Abstract
Introduction
Sodium-glucose cotransporter- 2 (SGLT2) inhibitors have become a cornerstone in heart failure (HF), Type 2 diabetes mellitus (T2DM), and cardiovascular disease (CVD) management. In the current retrospective study, we aimed to assess efficacy and safety of SGLT2 inhibitors early following acute myocardial infarction (AMI).
Methods
Patients with T2DM hospitalized for AMI in 2017-2020 were divided according to SGLT2 inhibitors therapy status on discharge (with vs without therapy). Primary outcome was defined as a composite of hospitalizations for HF, recurrent AMI, and cerebrovascular accident (CVA). Secondary outcomes included hospitalizations for any cause, total cumulative number of hospitalizations, and all-cause mortality.
Results
A total of 69 patients (mean age 59.2 ± 8.2 years) with AMI discharged with SGLT2 inhibitors were compared to 253 patients (mean age 62.5 ± 9.8) with no SGLT2 inhibitors. During the first year post-AMI, 4 (5.8%) patients in the treatment group and 16 (6.3%) in the control group were hospitalized for CV events (p = 1.0). Patients in the SGLT2 inhibitors group had lower rates of hospitalization for any cause (31.9% vs 47.8%, P = 0.02), with no change in mortality (0% vs 3.6%, P = 0.21). After multivariate regression analysis, only female gender was associated with increased risk for readmission, mainly due to urinary tract infections. No events of diabetic ketoacidosis (DKA) or limb amputation were reported.
Conclusions
We found that early initiation of SGLT2 inhibitors in T2DM patients following AMI is safe and decreases the risk of hospitalization for any cause.
Introduction
Type 2 diabetes mellitus (T2DM) is common among patients with cardiovascular disease (CVD) and is associated with increased risk of mortality.1–3 Sodium-glucose Co transporter-2 (SGLT2) inhibitors have become a cornerstone in treating patients with heart failure (HF) and CVD owing to their pleotropic effects.4–6 In the two pivotal trials, empagliflozin and dapagliflozin reduced CV events in patients with T2DM and CVD or at high risk for CVD.4,5 These drugs also showed positive outcomes in patients with HF across the whole spectrum of ejection fraction and in patients with chronic kidney disease (CKD).7–10 Suggested mechanisms for the cardioprotective properties of SGLT2 inhibitors include diuresis, weight reduction, anti-fibrotic effects, modulation of cardiac energy sources and neurohormonal pathways, reducing inflammation, anti-arrhythmic effects, and regulating endothelial function.11–17 Overall, the safety profile of these drugs is excellent with few side effects including genitourinary infections and rare cases of diabetic ketoacidosis (DKA) in patients with T2DM.18–21 The use of SGLT2 inhibitors in the acute setting of acute myocardial infarction (AMI) is still under investigation due to several safety concerns. First, patients with AMI may present with acute HF (particularly after anterior wall infarction), therefore SGLT2 inhibitors should be initiated only after achieving clinical stabilization.22–24 Second, SGLT2 inhibitors may increase the risk of contrast induced nephropathy in patients with AMI treated with percutaneous coronary intervention (PCI), particularly during complex procedures with large volumes of contrast media and in patients with T2DM and with concomitant use of diuretic therapy. 25 However, it should be noted that chronic use of SGLT2 inhibitors is associated with improved renal outcomes.9,10 Moreover, in recent study, patients on chronic SGLT2 inhibitors had lower risk for contrast nephropathy following PCI during the acute setting of AMI. 26 Third, patients on SGLT2 inhibitors referred to surgical revascularization following coronary angiography are at increased risk of DKA. In the current retrospective study, we aimed to evaluate the safety and efficacy of early SGLT2 inhibitors treatment (on discharge) in patients with T2DM hospitalized for AMI.
Materials and Methods
Study Design and Population
The study is retrospective, with the data collected from medical records of hospitalized patients. The cohort included patients with T2DM hospitalized for AMI in the Department of Cardiology at Galilee Medical Center, between 2017 and 2020. We divided the patients into two main groups: patients who received SGLT2 inhibitors on discharge (treatment group) and patients who did not receive this recommendation (control group) after the index event of AMI. Exclusion criteria included renal failure with estimated glomerular filtration rate (eGFR) <45 ml/min/1.73m2), and patients with missing data. Patients in whom myocardial injury was secondary to infectious disease, blood loss, or cardiac arrhythmia (type 2 AMI) were also excluded. Troponin level was measured using high-sensitivity ARCHITECT assay (Abbott) with cut-off values for abnormal levels above 20 ng/L and 30 ng/L for men and women, respectively. Left ventricular ejection fraction (LVEF) was calculated by the Simpson's biplane method in the apical four-chamber and two-chamber views using the formula: LVEF = Left ventricular end diastolic volume-Left ventricular end systolic volume)/ Left ventricular end diastolic volume X100. All echocardiographic studies were performed and interpreted by an expert cardiologist.
Definition of AMI
AMI was defined according to the fourth universal definition of myocardial infarction as a rise and/or fall of troponin level with at least one value above the 99th percentile upper range limit with at least one of the following: (a) symptoms of myocardial ischemia, (b) new ischemic ECG changes, (c) development of pathological Q waves in ECG, (d) evidence of new loss of viable myocardium or new regional wall motion abnormality on imaging modalities, (e) identification of coronary artery thrombus during angiography.
Outcomes
The primary outcome was defined as a composite of hospitalizations for heart failure (HF), recurrent AMI, and cerebrovascular accident (CVA) in two timeframes, during the first year and after the first year post-AMI. Secondary outcomes included hospitalizations for any causes, total cumulative number of hospitalizations, and all-cause mortality.
Statistical Analysis
Continuous variables are presented as mean ± standard deviation (SD) or median with interquartile range (IQR). Qualitative variables are presented as frequencies and percentages. Incidence of CV events was performed with a two-sided 95% confidence interval (CI) for proportions. Comparisons of continuous variables between the two groups were examined with either independent sample t-test or the Wilcoxon Rank-Sum test based on the sample size of the groups and the variables’ distribution shape. Categorical variables were analyzed using Pearson's chi-square or Fisher's exact test (if expectancy < 5). A two-tailed p-value < 0.05 was considered statistically significant. Multivariate logistic regression was used to adjust for possible confounders. All statistical analyses were performed using IBM SPSS Statistics software, version 27. The study was approved by the local ethics committee.
Results
Between 2017 and 2020, 371 patients with T2DM who were hospitalized for AMI were identified. After excluding 49 patients (missing data, type 2 AMI, and eGFR < 45 ml/min/1.73m2), a total of 322 patients (82% male, mean age 62 ± 12.7 years) were included in the final analysis. Of them, 69 (21.4%) were discharged with SGLT2 inhibitors and 253 (78.6%) patients were discharged without initiation or recommendation for SGLT2 inhibitors. The flow chart of the study is depicted in Figure 1.

Flow chart of the study. AMI, acute myocardial infarction; eGFR, estimated glomerular filtration rate; SGLT2, sodium-glucose cotransporter- 2; T2DM, type 2 diabetes mellitus.
29% of the patients were hospitalized for ST elevation AMI and 71% for non-ST elevation AMI. In the treatment group, 65 (94%) received empagliflozin (18 of them as a combined pill with metformin) and 4 (6%) received dapagliflozin. The median follow-up time was 2.6 years (IQR, 1.0-4.1 years). The baseline characteristics of the two groups are given in Table 1.
Baseline Characteristics of the Patients.

ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; CABG, coronary artery bypass grafting; CCB, calcium channel blocker; CVA, cerebrovascular accident; CRTD, cardiac resynchronization therapy; CVD, cardiovascular disease; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; ICD, implantable cardiac defibrillator; LVEF, left ventricular function; NSTEMI, non ST segment elevation myocardial infarction; PCI, percutaneous coronary intervention; SBP, systolic blood pressure; SD, standard deviation; STEMI, ST segment elevation myocardial infarction; TIA, transient ischemic attack.
Primary Outcomes
During the first year post-AMI, the primary outcome occurred in 4 (5.8%) and 16 (6.3%) patients in the treatment and the control group respectively, p = 1.0. Hospitalization rates per person for CV events were also similar between the groups, 0 [0-3], P = 0.99. Likewise, similar results were observed when comparing the rates of hospitalizations per person separately for recurrent AMI, HF, and CVA (median values of 0 [0-2] for AMI, 0 [0-3] for HF, and 0 [0-1] for CVA in the treatment and control groups, P = 1.0, 0.96, and 0.38, respectively). In the second frame of time (after the first-year post AMI), 2 (2.9%) and 11(4.3%) patients in the treatment and control groups, respectively were hospitalized for CV events, p = 0.74. Likewise, the median hospitalization rate per person for CV events during the same period was similar, 0 [0-1] and 0 [0-3] in the treatment and control groups, respectively, p = 0.67.
Secondary Outcomes
During the follow up period, patients in the treatment group had lower rate of hospitalization for any cause (31.9% vs 47.8%, p = 0.02). Hospitalizations per person for any cause during the first year were also lower in the treatment group, 0 [0-3] compared to 0 [0-8] in the control groups, p = 0.038. Hospitalization for any cause included chest pain (26%), urinary tract infections (13%), sepsis (20%), and other general symptoms (41%). Outcomes are summarized in Table 2. After adjustment for possible confounders (sex, age, LVEF, hypertension, BMI, CVD, and SBP), only female gender was associated with higher risk for hospitalization for any cause, mainly due to urinary tract infections (Table 3). Mortality rates were similar between the groups (0% vs 3.6% in the treatment and control group respectively, p = 0.22).
Summary of Outcomes.

AMI, acute myocardial infarction; CVA, cerebrovascular accident.
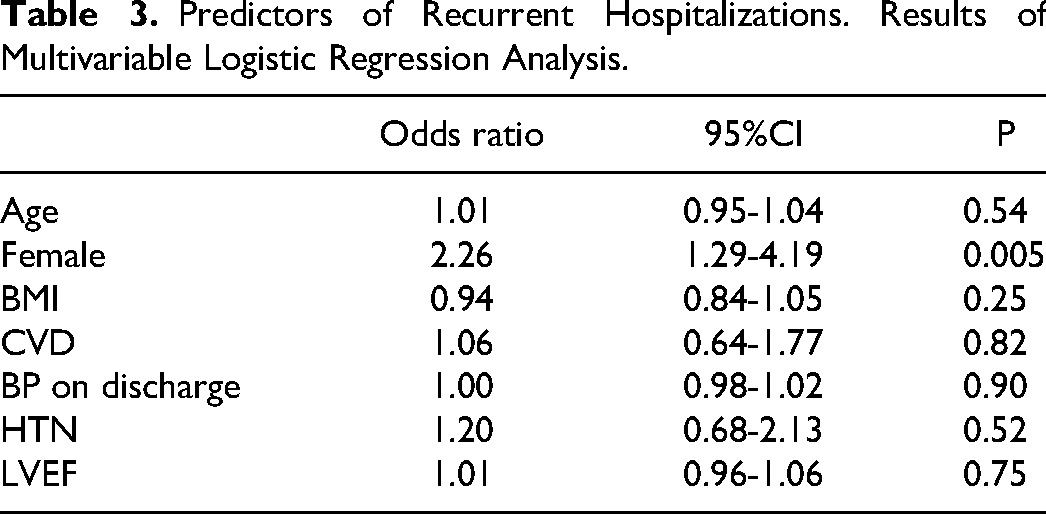
Predictors of Recurrent Hospitalizations. Results of Multivariable Logistic Regression Analysis.
Survival curve for all cause readmissions in the two groups is depicted in Figure 2.
Summary of SGLT2 inhibitors benefits and the potential risks in the setting of AMI are depicted in Figure 3.
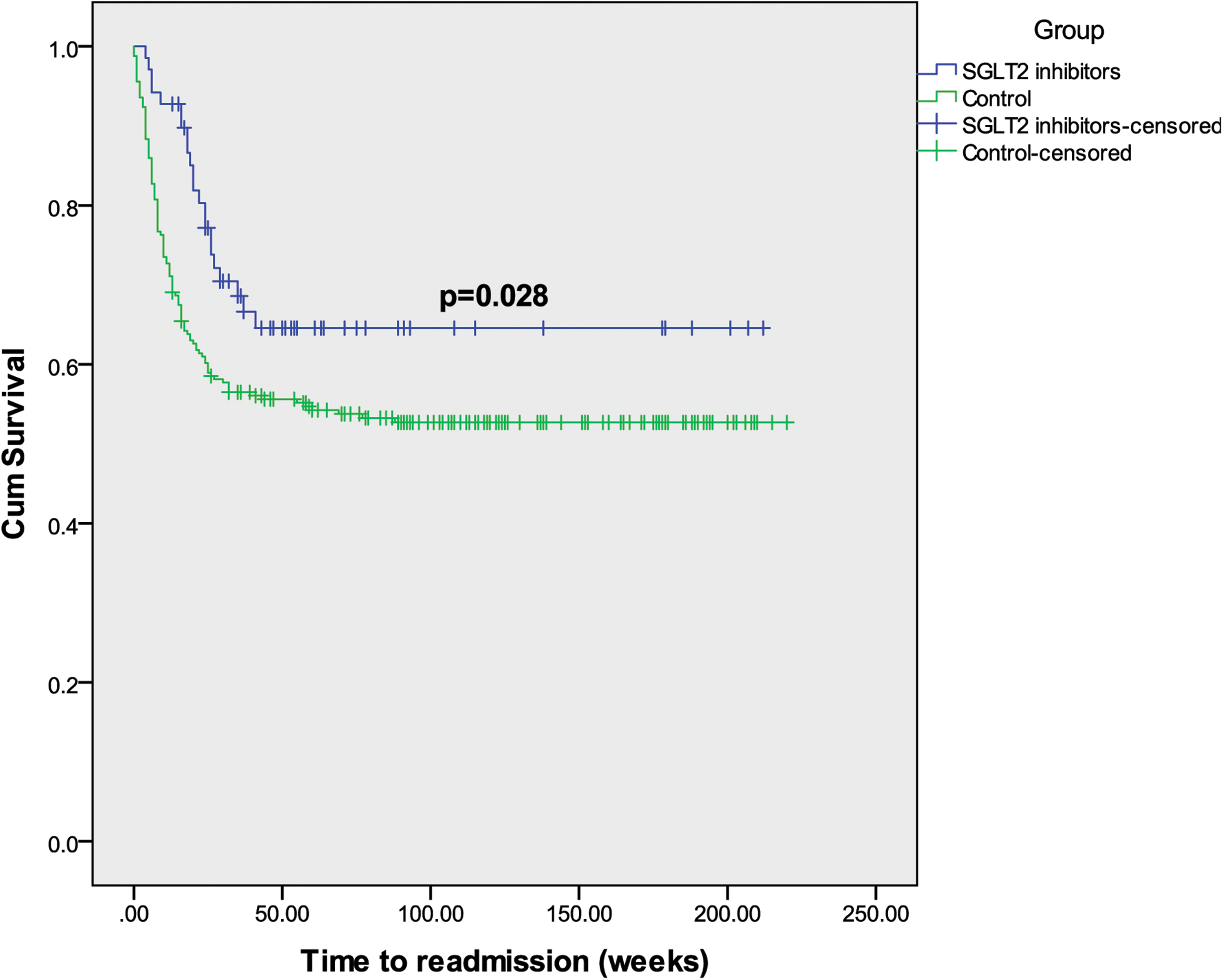
Kaplan Meier curve for hospitalization for any cause.

Summary of study rationale.
The rationale behind using SGLT2 inhibitors in the acute setting of acute myocardial infarction (AMI) is based on previous studies showing favorable outcomes in patients with ischemic heart disease or heart failure. Cumulative data indicates improved cardiac function due to anti remodeling and anti-fibrotic properties. Since the studies suggest very early benefits in large spectrum of patients and given the negligible risk of diabetic ketoacidosis (DKA) or contrast nephropathy, SGLT2 inhibitors administration in patients with AMI seems reasonable and beneficial.
Discussion
We examined the impact of early SGLT2 inhibitors administration (mainly empagliflozin) on CV outcomes in diabetic patients following AMI. We found that early SGLT2 inhibitors initiation in the setting of AMI was associated with lower rate of hospitalizations for any cause with no effect on CV events. Notably, a trend toward reduced mortality rate was observed in the SGLT2 inhibitors group, albeit not statistically significant. The small number of patients in the treatment group and the low rate of CV events in the study limit the statistical power and may explain the lack of difference in CV events and all-cause mortality. Following AMI, patients are at increased risk of developing HF due to neurohormonal activation and cardiac remodeling with altered myocardial metabolism. 27 Moreover, the diuretic effect along with the modest decline in blood pressure associated with SGLT2 inhibitors result in afterload reduction and decrease in filling pressures. 13 Several observations indicate the benefit of SGLT2 inhibitors early after AMI. In animal models, empagliflozin reduced cardiac fibrosis when initiated early after the induction of AMI by inhibiting TGF-β1/Smad3 fibrotic pathway, probably unrelated to the hemodynamic effects of the drug. 28 Canagliflozin infusion prior to coronary artery occlusion in swine model was also associated with improved cardiac work efficiency during ischemia. 29 Likewise, in the EMMY (Empagliflozin in myocardial infarction) trial, empagliflozin improved left ventricular function and diastolic parameters, and reduced N-terminal pro-brain natriuretic peptide (NT-proBNP) levels in patients with AMI when given 72 h after percutaneous coronary intervention (PCI) indicating possible improved cardiovascular outcomes in the long term although there was no effect on inflammatory burden.30–32 In a recent population-based study from Southern Korea, early use of SGLT2 inhibitors in diabetic patients following AMI treated with PCI was associated with lower risk of CV events, all-cause mortality, and HF hospitalization. 33 Sardu et al showed that SGLT2 inhibitors therapy exert anti-inflammatory effects and induce plaque regression and stabilization by reducing lipid core, increasing fibrous cap thickness, and decreasing inflammatory burden. 34 In our study, we included only diabetic patients since the indication for SGLT2 inhibitors at the time the study was conducted was for glycemic control. Diabetic patients taking SGLT2 inhibitors may have better cardiovascular outcomes following AMI as was shown recently in the study by Paolisso et al. 35 This early benefit is not related to glucose control, but rather to other pleotropic effects of the drugs.36,37 Despite the plenty of evidence with regard to the cardioprotective effects of SGLT2 inhibitors, some recent studies may raise several questions in the setting of AMI. In one study, SGLT2 inhibitors in AMI improved LVEF when compared to dipeptidyl peptidase IV inhibitors, however, without beneficial effects on CV events. 38 Likewise, dapagliflozin in AMI resulted in improved cardiometabolic outcomes in a recent study with no positive effect on CV events. 39 The discrepancy in the results may be attributed to possible different properties of drug types and variety in study populations. However, regardless of the difference in CV outcomes in abovementioned studies, SGLT2 inhibitors have no safety concerns and therefore should be initiated early following AMI owing to their cardiovascular and metabolic potential benefits once the patient is hemodynamically stable. In our study, there were no events of DKA or limb amputations in the SGLT2 inhibitors group. SGLT2 inhibitors were well tolerated without serious adverse events and their excellent safety profile may justify their use early following AMI. The EMPACT-MI (NCT04509674) trial and other ongoing trials will further shed the light on the role of SGLT2 inhibitors in the setting of AMI.
Study Limitations
The current study has several limitations that must be addressed. First, this is a small single-center study that included patients who were prescribed mainly empagliflozin and accordingly, the conclusions should be restricted to this drug. Second, because of the retrospective design, possible confounders cannot be excluded although we performed a multivariate regression analysis. As we mentioned before, the good safety profile of the drugs in this study with absence of serious adverse events is very promising given the known and consistent positive CV outcomes. Third, we were unable to confirm (despite significant efforts) that all the patients who had a recommendation to receive SGLT2i did receive it continuously albeit it was confirmed by pharmacy purchase history in all included patients. Fourth, we included diabetic patients and the results may not be applicable to non-diabetic patients. However, previous studies showed similar results in subgroup analysis, regardless of the presence or absence of diabetes.
Conclusions
In this small retrospective study, early initiation of SGLT2 inhibitors following AMI was safe and reduced the risk of all cause hospitalizations without effect on CV outcomes. The results of our study along with recent findings support early initiation of SGLT2 inhibitors following AMI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
