P = .087 vs I/R group) or ZnPP (27.96% ± 2.81%, P = .042 vs I/R group), respectively (Figure 2A and B).
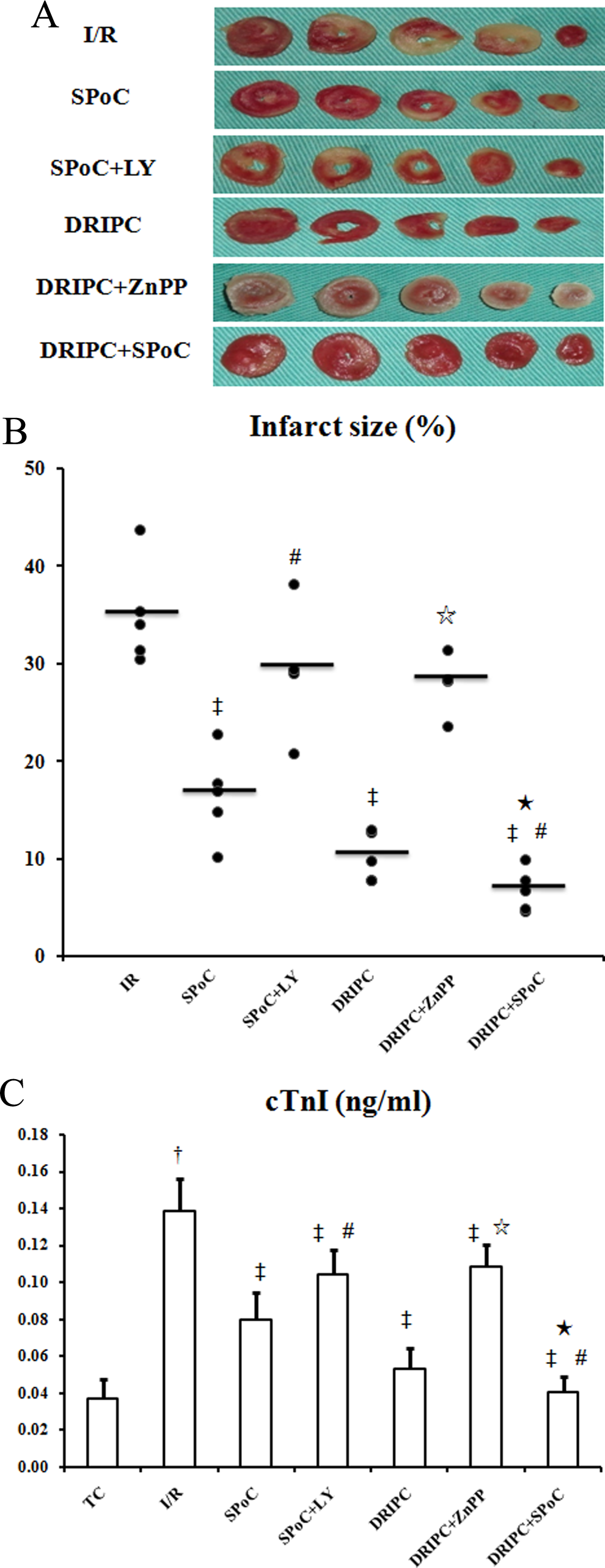
Myocardial injury in the experimental groups. A, Representative TTC staining of the hearts. B, Infarct size (n = 5) expressed as a percentage of total ventricular area. The transverse lines indicated the mean values. C, cTnI level (n = 10) in the coronary effluent. † P < .001 vs TC group, ‡ P < .001 vs I/R group, # P < .001 vs SPoC group, ⋆ P < .001 vs DRIPC group, ★ P < 0.05 vs DRIPC group. TTC indicates 2,3,5-triphenyltetrazolium chloride; cTnI, cardiac troponin I; I/R, ischemia/reperfusion; DRIPC, delayed remote ischemic preconditioning.
As a marker of myocardial enzymatic effect, the release of cTnI in the coronary effluent was largely increased in the I/R group whereas less release was observed both in the SPoC group and in the DRIPC group (P < .001 vs I/R group respectively). This release was further decreased in the DRIPC + SPoC group (P < .05; Figure 2C).
Phosphorylation of AKt and ERK1/2
The activation of RISK pathway including PKB/AKt and ERK1/2 was determined by Western blotting analysis. Sevoflurane postconditioning increased the AKt phosphorylation (P = .022 vs I/R group) with no effect on the ERK1/2 phosphorylation (P > .05 vs I/R group). This phosphorylation was inhibited by the specific PI3K inhibitor, LY294002 (P < .0001 vs SPoC group). Both DRIPC group and DRIPC + ZnPP group did not affect the RISK activation. Compared with the SPoC group, DRIPC + SPoC did not induce an enhanced phosphorylation of AKt or ERK1/2 (P > .05, respectively; Figure 3).

PKB/AKt, but not ERK1/2, was involved in the protection of SPoC and DRIPC + SPoC. A, Representative Western blotting of the phosphorylation of PKB/AKt and ERK1/2. B, Activation of PKB/AKt (normalized to T-AKt) was observed in the SPoC group and the DRIPC + SPoC group. C, No effect on ERK1/2 (normalized to T-ERK1/2) signal pathway was observed in each of all the groups. § P < .05 vs I/R group, ⋆ P < .001 vs DRIPC group, # P < .001 vs SPoC group. PKB indicates protein kinase B; ERK1/2, extracellular signal-regulated kinase 1 and 2; SPoC, sevoflurane postconditioning; DRIPC, delayed remote ischemic preconditioning; I/R, ischemia/reperfusion.
Expression of HO-1 and Nuclear Translocation of Nrf2
The HO-1 expression was largely increased in the DRIPC group (0.20 ± 0.04 vs 0.02 ± 0.006 in the I/R group, P < .0001) but not in the SPoC group (0.03 ± 0.02, P > .05). This increase was abolished by ZnPP (0.024 ± 0.015 vs 0.02 ± 0.006 in the I/R group, P > .05). Combination with DRIPC and SPoC presented an enhanced elevation in HO-1 expression (0.283 ± 0.067, P < .0001 vs SPoC group, P < .05 vs DRIPC group; Figure 4B). All these trends were further confirmed in the mRNA level (Figure 4C).

An enhanced cardioprotective effect of SPoC and DRIPC was partly mediated by the enhanced HO-1 level through Nrf2 translocation. A, Representative Western blotting of the HO-1 protein expression and Nrf2 translocation (C-Nrf2, cytosol Nrf2; N-Nrf2, nuclear Nrf2). B, Cardioprotection of DRIPC was abolished by a specific inhibitor of HO-1. A further expression of HO-1 was observed in the DRIPC + SPoC group (normalized to GAPDH). C, All these trends in HO-1 protein expression were further confirmed in the mRNA level (normalized to GAPDH). D, The increase in Nrf2 translocation was observed in both the SPoC group and the DRIPC group, which was further enhanced in the DRIPC + SPoC group (C-Nrf2 normalized to N-Nrf2). E, A positive correlation was observed between the HO-1 protein expression and the Nrf2 translocation. § P < .05 vs I/R group, ▵ P < .01 vs DRIPC group, # P < .001 vs SPoC group, ★ P < 0.05 vs DRIPC group. SPoC, sevoflurane postconditioning; DRIPC, delayed remote ischemic preconditioning; HO-1, heme oxygenase 1; Nrf2, nuclear factor erythroid 2-related factor 2; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
The mRNA regulation of HO-1 is partly mediated by the Nrf2/antioxidant response element (ARE) signal pathway. 39,40 Therefore, the nuclear translocation of Nrf2, expressed as the ratio of cytosol Nrf2 to nuclear Nrf2, was detected. The increase in Nrf2 translocation was of statistical significance in the SPoC group (1.31 ± 0.57 vs 0.56 ± 0.39 in the I/R group, P = 0.021), which was more obvious in the DRIPC group (3.00 ± 0.68, P = .002) . An enhanced increase was observed after the combination of SPoC and DRIPC than SPoC (P < .01) but not DRIPC (P > .05; Figure 4D). Moreover, the HO-1 expression was significantly positively correlated with the translocation of Nrf2 (Pearson correlation: r = 0.729, P < .001; Figure 4E).
Discussion
In the present study using Langendorff perfused isolated rat heart model, we found that: (1) activation of PKB/AKt, but not ERK1/2, was involved in the cardioprotection by SPoC; (2) HO-1 expression played an important role in the protection of DRIPC; (3) combination of DRIPC and SPoC could offer a superior cardioprotective potential than SPoC or DRIPC alone via an enhanced HO-1 level partly through the Nrf2 nuclear translocation.
There has always been a notion that cardioprotection by various conditioning protocols established in young and healthy animals could be translated in a negative way into the clinical population with various comorbidities (such as age 41,42 and gender 7 ) and/or cardiovascular medications (such as β-blockers 43 and statins 44 ). 45 Combination of 2 different conditioning protocols to obtain a superior cardioprotective effect may be an effective strategy. Several studies have been published with positive 18,19,46 –48 or negative 16,17 results since 2003. In our current study, both SPoC and DRIPC could offer protection from myocardial I/R injury. Moreover, we showed that DRIPC produced an additive effect to SPoC, as evidenced by significantly increased cardiac function recovery, reduced cTnI release, and in particular the limited IS.
Although the present data are unable to tell us the exact molecular mechanisms underlying the enhanced cardioprotective potential, we believe that the enhancement of HO-1 expression, but not RISK pathway, in reperfused myocardium may play an important role. Despite the controversy, 49 RISK pathway (PKB/AKt and/or ERK1/2) has been recognized as an important mechanism in the cardioprotective conditioning 50 –52 including SPoC. 21,53 Consistent with our previous finding, 28 this study showed that PKB/AKt pathway was involved in the cardioprotection by SPoC. Previous studies have reported the enhanced 18 or unchanged 19 RISK signal in the combination of remote ischemic perconditioning and ischemic postconditioning. In this study, a further phosphorylation in RISK pathway was not observed in the combined conditioning with an enhanced IS-limiting effect. In addition, we explored the potential role of HO-1 in this effect. As a heat shock protein protecting cell injury against excessive oxidative stress, HO-1 has been reported to be effective in protection against I/R injury of heart, 35,54 liver, 34 and brain. 55 In particular, involvement of HO-1 in delayed preconditioning with RIPC 34 or H2S 35 has been proposed. Accordingly, our study found that DRIPC mediated cardioprotection through the expression of HO-1. Moreover, the HO-1 level was further increased by adding DRIPC to SPoC, indicating its contributing role in the enhanced cardioprotection. Although HO-1 level was not increased in the SPoC group, a synergistic increase was shown in the DRIPC + SPoC group. This phenomenon suggests an underlying protective signal cross-talk between these 2 interventions, which needs further exploration in future studies.
The Nuclear factor erythroid 2-related factor 2/Antioxidant Response Element (Nrf2/ARE) signal pathway is a master regulator of the antioxidant response ubiquitously expressed in a wide range of tissues including myocardium. 39 This regulation is mediated by binding to a cis-acting regulatory element or enhancer sequence known as the ARE with a core nucleotide sequence of 5′-RTGACNNNGC-3′ to play the central role in the basal and inducible synthesis of antioxidant and phase II detoxifying enzymes, such as HO-1. 40 In the present study, for the first time, DRIPC was shown to induce cardioprotection by promoting the translocation of Nrf2 into the nucleus. This process was supported by the correlation analysis revealing a positive relationship between Nrf2 translocation and HO-1 protein expression. Such involvement of Nrf2 in cardioprotective effect was further suggested by the similar trend between the protein expression level and mRNA level of HO-1. However, this association should be verified using a specific inhibitor of Nrf2 or genetic knockout animal model in the future.
There are several limitations to our study. First, we used an isolated Langendorff perfused heart model to mimic the process of I/R injury, which could not reflect the potential role of neurohumor factors in the enhanced effect. Second, although we produced a superior protective strategy in young and healthy rat hearts, we still do not know whether it remains to be effective in aged or diabetic populations. Third, we did not focus on mitochondrial function, which has been recognized as the end effecter of various conditioning protocols. Fourth, we did not use the specific inhibitor-LY294002 or ZnPP in the combined conditioning group to explore the role of PKB/AKt or HO-1 in the enhanced cardioprotection, respectively. Fifth, we did not examine the role of Janus kinase/signal transducers and activators of transcription signal pathway in this combined conditioning. Sixth, 60-minute reperfusion may be too short for IS determination by TTC staining; however, Ferrera et al has confirmed that 60-minute reperfusion is sufficient to evaluate the cardiac function and IS in the Langendorff heart model. 56 Our previous studies also support this finding. 21,28,57 Finally, using inhibitors and dimethyl sulfoxide as controls, respectively, we have preliminarily found no influence in the IS (n = 3) as compared with the I/R group. However, we did not report this finding.
Conclusion
Using a Langendorff perfused rat heart model, the findings from this study suggests that the use of DRIPC produces an additive cardioprotection to SPoC through an enhanced HO-1 level partly via Nrf2 translocation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Specialized Research Fund for the Doctoral Program of Higher Education (Grant no. 20121106110013), the National Natural Science Foundation of China (Grant no. 81070098), and the Postgraduate Innovative Foundation of Peking Union Medical College (Grant no. 2011-1002-008).
