Abstract
Objective:
Acute lung injury is a common complication after cardiopulmonary bypass (CPB). Oxidative damage greatly impacts CPB-induced lung ischemic pathogenesis and may represent a target for treatment. We aimed to investigate whether curcumin upregulates heme oxygenase 1 (HO-1) expression and ameliorates lung injury in a rat CPB model.
Methods:
A total of 80 male Sprague-Dawley rats were divided into 2 sets of 5 groups (n = 8 per group): sham; control (CPB); vehicle; low-dose curcumin (L-Cur); and high-dose curcumin (H-Cur). Animals were pretreated with a single intraperitoneal injection of vehicle, L-Cur (50 mg/kg), or H-Cur (200 mg/kg) 2 hours prior to CPB. Lung tissue, serum, and bronchoalveolar lavage fluid was harvested 2 or 24 hours postoperatively. In the control group, CPB-induced lung injury was confirmed by histopathologic examination and a significantly increased wet-to-dry lung weight ratio and pulmonary permeability index value was observed (P < .05 vs sham group). Cardiopulmonary bypass was associated with a marked rise in the level of malondialdehyde and myeloperoxidase and a fall in superoxide dismutase 2 and 24 hours after surgery (P < .05 vs sham group). Administration of curcumin ameliorated lung damage and reversed the oxidative stress markers in a partially dose-dependent manner (P < .05 vs vehicle group). Furthermore, HO-1 gene transcription and protein expression were elevated to a greater extent in the lungs after curcumin pretreatment compared with the vehicle pretreatment.
Conclusions:
Curcumin has the potential to provide protection from CPB-induced lung damage reflected in the expression of oxidative stress markers. The antioxidant effect of curcumin may be partly related to upregulation of HO-1.
Introduction
Cardiac surgery with cardiopulmonary bypass (CPB) has been associated with an incidence of acute lung injury. Studies have shown that the etiology of lung injury is multifactorial, including pulmonary hypoperfusion, the induction of inflammatory mediators, hypothermia, and blood contact with the foreign surfaces of the CPB system. 1 These factors may collectively contribute to pulmonary hypoxic–ischemic and systemic inflammatory responses, which may result in generous formation of reactive oxygen species. Increasing evidence indicates that in the case of CPB, a large number of oxygen free radicals are generated that exceed the oxidant scavenging capacity of the endogenous antioxidant enzymes and thus cause cellular injury. 2 In response to these challenges, several stress proteins and antioxidant enzymes are activated to limit the damage at the cellular level. Recently it has been increasingly recognized that heme oxygenase 1 (HO-1) plays an important role in removing harmful free radicals. 3 A cytoprotective role for HO-1 has been supported by the accumulating data in various pulmonary disease models, including lung hypoxic–ischemic injury. 4 Our earlier research indicated that after CPB there is an increase in HO-1 protein in kidney tissue. 5 In view of these findings, it seems reasonable to extrapolate that the HO-1 system is related to the pathophysiologic progress of the lung after CPB.
Curcumin, a polyphenol found in the plant curcumin longa, has been shown to fortify the antioxidant system by inhibiting lipid peroxidation, scavenging superoxide and hydroxyl radicals, attenuating the reactivity of oxygen free radical species, and enhancing the activities of detoxifying enzymes. However, it remains unknown whether pretreatment with curcumin in a rat CPB model elevates the HO-1 protein level and alleviates lung injury. Emerging evidence suggests that curcumin upregulates HO-1 expression and protects rat brains against focal ischemia. 6 More importantly, an in vitro study demonstrated that curcumin boosts antioxidant defenses by increasing HO-1 levels in primary lung endothelial cells. 7 We hypothesize that administration of curcumin in a rat CPB model could upregulate the HO-1 protein level in lung tissues and subsequently attenuate oxidative injury.
Therefore, the aim of this study was to determine the potential effect of curcumin on the expression of oxidative stress markers and the impact on the activation of the HO-1 protein. As we all know, malondialdehyde (MDA) and myeloperoxidase (MPO) are markers of oxidative stress, and superoxide dismutase (SOD) is an antioxidant enzyme. Thus, we investigated the concentration of MDA, MPO, and SOD in the lungs. The effect of curcumin on the expression level of intrapulmonary HO-1 was examined.
Materials and Methods
Animals and Groups
Eighty male Sprague-Dawley rats weighing between 450 and 500 g were used in the investigation. All animals received humane care in compliance with “The Guide for the Care and Use of Laboratory Animals.” The experimental protocol was approved by the Committee of Animal Care of Nanjing University.
We designed 2 experimental sets according to previous studies 8 and our preliminary tests at the following time points: 2 and 24 hours after the termination of CPB. In each set, the rats were further subdivided randomly into 5 experimental groups (n = 8 per group): animals undergoing a sham CPB (sham group); animals undergoing CPB (control group); or animals undergoing CPB and pretreated with vehicle (vehicle group); pretreated with curcumin at 50 mg/kg (low-dose curcumin [L-Cur] group); or pretreated with curcumin at 200 mg/kg (high-dose curcumin [H-Cur] group). Curcumin (Sigma, St Louis, Missouri) dissolved in dimethyl sulfoxide was administered intraperitoneally 2 hours prior to CPB. The dose of curcumin used in the present study was based on previous experiments. 8 Animals received an additional dose of curcumin 12 hours postoperatively.
Surgical Procedures
The rat model of CPB was built as previously described in our laboratory with some modifications.
5
Briefly, rats were anesthetized with 5% isoflurane in 50% O2 in a plastic box. During subsequent surgical procedure, anesthesia was maintained with 1.5% and 2% isoflurane. After orotracheal intubation with a 14-gauge catheter, the animals were mechanically ventilated (Rodent respirator, DH-150, China) with a fraction of inspired oxygen (F
Specimen Collection
The animals were sacrificed by exsanguination 2 or 24 hours post-CPB. After the right main bronchus was clamped, the left main bronchus was cannulated, and the left lung was lavaged 4 times with 0.5 mL of Hanks balanced salt solution. Bronchoalveolar lavage fluid (BALF) was centrifuged at 1500 rpm for 10 minutes, and the supernatant was then stored at −80°C until analyzed. The right lung was used to determine the wet-to-dry (W/D) lung weight ratio and for histologic examination and biochemical analysis.
Assay of W/D Lung Weight Ratio and Pulmonary Permeability Index
The W/D lung weight ratio and pulmonary permeability index (PPI) were measured as previously described. 9 The W/D lung weight ratio served as an index of lung edema. The right upper lobe was dissected and weighed as “wet” weight, and then placed in an electric air blast drier at 70°C for 24 hours to obtain the “dry” weight. The PPI, as an indicator of pulmonary vascular permeability, was calculated as follows: BALF protein/plasma protein. The protein concentration of the blood and BAL were detected by Coomassie brilliant method according to the manufacturer’s instructions (Jiancheng Crop, Nanjing, China).
Light Microscope Examination
Portions of the middle lung lobes were immersed in 10% neutral-buffered formalin and processed routinely by embedding in paraffin. Tissue sections (4 mm thick) were stained with hematoxylin–eosin and examined under light microscopy. Criteria to evaluate the degree of lung injury included interstitial edema, hemorrhage, neutrophil infiltration, hyaline membrane formation, necrosis, and congestion. Each criterion was scored on a scale of 0 to 4, that is, 0 = normal; 1 = minimal (<25%); 2 = mild (25%-50%); 3 = moderate (50%-75%); and 4 = severe abnormalities (>75%). An experienced pathologist blinded to the treatment conditions made all histologic assessments.
Assay of Oxidative Stress Markers
The right lower lobe was used to determine pulmonary tissue MPO, MDA, and SOD levels. The homogenized samples were prepared as previously described. 10 Protein measurements were analyzed according to the method documented in previous studies. 11 The MPO activity was determined by the method of Knight and colleagues 12 and expressed as U/g protein. The levels of MDA in tissue homogenate were measured according to the method of Mihara and Uchiyama, 13 and values were expressed as nmol/mg protein. Total (Cu-Zn and Mn) SOD activity was determined by the method of Sun and coworkers 14 and expressed as U/mg protein.
Western Blot of HO-1
For Western blot analysis, the frozen lung tissues were homogenized in 200 μL of lysis buffer solution containing 20 mmol/L Tris (pH 7.5), 150 mmol/L NaCl, 1 mmol/L EDTA, 0.1% sodium dodecyl sulfate (SDS), 1% Nonidet P-40, and 0.5% sodium deoxycholate. The homogenates were centrifuged at 12 000g at 4°C for 20 minutes. An aliquot of the supernatant was used to determine the protein level. Protein aliquots were mixed with sample buffer, separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis in a 10% gel, and transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% nonfat dry milk for 1 hour at 37°C and then incubated with primary anti-HO-1 antibody and monoclonal anti-actin (Santa Cruz Biotechnology, Santa Cruz, California) overnight at 4°C. After washing with Tris Buffer Solution Tween (TBST), horseradish peroxidase–conjugated goat anti-rabbit secondary antibody at a 1:5000 dilution was added to the membranes and incubated for 1 hour at 37°C. The protein bands were detected by a chemiluminescent system (Renaissance, NEN Life Science, Zaventem, Belgium) and quantified by scanning densitometry using a GS-710 Imaging Densitometer (Bio-Rad, Hercules, California).
Expression of HO-1 Messenger RNA by Reverse Transcriptase–Polymerase Chain Reaction
Total RNA was extracted from lung tissue using TRIzol reagent with the manufacture’s protocol (Invitrogen, Karlsruhe, Germany). Reverse Transcriptase–Polymerase Chain Reaction (RT-PCR) system was adopted to amplify the HO-1 messenger RNA (mRNA). The primer sequences of HO-1 were as follows: forward: GGCGTCCATGTTGACTGA and reverse: AAGACAGCCCTACTTGGTTAG, with an amplification fragment of 183 bp. The PCR cycling program comprised an initial denaturation step at 95°C for 10 minutes, followed by 32 cycles of denaturation at 95°C for 20 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 30 seconds. After the last cycle, a final extension at 72°C for 10 minutes was performed. Amplification products were separated by 2% agarose electrophoresis. The images were photographed and analyzed with Stratagene analysis software. Relative gene expression quantification was calculated according to the comparative Ct method using β-actin as an internal standard.
Statistical Analysis
The results are expressed as mean ± standard deviation (SD) and analyzed by SPSS version 14.0 statistical software (SPSS for Windows 14.0; Chicago, Illinois). Comparisons between multiple groups were performed by 1-way analysis of variance (ANOVA) procedures, followed by the Bonferroni post hoc test for intergroup comparisons. The histologic semiquantitative analysis was compared by the nonparametric Mann-Whitney test. All P values less than .05 were considered to be statistically significant.
Results
Physiological Data
Three rats died during surgery due to technical difficulties (2 in the control group and 1 in the L-Cur group). Physiologic data before and after operation are illustrated in Table 1. In all groups that underwent CPB, the MAP fell significantly from baseline to approximately 80 mm Hg; pH levels declined, showing a trend to develop metabolic acidosis; and hematocrit was significantly decreased from the hemodilution of CPB. There was an increase in the pH values and hematocrit both 2 and 24 hours after surgery. However, no statistical differences existed at these time points between all groups that underwent CPB and the sham group. The arterial partial pressure of oxygen (PaO2)/F
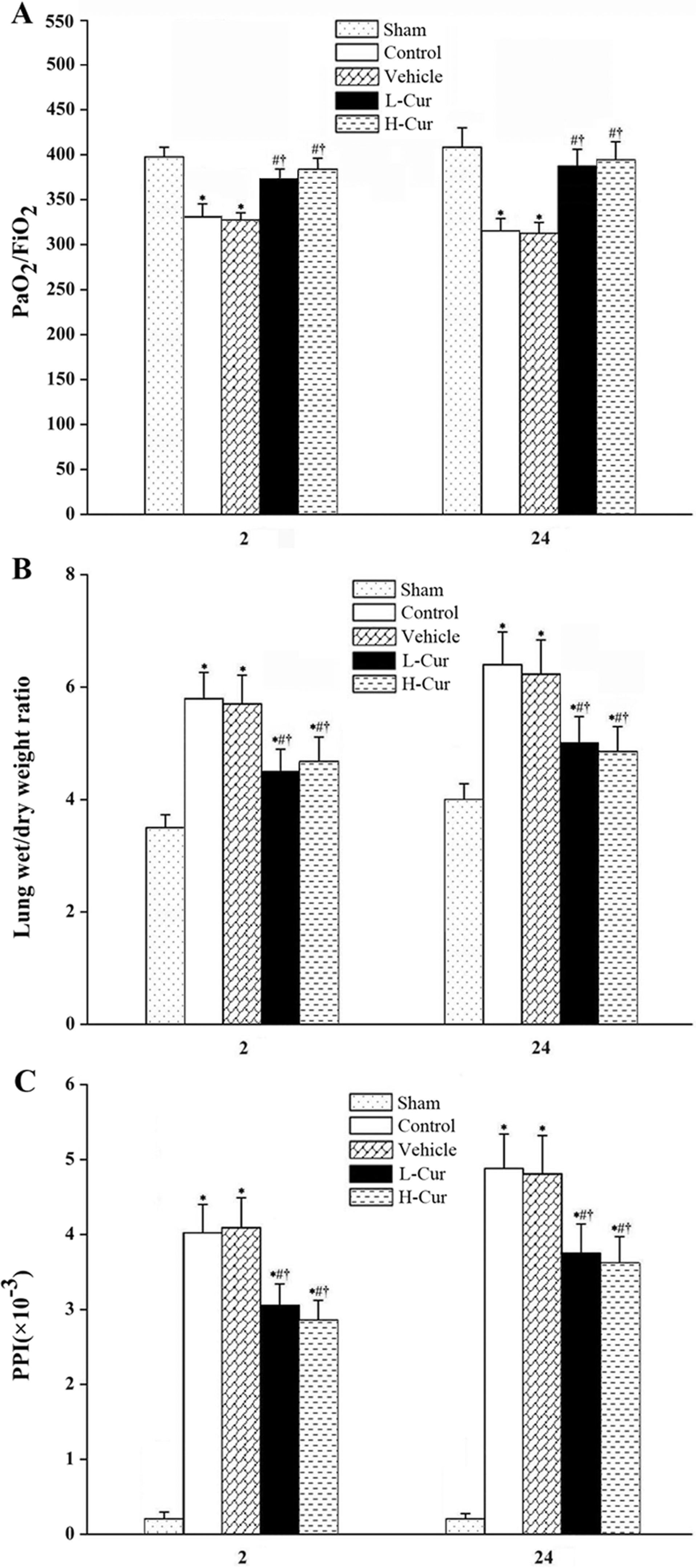
Effects of curcumin on (A) the PaO2/F
Physiologic Data Collected at Representative Time Points in Ratsa

Abbreviations: CPB, cardiopulmonary bypass; H-Cur, high-dose curcumin; L-Cur, low-dose curcumin; N/A, not assessed; T0, just before the initiation of CPB; T1, at the termination of CPB; T2, at 2 hours after CPB; T3, at 24 hours after CPB; ; SD, standard deviation.
aValues are mean ± SD; n = 8;
b P < .05 vs sham group.
c P < .05 vs control group.
d P < .05 vs vehicle group.
The W/D Lung Weight Ratio and PPI
The W/D lung weight ratio and PPI values were significantly elevated 2 and 24 hours after CPB (P < .05, respectively). Compared with the vehicle group, curcumin pretreatment markedly decreased the lung W/D ratio and PPI at both the 2 and 24 hours time points postoperatively (P < .05 for all comparisons; Figure 1B and C).
Light Microscopic Findings
In the vehicle group, the severity of lung injury, characterized by lung tissue edema, neutrophil infiltration, and hemorrhage, was more apparent at 24 hours post-CPB than that at 2 hours post-CPB. Administration of curcumin markedly reduced the lung injury score both 2 and 24 hours after surgery (P < .05 for comparisons). Although histologic changes showed that the animals in the H-Cur groups exhibited less lung hemorrhage and less hyaline membrane formation than those in the L-Cur group 2 hours post-CPB (P = .012), there were no differences between the 2 groups 24 hours post-CPB (P = .056; Figure 2).

Photomicrographs of lung 2 and 24 hours postoperatively in 4 groups (hematoxylin–eosin ×200). At post-CPB 2 hours, lung tissue samples from rats of control, vehicle, low curcumin-treated, and high curcumin-treated animals (A-D). At post-CPB 24 hours, lung tissue samples from rats of control, vehicle, low curcumin-treated, and high curcumin-treated animals (E-H). (I) Lung injury scores. CPB, cardiopulmonary bypass; H-Cur, high-dose curcumin; L-Cur, low-dose curcumin. *P < .05 vs control group; # P < .05 vs vehicle group; † P < .05 vs L-Cur group.
Oxidative Stress Markers
Compared with the corresponding sham group, there was a considerable increase in tissue concentration of MPO and MDA and a significant decrease in tissue concentration of SOD in the control group 2 and 24 hours post-CPB (P < .05 for all comparisons). Curcumin pretreatment was associated with reduced levels of MDA and MPO both 2 and 24 hours after surgery, while curcumin also prevented the decreased tissue levels of SOD (P < .05 for all comparisons). Furthermore, lung tissue concentrations of MPO and SOD were prominently different between the H-Cur and L-Cur groups, implying that curcumin modulated these oxidative stress markers in a dose-dependent manner. However, tissue MDA levels were not statistically different between the 2 groups (Table 2).
Biochemical Results of Oxidative Stress Markers in Experimental Groupsa

Abbreviations: CPB, cardiopulmonary bypass; MPO, myeloperoxidase; MDA, malondialdehyde; SOD, superoxide dismutase; SD, standard deviation.
aValues are as mean ± SD.
b P < .05 vs. sham group;
c P < .05 vs. control group;
d P < .05 vs.vehicle group.
e P < .05 vs. low-dose curcumin group.
HO-1 Expression
Western blot analysis indicated that CPB injury was associated with a slight increase in HO-1 protein 2 and 24 hours after surgery (P < .05 for both comparisons). A marked upregulation in HO-1 expression in both curcumin-treated groups was observed when compared with the vehicle groups 2 and 24 hours post-CPB (P < .05 for all comparisons). Meanwhile, there was a significant difference at these time points between the H-Cur and L-Cur groups, suggesting that curcumin regulates HO-1 expression in a dose-dependent way (Figure 3).
The expression of HO-1 mRNA in lung tissues was upreguated in animals treated with vehicle both 2 and 24 hours after surgery (P < .05 for both comparisons), which was in concordance with the results of Western blot analysis. Low dose of curcumin pretreatment elevated the HO-1 transcription level by 4.11- and 3.81-fold at 2 and 24 hours post-CPB, whereas a high dose of curcumin increased the levels of HO-1 mRNA by 4.95- and 4.63-fold at these time points (P < .05 for all comparisons; Figure 4).
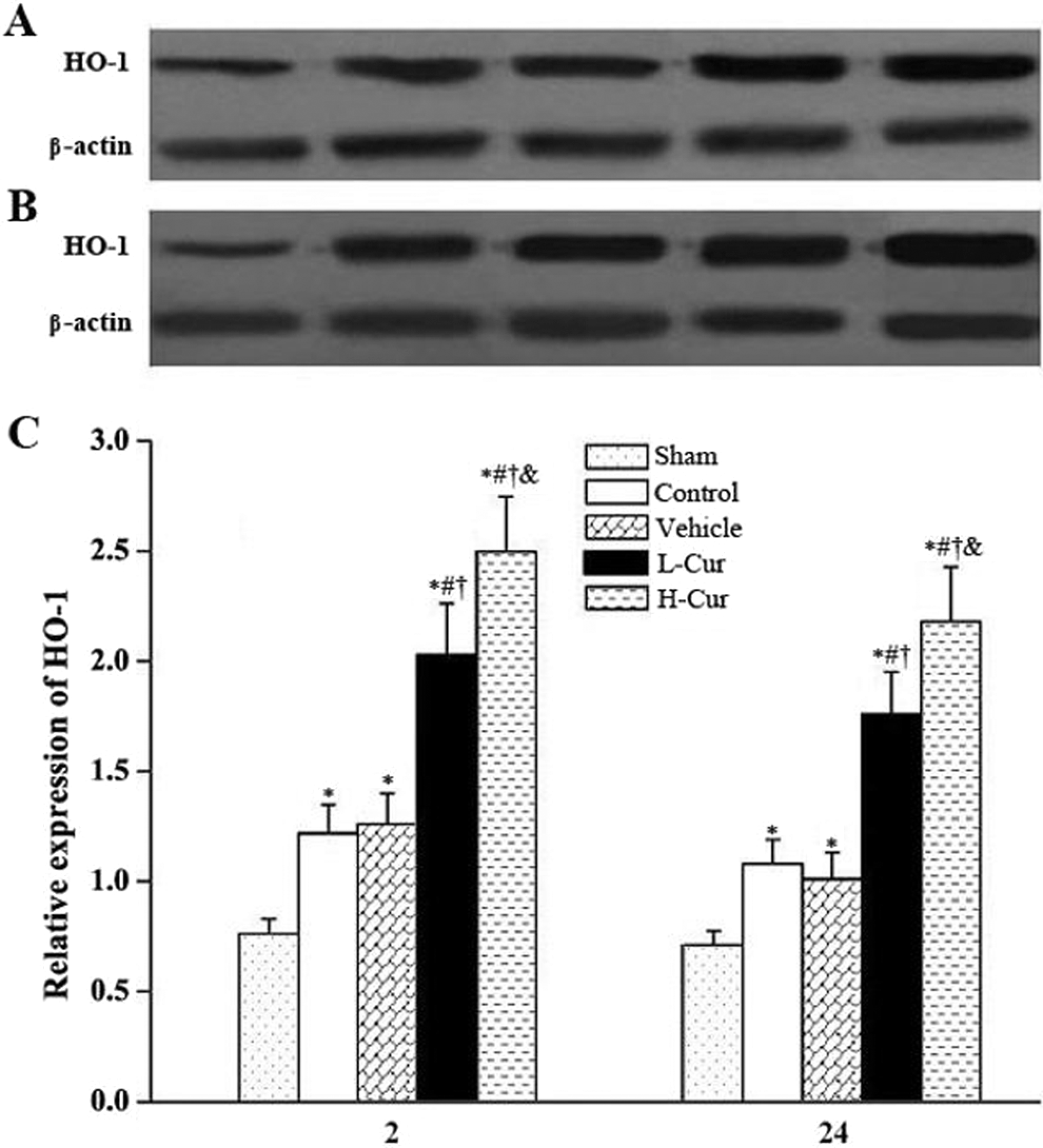
Effects of curcumin on the expression of HO-1 proteins. HO-1 proteins in the lungs detected by Western blot 2 hours (A) and 24 hours (B) post-CPB. (C) Relative expression of levels HO-1 proteins in different groups. HO-1, heme oxygenase 1; CPB, cardiopulmonary bypass; H-Cur, high-dose curcumin; L-Cur, low-dose curcumin. *P < .05 vs sham group; # P < .05 vs control group; † P < .05 vs vehicle group; & p < .05 vs L-Cur group.
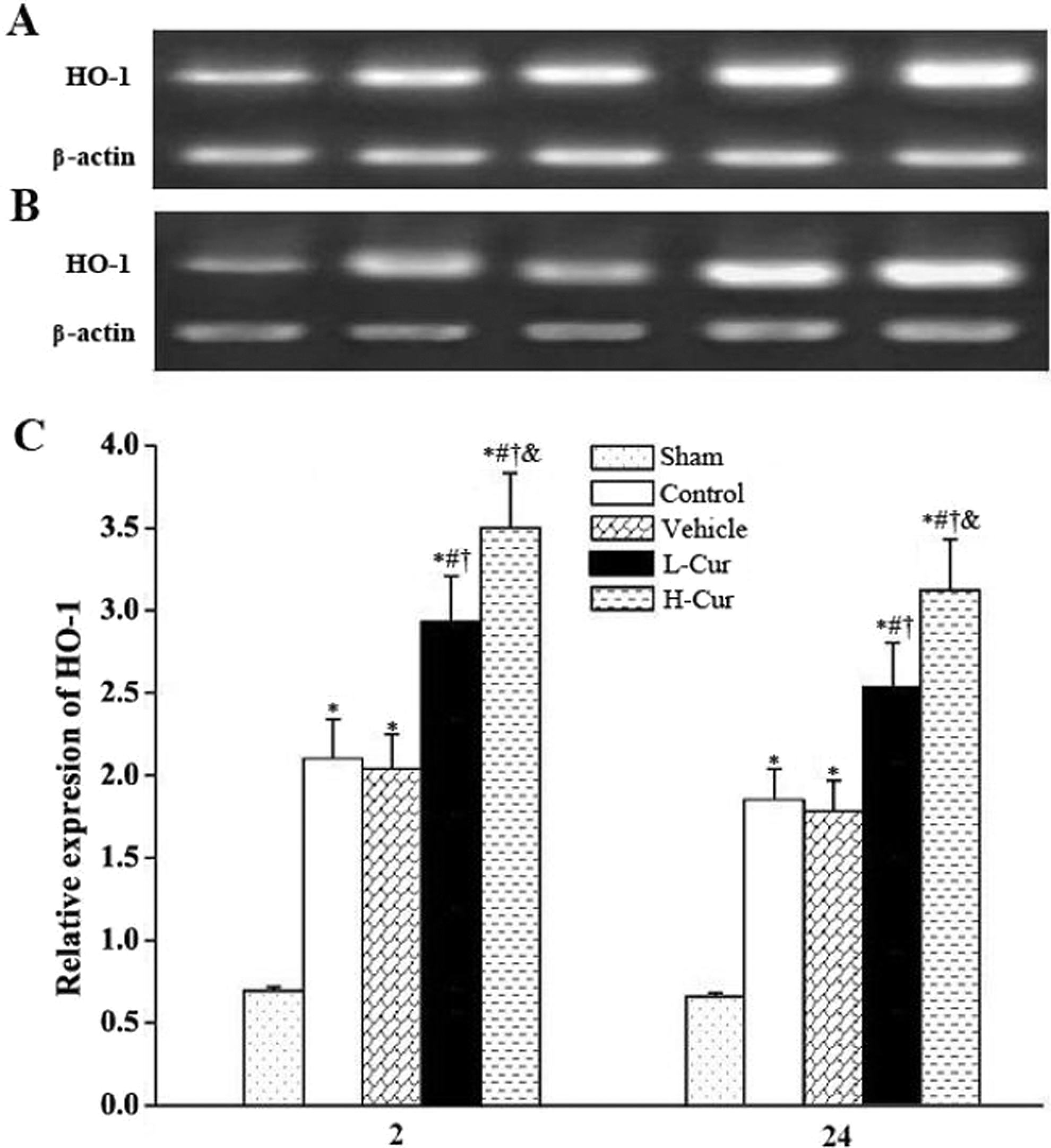
Effects of curcumin on the expression of the HO-1 gene. HO-1 gene in the lungs of rats detected by quantitative RT-PCR 2 hours (A) and 24 hours post-CPB (B). (C) Relative expression levels of the HO-1 gene in different groups. HO-1, heme oxygenase 1; CPB, cardiopulmonary bypass; H-Cur, high-dose curcumin; L-Cur, low-dose curcumin. *P < .05 versus sham group; # P < .05 versus control group; † P < .05 versus vehicle group; & P < .05 versus low-dose curcumin group.
Discussion
This study demonstrates that the administration of curcumin ameliorated the effect on CPB-induced lung injury as indicated by the histopathologic and biochemical results. Pretreatment with curcumin attenuated the infiltration of neutrophils in the pulmonary interstitium, ameliorated pulmonary interstitial edema, reduced oxidative stress marker production, and appeared to upregulate antioxidant enzyme expression. Furthermore, we found a significant elevation of the HO-1 protein level in the lungs of curcumin-treated rats, which could be the mechanism underlying the protective effects of curcumin.
There are several studies concentrating on the organ-protective effect of curcumin. Owing to ethics restrictions, however, it is difficult to obtain both the morphologic data and the level of oxidative stress markers from tissues having clinical status. The experimental design in this rat CPB model allowed for the investigation of the levels of MPO, MDA, and SOD in lung tissues, especially the expression of HO-1 in lung tissues.
A recent review indicates that the HO-1 system has been gaining recognition as a vital contributor to the cellular response to diverse oxidative stimuli. 3 HO-1-mediated cytoprotection may be critical for lung tissue vulnerable to oxidative stress. 4 Nonetheless, there are few data available that demonstrate the relationship between the HO-1 system and lung protection after CPB. Thus, the present study evaluated the role of HO-1 in rat lung tissue subjected to CPB. Our data showed that CPB induced an increase in the expression of HO-1 with a corresponding decrease in MPO and MDA production 2 and 24 hours post-CPB, suggesting that the HO-1 system might play a role in regulating oxidative stress in the early phase after CPB.
There is increasing evidence demonstrating that there is a role for curcumin in regulating the expression of HO-1 in vivo. 6 However, it remains unclear whether curcumin reduces CPB-derived oxidative damage via the HO-1 system. The fact that curcumin exhibits a potent cytoprotective property against oxidative injury in lung tissues after CPB possibly through the induction of HO-1 is a novel finding. In addition, our data further revealed that curcumin administered at a dose of 200 mg/kg may be more effective in the attenuation of oxidative injury than a dose of 50 mg/kg.
With respect to the underlying mechanisms by which curcumin elevates HO-1 expression, at least 2 separate explanations are plausible. First, induction of HO-1 by curcumin could be due to the activation of nuclear factor erythroid 2-related factor 2 (Nrf2), which controls the coordinated expression of important antioxidant and detoxification genes through a promoter sequence termed the antioxidant response element (ARE). The Nrf2 is sequestered in the cytoplasm as an inactive complex with its repressor Keap 1. 6 Upon stimulation by curcumin, Nrf2 dissociates from Keap 1 and translocates into the nucleus where it binds to ARE sites in the HO-1 promoter, resulting in the activation of HO-1. 15 Second, inhibition of nuclear factor-kappaB (NF-κB) plays an important role in CPB-induced lung damage. Convincing evidence has indicated that NF-κB is involved in regulating the expression of the HO-1 gene. It is currently understood that NF-κB activity is mediated by the inhibitory protein IκB. Curcumin has been shown to inhibit phosphorylation and degradation of IκB through the inactivation of IκB kinase activity and to further attenuate the release and nuclear transmigration of NF-κB, thus promoting HO-1 gene transcription. 16 Since HO-1 gene expression is regulated via various transcriptional factors, further experiments are required to elucidate the precise mechanisms by which curcumin induced HO-1 expression in the case of CPB.
In a study of patients undergoing cardiac surgery with CPB, the level of lipid peroxidation products is elevated in the lungs and plays an important role in lung oxidative injury. 17 Malondialdehyde is a major product of oxidative degradation and is an index of the rate of lipid peroxidation. In a recent article, it was documented that curcumin reduced the levels of MDA involving HO-1 induction in a model of ethanol-induced hepatocyte oxidative damage. 18 In our model, administration of curcumin also elevated the HO-1 expression, concomitant with the decrease in MDA production. These results further suggest that curcumin likely reduces oxidative stress through the induction of HO-1 in lung tissues.
It has been increasingly recognized that neutrophil infiltration is a major cause of CPB-induced lung injury. The activity of MPO is correlated with the number of neutrophils and can be used as a neutrophil indicator. A substantial amount of data indicates that CPB causes a remarkable augmentation in the MPO activity in the lungs, 19 a finding supported by our data as well. Moreover, we further corroborated that curcumin could downregulate the concentration of MPO after CPB. As stated previously, this effect may be the result of upregulation of HO-1 expression. Another potential explanation is that curcumin may modify the MPO structure. It is known that the active site of human MPO is located in a hydrophobic cavity with a narrow oval-shaped opening. 20 The inhibitory effect of curcumin on MPO secretion is attributed to the blockage of the entry of this cavity. 21
The SOD is an antioxidant enzyme that scavenges the superoxide free radical by enzymatic dismutation. According to our findings, curcumin pretreatment elevated the activity of SOD in lung tissues, suggesting that curcumin may attenuate CPB-induced lung injury by upregulating the level of antioxidative enzyme. However, the exact mechanism of curcumin’s action on SOD activity after CPB has not yet been elucidated. It has been well documented that carbon monoxide, an end product of HO-1, results in the activation of mitogen-activated protein (MAP) kinase in endothelial cells. 22 In a study of Qadri and coworkers, 23 MAP kinase had a direct stimulatory effect on cellular SOD. Based on these findings, it seems reasonable to extrapolate that curcumin increases SOD expression, at least in part, through upregulation of HO-1.
A new finding in our study was the salutary effects of curcumin on the PaO2/F
One limitation in the current study is that we only observed the lung function for 24 hours after the operation. Further long-term studies should be performed to investigate the protective effects of curcumin in CPB models. Moreover, the dose for the administration of curcumin in the present study was based on previous experiments. It is not known whether curcumin pretreatment in a higher dose has further beneficial effects after CPB. Recently, a clinical study 25 has used curcumin at 8000 mg/d for 3 months, and no side effects have been observed, suggesting a potential use of curcumin for long-term therapy for lung injury.
In summary, the present study confirmed that pretreatment with curcumin alleviated lung injury associated with CPB. The beneficial properties of curcumin may be attributable to its ability to improve CPB-induced oxidative stress and upregulation HO-1 expression. Although the exact mechanism of the action of curcumin in terms of HO-1 regulation is far from clear, our investigation suggests that curcumin may acts as an antioxidant in the prevention of lung damage after CPB.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
