Abstract
Aims:
We determined whether implantation of heart tissue-derived decellularized matrix, which contains native biochemical and structural matrix composition, could thicken the infarcted left ventricular (LV) wall and improve LV function in a rat myocardial infarction model.
Methods and Results:
Myocardial infarction was induced by left coronary ligation in Fischer rats. One week later, saline (75 μL, n = 17) or matrix (75 μL, n = 19) was directly injected into the infarcted area. At 6 weeks after injection, cardiac function was assessed by left ventriculogram, echocardiography, and Millar catheter. The hearts were pressure fixed to measure postmortem LV volume and processed for histology. Left ventriculogram demonstrated that LV ejection fraction (EF) was significantly greater in the matrix-treated (56.7% ± 1.4%) than in the saline-treated group (52.4% ± 1.5%; P = .043), and paradoxical LV systolic bulging was significantly reduced in the matrix-treated group (6.2% ± 1.6% of the LV circumference) compared to the saline-treated group (10.3% ± 1.3%; P = .048). Matrix implantation significantly increased the thickness of infarcted LV wall (0.602 ± 0.029 mm) compared to the saline-treated group (0.484 ± 0.03 mm; P = .0084). Infarct expansion index was significantly lower in the matrix-treated group (1.053 ± 0.051) than in the saline-treated group (1.382 ± 0.096, P = .0058). Blood vessel density and c-kit positive staining cells within the infarct area were comparable between the 2 groups.
Conclusions:
Implantation of heart tissue-derived decellularized matrix thickens the LV infarcted wall, prevents paradoxical LV systolic bulging, and improves LV EF after myocardial infarction in rats. This benefit was not dependent on the enhanced angiogenesis or the recruitment of endogenous stem cells to the injury site.
Introduction
Myocyte necrosis resulting from ischemic injury is replaced by fibrotic scar tissue, a process characterized by a cascade of events including breakdown of the extracellular matrix (ECM), myocyte slippage, ventricular wall thinning and dilatation, and infarct expansion. According to Laplace's law, ventricular wall thinning may play a crucial role in postmyocardial infarction remodeling, because thinning of the myocardial wall can cause an increase in wall stress that contributes to infarct expansion and ventricular dilation. Increasing the thickness of the infarcted ventricular wall might limit the adverse left ventricular (LV) remodeling. 1,2 A popular therapy in recent years is to utilize injection of stem cells into the infarct. However, stem cells require time for cell processing, allogeneic cells may induce immune rejection, and in some studies transplanted cells have a poor rate of retention. Another approach is to utilize injection of acellular material. In recent years, decellularized ECM that is derived from different tissues, such as small intestine, 3 pericardium 4 and myocardial tissue, 5 –7 has been developed for repair of damaged myocardium.
Specificity of tissue-derived ECM as a substrate exists for the cell type that originated from that tissue. 8 Recent studies by Dr Zhang 8 –11 have demonstrated that tissue-specific ECM significantly improves the adhesion, growth rates, and phenotypes of skin, muscle, and liver cells in vitro. They demonstrated that each cell type grew best on ECM from its tissue of origin. Liver cells grew best on liver ECM; skin cells grew best on dermal ECM; muscle cells grew best on muscle ECM; and these matrices served to maintain phenotype and function of these cells. Moreover, they showed that growth rates and cell function were not as high when the cell and ECM were mismatched, and importantly, that this was especially true for complex tissues such as liver. Although cells were removed from these organs or tissues, the most important biochemical components of ECM (ie, collagen, glycosaminoglycans, fibronectinin/laminin) remained. 8 –11 Therefore, we hypothesize that heart-derived ECM can enhance heart tissue regeneration via recruitment of stem cells from local tissue to repair the scarring tissue.
Recently, Singelyn et al 7 made an injectable hydrogel derived from pig ventricular ECM and injected this material for the first time into 2-week-old infarcted rat hearts, which had been subjected to 25 minutes of coronary occlusion followed by reperfusion. At 4 weeks, magnetic resonance imaging demonstrated that ECM improved cardiac function. Histology showed that ECM increased the average area of cardiomyocyte islands in the infarct area. The c-kit+ cells were observed in low number within the myocardial matrix scaffold, but the authors did not quantitate the number of c-kit+ cells between the groups. The infarcted LV wall thickness and regional LV wall motion were not examined in this study. In our present study, we determined whether implantation of heart tissue-derived ECM, which contains native biochemical and structural matrix composition, could thicken the infarcted LV wall, increase neovascularization, promote recruitment of c-kit+ cells, prevent paradoxical LV systolic bulging, and improve LV function in a rat myocardial infarction model.
Materials and Methods
All the experiments were approved by the Institutional Animal Care and Use Committee and were performed in accordance with the guidelines for the care and use of laboratory animals (NIH publication No. 85-23, National Academy Press, Washington, District of Colombia, revised 1996). The Heart Institute at Good Samaritan Hospital is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Preparation of Rat Heart-Derived Decellularized ECM
Fischer rat hearts were harvested and were cleaned of adipose tissue and cut into 1-mm thick sections. Sections were placed in a 50 mL conical tube and rinsed in Dulbecco phosphate-buffered saline (PBS) with 1% antibiotic and antimitotic solution (AA; Sigma Aldrich, St Louis, Missouri). Heart tissues were thoroughly homogenized into fine pieces with the T10 basic ULTRA-TURRAX Disperser (IKA, Wilmington, North Carolina) in a 3.4 mol/L sodium chloride (NaCl2) solution followed by three 30-minute treatments in 3.4 mol/L NaCl2 solution. For decellularization, heart tissue samples were exposed to a PBS nuclease solution (50 units/mL DNAse and 10 mg/mL of RNAse) for 1 hour to remove any remaining nucleic content. After 1-hour treatments in 1% Triton X-100, the heart tissues were washed in PBS solution 3 times (1 hour per washing) to remove any residual substances. During all washing and treating procedures, samples were placed on the 4°C shaker (130 rpm). During each solution change, samples were centrifuged at 3000 rpm for 10 minutes followed by the removal of the supernatant to keep the ECM in pellets. The sizes of final ECM particulates were arranged from 40 µm to 100 µm. The heart ECM pellets were stored at −80°C until use.
Surgical Procedures for Coronary Ligation and Matrix Injection
Myocardial infarction was induced according to the methods described previously. 12 Briefly, female Fischer rats were anesthetized with 1 intraperitoneal administration of ketamine (75 mg/kg) and xylazine (5 mg/kg), intubated, and mechanically ventilated with room air. The adequacy of anesthesia was monitored by responsiveness to painful stimuli, loss of muscular tone, character, and rate of breathing. Under aseptic conditions, the heart was exposed by a left thoracotomy through the fourth intercostal space. The left coronary artery was encircled with a silk suture and permanently ligated. Then, the incision of the chest was closed by suturing the muscle and stapling the skin, and the rat recovered under care. Buprenex (0.001 mg/100 g body weight) was subcutaneously given for 2 days (twice daily) as postoperative analgesic.
At 1 week after coronary ligation, the rats were reanesthetized, and the hearts were exposed by a second left thoracotomy through the fifth intercostal space as described above. Matrix (75 μL) or saline (75 μL) was directly injected into the LV infarcted area with a 28-gauge needle attached to an insulin syringe. The incision of the chest was closed and the rats were allowed to recover. During the 6 weeks follow-up, the rats were housed in a clean laboratory room with temperature-controlled environment under a 12-hour light–dark cycle, and fed with free access to food and water.
Assessment of Cardiac Function by Echocardiography and Left Ventriculography
A Sonos 5500 ultrasound system (Philips Medical System, Andover, Massachusetts) with a 15-MHz transducer (Philips Medical System) was used to assess LV fractional shortening (FS) prior to coronary artery occlusion, at 1 week after myocardial infarction and at 6 weeks after treatment in all animals. Two-dimensional images from short-axis views of the LV cavity were obtained at the level of the papillary muscle (Figure 1). Motion-mode (M-mode) images were used to assess global LV function by calculating LV FS, which was performed on 3 consecutive beats and averaged. The FS (%) was expressed as ([LV end-diastolic diameter − LV end-systolic diameter]/LV end-diastolic diameter) × 100%.

Echocardiographic analysis of cardiac function. Representative transthoracic left ventricular (LV) M-mode echocardiograms at the level of papillary muscle in a saline-treated (A, B) and a matrix-treated (D, E) heart before coronary occlusion (A, D) and at 6 weeks after treatment (B, E). Note the changes in the LV wall motion between the normal heart (A, D) and the infarcted heart (B, E), showing reduction in wall motion of the anterior wall. Panel C showed the paradoxical systolic bulging (red arrows) of the infarcted LV segment at 6 weeks after saline treatment. In panel F, the paradoxical systolic bulging was not observed (yellow arrow). Panel C enlarges the paradoxical systolic bulging portion pointed by the red arrows in panel B to show more clearly the motion (red arrows) of the anterior LV infarcted wall. The paradoxical systolic bulging was not observed in panel F, which was the magnification of the portion pointed by the yellow arrow in panel E.
Since M-mode echocardiography provides an “ice-pick” view of the heart only, we also used LV ventriculography as previously described 13 in order to visualize the entire LV cavity in end diastole and end systole. Briefly, a XiScan 1000 C-arm x-ray system (XiTec, Inc East Windsor, Connecticut; 3-inch field of view) was used for LV contrast ventriculography at 6 weeks after treatment. Rats were anesthetized by 1 intraperitoneal administration of ketamine and xylazine, and monitored by the methods as described above. A catheter was inserted into the left jugular vein. Video images (anterior-posterior and lateral projections) were acquired on half-inch super-VHS video tape at 30 frames per second under constant fluoroscopy, following the injection of 1 mL nonionic contrast into the left jugular vein via the catheter. The video images were analyzed in a blinded manner to calculate end-systolic and end-diastolic LV volumes. All parameters were averaged over 3 consecutive cycles in both anterior-posterior and lateral projections. The LV ejection fraction (EF, %) was calculated as ([end-diastolic LV volume − end-systolic LV volume]/ end-diastolic LV volumes) × 100%, and averaged over both anterior-posterior and lateral projections.
The paradoxical systolic bulging of the infarct LV wall was assessed based on the LV ventriculography. Tracings of the LV cavity images at end diastole and end systole of the same cardiac cycle were superimposed on transparent film. Paradoxical systolic bulging is identified as that portion of the end-systolic tracing that extends outside the end-diastolic tracing. The extent of paradoxical systolic bulging (%) is expressed as (circumference of the bulging segment/total LV systolic circumference) × 100%. For each heart, 3 consecutive beats in both the anterior-posterior and lateral views were calculated and averaged.
Measurements of Hemodynamics
At 6 weeks after treatment, rats were anesthetized by 1 intraperitoneal administration of ketamine and xylazine and monitored by the methods described as above. The right carotid artery was exposed, and a small incision was made in the artery wall. A 2F high-fidelity, catheter-tipped micromanometer (model SPR-869, Millar, Inc, Houston, Texas
Assessment of Postmortem LV Volumes and Histological Parameters
After 0.6 mL 50% Unisperse blue dye, a suspension of blue particles (Ciba Geigy, Hawthorne, New York), was intravenously injected to stain the perfused blood vessels in the LV wall, the rats were euthanized with an intravenous injection of 1 mL of potassium chloride (149 mg/mL) to arrest the heart in diastole under deep anesthesia (monitored by no response to painful stimuli). The hearts were harvested and were pressure fixed (pressure equal to 13 cm water column) in formalin. The LV volumes were measured by filling the cavity with water and weighing; this process was repeated 3 times and the 3 volume measurements were averaged.
Formalin-fixed hearts were cut into 3 transverse slices. The middle slice was embedded in paraffin for histology. The paraffin-embedded tissue was sectioned (5 μm thickness) and stained with hematoxylin and eosin and Picrosirius red. Histological images of the stained sections were traced with computerized planimetry and the following parameters were measured: (1) scar thickness (average of 5 equidistant measurements) and septum thickness (average of 3 equidistant measurements); (2) total LV epicardial circumference and endocardial circumference; (3) epicardial circumference and endocardial circumference of the LV infarcted segment; and (4) LV cavity area and total LV area. The blood vessel density in the scar area was also calculated on hematoxylin and eosin-stained slides by counting stained blood vessels that contained perfused blue particles. Ten microscopic fields at ×400 magnification within the infarcted area from each heart were randomly chosen for counting and the total blood vessel density was expressed as vessel number/mm2.
Immunohistochemical C-Kit Staining
In order to identify cardiac stem cells within the infarcted LV wall, slices of the paraffin-embedded heart tissue were stained with antibodies against c-kit, a marker for stem cells. Briefly, deparaffinized sections underwent epitope retrieval using EDTA pH 9.0 (Fisher Scientific, Pittsburgh, Pennsylvania); permeabilized with 0.01% Triton; kept in peroxidase inhibitor solution; incubated with rabbit anti-c-kit primary antibodies (Abcam, Boston Massachusetts) at 4°C overnight. The sections were then incubated with biotinylated horse anti-rabbit secondary antibodies (Vector Labs, Burlingame, California) and treated with 3,3'-diaminobenzidine. Quantification of c-kit+ cells was performed by Image Pro Plus software (Bethesda, Maryland) based on images taken at magnification ×200. Size gating was used in order to eliminate false positive counts and to include clustered cells. Four microscopic fields at ×200 magnification within the infarcted area were randomly chosen for counting c-kit+ cells from each heart (n = 6 in each group). The average of positive c-kitstained cells per ×200 image field of microscope was compared between the 2 groups.
Statistical Analysis
Data are presented as mean ± standard error of the mean. Student t test was performed for comparison of results between the 2 groups. Significance was accepted at P < .05.
Results
The LV FS by Echocardiography
The percentage of FS at baseline before coronary occlusion was similar between the saline group (47.3% ± 1.3%) and matrix group (46.5% ± 0.9%). The percentage of FS at 1 week after myocardial infarction fell but remained similar between the 2 groups (21.4 ± 1.6 in saline group and 20.7 ± 2 in matrix group). At 6 weeks after treatment, the percentage of FS was significantly higher in the matrix group (26.2% ± 2.2%) than in the saline group (17.8% ± 1.5%, P = .0034; Figure 2).
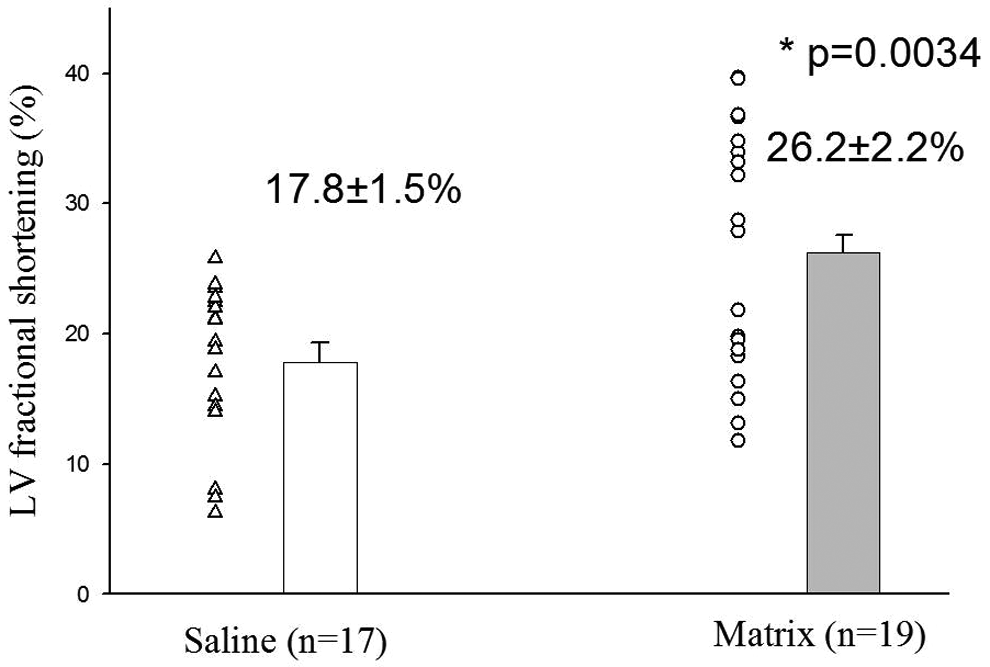
Matrix injection significantly improved the left ventricular (LV) fractional shortening compared to saline at 6 weeks after treatment (P = .0034).
The LV EF and Paradoxical Systolic Bulging by LV Ventriculography
At 6 weeks after treatment, there was significantly higher LV EF (56.7% ± 1.4%) in the matrix-treated group compared to the saline group (52.4% ± 1.5%, P = .043; Figure. 3). The extent of systolic bulging, expressed as percentage of total LV systolic circumference, was significantly smaller in the matrix-treated group (6.2% ± 1.6%) compared to the saline group (10.3% ± 1.3%; P = .048).
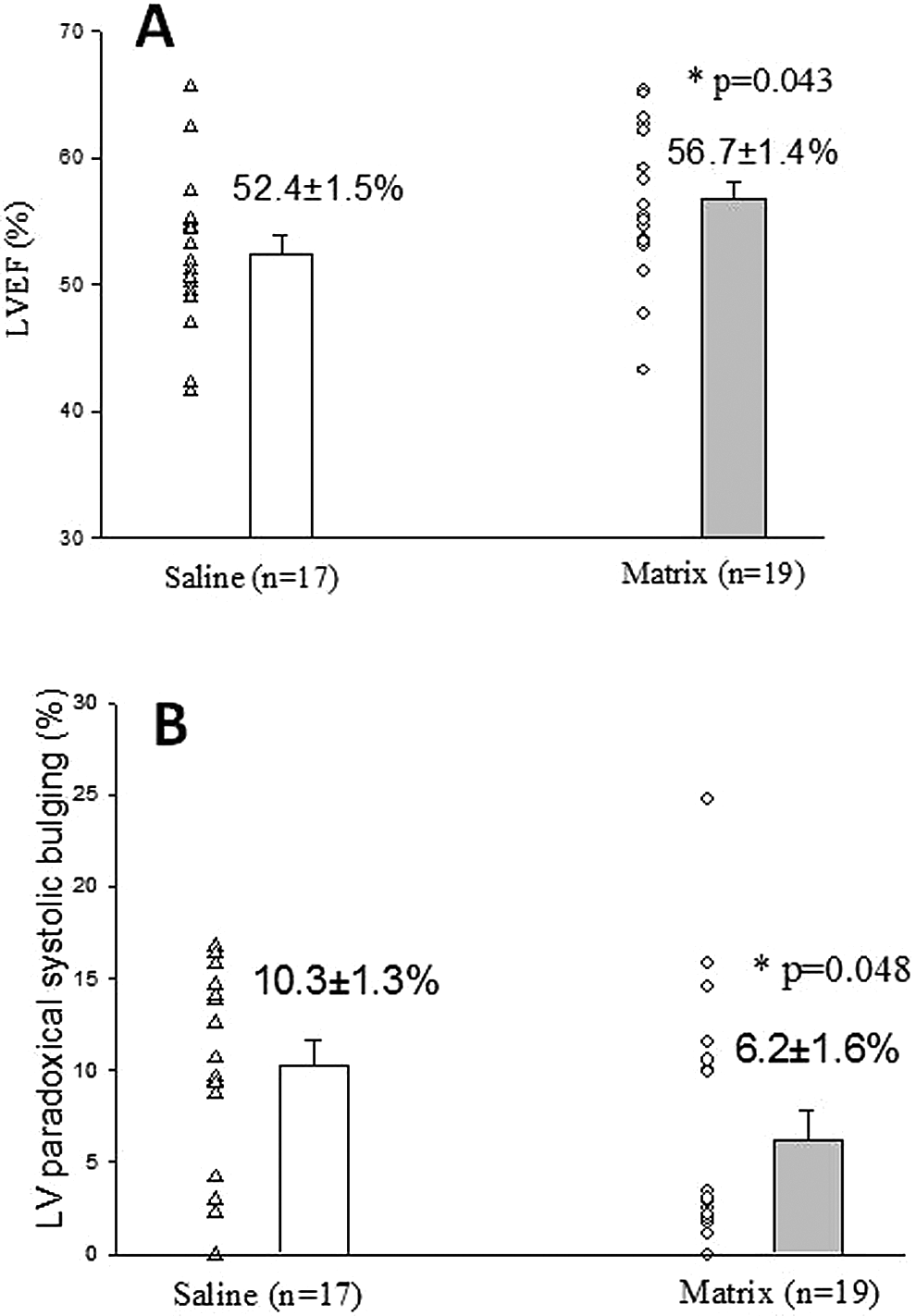
Cardiac function and left ventricular (LV) wall motion assessed by LV ventriculography. Panel A: left ventricular ejection fraction (LVEF) calculated by angiography at 6 weeks after matrix or saline injection directly into the scar area of myocardial infarction in rats. Matrix significantly increased the LVEF (P = .043). Panel B: The extent of paradoxical systolic bulging is expressed as (circumference of the bulging segment/total LV systolic circumference)×100%. Matrix implantation significantly prevented paradoxical LV systolic bulging in the matrix-treated group compared to the saline-treated group (P = .048).
Hemodynamics
No significant differences were noted in heart rate, systolic and diastolic blood pressure, and ±dp/dt between the 2 groups (Table 1).
Parameters of LV Function and Postmortem Morphometry.

Abbreviation: LV, left ventricle.
aStatistically significant.
Postmortem LV Volumes, Scar Thickness, Infarct Sizes, and Expansion Index
There was a nonsignificant trend toward smaller postmortem LV volume in the matrix-treated group (349 ± 12 μL) compared to the saline group (371 ± 11 μL; P = .20). At 6 weeks, the scars in the control group appeared to be composed primarily of collagen and were thin. Histological analysis revealed visibly thicker scars (stained red for collagen by Picrosirius red staining) in the matrix group by qualitative assessment (Figure 4). By quantitative analysis, matrix injection significantly increased scar thickness (602 ± 29 μm) compared to the saline group (484 ± 30 μm, P = .0084; Table 1, Figures 4 and 5). The infarct size, expressed as percentage of total LV circumference, was similar between the saline group (48.0% ± 1.2%) and the matrix group (45.7% ± 2.2%, P = .37). The expansion index as defined by Hochman and Choo, 14 which is expressed as (LV cavity area/total LV area × septum thickness/scar thickness), was significantly reduced in the matrix group (Table 1), mainly due to the increased wall thickness in the matrix-treated group. No evidence of an inflammatory response was observed within the infarct area in either of the groups. By 6 weeks after infarction, inflammation is not expected.

Representative slices of hearts stained with Picrosirius red staining. Muscle cells stain yellow while collagen stains red. A, Heart received saline. B, Heart received matrix. Note that in the red stained scar area, the infarcted wall is thicker in the heart that received matrix (B) compared to the heart that received saline (A). Scale bar = 5 mm.

Scar thickness, average of the measurements at 5 equidistant points of the infarcted left ventricular wall, is significantly higher in the matrix group compared to the saline group (P = .0084).
Blood Vessel Density and Recruitment of Endogenous Stem Cells in Infarcts
The density of blood vessels that contained perfused blue particles in the scar area was 189 ± 12 vessels/mm2 in the control group (n = 17) and 165 ± 23 vessels/mm2 in the matrix group (n = 19; P = .077). The number of c-kit+ cells was similar between the control group (105 ± 13 positive-stained cells per ×200 image field, n = 6) and the matrix-treated group (111 ± 11 positive-stained cells per ×200 image field, n = 6; Figure 6).

Immunohistochemical staining with primary antibody against c-kit of the rat hearts (A) control received saline and (B) received matrix implantation. The red arrows identify positive c-kit stained cells (brown color stained) within the infarct area (magnification 400×, scale bar = 20 µm).
Discussion
This study demonstrated that implantation of heart tissue-derived decellularized matrix into the scar of a myocardial infarction significantly increased scar thickness, prevented infarct expansion and paradoxical LV systolic bulging, and improved LV EF after myocardial infarction compared to saline injection in a rat model. This beneficial effect was observed independent of recruiting c-kit+ stem cells to the infarct.
Biomaterial Implantation Has Similar Effects to That of Cell Therapy for Myocardial Infarction
Our research group has previously demonstrated that cell transplantation therapy is able to thicken the LV infarct wall, reduce paradoxical systolic bulging of the infarct wall, and enhance LV EF in experimental myocardial infarction model (for review, see Ref 15). The limitations of cell therapy include the time it takes to process cells; immune rejection to the donor cells; poor retention of cells with loss through the venous vasculature and/or death of cells; potential of infection, and potential of teratomas. Compared to cell transplantation, biomaterial implantation has none of these limitations, and can be made ready-to-use. For now, numerous noncellular injectable biomaterials, such as hyaluronic acid, 2 fibrin glue, 16 collagen, 17 alginate, 18 small intestine ECM, 3 pericardial ECM, 4 self-assembling peptides, 19 platelet gel, 20 and ventricular ECM, 7 have been evaluated in different animal myocardial infarction models, and some similar effects to that of cell therapy have been observed (Table 2). Among these biomaterials, ECM may be one of the most promising materials for the repair of damaged myocardium. Our present study demonstrated that myocardial ECM implantation improved cardiac function in a coronary ligation rat myocardial infarction model, and for the first time showed that ECM implantation prevented paradoxical LV systolic bulging. This may be a very important aspect of how ECM improved LV EF, and eventually reduced infarct expansion and remodeling. The reduction of paradoxical LV systolic bulging may be an important but passive mechanical phenomenon. By reducing paradoxical LV systolic bulging, ECM helps prevent negative work and reduces LV wall stress. In our study, the recruitment of stem cells or blood vessels was not necessary to prevent paradoxical LV systolic bulging.
Main Findings in Experimental Biomaterial Therapy for Myocardial Infarction.

Abbreviations: ECM, extracellular matrix; LV, left ventricular; MI, myocardial infarction; NF, nanofiber.
New Blood Vessel Formation Within the Implanted Biomaterials
Injected biomaterials might promote endothelial cell infiltration and neovascularization within the microenvironment of implanted biomaterials. Davis et al 21 injected self-assembling peptides into the normal LV-free wall in mice. The injected peptides formed detectable nanofiber microenvironments within the myocardium. The self-assembling peptide nanofiber microenvironments recruited progenitor cells that expressed endothelial and smooth muscle cell markers. Functional α smooth muscle actin-positive vessels within the myocardium formed 4 weeks later, including some with red blood cells within, suggesting that the new blood vessels were capable of connection to the host vasculature. Huang et al 22 occluded the left coronary artery for 30 minutes followed by reperfusion in the rat model. One week later, collagen, matrigel, fibrin glue, or saline were injected into the infarcted area. Injection of collagen, matrigel, and fibrin glue into the infarct zone yielded significantly higher levels of capillary formation compared to the saline at 5 weeks posttreatment. However, the degree of angiogenesis was not significantly different among the collagen, matrigel, and fibrin glue. Singelyn et al 5 harvested hearts from pigs and decellularized cardiac tissue to obtain injectable myocardial decellularized matrix. In vitro cell culture studies showed that the myocardial matrix promoted migration of both cultured rat aortic smooth muscle cells and human coronary artery endothelial cells. When the myocardial matrix was injected into the normal LV wall of male Sprague-Dawley rats, a significant increase in arteriole formation within the matrix was observed at 11 days postinjection. In our present study, injected materials did not promote neovascularization. Some characterization of the materials, such as the composition, structural properties of the gelled matrix, and concentration of the materials, may have affected the cellular infiltration and blood vessel formation. For example, our previous studies 17,23,24 demonstrated that the concentration of implanted collagen can influence blood vessel formation. Our study suggests that the cardiac ECM that we utilized prevented LV remodeling without requiring an increase in neovascularization.
Recruitment of Endogenous Stem Cells by Implanted Biomaterials
Another theoretical benefit of biomaterial injection is to stimulate the recruitment of endogenous stem cells to the injury site to contribute to endogenous tissue regeneration and improvement of cardiac function. C-kit is a receptor tyrosine kinase family member that is expressed in cardiomyocyte precursors in the adult heart. C-kit+ cardiac cells may possess the fundamental properties of stem cells. 25 Endogenous c-kit+ cells may be stimulated to amplify and differentiate into cardiomyocytes contributing to myocardial repair in infarcted hearts. 26 Zhao et al 3 subjected rats to 45 minutes of left coronary occlusion followed by reperfusion for 42 days. Small intestine ECM or saline was injected into the previously ischemic myocardium after reperfusion. Compared to saline, implanted matrix significantly increased the population of c-kit+ cells, myofibroblasts, and macrophages in infarcted myocardium and enhanced angiogenesis. The results suggested that matrix injection improved cardiac function through enhanced recruitment of c-kit+ cells, myofibroblasts, and macrophages. Ou et al 27 injected matrigel or saline into myocardial infarction immediately after left coronary ligation in rats. Four weeks later, matrigel significantly increased the infarct wall thickness and improved LV function. Hearts that received matrigel exhibited higher capillary density and significantly more numerous c-kit+ stem cells, suggested that intracardiac matrigel injection improved myocardial function by the improved recruitment of CD34+ and c-kit+ stem cells. In contrast, in our study while we did observe c-kit+ cells within the infarcted scar, there was no evidence that ECM enhanced recruitment of these cells above and beyond infarction alone. Therefore, any benefit of the ECM cannot be attributed to an increase in c-kit+ cells in our study.
Biomaterial Implantation Prevents Postinfarction Paradoxical LV Systolic Bulging
Paradoxical LV systolic bulging of due to coronary artery occlusion is a passive movement, 28 has been considered to be a potential adverse factor for LV function, 29 and contributes to myocardial infarction expansion and aneurysm formation. 30 Biomaterial implantation thickened the infarcted wall, acting as a baffle that prevented or lessened paradoxical LV systolic bulging, which then improved LV systolic function and EF, and helped to attenuate pathologic LV remodeling. 17 Using a 3-dimensional finite element model simulation of an ovine heart with an anteroapical infarct, Wall et al 31 investigated the short-term ventricular mechanical effects of implanted noncontractile materials such as synthetic ECMs or cellular masses. The results of this simulated study indicated that the injection of small volumes of noncontractile material to a damaged LV wall had important effects on cardiac mechanics. However, Rane et al 32 injected nondegradable synthetic polyethylene glycol (PEG) gels or saline into 1-week-old myocardial infarction in female Sprague Dawley rats. Seven weeks later, compared to saline, PEG gel injection significantly increased infarct wall thickness, but did not prevent negative LV remodeling or improve cardiac function assessed using magnetic resonance imaging. Histology showed that PEG gel did not induce postmyocardial infarction neovascularization and cell infiltration within the infarct area. These results suggest that not all acellular materials confer a benefit. In our present study, we demonstrated that implantation of heart tissue-derived decellularized matrix thickened the LV infarcted wall, prevented paradoxical systolic bulging, and improved LV EF after myocardial infarction in rats, which is consistent with what has been previously seen with other materials such as collagen. 17 Unlike other studies, our data support the concept that thickening the infarct wall by biomaterials could preserve cardiac function through passive structural reinforcement, which was independent of recruiting c-kit+ stem cells or neovascularization.
In conclusion, our data showed that implantation of heart tissue-derived decellularized matrix into the scar after myocardial infarction thickened the infarcted ventricular wall, prevented paradoxical LV systolic bulging, and improved LV EF compared to saline-treated animals. However, these benefits were not associated with enhanced angiogenesis or the recruitment of endogenous stem cells to the injury site. Although this study suggests that implantation of biomedical materials into a myocardial infarction as an internal support can provide a novel approach to preserve cardiac function after myocardial infarction at the earlier stage of postinfarction, the long-term effects and underling mechanisms need further investigation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Los Angeles Thoracic and Cardiovascular Foundation and Celavie Biosciences.
