Abstract
There is limited data on the use of cardiovascular disease (CVD) risk factor medications following renal transplant, especially when comparing use across ethnicities. The aim of this study was to compare the incidence, treatment, and impact of CVD between ethnicities in kidney transplant recipients. This was a retrospective cohort study of adults who underwent transplant between 2000 and 2008 within our academic medical transplant center. Pediatrics, multiorgan transplants, and those lost to follow-up were excluded. Data collection included all transplant and sociodemographic characteristics, medication use, CVD risk factor management, and follow-up events, including acute rejection, graft loss, and death. A total of 987 patients were included and followed for a mean of 6.7 ± 3.0 years. The baseline demographics revealed black patients were equally likely to have preexisting CVD (24% vs 25%, P = .651), but more likely to have preexisting diabetes (35% vs 23%, P < .001) or hypertension (97% vs 94%, P = .029). Black patients had poorer treatment of CVD risk factors, with lower rates of control of diabetes (35% vs 51%, P < .05) and dyslipidemia (37% vs 42%, P < .05). Black renal transplant recipients who had preexisting CVD had reduced graft survival rates compared to white patients (10-year rate 50% vs 60%, P = .033), but similar rates of graft survival were found in those without CVD (10-year rate 70% vs 71% in white patients, P = .483). CVD is common in transplant recipients, with black patients having higher rates and poorer control of diabetes and dyslipidemia.
Introduction
Black renal transplant recipients experience a disproportionately higher incidence of graft failure. Based on the most recently published scientific registry of transplant recipients data, black recipients have a 9.8% lower rate of graft survival at 5 years posttransplant (76.1% vs 66.3%), which increases to 13.8% at 10 years (50.9% vs 37.1%). Numerous studies have been conducted over the past 2 decades unraveling the likely mechanisms involved with this disparity with most focusing on immunologic factors, access to transplant barriers, and socioeconomic elements. 1 –7 Despite this, the impact of ethnicity on graft outcomes has not diminished, and there are few interventional studies demonstrating improved outcomes in black transplant recipients. 8
Although it is well known that black renal transplant recipients are significantly more likely to have preexisting cardiovascular disease (CVD) risk factors, including hypertension and diabetes at the time of transplant, 2 there is little research analyzing the direct effects these comorbidities contribute to the overall disparity seen in transplantation. Therefore, the aim of this study was to analyze the incidence, treatment, and goal attainment of hypertension, diabetes, and dyslipidemia in black and white renal transplant recipients and to determine the impact CVD has on graft survival.
Methods and Materials
Study Design and Patients
This was a single-center retrospective cohort study of renal transplant recipients who underwent transplantation between 2000 and 2008. Patients were excluded if they were less than 18 years of age at the time of transplant, received a multiorgan transplant, or were of a race/ethnicity other than white or black. Data were collected at baseline, from time of kidney transplant, to death, loss to follow-up, or end of the study (April 2012). If multiple key factors or outcomes were missing or unobtainable for a specific patient, they were also excluded from this study. For categorization and analysis of data, patients were divided into 2 groups: black and white renal transplant recipients.
Data Collection
All data were collected in a retrospective cohort manner from patient electronic and hard copy medical records. Initially, baseline patient demographics and transplant characteristics were recorded from the past medical history taken at the time of evaluation and updated at the time of transplant. All clinical posttransplant events were captured, including acute rejection rates, graft failure, and death. Laboratory, vitals signs, and clinical assessments were captured daily for the first week posttransplant, then weekly for 1 month, then at 3 and 6 months, and finally yearly thereafter. For analysis, values were averaged for each year independently and then averaged across the entire follow-up time period for each patient.
Variable Definitions
Preexisting diabetes
This was defined as patients who had a clinical diagnosis of diabetes mellitus (DM) prior to transplant. The diagnosis of DM was documented in the medical record and included patients with both diet control and those who required oral hypoglycemic agents or insulin for DM management.
Hypertension control
Control of blood pressure was defined based on the National Kidney Foundation, 9 Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7), 10 and American Society of Transplantation (AST) guideline recommendations, 11 which is achieving an average systolic blood pressure of less than 130 mm Hg and a diastolic blood pressure of less than 80 mm Hg across the follow-up period. For treatment comparisons, medications were grouped by class in a similar fashion to JNC 7 guidelines.
Compelling indications for hypertension medications
The need for medications to treat hypertension with compelling indications was based on the current JNC 7 recommendations. 10 For β-blockers, the compelling indications were a history of congestive heart failure or acute myocardial infarction; for angiotensin-converting enzyme inhibitors (ACE-Is) or angiotensin receptor blockers (ARBs), the indications include a history of congestive heart failure, acute myocardial infarction, diabetes, or chronic kidney disease (estimated glomerular filtration rate [eGFR] <60 mL/min based on the modified Modification of Diet in Renal Disease [MDRD] equation and averaged across the follow-up period).
Dyslipidemia control
Control of lipoproteins was defined based on the National Cholesterol Education Program (NCEP) III 12 and AST guidelines, 11 when the low-density lipoprotein (LDL) level was less than 100 mg/dL, unless the patient has diabetes with known CVD, in which case the goal LDL was less than 70 mg/dL. If a patient had an LDL of greater than either of those values at least twice, then statin therapy was considered a compelling indication.
Diabetes control
Controlled diabetes was defined based on a modified American Diabetes Association recommendation, 13 which was an average hemoglobin A1c of less than 7.0% or an average blood glucose less than or equal to 150 mg/dL throughout the study period (for those who did not have hemoglobin A1c recorded in the medical records).
Graft failure
Graft failure was defined as return to chronic dialysis, retransplant, or death.
Immunosuppression
Patients received induction therapy with either thymoglobulin 1.5 mg/kg intravenously (IV) daily for 3 to 5 doses, daclizumab 1 mg/kg IV on day 0 and day 7 posttransplant, or basiliximab 20 mg IV on day 0 and day 4 posttransplant. Choice of induction therapy was based on the protocols that utilized thymoglobulin for patients with high immunologic risk (retransplantation, cold ischemia time >24 hours, or panel reactive antibody >20%) and an interleukin 2 receptor antibody in all other patients. Maintenance immunosuppression from 2000 to 2004 consisted of cyclosporine adjusted to maintain target through whole blood concentrations between 200 and 275 ng/mL for weeks 1 through 6, 125 to 225 ng/mL for weeks 7 through month 12, and >70 ng/mL after 1 year. Tacrolimus became the agent of choice in 2005. Tacrolimus was adjusted to maintain target through whole blood concentrations between 8 and 12 ng/mL for weeks 1 through 6, 6 to 12 ng/mL for weeks 7 through month 12, and >5 ng/mL after 1 year. In addition, all patients received mycophenolate mofetil 1 g twice daily and prednisone titrated to 5 mg daily by day 90 posttransplant, with tapers below 5 mg occurring rarely. Very few patients were taken off corticosteroids.
Outcome Measures
The primary outcomes for this study were to analyze the incidence and treatment of CVD and associated risk factors, which included hypertension, diabetes, and dyslipidemia between black and white transplant recipients. Analysis of graft survival rates between groups based on a history of CVD or risk equivalent was also conducted.
Statistical Analysis
Sample characteristics were compared across the groups using the Student t test for continuous variables with normal distribution and the Wilcoxon rank sum test for variables without normal variance. The Fisher exact test was used to compare the groups for categorical variables. Univariate, unadjusted Kaplan-Meier survival analysis was used to compare graft survival of the 2 groups (black and white recipients) for patients with and without preexisting CVD or CV risk equivalence. The log-rank test was used to compare factors. Statistical significance was based on a P value of less than .05. All data were collected manually by review of medical records; patient-specific information was deidentified and recorded on data collection sheets and input into a spreadsheet (Excel, MS Office, version 2010, Microsoft Corporation, Seattle, Washington); SPSS (version 20, SPSS Inc, Chicago, Illinois) was used for the analysis.
Results
A total of 1187 renal transplants occurred between 2000 and 2008. After removing excluded patients (pediatrics and multiorgan recipients) and patients of non-black or non-white origin, a total of 987 patients were included in this analysis (578 [59%] black and 409 [41%] white recipients). Table 1 displays the baseline characteristics of the 2 groups. Demographic comparisons revealed black renal transplant recipients were, on average, less likely to be male (56% vs 62%, P = .011), younger (48 vs 50 years, P < .001), and had a higher body mass index (26 vs 25, P = .039). Baseline transplant characteristics confirmed that black renal transplant recipients had higher human leukocyte antigen mismatches (4 vs 3, P < .001), waited a longer time on dialysis prior to transplant (3.9 vs 2.0 years, P < .001), and received less living donor transplants (7% vs 31%, P < .001). Finally, comparisons of baseline comorbidities reveal black recipients had a lower incidence of smoking (25% vs 35%, P < .001), but a larger burden of significant CVD and CVD risk factors, with a higher incidence of heart failure (4% vs 2%, P = .047), hypertension (97% vs 94%, P = .029), and pretransplant diabetes (35% vs 23%, P < .001).
Baseline Demographic and Transplant Characteristics.
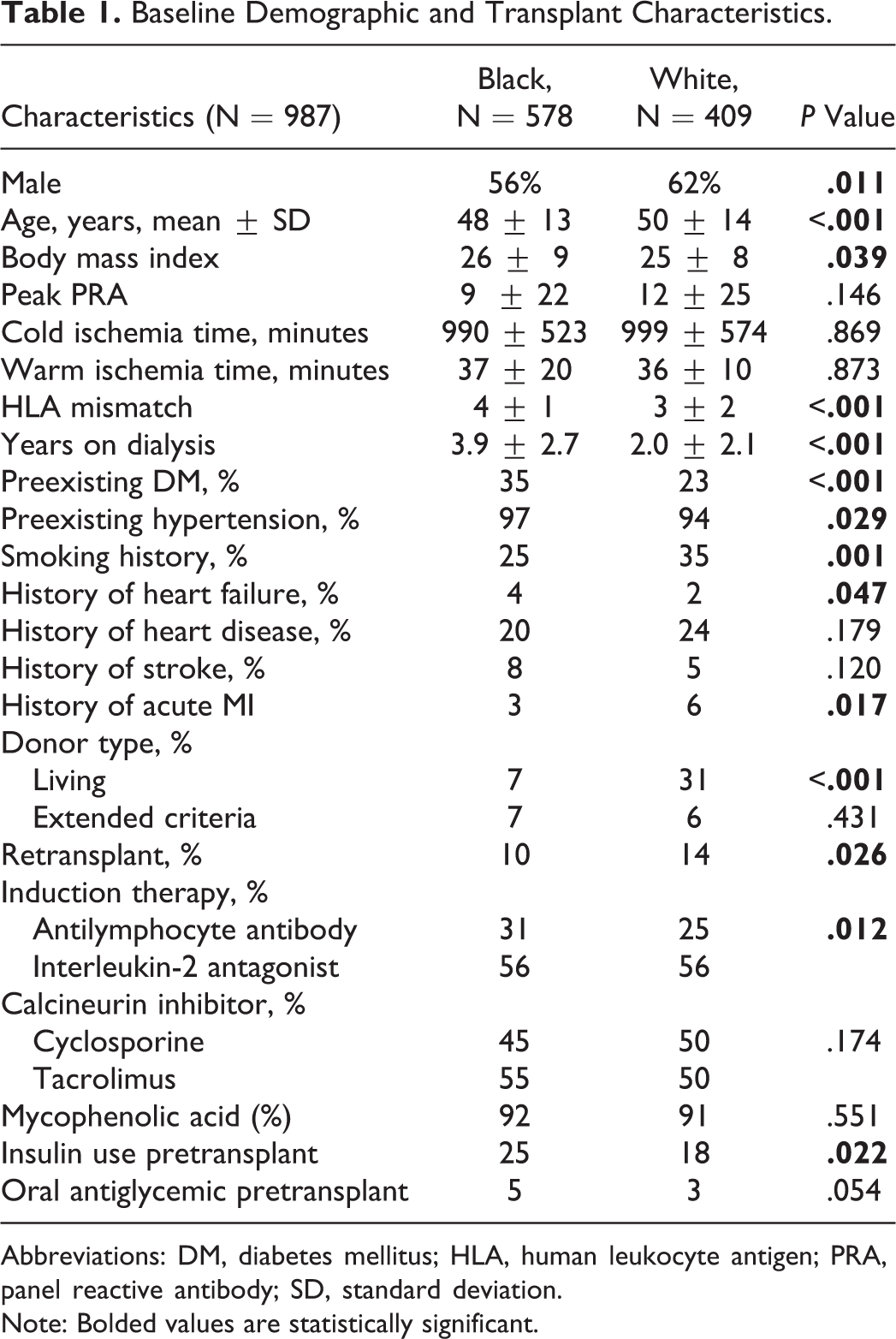
Abbreviations: DM, diabetes mellitus; HLA, human leukocyte antigen; PRA, panel reactive antibody; SD, standard deviation.
Note: Bolded values are statistically significant.
Figure 1 displays the medications used for the treatment of CVD risk factors and the rates of achievement of risk factor control. For hypertension management, black recipients were more likely to receive vasodilators, clonidine, dihydropyridine (DHP), and non-DHP calcium channel blockers (CCB), and averaged more antihypertensive agent utilization (2.3 ± 1.2 vs 1.9 ± 1.1, P < .001). Use of β-blockers, diuretics, ACE-Is, and ARBs were similar between the groups. The use of insulin and oral antihyperglycemic agents for the management of diabetes was similar between the groups. For the treatment of dyslipidemia, white recipients were more likely to receive a fibrate (24% vs 11%, P < .05) or fish oil (14% vs 10%, P < .05), with similar use of statins, ezetimibe, and niacin between the groups. Attainment of CVD risk factor control revealed that black recipients were less likely to be at desired goals for both diabetes (35% vs 51%, P < .05) or LDL (37% vs 42%, P < .05), with a trend toward reduced control of hypertension (32% vs 37%, P = .117, respectively).
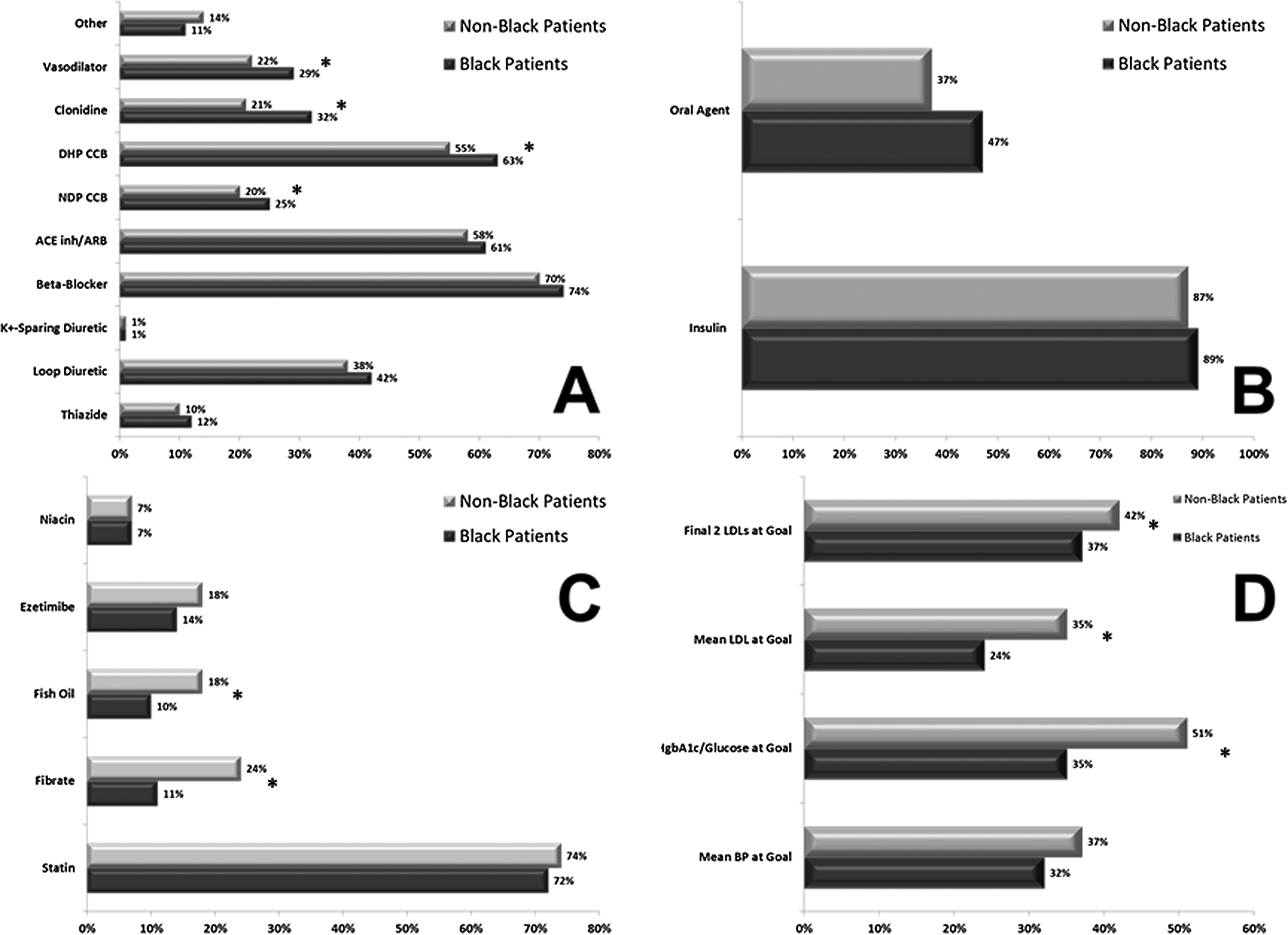
Hypertension, diabetes, and dyslipidemia medication use and attainment of goals. These charts display the medication utilization for hypertension (A), diabetes (B), and dyslipidemia (C) and the ability to achieve goals for each cardiovascular disease (CVD) risk factor (D) between black and white transplant recipients. *P < .05.
Table 2 displays the utilization of medications for compelling indications and a number of clinical surrogate markers. Black kidney transplant were more likely to have a compelling indication for statin therapy (45% vs 33%, P < .001), with the utilization of statins for compelling indication being similar in these patients (69% vs 71%, P = .646). Black patients were equally likely to have a compelling indication for β-blocker therapy (18% vs 22%, P = .075) and utilization of this therapy when a compelling indication was present (64% vs 74%, P = .163). Similar equality between the groups was noted for ACE-I or ARB indication and use. Analysis of surrogate markers for control of the disease states demonstrated that black renal transplant recipients had higher mean systolic and diastolic blood pressures and LDL cholesterol levels (Table 2), with more white patients having chronic kidney disease (eGFR <60 mL/min, 74% vs 64%, P = .032), despite black recipients having a higher mean serum creatinine (1.9 ± 0.8 vs 1.7 ± 0.6 mg/dL, P < .001).
CVD Medication Utilization and Associated Outcomes.
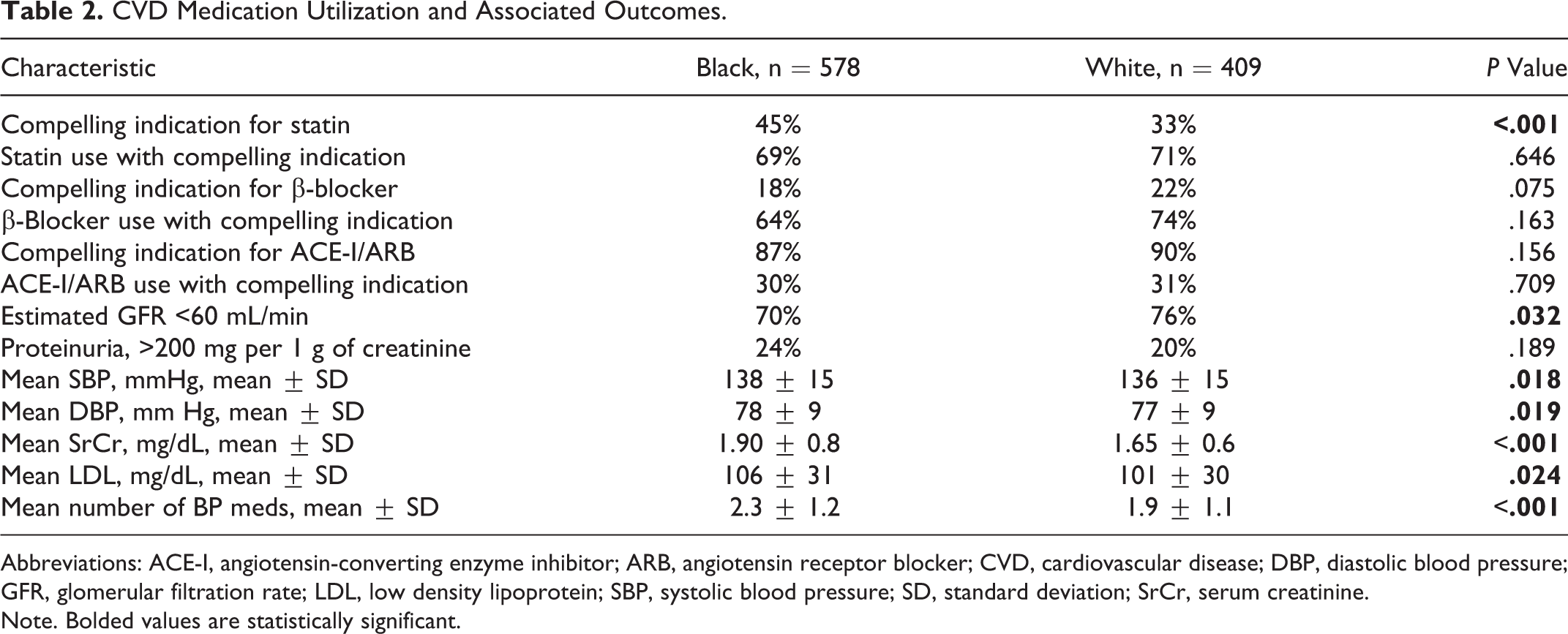
Abbreviations: ACE-I, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CVD, cardiovascular disease; DBP, diastolic blood pressure; GFR, glomerular filtration rate; LDL, low density lipoprotein; SBP, systolic blood pressure; SD, standard deviation; SrCr, serum creatinine.
Note. Bolded values are statistically significant.
Figure 2 displays the Kaplan-Meier graft survival analysis comparing groups based on the presence (Figure 2A) or absence (Figure 2B) of CVD or risk equivalence (defined as a pretransplant history of acute myocardial infarction, revascularization, coronary artery bypass graft, stroke, peripheral vascular disease, aortic aneurysm, or diabetes). Black transplant recipients had significantly reduced graft survival rates if CVD or risk equivalence was present (10-year graft survival 50% in black patients [n = 309] and 60% in white patients [n = 185], P = .033), whereas the graft survival rates were similar between transplant patients without preexisting CVD present (10-year graft survival 70% in black patients [n = 224] and 71% in white patients [n = 269], P = .483).

Graft survival rates. These charts display the Kaplan-Meier curves for graft survival in both black and white transplant recipients dichotomized for patients with preexisting cardiovascular disease (CVD) or risk equivalence (A) and those without (B).
Discussion
The results of this study demonstrate that black renal transplant recipients have an increased incidence of CVD risk factors, including diabetes, hypertension, and compelling indications for statin therapy. Additionally, these data reveal that black recipients have reduced CVD risk factor treatment and goal attainment, especially as it pertains to diabetes and dyslipidemia. Finally, the Kaplan-Meier analyses demonstrate that the sheer presence of preexisting CVD reduces graft survival rates in a more substantial manner in blacks, when compared to white renal transplant recipients. These data, taken in their entirety, provide evidence that CVD and risk factor treatment is likely a prominent factor in the graft survival disparities seen within black renal transplant patients.
Graft survival disparities seen in black renal transplant patients is well documented 8 and has been an extensive area for exploratory mechanistic studies since the landmark study performed by Opelz and colleagues 14 brought this public health issue to light. However, the vast majority of studies analyzing graft disparities in renal transplantation based on ethnicity have focused on immunologic, socioeconomic, or barriers to access as prime mechanisms to account for this issue. 2,15 Yet, there is limited data exploring the impact of CVD or risk factor treatment on this well-known disparity. Hypertension has probably been the most robustly explored area of analysis within this realm, and previous studies have demonstrated that black patients who underwent renal transplant have more aggressive hypertensive patterns, which require more intensive treatment and despite this, do not reach goal values as often compared to white recipients. 15 –19 The data we present in this study validate these previous findings with a much larger data set, with higher numbers of black recipients.
The detrimental impact of diabetes on graft survival is also well documented 20,21 ; however, there are extremely limited analyses exploring this in the context of ethnic disparities. Most studies fail to capture enough black transplant recipients to adequately power this type of analysis. 22 –24 Additionally, this is the first study that we are aware of that captures the rates of control of diabetes following renal transplantation and compares this across patient ethnicity. These findings provide further evidence, which is well documented in the general nontransplant population demonstrating black patients have reduced rates of control of diabetes. 25 –31 Since it is well known that diabetes reduces graft survival, 20 the impact of this disease state on ethnic disparities should not be discounted, and may be one of the major nonimmunologic or socioeconomic contributors to the reduced graft survival rates.
The ethnic disparities seen with the identification, treatment, and goal attainment of dyslipidemia demonstrated within this study are also novel, and to our knowledge has not been reported within the renal transplant population. However, the results of this study do correlate well with studies in the general population with regard to need for statin therapy and the under treatment of dyslipidemia within black patients. 19,32,33 Because there is well-designed trials demonstrating black patients, when compared to white patients, have equivalent reductions in CVD events and CVD-related death with statin therapy, the disparity in treatment of dyslipidemia represents an area for targeted intervention to eliminate this bias. 28,34,35
Recently, the results, with regard to the use of CVD medications, of the Patient Outcomes in Renal Transplantation (PORT) study were published. 36 This was a multicenter, large-scale, retrospective analysis of 10 transplant centers studying the CVD outcomes and medication use of adult renal transplant recipients. Although only 8.5% of the patients in this study were black, the data presented in the PORT study are similar to the results displayed here. Black patients were less likely to receive statin therapy and more likely to receive CCB antihypertensive therapy. However, obtainment of goals or analysis of diabetes outcomes was not assessed in the PORT study. The PORT results, taken in context with previous studies and the results of the data presented in this study, demonstrate that black renal transplant recipients clearly have a significantly greater burden of CVD risk factors, which are not adequately treated to well-defined goals. The impact of this disparity on graft outcomes across the renal transplant population, and demonstrated in the Kaplan-Meier survival curves within this study, is likely substantial. The CVD risk factor treatment and goal obtainment may represent one of the more important nonimmunologic factors associated with reduced graft survival rates among this high-risk population of renal transplant recipients. 2
There is nearly a complete lack of interventional studies within kidney transplant recipients demonstrating improved outcomes with a focused approach on treating CVD risk factors, including hypertension, diabetes, and dyslipidemia. One small prospective interventional trial conducted by Chisholm and colleagues 37 demonstrated improved blood pressure control in black renal transplant recipients through a pharmacist’s interventions in improving the identification and resolution of medication-related problems. Interventional studies, conducted in a similar manner but on a larger scale, are desperately needed to improve the identification and treatment of these CVD risk factors.
There are several limitations in this study worthy of discussion. First, because this was not a prospective study, we were not able to capture all possible confounding variables, including, and most importantly, socioeconomic status (SES). Since this has been demonstrated in the previous studies to contribute to graft disparities, 15,38,39 we felt that including SES within this data was not essential, but admittedly, would add strength to the overall analysis. Second, data collection was performed by chart review and relied on patient’s recall of medical history for baseline data, which makes our findings somewhat prone to recall bias, especially as it relates to family history or previous CVD medical history. Third, hemoglobin A1c was not available for all patients and was sparsely measured in others; therefore, glycemic control was determined by either mean A1c or average serum glucose if the A1c was not available for analysis. By using average blood glucose for some patients, we likely underestimated actual glycemic control since there were limited numbers of values for patients who were more than 1 year from transplant. Another limitation is that the survival analysis conducted for patients with or without CVD risk equivalence was determined in an unadjusted fashion, and there are known differences between black and white kidney transplant recipients which may also significantly influence graft survival. Therefore, this analysis cannot feasibly conclude that the presence of CVD explains the increased rates of graft loss in black patients. Finally, after the first year posttransplant, the data were captured at annual transplant clinic visits, and therefore, changes to medications that occurred between clinic visits may not have been accounted for by this study. Due to the retrospective nature of this analysis, additional data points could not be captured. However, the vast majority of changes to CVD risk factor medications usually occurs early posttransplant and these data were captured.
Conclusion
The CVD is common in transplant recipients, with black patients having higher rates and poorer control of diabetes and dyslipidemia. Black kidney transplant recipients with preexisting CVD have poorer graft survival rates compared to white patients. These data demonstrate the need to study the impact of improved CVD risk management in black renal transplant recipients as a potential mechanism to mitigate graft disparities within this population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was conducted with the support of Novartis Pharmaceuticals.
