Abstract
Background:
Menopause is associated with increased arterial stiffness, an independent marker of cardiovascular risk. Omega-3 polyunsaturated fatty acids (N3-PUFAs) are thought to have multiple cardiovascular benefits, including prevention of arterial stiffness. We investigated whether treatment with N3-PUFA prevents increase in arterial stiffness in ovariectomized rats, an animal model of experimental menopause.
Methods:
A total of 43 Wistar rats, 2 months old, were divided into 3 groups, control, sham surgery, normal diet (CTRL, n = 15); ovariectomy, normal diet (OVX, n = 14); and ovariectomy with N3-PUFA supplementation (0.8 g/kg/d in daily gavages administration; OVX + O3, n = 14). Two months after surgery, carotid–femoral pulse wave velocity (PWV) and arterial blood pressure (BP) were measured by carotid and femoral cannulation. Aortic morphometric measurements were performed after dissection.
Results:
Ovariectomy caused a significant increase in BP (P < .05), PWV (P < .0001), and elastic modulus (P = .001) compared to CTRL. After ovariectomy, N3-PUFA supplementation completely prevented increase in arterial stiffness (P < .0001 vs OVX) and BP (P < .05 vs OVX) and resulted in a significant increase in body weight and aortic thickness.
Conclusions:
In an experimental model of menopause, N3-PUFA supplementation prevents arterial stiffening and other vascular changes induced by ovariectomy. These results represent a therapeutic benefit of N3-PUFAs in prevention of postmenopausal cardiovascular disease.
Introduction
Cardiovascular disease is the leading cause of morbidity and mortality in postmenopausal women. A sharp increase in the risk of cardiovascular events is seen after menopausal transition, both after natural menopause 1 and bilateral oophorectomy. 2 The biological explanation of this phenomenon has been attributed to the loss of sex steroid hormones that exert a wide range of properties on the cardiovascular system. 3 Estrogens are thought to have vasoprotective activity, and their deficiency is associated with negative changes in lipid metabolism 4 and hemodynamic profile. 5 Despite this, studies on hormone replacement therapy in postmenopausal women has led to contrasting results about cardiovascular outcomes. 6
Arterial stiffness is one of the earliest manifestations of adverse functional and structural changes in the vascular system. Measurement of arterial stiffness with the gold standard method of pulse wave velocity (PWV) has proved to be a potent predictor of cardiovascular events. 7 Menopause is considered an independent factor increasing arterial stiffness, 8 and this effect is related, at least in part, to estrogen deficiency. 9,10 Previous studies showed that the loss of estrogen activity may influence the vascular system through endothelial dysfunction 11 and dysregulation in the renin-angiotensin system activity, 12,13 resulting in promotion of atherosclerosis. Furthermore, an unfavorable influence of multiple cardiovascular risk factors, such as atherogenic lipid profile, hypertension, or increase in body weight, as seen after menopausal transition, can explain the increase in arterial stiffness. 14
A large body of evidence from experimental, clinical, and epidemiological research has demonstrated the benefits of omega-3 polyunsaturated fatty acids (N3-PUFAs) on cardiovascular health. 15 Membrane content of N3-PUFA, both as eicosapentaenoic (EPA) acid and as docosahexaenoic (DHA) acid, has been suggested, in observational studies, as a risk factor for cardiovascular outcomes. 16 Treatment with N3-PUFA is able to prevent endothelial dysfunction and atherosclerosis, 17 and their use is associated with a reduction in arterial stiffness 18 that may account for some of the cardioprotective properties of N3-PUFA. A wide range of physiological and molecular pathways are affected by N3-PUFA and possibly implied in the regulation of arterial stiffness.
Despite the reported benefits of N3-PUFA, few studies investigated their role in cardiovascular prevention after menopausal transition. A higher dietary intake of N3-PUFA was associated with a reduced progression of coronary artery disease and heart failure in postmenopausal women, 19,20 and a reduction in plasma levels of N3-PUFA in postmenopausal women identifies a population with higher cardiovascular risk. 21 Our group recently demonstrated that treatment with N3-PUFA after experimental menopause can reverse endothelial dysfunction and oxidative stress. 22
The aim of our study was to investigate the influence of N3-PUFA treatment on vascular structure and properties, through evaluation of PWV, in estrogen-deficient ovariectomized rats, an animal model widely employed to study menopause.
Methods
Animals and Diets
A total of 43 female Wistar-Kyoto rats, 2 months old, initially weighing 140 to 170 g, were fed with a standard rat chow (Harlan 2018, 3.4 cal/g, supplied by Harlan, San Pietro al Natisone, Italy), for at least 2 weeks. The animals were kept in temperature-controlled facilities on a 12-hour light/dark cycle. Animals were then randomly assigned to 3 experimental groups, (1) control group of sham-operated rats receiving normal diet (CTRL, n = 15), (2) ovariectomy group receiving normal diet (OVX, n = 14), and (3) ovariectomy group receiving normal diet plus daily esophageal gavages administration of EPA and DHA (OVX + O3, n = 14). Supplementation of omega-3 fatty acids was started on day 7 after surgical ovariectomy. Diets were prepared weekly and stored to prevent degradation, and food was provided and removed daily. To standardize study protocol, CTRL and OVX rats received an equal volume of normal saline solution by gavage per day. Animals were sacrificed after 2 months since the surgical procedure. Part of the total population of the present study (30 animals, 10 per group) was also included in a different experiment, the results of which were published in a previous article. 22 All experiments were performed according to the guidelines and protocols approved by the European Union (Council 86/609; DL January 27, 1992, no. 116) and by the Animal Research Ethics Committee of the University of Trieste, Italy.
Drugs and Reagents
Omega-3 polyunsaturated fatty acids treatment was carried out with a commercially available preparation (Eskim), manufactured by Sigma-Tau (Italy). The manufacturing process of the drug fulfilled the good manufacturing practice standards, and the production was optimized to improve the stability of the compound. The average content of EPA and DHA was, respectively, 450 ± 50 and 395 ± 55 mg/g. A daily dose of 0.8 g/kg/d was administered to rats in the OVX + O3 group, corresponding to a 0.65 g/kg/d of purified active compound (EPA + DHA). The study dose corresponded to a human equivalent dose of about 7 g/d. Saline solution (0.9% NaCl) was manufactured by Diaco (Italy).
Surgical Procedure
Bilateral ovariectomy and sham surgery were performed via a mid-abdominal route under xylazine (10 mg/kg, intraperitoneal [IP] injection) and Zoletil (40 mg/kg, IP) anesthesia. Anesthesia was assessed by complete absence of limb retraction upon painful stimulation. The fallopian tube was ligated with absorbable suture, and the ovary was removed. This model of surgical menopause was validated in a previous experiment, 22 which proved a significant decrease in serum estradiol levels after surgical bilateral ovariectomy, compared with sham-operated rats.
Hemodynamic Measurements
After 2 months since the surgical procedure, the animals were anesthetized with xylazine (5 mg/kg IP) and Zoletil (10 mg/kg IP). Nylon cannulas were introduced into the right common carotid and left femoral arteries up to the aortic ostia. The anatomic locations of the tips of the cannula were checked by postmortem dissection. Cannulas were connected to low-volume blood pressure (BP) transducers (STATHAM range −50 to +300 mm Hg), linked to a Coulbourn analog-to-digital convertor plus computer (Coulbourn Instruments; Lablink, Allentown, Pennsylvania). The frequency response of the cannula plus pressure transducer was minimum 100 Hz with 1% FS/24 Hz.
Systolic and diastolic arterial BPs and BP wave from the carotid and femoral levels were recorded. Aortic PWV was measured by determining the time delay between the crest of the systolic arterial BP wave at the 2 levels. The distance between the 2 cannula tips was determined by direct measurement after dissection of the aortic pathway, with a cotton thread applied on the surface of aorta between the tips of the 2 cannulas. The personnel who performed hemodynamic measures was blinded to study group assignment of each animal.
Aortic Thickness and Elastic Modulus Calculation
Immediately after dissection, in each animal, the thoracic aorta was divided into rings (3 mm long) used for assessing aortic thickness (AT) and inner diameter (ID). The elastic modulus was calculated using the following equation (Moens-Korteweg): Elastic Modulus = PWV2 × BD × ID/AT. Blood density (BD) was estimated as a standard value of 1060 kg/m3.
Determination of Blood Glucose, Plasma Lipids, and Omega-3 Index
Levels of glucose, plasma lipids, and membrane omega-3 index were obtained only on 30 of 43 animals considered in the present study. These data were already published as referenced. 22 Briefly, in this population of 30 animals, ovariectomy significantly affected total cholesterol level that was normalized with N3-PUFA treatment. Membrane omega-3 index was significantly reduced after menopause, with a remarkable increase in the N3-PUFA-treated group.
Statistical Analysis
All data are expressed as mean ± standard deviation, unless otherwise specified. The Kolmogorov-Smirnov test for normality was initially performed for all data. One-way analysis of variance was used to assess group differences. If significant differences were observed with analysis of variance, Fisher least significant difference post hoc analysis for multiple comparisons was performed to identify the differences among the mean values in the groups. A group-specific analysis of covariance (ANCOVA) was used to determine the differences in PWV using covariate mean BP, and residual plots were inspected. All statistical analyses were conducted using commercially available statistical software (IBM SPSS Statistics v20). A P value of less than .05 was considered statistically significant.
Results
Body Weight and Aortic Morphometry
At the end of the experiment, body weight was higher in OVX rats compared to CTRL, and it further increased in animals treated with N3-PUFA (Table 1). Administration of N3-PUFA in ovariectomized rats led to a significant increase in AT when compared to other experimental groups. Aortic ID did not vary significantly between the 3 groups (Table 1).
Hemodynamic and Morphological Measures by Group.a

Abbreviations: N3-PUFA, omega-3 polyunsaturated fatty acids, BP, blood pressure.
a Data reported as mean ± SD. Means in the same row that share a common superscript letter do not differ significantly, while those that do not share a letter differ at least by P < .05.
Blood Pressure
Both systolic and diastolic BP levels were significantly increased in rats after ovariectomy (Table 1). Treatment with N3-PUFA prevented this hypertensive effect after ovariectomy in OVX + O3 rats, leading to BP levels comparable to CTRL rats and significantly lower than OVX rats.
Arterial Stiffness
The PWV in the sham-operated control group was 3.78 ± 0.32 m/s. Ovariectomy markedly increased PWV to 5.61 ± 0.62 m/s, compared to the control group (P < .0001, Figure 1A). Treatment with N3-PUFA prevented increase in PWV, with values not significantly different from the control group (OVX + O3 3.87 ± 0.51 m/s, P = .61) and lower than the OVX group (P < .0001). In the ANCOVA model, PWV values were adjusted for BP value ([estimated mean ± standard error, m/s] CTRL 388.2 ± 13.6, OVX 553.2 ± 15.3, and OVX + O3 398.9 ± 13.6). In this analysis, significant differences were observed from OVX group and both CTRL and OVX + O3 groups (P = .001 CTRL vs OVX and P = .001 OVX vs OVX+O3). At the time point of PWV measurement, the heart rate in the control group was 285 ± 16 bpm, and there was no significant difference in heart rate between the groups investigated (OVX 277 ± 17 bpm and OVX+O3 291 ± 10 bpm).
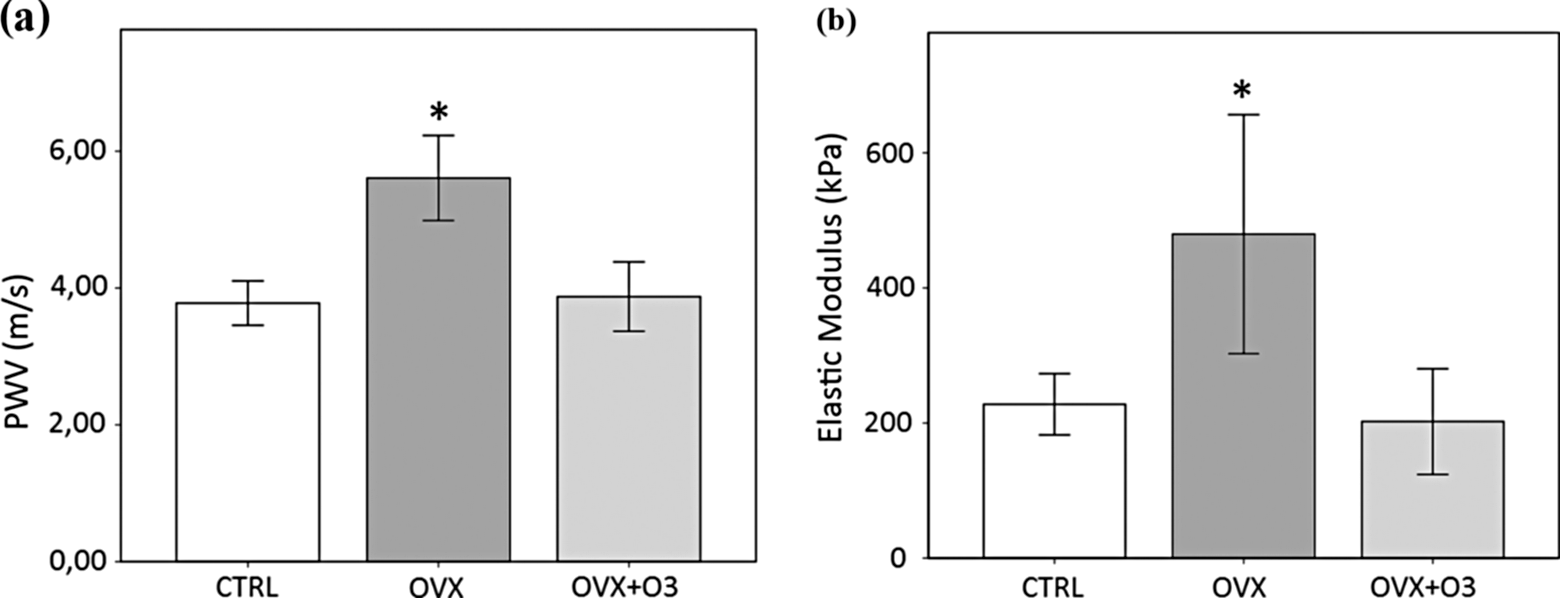
The effect on arterial stiffness in ovariectomized rats (OVX), ovariectomized rats receiving omega-3 polyunsaturated fatty acid (N3-PUFA) supplementation (OVX + O3), and control sham-operated rats (CTRL) at the end of the experimental period. A, Aortic pulse wave velocity (PWV) determined from the time taken for the arterial pulse wave to propagate from carotid to femoral artery. B, Elastic modulus was calculated using Moens-Korteweg equation from PWV, blood density, and aortic diameter and thickness. Data reported as means. Error bars ±1 SD. *P < .0001 versus CTRL or OVX+O3.
Elastic Modulus Calculation
Elastic modulus increased significantly in ovariectomized rats (479.60 ± 176.84 kPa) compared to control group (227.67 ± 45.39 kPa, P = .001 vs OVX), but treatment with N3-PUFA led to values comparable to control group (P = .64) and significantly lower to OVX group (202.13 ± 78.21 kPa, P < .0001 vs OVX, Figure 1B).
Discussion
In this study, we found that in female rats ovariectomy produced an increase in arterial stiffness, measured as PWV, which could be prevented through administration of N3-PUFA. This treatment also prevented adverse hemodynamic changes associated with experimental menopause, such as increase in BP and elastic modulus, although a significant thickening of aorta and weight gain were seen in the OVX + O3 group compared to the OVX group.
Menopause is notoriously associated with negative cardiovascular outcomes. 1,2 Arterial stiffness, measured through the gold standard method of PWV, is considered by current guidelines 23 as an indicator of subclinical target organ damage and a strong predictor of future cardiovascular events. Previous cross-sectional and longitudinal studies, 8–10,24 conducted both in humans and in animal experimental models, demonstrated that menopausal state adversely affects vascular mechanical function and increases arterial stiffness.
In our current study, ovariectomy led to a significant increase in PWV. In addition, a significant increase in body weight, BP, total and low-density lipoprotein cholesterol, and AT, and a decrease in membrane omega-3 index were seen. Because of the multiple etiologies of arterial stiffness, all of the factors mentioned earlier, and possibly others, could play a role in the vascular changes seen after menopause. Hypertension is considered the main determinant of arterial stiffness, both in experimental 25 and in clinical conditions, 26 and in our experiment, systolic and diastolic BP were higher in ovariectomized rats. Etiology of postmenopausal hypertension is complex, involving activation of renin–angiotensin system, increased sympathetic reactivity and vasoconstriction, inflammation, and possibly other mechanisms. 27 Some of these mechanisms can also influence arterial stiffness independent of hypertension, through increasing expression of angiotensin-converting enzyme (ACE) and angiotensin II type 1 receptor, and leading to increased oxidative stress and endothelial dysfunction. 11 A proinflammatory change in cytokine activity has been observed after menopausal transition 28 that can produce an unfavorable change in the vascular system, through endothelial dysfunction and change in aortic wall composition. 29 Estrogen deprivation is also associated with adverse changes in metabolic profile after menopause, through an increase in body weight and an atherogenic lipid profile, 30 and both of these alterations were observed in our experimental animals.
Omega-3 fatty acids have been suggested as an evidence-based means of reducing arterial stiffness in clinical 18,31,32 and experimental conditions. 33 The EPA and DHA supplementation in the diet, both as regular consumption in fatty fish and as pharmaceutical supplement, has been demonstrated to have several beneficial effects on cardiovascular health, without significant adverse effects, although a definitive effect on cardiovascular event prevention and mortality is still a matter of debate. 34 The mechanisms responsible for the improvement in arterial stiffness after N3-PUFA administration are multiple and involve BP reduction, modification of lipid profile, restoration of endothelial dysfunction, and autonomic modulation. It is likely that arterial stiffness in our experimental model was affected by each of the above-mentioned mechanism through a synergistic effect. The BP levels are the main determinant of arterial stiffness, which may be reversed by lowering systemic pressure. 35 The observed effect in PWV could be a secondary consequence of the significant BP lowering observed in OVX + O3 group. In order to control this possible confounder, we adjusted PWV for mean BP. In this analysis, significant differences were maintained in PWV comparison between the groups, suggesting that N3-PUFA effect in reducing PWV is not mediated solely by BP reduction. Administration of EPA and DHA has been associated with a significant reduction in triglyceride level and a modest increase in high-density lipoprotein (HDL). 36 In our previous experiment, 22 we found an unexpected decrease in total cholesterol and essentially unchanged values of triglycerides and HDL. These changes in lipid profile are unlikely to have influenced arterial stiffness in a short period of time as that of our study. Most data suggest that reduction in arterial stiffness with N3-PUFA supplementation occurs independent of body mass index reduction or heart rate. 18 Interestingly, in our experimental animals, an increase in body weight after supplementation of N3-PUFA, probably due to an increased energy intake, was coupled with a reduction in PWV. No significant difference was observed in heart rate between the groups at the time point of PWV measurement.
In addition to being determined by cardiovascular risk factors, arterial stiffness is influenced by the vessel geometry via the hemodynamic overload on the arterial wall. We observed that treatment with N3-PUFA produced an increase in AT, while the simple ovariectomy did not. The elastic modulus, calculated from aortic diameter and thickness, was higher in the ovariectomy group compared to the treatment group. This could reflect a change in the elastic properties of the arterial wall associated with menopause which was prevented by N3-PUFA administration, although some limitation must be acknowledged in the ex vivo experimental measurements, and the derived calculation should therefore be considered with caution. Considering the components of arterial wall, collagen has the highest elastic modulus. 37 An increased collagen deposition in the arterial wall could therefore represent an adaptation to the altered hemodynamic balance produced by menopause and determine the observed increase in arterial stiffness.
Dietary supplementation with N3-PUFA changed membrane phospholipid composition in our experimental animals. As evidenced in our previous experiment, 22 membrane omega-3 index increased after treatment, while the same index was markedly decreased in the untreated group. Membrane omega-3 index in erythrocytes represents a surrogate marker of individual EPA + DHA status, and in clinical content, a marker of increased cardiovascular risk. 16 It is possible that influence in cell membrane fluidity, caused by the increased presence of N3-PUFA, 38 could affect the tensile and viscoelastic properties of the arterial wall leading to a reduced stiffness, although this consideration is purely speculative and cannot be demonstrated by our data. We have already demonstrated that enrichment of N3-PUFA in cell membranes exerts per se an effect on endothelial properties, mainly interacting with endothelial nitric oxide synthase and nicotinamide adenine dinucleotide phosphate oxidase and thus reversing endothelial dysfunction. 22 Given the short duration of the experimental phase, these mechanisms, involving enhancement of NO production, are likely to be related to the regulation of arterial stiffness. The observed reduction in arterial stiffness in our experiment probably resulted from both these functional properties and structural changes in the arterial wall, induced by multiple intermediate determinants of atherosclerosis promotion.
Some concerns may be raised about the potential occurrence of side effects of high-dose N3-PUFA administered in our experiment. Except for weight gain, no relevant side effects (ie, hemorrhages and gastrointestinal disorders) were observed in rats treated with N3-PUFA. Since such high doses as 0.65 g/kg/d of EPA + DHA (corresponding to an approximate human equivalent dose of 7 g/d) have not been tested in clinical trials involving humans, the generalizability of our results must be further evaluated.
Other strategies have been tested with the end point of reducing arterial stiffness, specifically in postmenopausal women, as exercise training, 39 administration of tetrahydrobiopterin, 40 low-dose statins, 41 and isoflavones, 42 all with some encouraging results. Hormone replacement therapy was suggested to decrease stiffness of the aorta and large arteries in postmenopausal women, 43 but the effective benefits of hormone therapy on cardiovascular health are still under debate, 44,45 and its use for cardiovascular prevention is actually discouraged in some patients for side effects and risks. The N3-PUFA supplementation may represent a useful treatment in cardiovascular prevention in these patients, with a favorable risk–benefit profile and multiple beneficial properties, but a confirmation is required in clinical field studies.
Conclusion
In the light of recent clinical trial results, 46,47 clinical beneficial effects of N3-PUFA reduce the risk of cardiovascular disease in high-risk populations still remains inconclusive. Encouraging results were provided in the use of N3-PUFA for reducing arterial stiffness, a parameter that is widely used in the clinical setting, because of the good predictive value for cardiovascular morbidity and mortality. Our results represent preliminary evidence for the possible use of N3-PUFA in reducing arterial stiffness in women during menopausal transition and encourage clinical trials considering arterial stiffness as an end point for cardiovascular risk reduction in postmenopausal women.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
