Abstract
The effectiveness of ischemic preconditioning (IPC) to protect the heart against ischemia/reperfusion injury (IRI) declines with age. The deacetylase protein sirtuin 1 (Sirt 1) confers myriad functions including longevity and cardioprotection against IRI. As such, Sirt 1 may be a potential candidate to explain the protective effect of IPC. We aim to explore the role of Sirt 1 in the loss of the cardioprotective effect of IPC with age. Isolated hearts from young (9 weeks) and older (12-18 months) Long-Evans rats were subjected to 30 minutes of global ischemia and 60 minutes of reperfusion. Preconditioning stimuli were applied with either 2 cycles of 5-minute ischemia/reperfusion or with the potent Sirt 1 agonist resveratrol (RSV, 10 µmol/L) for 15 minutes followed by a 10-minute washout before the sustained ischemia. Both IPC and RSV significantly enhanced the functional recovery of young hearts by 168% (P < .001 vs control) and 65% (P < .01 vs control), respectively, and concomitantly reduced the infarct size by 65% and 45%, but the effect was blunted in older hearts. Administration of the selective Sirt 1 inhibitor III to young hearts did not alter the protective effect of IPC. Following ischemia/reperfusion, higher Sirt 1 deacetylase activity was detected in older hearts compared to young hearts (0.48 ± 0.13 arbitrary units [AU] vs 0.17 ± 0.03 AU, P < .01) and IPC did not alter Sirt 1 deacetylase activity. In conclusion, although Sirt 1 deacetylase activity is increased with age during ischemia/reperfusion, our data suggest that the loss of the cardioprotective effect of IPC in older animals is likely to be independent of Sirt 1.
Introduction
Age progression induces significant biological and morphological changes in the heart that can give rise to diminished contractile function and cardiovascular complications. 1 Innate mechanisms that protect young hearts from damage due to myocardial infarction are compromised in aging hearts. Of particular importance is the efficacy of ischemic preconditioning (IPC), 2 a well-established strategy that limits myocardial injury by exposure to sequences of brief ischemia/reperfusion (I/R) episodes prior to the main ischemic event, declines during aging. 3 Discovering endogenous factors and/or cellular signaling cascades that can restore the cardioprotective processes impaired with aging may therefore be vital for clinical therapy.
Sirtuin 1 (silent information regulator [Sirt 1]) is a widely studied class III histone deacetylase that is naturally expressed in the human body and relies on intracellular sources of nicotinamide adenine dinucleotide (NAD+) to promote its pleiotropic actions, including deceleration of the aging process. The upregulation of Sirt 1 by reduced nutrient intake and pharmacological sirtuin agonists results in prolonged survival of lower life forms. 4 –7 In mammals, the polyphenol resveratrol (RSV), known to mimic IPC and protect the heart from ischemia/reperfusion injury (IRI), 8 –10 was suggested to enhance longevity of mice on a calorie-rich dietary regimen via Sirt 1 activation. 11 In contrast, surviving mice with whole-body Sirt 1 deficiency display an altered phenotype with deformities occurring during development in the heart as well as other organs. 12 The functions of Sirt 1 under physiological conditions also extend to gene silencing, DNA repair, and metabolism. Furthermore, Sirt 1 prevents apoptosis and raises the tolerance to oxidative stress in cardiomyocytes. 13 Cardiac hypertrophy, fibrosis, apoptosis, and ventricular dysfunction observed with aging are abrogated in mouse strains that overexpress the Sirt 1 gene at a low-to-moderate (2.5- to 7.5-fold) degree above baseline in the heart. 14 On the other hand, a high magnitude (12.5-fold) of cardiac-restricted Sirt 1 overexpression counters these protective effects and promotes cardiomyopathy. 14 These hormetic effects thus indicate that the ability of Sirt 1 to positively impact the heart is dosage dependent.
There is however limited information about the relevance of Sirt 1 in IRI and IPC with advanced age. Interestingly, a 7.5-fold increase in cardiac-specific Sirt 1 gene expression confers protective effects against IRI, by reducing infarct size and ameliorating functional recovery of mouse hearts. 15 Nadtochiy et al noted a lack of IPC-mediated cardioprotection in mice with attenuated Sirt 1 gene expression. 16 Since animal experiments provide evidence that attenuation of the beneficial features evoked by IPC in rat hearts occurs from middle age (12 months), 17 , 18 we therefore investigated whether Sirt 1 is required for the cardioprotective actions of IPC, and whether loss of IPC’s favorable effects with age is attributed to changes in Sirt 1 deacetylase activity. This was carried out using the isolated rat heart model (9 weeks old vs 12-18 months old animals).
Methods and Materials
Animal Experiments
Animal experiments were conducted on male Long-Evans rats at the ages of 9 weeks (young) and 12 to 18 months (older). All procedures were implemented in accordance with the Guide of the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, Revised 1996). The Faculty of Health Sciences Animal Ethics Committee of the University of Cape Town approved all the experimental protocols used in this study. A total of 265 animals were used for this study.
Langendorff-Perfused Rat Hearts
Rats were anesthetized with sodium pentobarbitone (60 mg/kg) by intraperitoneal injection and received the anticoagulant, heparin (200 IU). Hearts were subsequently harvested for retrograde perfusion on the Langendorff system at a constant pressure of 100 cm H2O with oxygenated (95% O2, 5% CO2, 37°C) Krebs-Henseleit buffer (pH 7.35-7.45), as previously described. 19 Hemodynamic measurements for heart rate, coronary flow, left ventricular end systolic pressure (LVESP), left ventricular end diastolic pressure (LVEDP), and left ventricular developed pressure (LVDP) were assessed during the protocols. This was achieved by means of a water-filled balloon positioned in the LV that was inflated to settings of 0 to 12 mm Hg for LVEDP and coupled to a pressure transducer. Exclusion criteria for the Langendorff-perfusion model were as previously described. 20
The experimental protocols are depicted in Figure 1. All hearts were equilibrated for 30 minutes before undergoing treatments. Control hearts were subjected to 30 minutes of global ischemia and 60 minutes of reperfusion (I/R). Preceding global ischemia, interchanging cycles of I/R for 2 × 5 minutes followed by a 10-minute washout period was applied to induce IPC. The sirtuin agonist RSV (10 µmol/L, Sigma-Aldrich, Germany) was given for 15 minutes followed by a 10-minute washout period before sustained global ischemia to promote pharmacological preconditioning. To explore whether Sirt 1 is required for preconditioning, 2 sirtuin antagonists, that is nicotinamide (NAM, 50 µmol/L, Sigma-Aldrich, Germany) or Sirt 1 inhibitor III (SIII, 100 nmol/L, Merck, Germany) were administered during the preconditioning stimulus. Additional groups consisted of hearts pretreated for 23 minutes with either NAM or SIII alone, a 5-minute washout period, and then global I/R. For perfusion of the pharmacological compounds through the heart, stock solutions were prepared in dimethylsulfoxide (DMSO) and then diluted in Krebs-Henseleit buffer. At the end of reperfusion, quantification of infarct size was carried out by 2,3,5-triphenyltetrazolium chloride staining (Sigma-Aldrich, Germany) and by using the Planimetry + computer software program.
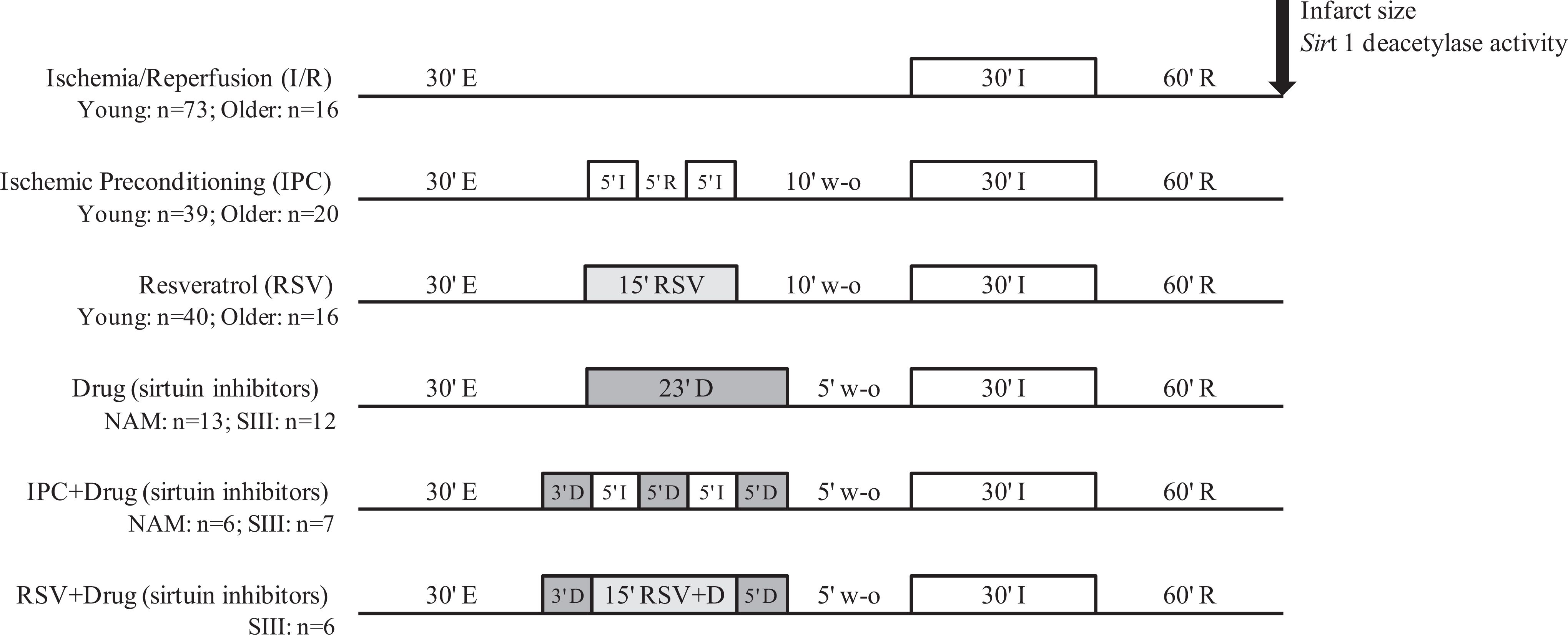
Schematic representation of Langendorff retrograde perfusion protocols. E indicates equilibration; I/R, ischemia/reperfusion; IPC, ischemic preconditioning; RSV, resveratrol (10 µmol/L); D, drugs (nicotinamide 50 µmol/L; or Sirt 1 inhibitor III 100 nmol/L); w-o, washout; n, total number of animals used for each treatment group.
Sirt 1 Deacetylase Activity
In a separate set of additional experiments, heart tissues were collected at the end of Langendorff perfusion protocols, freeze clamped in liquid nitrogen, and purification of nuclear proteins was prepared at 4°C. Approximately 100 mg of heart tissue was homogenized in lysis buffer (10 mmol/L Tris-HCl pH 7.5, 10 mmol/L sodium chloride, 15 mmol/L magnesium chloride, 250 mmol/L sucrose, 0.5% nonidet P-40, 0.1 mmol/L EGTA), briefly vortexed, and incubated on ice for 15 minutes. Centrifugation of the homogenate at 1300g for 10 minutes was initiated through a sucrose cushion (30% sucrose, 10 mmol/L Tris-HCl pH 7.5, 10 mmol/L sodium chloride, 3 mmol/L magnesium chloride). The supernatant was disposed of and the pellet containing the nuclei was washed with 10 mmol/L Tris-HCl pH 7.5 and 10 mmol/L sodium chloride. The nuclei were then suspended in extraction buffer (50 mmol/L HEPES KOH pH 7.5, 420 mmol/L sodium chloride, 0.5 mmol/L EDTA, 0.1 mmol/L EGTA, 10% glycerol) and subjected to sonication for 30 seconds. After 30 minutes on ice, the nuclei were then centrifuged at 13 000 rpm for 10 minutes. Crude nuclear proteins were obtained from the resulting supernatant. Protein concentrations were determined from heart samples with the conventional Lowry method. 21
Sirt 1 deacetylase activity was detected in protein extracts using the CycLex Sirt 1/Sir 2 Fluorometric assay kit (MBL International, Woburn, Massachusetts), as reported in the manufacturer’s manual. Fluorescence was recorded at an excitation wavelength of 340 nm and emission wavelength of 440 nm every 2 minutes for a duration of 60 minutes. Sirt 1 deacetylase activity expressed in arbitrary units (AU) was calculated from the difference between the fluorescent signal of the experimental samples and no enzyme control, and normalized to the protein concentration.
Statistical Analysis
The results are presented as the mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) and Tukey post hoc test was used for comparison of multiple treatments in the same age group. Differences between age and various treatments were analyzed with the 2-way ANOVA and Bonferroni posttest. A P < .05 indicated that the values reached statistical significance.
Results
Cardioprotective Effects of Preconditioning is Reduced With Age
The effects of age were tested on rat hearts preconditioned with ischemia or pharmacologically with the sirtuin agonist, RSV, prior to undergoing global I/R on the Langendorff system. Functional parameters depicted in Table 1 revealed that heart rates were lower in older animals than in young animals (P < .001). Young hearts displayed improved functional recovery following IPC (47.2% ± 4.7%; P < .001) and RSV pretreatment (29.0% ± 1.9%; P < .01) compared with I/R (17.6% ± 1.3%; Figure 2A). In parallel, infarct size decreased from 51.0% ± 4.2% for I/R to 17.6% ± 5.6% for IPC (P < .001) and 28.2% ± 3.1% for RSV (P < .01; Figure 2B). However, these significant cardioprotective effects were not observed in older hearts (P = ns vs I/R of older hearts).
Hemodynamic Measurements of Langendorff Perfused Rat Heartsa
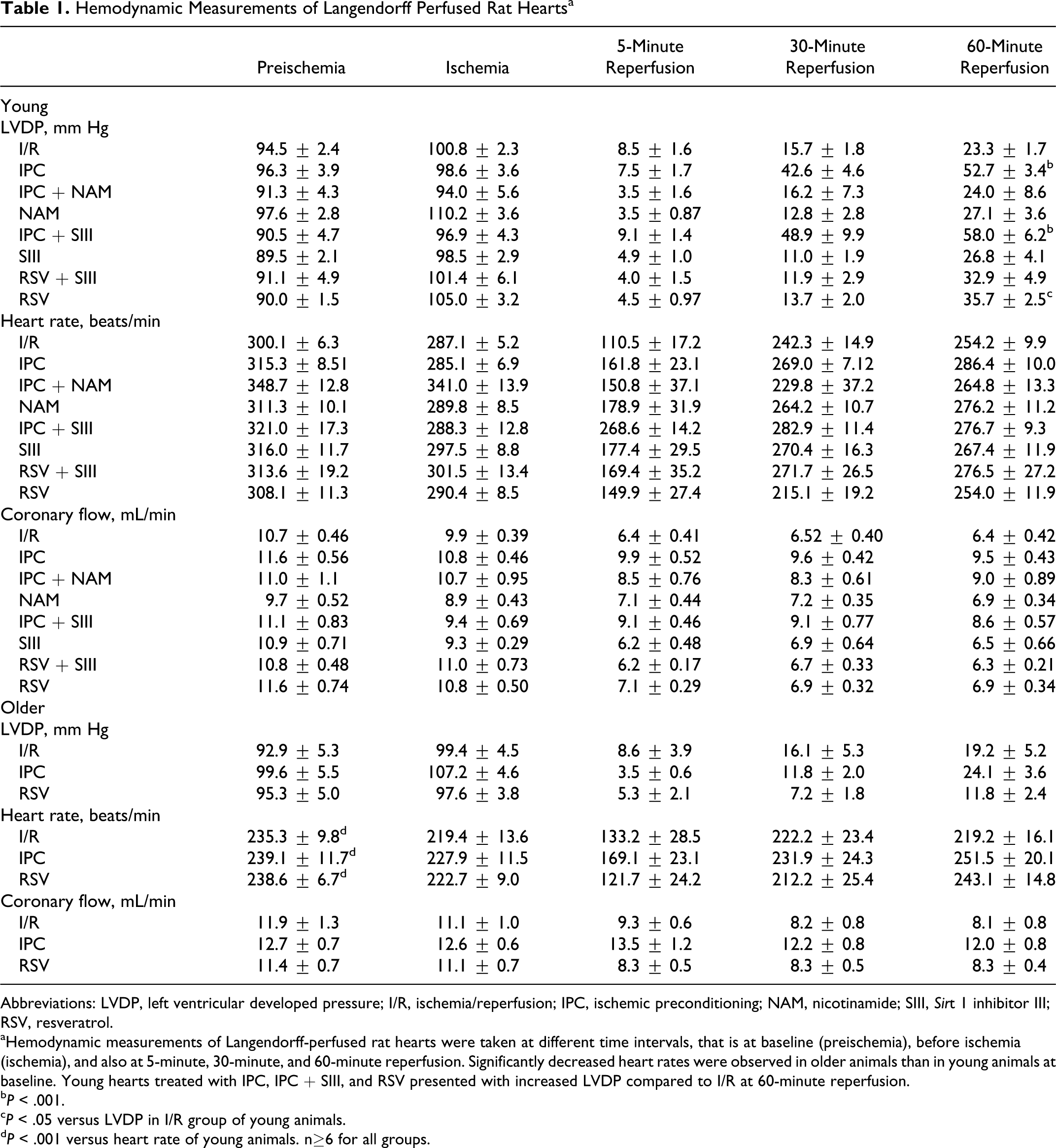
Abbreviations: LVDP, left ventricular developed pressure; I/R, ischemia/reperfusion; IPC, ischemic preconditioning; NAM, nicotinamide; SIII, Sirt 1 inhibitor III; RSV, resveratrol.
aHemodynamic measurements of Langendorff-perfused rat hearts were taken at different time intervals, that is at baseline (preischemia), before ischemia (ischemia), and also at 5-minute, 30-minute, and 60-minute reperfusion. Significantly decreased heart rates were observed in older animals than in young animals at baseline. Young hearts treated with IPC, IPC + SIII, and RSV presented with increased LVDP compared to I/R at 60-minute reperfusion. b P < .001.
c P < .05 versus LVDP in I/R group of young animals.
d P < .001 versus heart rate of young animals. n≥6 for all groups.
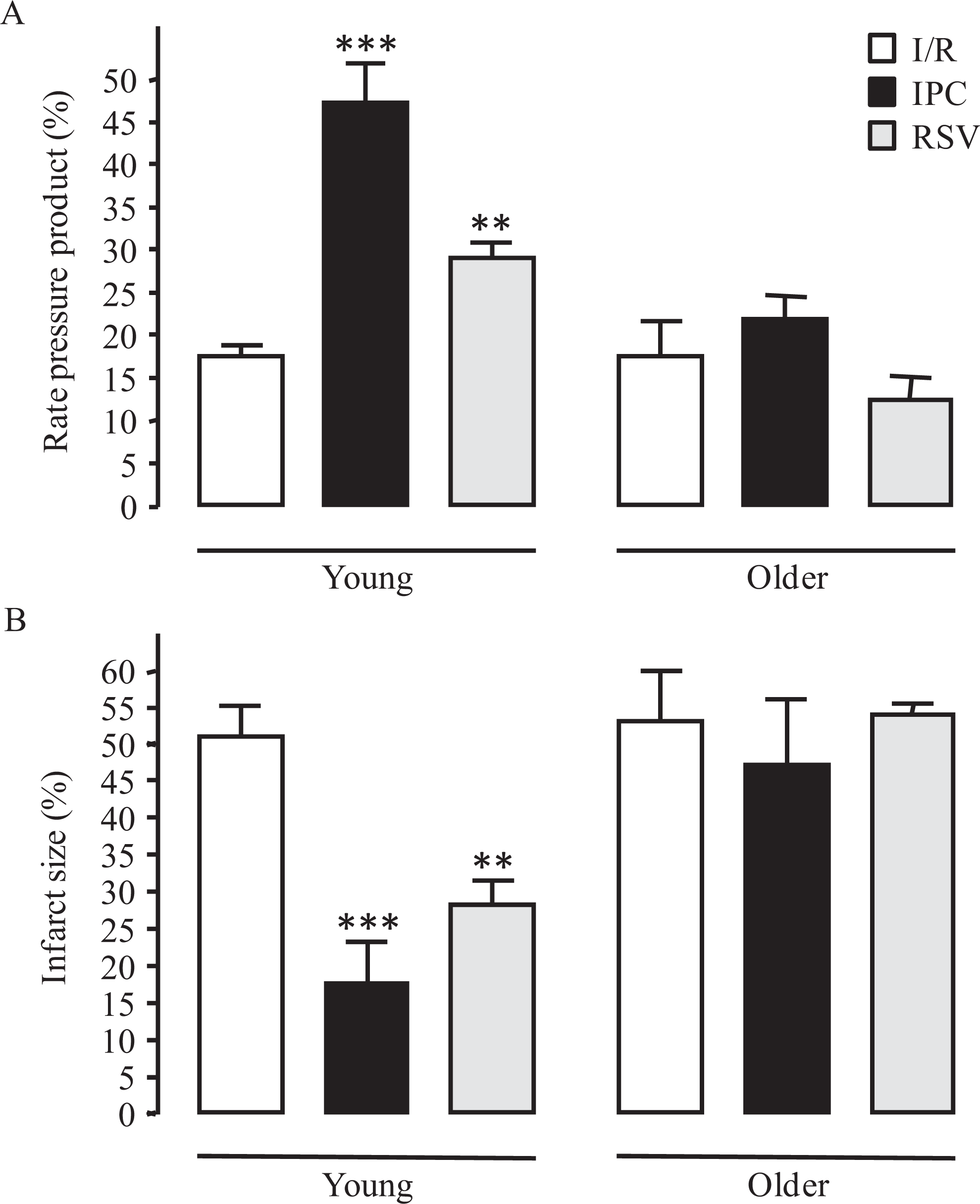
Effects of preconditioning and age on myocardial I/R injury. Improved functional recovery (A) (expressed as percentage rate pressure product) and reduced infarct size (B) in young hearts preconditioned with ischemia (IPC) or RSV (10 µmol/L) prior to undergoing I/R on the Langendorff system was attenuated in older hearts. I/R indicates ischemia/reperfusion; IPC, ischemic preconditioning; RSV, resveratrol. ***P < .001 and **P < .01 versus I/R of young hearts, n = 4 to 17 per group.
The IPC- and RSV-Mediated Protection Against IRI is Not Prevented by Sirtuin Inhibition in Langendorff Rat Hearts
To establish whether Sirt 1 plays a role in preconditioning, rat hearts were perfused with the nonspecific sirtuin inhibitor, NAM, or the selective Sirt 1 antagonist, SIII (Figure 3). Here, NAM markedly reduced the functional recovery of IPC (16.6% ± 5.5% for IPC + NAM vs 49.9% ± 3.1% for IPC; P < .001) but failed to reduce the protective effect of IPC toward infarct size (32.3% ± 12.1% for IPC + NAM vs 27.8% ± 4.1% for IPC; P = ns). In contrast, the addition of SIII during IPC or RSV pretreatment was not associated with changes in functional recovery (57.7% ± 5.7% for IPC + SIII, 31.6% ± 6.7% for RSV + SIII; P = ns) and infarct size (23.6 ± 3.2% for IPC + SIII, 24.6 ± 2.7% for RSV + SIII; p = ns). Furthermore, administration of NAM and SIII alone presented a functional recovery of 26.3% ± 4.5% and 23.4% ± 3.8%, with an infarct size of 36.2% ± 7.2% and 43.9% ± 6.9%, respectively, which were similar to I/R group.

Effects of sirtuin inhibition on IPC- and RSV-mediated cardioprotection in young animals. Nonspecific inhibition of sirtuins with NAM (50 µmol/L) during IPC markedly decreased functional recovery (A) but failed to increase infarct size (B). Both functional recovery and infarct size were preserved for IPC or RSV (10 µmol/L) hearts treated with the selective Sirt 1 antagonist, SIII (100 nmol/L). I/R indicates ischemia/reperfusion; IPC, ischemic preconditioning; NAM, nicotinamide; SIII, Sirt 1 inhibitor III; RSV, resveratrol. ***P < .001 and *P < .05 vs I/R. ### P < .001 versus IPC, n = 6 to 36 per group.
Sirt 1 Deacetylase Activity is Upregulated by I/R in Older Rat Hearts But Unchanged by IPC and RSV in Both Young and Older Rat Hearts
As shown in Figure 4, Sirt 1 deacetylase activity was evaluated in nuclear extracts of treated hearts after 1 hour of reperfusion. Interestingly, higher Sirt 1 deacetylase activity levels were obtained for I/R in older hearts (0.48 ± 0.13 AU) than in young hearts (0.17 ± 0.03 AU; P < .01). Neither an IPC stimulus nor an RSV pretreatment for both age groups altered Sirt 1 deacetylase activity. Although there was a trend for RSV to increase Sirt 1 deacetylase activity of young hearts, the values did not reach statistical significance. Sirt 1 deacetylase activity values for IPC and RSV, respectively, were 0.12 ± 0.04 AU and 0.68 ± 0.41 AU for young hearts and 0.17 ± 0.02 AU and 0.17 ± 0.01 AU for older hearts (P = ns vs I/R).

Sirt 1 deacetylase activity measured at the end of reperfusion in nuclear extracts of heart tissue was significantly higher for I/R in older animals compared to young animals. In contrast, IPC or pretreatment with RSV (10 µmol/L) did not alter Sirt 1 deacetylase activity for both age groups. NC indicates normoxic control; I/R, ischemia/reperfusion; IPC, ischemic preconditioning; RSV (10 µmol/L), resveratrol; NAM (50 µmol/L), nicotinamide; SIII (100 nmol/L), Sirt 1 inhibitor III; AU, arbitrary units. *P < .05 versus I/R of young hearts, n = 3 to 20 per group.
Discussion
This study revealed that the effects of preconditioning with ischemia (IPC) or RSV on IRI were age dependent, with cardioprotection evident in young animals but lacking in older animals. Preconditioned young hearts exhibited no changes in response to selective Sirt 1 inhibition. Also, Sirt 1 deacetylase activity was enhanced by I/R with aging, whereas it was unaffected by IPC and RSV in both young and older hearts.
With aging, there is a reduced capacity to preserve and salvage the heart from injury induced by I/R. 22 As a result, the aging heart becomes less resilient to cardioprotective approaches such as IPC. Using the Langendorff retrograde perfusion model, our data show that older rat hearts displayed decreased heart rates, a functional feature of aging hearts. 23 Furthermore, the potential of IPC and RSV, a pharmacological mimetic of IPC, 9 , 10 in minimizing IRI in young hearts was attenuated in older hearts. This is consistent with the earlier studies demonstrating that increased susceptibility to myocardial damage triggered by I/R is detectable from the age of 12 to 13 months and even more apparent at the age of 18 to 24 months in rodents. 17 , 24 However, Schulman et al found that increasing the IPC potency from 1 cycle to 3 cycles of 5 minutes of I/R maintained the cardioprotection conferred by IPC in the 12-month-old rat hearts yet remained ineffective under the same conditions in the 18- to 20-month-old rat hearts. 25 Therefore, the intensity of the IPC stimulus should be carefully considered when examining age-related effects. The older animals used in our study were 12 to 18 months old and IPC was elicited with 2 cycles of 5 minutes of I/R. The application of a stronger IPC stimulus may recover the protection, and this requires further investigation. A plethora of mechanisms is suggested to account for the attenuation of preconditioning-mediated cardioprotection with aging. Mechanisms such as impaired mitochondrial function, downregulated gene and protein expression of extracellular ligands and receptors, and alterations of signaling molecules involved in the reperfusion injury salvage kinase (RISK) pathway and survivor activating factor enhancement (SAFE) pathway, influence the survival of aging hearts. 1
Since Sirt 1 exerts antiaging properties and it has recently been implicated in cardioprotection, 26 , 27 this key factor was employed in this study to determine whether it contributes to IPC-induced cardioprotection and whether activation of Sirt 1 with RSV would restore cardioprotection in aging hearts. In perfused hearts, we found that sirtuin inhibition with NAM during IPC significantly reduced functional recovery, whereas infarct size was unaltered. Even though NAM acts nonspecifically, it is relevant as an endogenous compound commonly used as a Sirt 1 inhibitor in both in vivo and in vitro experiments. 28 –31 The concentration of NAM administered to the hearts was at the IC50 for sirtuins and exceeds the levels (∼50 times) naturally present in mammalian tissues. 32 The NAM is documented to impede nitric oxide production and vasodilation by preventing deacetylation of the Sirt 1 targeting endothelial nitric oxide synthase and this role of NAM may possibly explain the result obtained for functional recovery. 33 Because of the nonspecificity of NAM, experiments were also undertaken with the selective Sirt 1 antagonist, SIII, in order to clarify whether Sirt 1 is involved in IPC. The SIII is a stable compound with minimal toxicity that inhibits Sirt 1 activity at low concentrations and with an efficacy that is strikingly higher than NAM (∼1000-fold) as well as other compounds. 34 The IC50 of SIII was used in our experiments and this dose conformed to previous literature. 34 , 35 Our results show that SIII did not reverse the improvement in functional recovery or the infarct-sparing effect of hearts preconditioned with ischemia or RSV. This data conflicts with a recent study conducted in murine hearts, in which the Sirt 1 and Sirt 2 inhibitor, splitomycin abolished the favorable effects of IPC. 36 However, the same group also showed that cardioprotection against IRI was not achieved by pharmacologically stimulating Sirt 1. A subsequent publication revealed a lack of protection in the hearts of heterozygous Sirt 1+/− mice subjected to IPC. 16 Furthermore, Hsu et al reported that sensitivity to myocardial injury caused by I/R was decreased in mice with forced expression of the Sirt 1 gene in the heart. 15 It has been proposed that while Sirt 1 alone is not adequate to emulate IPC, it does contribute to the cardioprotective effect of IPC. The inconsistencies between these studies may therefore be due, in part, to variations in the animal models (rat vs mouse) and/or inhibitor specific. The use of pharmacological inhibitors is a limitation of our investigation and the conclusion of this study needs to be taken with caution.
In the subcellular milieu, Sirt 1 resides in different locations according to the physiological setting or stress signal. During development, Sirt 1 expression is confined to the nucleus of the fetal heart and distributed in both the nucleus and the cytoplasm of the adult heart. 37 Sirt 1 is reported to mobilize to the nucleus of adult cardiomyocytes in response to stressors such as myocardial infarction and dilated cardiomyopathy. 27 , 38 , 39 In the present study, Sirt 1 deacetylase activity was therefore measured in nuclear extracts and was higher in older hearts than young hearts following exposure to I/R. This correlates with the earlier work by Alcendor et al where the Sirt 1 level in the heart was enhanced by pathologic stimuli, namely left ventricular hypertrophy and heart failure, pressure overload, and oxidative stress. 13 , 14 This research group also found that characteristic changes with age like increases in cardiac hypertrophy, fibrosis, apoptosis, and ventricular dysfunction were abrogated in genetically altered mice with a modest degree of cardiac-restricted Sirt 1 overexpression. Conversely, Sirt 1 that was expressed at a high level exclusively in the heart opposed these cardioprotective effects. The authors suggested that aging and apoptosis in the myocardium may be offset by a moderate Sirt 1 expression. However, Sirt 1 gene dose-dependent cardioprotection is paradoxical in light of the evidence by Kawashima et al 40 demonstrating a decline in cardiac diastolic function, derangements in mitochondrial morphology, and impaired mitochondrial function in mouse hearts with a 6.8-fold upregulation of Sirt 1 expression. Furthermore, 2 publications reported a detrimental effect of low Sirt 1 overexpression on cardiac function following exposure to pressure overload. 40 , 41 It has been suggested that a favorable function for Sirt 1 on the heart may hinge on the severity and type of pathological condition. 42 Intriguingly, IPC had no effect on Sirt 1 deacetylase activity as predicted, while RSV tended to increase Sirt 1 deacetylase activity but the results did not reach statistical significance. The ability of RSV to directly stimulate Sirt 1 has been questioned due to controversies associated with the fluorometric deacetylase assay method, despite numerous studies showing that RSV can enhance Sirt 1 deacetylase activity. 43 –47 In addition, Raval and colleagues showed that protection stimulated by IPC and RSV was accompanied with a timely Sirt 1 activation in neuronal cells. 48 The RSV exhibited a transient effect and Sirt 1 deacetylase activity increased at 30 minutes of a 1-hour treatment and reached basal levels at 48 hours of reperfusion before the onset of ischemia, but Sirt 1 activation was only detected at 48 hours after IPC. This time frame represents second window of preconditioning, a delayed stage of cardioprotection that is attained after 24 to 72 hours. Our study typifies the first window of preconditioning, an acute stage of cardioprotection, which arises during 1 to 3 hours of reperfusion, and it is likely that gene activation of Sirt 1 does not have time to occur in our isolated heart model. Different time points or altering the reperfusion time may be needed to detect a possible effect of IPC on Sirt 1 deacetylase activity in our model and this may give different results.
In conclusion, our results suggest that loss of IPC-mediated cardioprotection with aging occurs independently of Sirt 1 activation. Given that the overall evidence argues against the use of pharmacological inhibitors and that genetically modified animals also show conflicting results for Sirt 1 on the heart, further work is required to obtain a definitive link between Sirt 1 and IPC with age.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Support for this work was granted by the University of Cape Town, the Medical Research Council of South Africa, and the National Research Foundation.
