Abstract
Background:
Vascular calcification is a potent predictor of plaque instability and cardiac events. Osteoprotegerin (OPG), well-known vascular calcification mediator, is a signaling molecule involved in bone remodeling, which has been implicated in the regulation of vascular calcification and atherogenesis. The purpose of this study was to compare the combination treatments of olmesartan/azelnidipine and olmesartan/diuretics on serum bone-related markers in patients with essential hypertension.
Methods and Results:
A total of 48 patients with hypertension treated with 20 mg olmesartan were randomized to receive combination treatment with 16 mg azelnidipine (O/A group) or diuretics (1 mg indapamide; O/D group) for 12 months. Osteoprotegerin, matrix metalloproteinase 2 (MMP-2), and high-sensitive CRP (hs-CRP) were measured after 3 and 12 months of treatment. Cardio-ankle vascular index (CAVI) was measured as the arterial stiffness using a VaSera CAVI instrument at the same time points. In both groups, the systolic and diastolic blood pressure reduction is similar. Serum OPG, MMP-2, and hs-CRP were significantly decreased at 12 months in the O/A group (P < .05), while there were no significant reductions in the O/D group. CAVI was significantly improved at 12 months in both the treatment groups. The improvement in CAVI was significantly greater in the O/A group than in the O/D group.
Conclusion:
Azelnidipine, but not indapamide, combined with olmesartan, improved arterial stiffness and were associated with significant decrease in OPG, MMP-2, and hs-CRP concentrations. These results suggest that the beneficial effects of the combination treatments of olmesartan/azelnidipine on arterial stiffness are mediated by alteration in bone-remodeling and inflammatory markers.
Introduction
Arteriosclerosis is often complicated by vascular calcification, an important factor contributing to the onset and progression of cardiovascular diseases as well as an independent predictive factor for plaque instability and cardiovascular events. 1 Vascular calcification was formerly regarded as only a part of the developmental process of arteriosclerosis and to be characteristic of advanced aging; however, recent studies have shown that calcification is caused by changes in vascular wall cells and the extracellular matrix. 2 Currently, vascular calcification is regarded as a treatment target. However, no effective treatment approach has as yet been established for vascular calcification and this may reduce the efficacy of other cardiovascular treatments such as coronary intervention. Serum osteoprotegerin (OPG), 3 a bone turnover marker involved in coronary calcification and atheroma formation, is thought to be a useful serum marker of vascular calcification.
The combination of an angiotensin receptor blocker (ARB), a diuretic-antihypertensive, and a calcium-channel blocker (CCB) or concurrent uses of an ARB and a combination drug are anticipated to become the standard treatment for hypertension. However, the ARB/diuretic combination and the ARB/CCB combination have not been directly compared in the changes of bone-related markers in a clinical study. The purposes of this study are to investigate the effects of ARB/CCB versus ARB/diuretic combination on bone turnover markers, inflammation, and arteriosclerosis.
Methods
Our study included 48 patients with hypertension, 32 men and 16 women, all at least 20 years of age, who had been given olmesartan monotherapy (20.0 mg once-daily) at our outpatient clinic and had failed to satisfy antihypertensive criteria after 8 weeks of treatment. Hypertension was defined as 2 or more systolic blood pressure (SBP) measurements more than 140 mm Hg and/or diastolic blood pressure (DBP) more than 90 mm Hg, or documented hypertension currently treated with antihypertensives. All antihypertensive therapies used by the enrolled patients had initially been switched to olmesartan monotherapy. Patients were excluded from the study if their SBP exceeded 200 mm Hg and/or DBP exceeded 115 mm Hg. Patients with 2 or more SBP measurements more than 140 mm Hg and/or DBP measurements more than 90 mm Hg, after 8 weeks of olmesartan monotherapy, were included in the analysis. The exclusion criteria were secondary hypertension, arrhythmias, currently being treated for congestive heart failure, peripheral artery disease (ankle-brachial index of lower than 0.9), cardiovascular diseases (those with previously treated angina pectoris or myocardial infarction, those with reproducible chest pain on exercise, or those with chest pain at rest), clinically significant valvular disease, renal dysfunction (serum creatinine > 2.0 mg/dL), hyperkalemia, serious hepatic dysfunction, cancer, connective tissue disease, severe infection, being perioperative, serious trauma, hypersensitivity to any ingredients of the study drugs, confirmed or possible pregnancy, and being otherwise considered ineligible by the primary physician. The study was approved by the institutional review board of University of Fukui. All patients were fully informed of the study objectives and methods and consented to participate.
Patients with 2 or more nonconsecutive outpatient SBP of 140 mm Hg or higher and/or DBP of 90 mm Hg or higher were included in the analysis of data from this prospective, randomized, investigator-blinded, open-label, controlled study. The patients were assigned to receive olmesartan plus azelnidipine (O/A) 16 mg or olmesartan plus indapamide (O/D) 1 mg for 12 months. Azelnidipine is retained in the vascular wall due to its high lipophilicity; its efficacy for vascular remodeling is superior to that of other calcium channel antagonists. 4 Similarly in the diuretics selection; we chose “indapamide” for the control medicine, since indapamide exerts antioxidative, antiatherogenic actions, and enhances endothelial function compared to other diuretic-antihypertensive medications. 5 Changing the doses of study drugs was not allowed.
The doses of azelnidipine and indapamide in this study were indirectly selected on the basis of previous reports. Azelnidipine 16 mg and amlodipine 5mg had the similar blood pressure-lowering effects. 6 Hydrochlorothiazide 12.5 mg and amlodipine 5 mg were used for the starting doses in combination with an angiotensin converting enzyme inhibitor. 7 In combination with ARB, indapamide 0.65 mg to 1.0mg and hydrochlorothiazide 12.5 mg had the comparable blood pressure-lowering effects. 8 The study drugs were to be taken after breakfast. Use of other antihypertensives or any drugs that might affect the evaluation of study measurements was not allowed.
The study measurements included serum OPG, serum high-sensitivity C-reactive protein (hs-CRP), matrix metalloproteinase 2 (MMP-2), and tissue inhibitor of matrix metalloproteinase (TIMP) 2. Blood samples were drawn from the cubital vein at 3 and 12 months. As the first blood pressure measurements for the analysis, we used the levels obtained 3 months after the start of the treatment. Because we intended to exclude the early antihypertensive effects on the markers, and the sufficient antihypertensive effects would have been obtained after 3 months. Latex immunonephelometry was used for the measurement of hs-CRP, and an enzyme-linked immunosorbent assay (ELISA) kit (Daiichi Fine Chemical Co, Ltd, Japan) for MMP-2 and TIMP-2 measurements. Osteoprotegerin was measured using commercial ELISA kits; OPG by Immundiagnostic AG (intra- and interassay coefficient of variation: 4.0% and 4.8%; Bensheim, Germany).
For the evaluation of arteriosclerosis, the cardio-ankle vascular index (CAVI) was determined after a 10-minute rest during an outpatient visit using the vascular screening system VaSera VS-1000 (Fukuda Denshi Co, Ltd, Japan). The patients were not allowed to eat or smoke immediately before the examination.
Statistical Analysis
For the evaluation of baseline intergroup differences, the unpaired t test was used for continuous variables and the Chi-square test for categorical variables. The Wilcoxon signed rank test, a nonparametric method, was used for the within-group comparison of baseline and posttreatment data because data normality could not be expected due to the small sample size. The Mann-Whitney U test was used for intergroup comparisons of posttreatment data. All data are shown as means ± standard deviation, using a P < .05 significance level. In addition, the change in bone-rerated markers, proinflammatory cytokines, and CAVI were illustrated using the boxplots with whiskers method. The lower hinge, median, and upper hinge of the box correspond to the 25th, 50th, and 75th percentiles, respectively.
Results
The 32 men and 16 women in this study ranged in age from 43 to 78 (mean 68.3 ± 14.9) years and had an average body mass index (BMI) of 23.3 ± 6.4 kg/m2. There were no significant differences in baseline characteristics (age, sex, BMI, blood pressure, pulse rate, cardiovascular risks, estimated glomerular filtration rate [GFR], which is calculated using the Modification of Diet in Renal Disease Study equation [GFR = 186.3 × [serum creatinine in mg/dL] − 1.154 × [age] − 0.203 × [0.742 if female]], medication usage in diabetes mellitus or hyperlipidemia) between patients treated with O/A group and those treated with the O/D group (Table 1).
Baseline Characteristics of the 2 Groups.
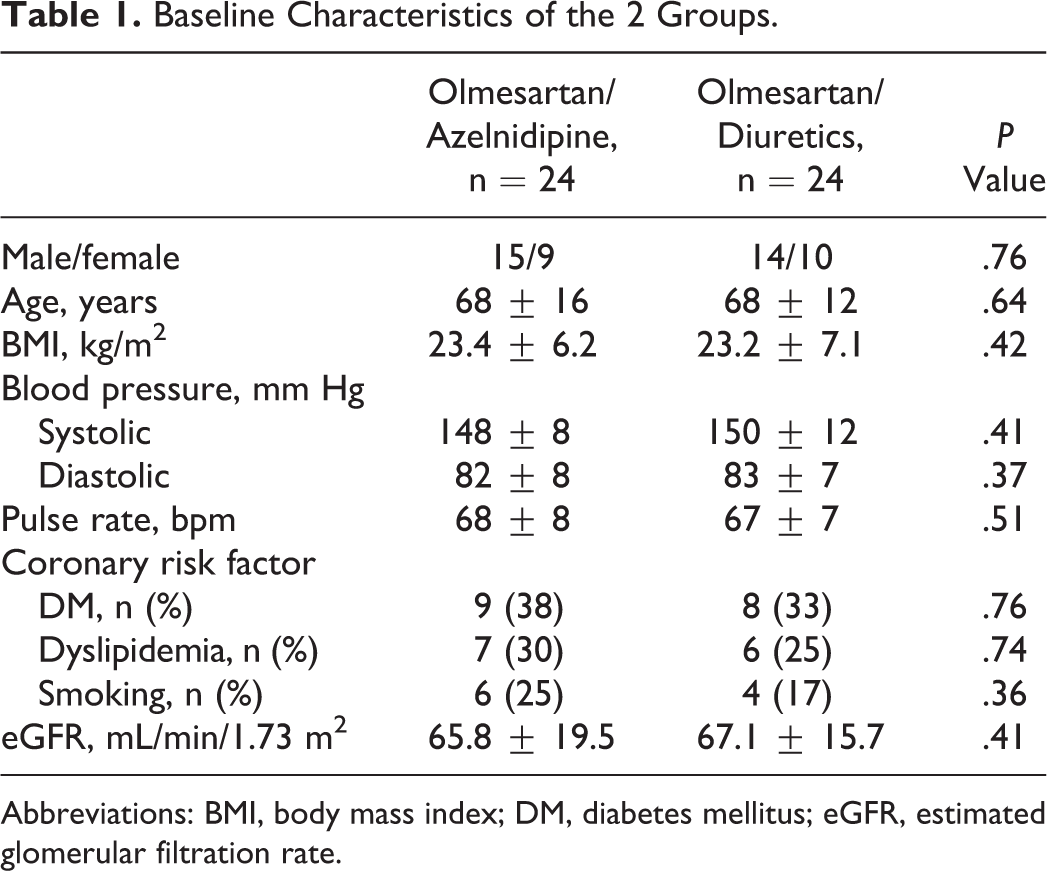
Abbreviations: BMI, body mass index; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate.
After 12 months of treatment, blood pressures were significantly decreased in both groups, and there was no significant intergroup difference (SBP: 18 ± 7 mm Hg and DBP: 6 ± 5 mm Hg in the O/A group; SBP: 19 ± 11 mm Hg and DBP: 8 ± 2 mm Hg in the O/D group). The pulse rate was significantly decreased in the O/A group (−4.4 ± 4.2 bpm) than in the O/D group (Table 2). Changes in blood low-density lipoprotein (LDL) cholesterol and high-density lipoprotein (HDL) cholesterol levels did not differ significantly between the 2 treatment groups (Table 2). During the follow-up period, no new onset of diabetes mellitus and worsening renal function were observed in either group.
Comparison of Changes in the 2 Groups.

Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Serum OPG was significantly decreased at 12 months, as compared with the data at 3 months, in the O/A group (P < .05) while there was no significant reduction in the O/D group (Figure 1).

Box plots for the changes in bone-rerated markers in the 2 groups. This graph shows the serum OPG: OPG levels at 3 and 12 months after treatment. Serum OPG was significantly decreased at 12 months, as compared with the data at 3 months, in the O/A group (P < .05) while there was no significant reduction in the O/D group. OPG indicates osteoprotegerin; O/A, olmesartan plus azelnidipine; O/D, olmesartan plus indapamide.
At 12 months, hs-CRP was significantly reduced as compared with the data at 3 months in the O/A group while there was no significant reduction in the O/D group (Figure 2A). Matrix metalloproteinase 2 also showed a significant decrease at 12 months as compared with the data at 3 months in the O/A group, while there was no significant change in the O/D group (Figure 2B). Cardio-ankle vascular index was significantly improved at 12 months, as compared with the data at 3 months, in both the treatment groups. The improvement was significantly greater in the O/A group than in the O/D group (Figure 3).

Box plots for the changes in serum hs-CRP and serum MMP-2 levels after treatments. A, At 12 months, hs-CRP was significantly reduced as compared with the data at 3 months in the O/A group while there was no significant reduction in the O/D group. B, Matrix metalloproteinase 2 also showed significant decrease at 12 months as compared with the data at 3 months in the O/A group, while there was no significant change in the O/D group. hs-CRP indicates high-sensitive CRP; MMP-2, matrix metalloproteinase 2; O/A, olmesartan plus azelnidipine; O/D, olmesartan plus indapamide.

Box plots for the changes in CAVI in the 2 groups after treatment. Cardio-ankle vascular index was significantly improved at 12 months, as compared with the data at 3 months, in both the treatment groups. The improvement was significantly greater in the O/A group than in the O/D group. CAVI indicates cardio-ankle vascular index; O/A, olmesartan plus azelnidipine; O/D, olmesartan plus indapamide.
Discussion
The major finding in this study was that serum OPG levels were significantly reduced by combination therapy in the O/A group compared to the O/D groups, despite there being no significant difference in brachial SBP decreases between these 2 groups. Although there were no intergroup differences in brachial blood pressure decreases or lipid levels, the CAVI improvement was significantly greater in the O/A group than in the O/D group.
Calcification of the abdominal aorta and carotid artery correlates with arteriosclerosis. 9 Recent studies have found coronary artery calcification to correlate with arteriosclerosis. 10 Vascular calcification is now regarded as an important predictor of cardiovascular events. In the present study, the bone-related marker OPG decreased in patients treated with O/A as compared with that in patients treated with O/D. Osteoprotegerin is a secretory protein belonging to the tumor necrosis factor receptor gene super family and inhibits both the induction of osteoclast differentiation and bone resorption. High serum OPG is associated with increased risk of cardiovascular death. 11 Serum OPG increases in proportion to coronary calcification in type 2 diabetics. 12 Therefore, serum OPG is considered to be very useful as a vascular calcification marker. Only a few studies have examined the effects of drug therapies on serum OPG levels. One study showed that pioglitazone reduced serum OPG and hs-CRP in type 2 diabetics. 13 The authors suggested that the serum OPG level may be decreased by the anti-inflammatory action of pioglitazone. Whether decreases in OPG directly reflect amelioration of vascular calcification remains uncertain. Statins and bisphosphonates are thought to reduce vascular calcification based on anti-inflammatory action as well as antioxidative action. 14 Since azelnidipine exerts both a strong anti-inflammatory action and an antioxidative action on the vascular wall, 15 the calcification inhibitory factor OPG may decrease in parallel with reduced vascular stress.
Improvements in blood pressure, lipid levels, and glucose metabolism are required to reduce the arteriosclerosis indicator CAVI. 16 In regard to antihypertensive drugs with a vasodilatory effect, both ARBs and CCBs have been shown to reduce the arterial stiffness. However, the extent of reduction in the arterial stiffness varies between the 2 classes of drugs. 16,17 Furthermore, it has also been reported that statins reduce the arterial stiffness. 18 Thus, although antihypertensive effects may contribute to reduction in the arterial stiffness, other factors may also be important. 19,20 . The CAVI improvement in our patients could be explained by mechanisms of action of azelnidipine other than its hemodynamic effects, because the blood pressure and lipid level reductions were unchanged compared with diuretics. One of the pharmacological characteristics of azelnidipine is high-vascular tissue affinity. Azelnidipine exerts a continuous action on vascular tissue independently of its blood concentration, and, in contrast to other CCBs, the blood pressure-lowering effect of azelnidipine is thought to be minimally associated with its elimination half-life or blood concentration. 4 The free radical scavenging ability to reduce systemic oxidative stress of azelnidipine is greater than that of other dihydropiridine calcium antagonists or diuretics. 15,21,22 The intergroup difference in CAVI improvement in our study may be attributable to these antiatherogenic actions and antioxidant effects of azelnidipine. In the Japan-Combined Treatment with Olmesartan and a Calcium Channel Blocker Versus Olmesartan and Diuretics Randomized Efficacy (J-CORE) study, 23 in which olmesartan was administered in combination with azelnidipine or hydrochlorothiazide, the aortic pulse wave velocity was measured, and significant improvement in central arterial change and arterial stiffness with combined use of olmesartan and azelnidipine has already been reported. In our study, we consider that the CAVI results were similar to those in the J-CORE study, and we speculate that changes in central arterial blood pressure may also contribute to the improvement in arterial stiffness and bone-rerated markers.
Limitations
Because our sample size was small, it might be difficult to conclude that the combination treatment of olmesartan with azelnidipine is better for arterial stiffness improvement than with indapamide and that we were not able to demonstrate reduced vascular calcification directly in this study. A study consisting of a larger number of patients and longer observation period and quantification of vascular calcification using multidetector-row computed tomography should be conducted in a future study. The duration of observation was too short to evaluate structural arterial wall changes in patients treated with a CCB plus a diuretic. A longer observation period, intravascular ultrasound, and optical coherence tomography will be required to assess such changes. We did not evaluate the systemic oxidative stress. In the previous study, 18 the improvement in oxidative stress was also impaired the vascular stiffness. We assume that the oxidative stress reduction was also associated with vascular stiffness improvement in our study group.
In conclusion, the combination therapy with olmesartan and azelnidipine more significantly decreases serum OPG levels and CAVI than olmesartan and indapamide combination in addition to its blood pressure-lowering effects. This decrease in OPG level and CAVI is associated with proinflammatory cytokine improvement. Further studies are needed to further elucidate the relations between the change in serum bone-related markers and the improvement in vascular calcification.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by a Research Grant from the University of Fukui.
