Abstract
Type 2 diabetes mellitus (T2DM) is a progressive and multifactorial metabolic disease mainly characterized by hyperglycemia and insulin resistance. Abnormal platelet reactivity associated with an increased risk of cardiovascular disease (CVD) is also a feature characteristic of patients with T2DM. Dual antiplatelet therapy consisting of aspirin and an adenosine diphosphate platelet P2Y12 receptor antagonist, such as clopidogrel, represents the standard antithrombotic regimen for the secondary prevention of CVD risk in T2DM. However, a high proportion of patients with T2DM exhibit high on-treatment platelet reactivity to aspirin and/or clopidogrel, associated with a greater risk of adverse cardiovascular events compared with nondiabetic patients. Consequently, novel antiplatelet therapeutic approaches may be required in order to avoid such events. Vorapaxar is a novel antiplatelet agent that targets the platelet protease-activated receptor 1 and inhibits thrombin-induced platelet activation. Vorapaxar has been studied in 2 phase III clinical trials and has been approved for use in the secondary prevention of atherothrombotic events in patients with a previous myocardial infarction (MI) or peripheral arterial disease. New data from the Thrombin-Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P)-TIMI 50 trial MI cohort demonstrate that the subgroup of patients with T2DM exhibits increased benefit from vorapaxar use compared with non-T2DM patients. The aim of the present work is to critically review the current knowledge concerning vorapaxar use in patients with T2DM as well as to discuss the possible mechanism(s) underlying vorapaxar’s beneficial effect in T2DM.
Introduction
Type 2 diabetes mellitus (T2DM) is a progressive and multifactorial metabolic disease, mainly characterized by hyperglycemia and insulin resistance. Thrombotic abnormalities and an increased risk of microvascular and macrovascular complications are also features characteristic of patients with T2DM. 1,2 Over the years, diabetes has been evolved in a pandemic, since more than 380 million people have been affected by it and its prevalence is expected to increase even more in the future, 1 as sedentary lifestyle and a hypercaloric diet tend to be adopted worldwide. 3 Meanwhile, the economic burden of diabetes is huge, given that the global health-care expenses for disease prevention and treatment are hundreds of billions of dollars, a cost that is expected to further increase prospectively. 2
Patients with T2DM have 2 times higher risk of cardiovascular disease (CVD) 4 and its clinical manifestations (eg, death from cardiovascular causes, myocardial infarction [MI], and ischemic stroke). Thus, diabetic patients are at high risk and are prone to increased morbidity and mortality. The greater CVD risk in patients with T2DM compared with nondiabetic patients is attributed, among others, to their prothrombotic state, predominantly to the increased platelet reactivity. Indeed, the presence of hyperglycemia and insulin resistance in these patients leads to increased arterial vessel wall platelet adhesion and increased platelet activation either by primary (eg, thrombin) or by auxiliary agonists (eg, adenosine diphosphate [ADP] and serotonin that are secreted from the platelet-dense granules). 5 -9
Dual antiplatelet therapy (DAPT) consisting of aspirin and an ADP platelet P2Y12 receptor antagonist, such as clopidogrel, in high-risk patients (patients with an acute coronary syndrome [ACS] or undergoing percutaneous coronary intervention [PCI]) or clopidogrel monotherapy in patients who cannot take aspirin is the antithrombotic regimen of choice for patients with T2DM. 10 However, despite the use of DAPT, 21.9% of diabetic patients treated with aspirin 11 and 36% treated with clopidogrel 12 have recurrent ischemic events. Responsiveness variability or high on-treatment platelet reactivity (HTPR) to aspirin and/or clopidogrel has been implicated in event recurrence. The presence of diabetes itself is a factor of HTPR. This could be explained by mechanisms such as low bioavailability of the clopidogrel active metabolite, lack of platelet-insulin responsiveness, hyperglycemia, increased oxidative stress, or alterations in calcium metabolism in platelets. 10 Thus, in order to optimize platelet inhibition in T2DM, several modifications of the aforementioned standard regimen have been proposed. These include dose modification of clopidogrel, use of more potent P2Y12 antagonists, such as prasugrel, and/or addition of a third antiplatelet agent. 13
Thrombin is the most potent platelet agonist that activates platelets via protease-activated receptors (PARs)-1 and -4. PAR-1 is considered the primary thrombin receptor, since it exhibits higher thrombin affinity compared with PAR-4. 14 Vorapaxar is a potent, selective orally administered PAR-1 antagonist, which has been tested in 2 large-scale phase III clinical trials and has been approved for use in selected patients (for the secondary prevention of atherothrombotic events in patients with a history of MI or peripheral arterial disease [PAD]) on top of DAPT. 14
The aim of the present review is to illustrate in-depth current knowledge on the clinical usefulness of vorapaxar in the subset of patients with MI who have a history of T2DM as well as to provide possible mechanisms underlying vorapaxar’s action in these patients. Furthermore, the mechanisms responsible for the increased platelet reactivity in T2DM and the strategies of antiplatelet therapy applied in current clinical practice are also reported.
Increased Platelet Reactivity in Diabetes Mellitus
Patients with T2DM exhibit various alterations in platelet functionality, which renders them more susceptible to activation. These platelets are denoted as “diabetic platelets.” Platelet dysfunction is observed soon after the development of diabetes and long before vessel impairment. 15,16 Diabetic platelets are characterized by an increased turnover, that is, the increased rate of elimination and generation of newborn platelets in the circulation, 7,9,17 a proportion of which consists of immature platelets. 18 Another platelet disorder in T2DM is the heightened thromboxane A2 (TxA2) biosynthesis from arachidonic acid via the cyclooxygenase 1 pathway. Importantly, TxA2 levels are greatly reduced after insulin administration in these patients. 16,19 Activation of diabetic platelets leads to increased secretion of cytokines and chemokines from the platelet alpha granules which, in turn, display proinflammatory and proatherogenic roles. Moreover, platelets from patients with T2DM exhibit an enhanced membrane expression of the biologically active CD31, CD36, CD49b, CD62P (P-selectin), and CD63 as well as the glycoprotein (GP) Ib and integrin aIIbβ3 receptors which are responsible for platelet adhesion and aggregation, respectively (Figure 1A). 7 -9 Type 2 DM is also characterized by a hypercoagulable state, despite the fact that the coagulation pathways are often dysfunctional in diabetic patients. 10,20,21 Indeed, there is vast evidence supporting that T2DM is associated with upregulation of the coagulation cascade and thus enhanced thrombin formation. 22 -24 Since thrombin is the strongest platelet agonist, the increased thrombin generation in patients with T2DM may play a prominent role in the platelet hyperreactivity observed in these patients (Figure 1A).

A, Platelet abnormalities in T2DM. Thrombin is the strongest platelet agonist, thus presumably plays a dominant role in the aforementioned abnormalities via its main receptor, PAR-1. Consequently, in patients with T2DM, thrombin may lead to increased membrane expression of CD31, CD36, CD49b, CD62P (P-selectin), and CD63 as well as GPIb and integrin aIIbβ3 receptors. The 2 latter receptors are responsible for platelet adhesion and aggregation. Moreover, thrombin may lead to increased secretion of ADP and serotonin from the dense granules that enhance platelet activation and aggregation as well as increased secretion of cytokines and chemokines from alpha granules, which exhibit proinflammatory and proatherogenic roles. Thrombin may also increase the concentration of intracellular Ca2+, thus leading to enhanced platelet activation. B, Hypothetical scheme representing the potential role of vorapaxar in inhibiting the platelet reactivity in T2DM that is much higher compared with nondiabetic patients. ADP indicates adenosine diphosphate; GP, glycoprotein; PAR-1, protease-activated receptor 1; T2DM, type 2 diabetes mellitus.
The mechanisms underlying the platelet abnormalities and the hypercoagulable state in T2DM are mainly the metabolic disorders (ie, hyperglycemia) that accompany diabetes as well as insulin resistance. Hyperglycemia is responsible for the increased platelet turnover, whereas it causes osmotic changes in the platelet environment and increased nonenzymatic glycosylation of the platelet membrane, which results in the reduction of the platelet membrane fluidity. Hyperglycemia is also associated with disorders in the signaling pathway of the insulin receptor (IR), the activation of protein kinase C, the upregulation of the P2Y12-signaling pathway as well as the increased membrane expression of P-selectin and the integrin-receptor aIIbβ3. 7 -9 Meanwhile, insulin resistance increases platelet susceptibility to aggregation and diminishes their response to the physiological inhibitors of platelet activation, nitric oxide (NO), and prostaglandin I2 (PGI2; prostacyclin), 25,26 since insulin (via IR and insulin-like growth factor 1) reduces platelet response to various agonists, increasing at the same time their sensitivity to the action of NO and PGI2. 27,28 Moreover, insulin resistance increases the intracellular levels of Ca2+, which in turn lead to increased platelet activation. 29 Due to the aforementioned platelet abnormalities, HTPR to aspirin and/or clopidogrel is observed more often in patients with T2DM. 30,31 Therefore, alternative to the established DAPT with aspirin and clopidogrel regimens should be considered in diabetic patients.
Standard-of-Care Antiplatelet Therapy in the Secondary Prevention of Diabetic Patients
Aspirin represents the cornerstone of antiplatelet therapy for the secondary prevention of atherothrombotic events in diabetic patients. 32 -34 Aspirin at a dose of 75 to 160 mg daily has proven to be equally effective in reducing adverse cardiovascular events in T2DM as high-dose aspirin (300-325 mg daily), albeit with less bleeding complications. 33,35,36 Since a higher rate of platelet turnover is observed in T2DM, aspirin administration twice daily could suppress platelet activation more efficiently. 37
Dual antiplatelet therapy consisting of aspirin and clopidogrel is the standard antithrombotic regimen in patients with ACS who have a T2DM background. The existence of HTPR to clopidogrel in a relatively high proportion of patients with T2DM and the adverse clinical outcome that these patients experience have led to the study of alternative therapeutic approaches regarding antiplatelet therapy. The Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) trials have investigated the efficacy of various antiplatelet combinations in diabetic patients with HTPR to clopidogrel. Thus, OPTIMUS-1 compared high (150 mg daily) versus low clopidogrel dose (75 mg daily) given for 30 days in patients with T2DM with poor clopidogrel response. The high clopidogrel dose was more effective in inhibiting platelet activation; however, a proportion of patients still exhibited HTPR. 38 The OPTIMUS-2 trial investigated the efficacy of cilostazol addition (100 mg twice daily) to DAPT for 2 weeks in patients with T2DM and showed that this triple regimen significantly reduces platelet reactivity. 39 The OPTIMUS-3 trial compared prasugrel (60 mg loading dose [LD] plus 10 mg maintenance dose [MD] daily) with high clopidogrel dose (600 mg LD plus 150 mg MD daily) for 1 week in patients receiving aspirin and showed that prasugrel was superior to clopidogrel in suppressing platelet reactivity. 40 The Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel (TRITON)-TIMI 38 study compared prasugrel (60 mg LD plus 10 mg MD daily) with clopidogrel (300 mg LD plus 75 mg MD daily) for 6 to 15 months in patients with ACS with scheduled PCI, 23% of whom had a history of T2DM. 41 Prasugrel significantly reduced the incidence of the composite primary efficacy end point (death from cardiovascular causes, nonfatal MI, or nonfatal stroke) compared with clopidogrel (9.9% vs 12.1%, respectively, hazard ratio [HR] = 0.81, 95% confidence interval [CI]: 0.73-0.90, P < .001). 41 Prasugrel also reduced significantly the incidence of MI (7.4% vs 9.7%, respectively, P < .001) and stent thrombosis (1.1% vs 2.4%, P < .001) compared with clopidogrel. 41 In the cohort of patients with T2DM of the TRITON-TIMI 38, prasugrel significantly reduced the primary efficacy end point compared with clopidogrel (12.2% vs 17.0%, respectively, HR = 0.70, P < .001) as well as the incidence of MI by 40% (8.2% vs 13.2%, respectively, HR = 0.60, P < .001). 42 The Platelet Inhibition and Patient Outcomes (PLATO) trial compared the efficacy and safety of ticagrelor (180 mg LD plus 90 mg MD twice daily) with clopidogrel (300-600 mg LD plus 75 mg MD daily) for 12 months in patients with ACS, 25% of whom had a history of diabetes. 43 In the overall PLATO trial, ticagrelor significantly reduced the composite primary efficacy end point (death from cardiovascular causes, MI, or stroke) compared with clopidogrel (9.8% vs 11.7%, respectively, HR = 0.84, 95%CI: 0.77-0.92, P < .001). 43 Moreover, ticagrelor, compared with clopidogrel, reduced the incidence of MI (5.8% vs 6.9%, respectively, P = .005) as well as the incidence of vascular death (4.0% vs 5.1%, respectively, P = .001). 43 However, ticagrelor benefits did not differ among patients with or without diabetes (P for interaction [P interaction] = .49 for the primary end point; P interaction = .32 for MI). 44 Conclusively, in patients with T2DM who undergo PCI, DAPT with prasugrel and aspirin is the regimen of choice.
Vorapaxar as an Adjunctive Antiplatelet Agent in Diabetes Mellitus
Vorapaxar Pharmacokinetics and Pharmacodynamics
Vorapaxar is derived from the natural product himbacine and has been modified as a crystalline salt to be used as a drug. 45 Vorapaxar is a potent and competitive antagonist of PAR-1, which binds with a high affinity to a specific binding pocket of the receptor 46 (the half-life of disengagement is estimated to be 20 hours). 47 Consequently, vorapaxar is selective for PAR-1, inhibiting thrombin-induced platelet aggregation but has no effect on the aggregation mediated through other receptors such as ADP, TxA2, collagen, or PAR-4 receptors. 48 Moreover, vorapaxar does not affect the thrombin-induced fibrin generation from fibrinogen, thus leaving intact the coagulation cascade. 49
Vorapaxar exhibits high oral bioavailability (more than 90%), and it is rapidly absorbed from the gastrointestinal tract. 49 Vorapaxar is slowly but extensively metabolized in the liver by the cytochrome P (CYP) 450 members, CYP3A4 and CYP2J2 to 2 main products: the inactive metabolite M19 and the active metabolite M20, which exhibits an activity similar to vorapaxar. 50 Accordingly, vorapaxar has a long plasma half-life of 159 to 311 hours, 14 thus offering a prolonged antiplatelet effect. Vorapaxar is excreted mainly through the feces, while less than 5% is excreted by renal clearance. 51 Due to its metabolic characteristics, vorapaxar should not be coadministered with CYP3A4 inhibitors (such as ketoconazole) or inducers (such as rifampin), since coadministration could affect its antiplatelet activity. 52,53 On the contrary, vorapaxar can be coadministered with warfarin or other CYP2C9/CYP2C19-metabolized drugs. 54 Finally, food intake does not affect vorapaxar’s efficacy. 49
Another potent and selective PAR-1 antagonist that was developed concurrently with vorapaxar was atopaxar. The efficacy and safety of atopaxar has been evaluated in the 2 phase II clinical trials Japanese-Lesson from Antagonizing the Cellular Effect of Thrombin and Lessons from Antagonizing the Cellular Effect of Thrombin-Acute Coronary Syndromes. Nonetheless, adverse effects such as rise of hepatic enzyme levels and prolongation of the QTc interval have suspended further testing of atopaxar. 14
Vorapaxar in Patients With a History of MI and Diabetes Mellitus
The safety and efficacy of vorapaxar was evaluated in The Thrombin Receptor Antagonist for Clinical Event Reduction in Acute Coronary Syndrome clinical trials (TRA-CER) 55,56 and the Thrombin-Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P)-TIMI 50 57,58 clinical trials. In the TRA-CER trial, vorapaxar reduced the composite primary efficacy end point (cardiovascular death, MI, stroke, recurrent ischemia with rehospitalization, or urgent coronary revascularization) compared with placebo, but this reduction was not accompanied with statistical significance (18.5% vs 19.9%, respectively, HR = 0.92, 95% CI: 0.85-1.01, P = .07). 56 About 31% of TRA-CER participants had a history of T2DM, which was one of the prerequisites for study enrollment. However, according to our knowledge, there is no specific analysis available until today, describing the effect of vorapaxar on this subpopulation. On the contrary, vorapaxar’s efficacy and safety was evaluated in the subgroup of patients with T2DM of the prespecified MI cohort of the TRA 2°P-TIMI 50 trial. 59
In this context, the effect of vorapaxar, on top of the standard antiplatelet therapy (aspirin plus clopidogrel), was evaluated in patients who presented diabetes and a previous MI but did not have a previous stroke or transient ischemic attack (n = 3623) in comparison with nondiabetic patients with MI (n = 13 273). 60 Of the majority of MI patients with T2DM, 84% were treated with either insulin or noninsulin therapies for hyperglycemia. Patients with T2DM, compared with non-T2DM patients, had approximately 2 times higher rate of death incidence from cardiovascular causes, MI, or stroke (14.3% vs 7.6%, respectively, P < .001). Even after adjustment for potential confounders (such as treatment allocation, age, history of hypertension, history of hyperlipidemia, and history of PAD, etc), diabetic patients had still 47% higher risk for the aforementioned events. In patients with T2DM treated with vorapaxar, a statistically significant reduction in the primary efficacy end point was observed compared with placebo (11.4% vs 14.3%, respectively, P = .002), while in nondiabetic patients, the respective reduction was less pronounced (6.3% vs 7.6%, respectively, P = .003; Table 1). Thus, vorapaxar treatment, compared with placebo, reduced the primary efficacy end point regardless of diabetes existence (P interaction = .40). Importantly, the absolute risk reduction in T2DM was greater compared with nondiabetic patients, which translates to 1 of 29 number needed to treat to avoid a major cardiovascular event among diabetic patients and 1 of 74 number needed to treat to avoid such an event in nondiabetic patients. However, we should point out that in diabetic patients treated with vorapaxar, all-cause mortality and cardiovascular mortality were not significantly different compared with placebo (P = .36 and .43, respectively). Moderate or severe bleeding events were more frequent in patients treated with vorapaxar, compared with placebo, in diabetic (4.4% vs 2.6%, respectively, P = .02) and nondiabetic patients (2.9% vs 1.9%, respectively, P < .001; Table 1), regardless of diabetes existence (P interaction = .93). Intracranial hemorrhage rates were not increased in either the diabetic or the nondiabetic cohort. Collectively, 2 prespecified composite end points of net clinical outcome were evaluated: among patients with T2DM, vorapaxar compared with placebo improved the composite outcome of death from cardiovascular causes, MI, stroke, or recurrent ischemia leading to revascularization plus moderate/severe bleeding (16.7% vs 19.6%, respectively, P = .005; this composite end point of clinical outcome was not significantly improved in nondiabetic patients), while it improved the composite outcome of death from cardiovascular causes, MI, stroke, or severe bleeding (13.6% vs 16.4%, respectively, P = .006; Table 1). Thus, the absolute risk difference in the first composite end point of net clinical outcome for the diabetic patients was −3.89% (95% CI: −1.34 to −6.11) and for the nondiabetic patients was −0.53% (95% CI: 0.61 to −1.57). 60
Comparison of Efficacy and Safety End Points As Well As Net Outcome Between T2DM (Vorapaxar vs Placebo) and Non-T2DM Patients (Vorapaxar vs Placebo) From the TRA 2°P-TIMI 50 Trial.
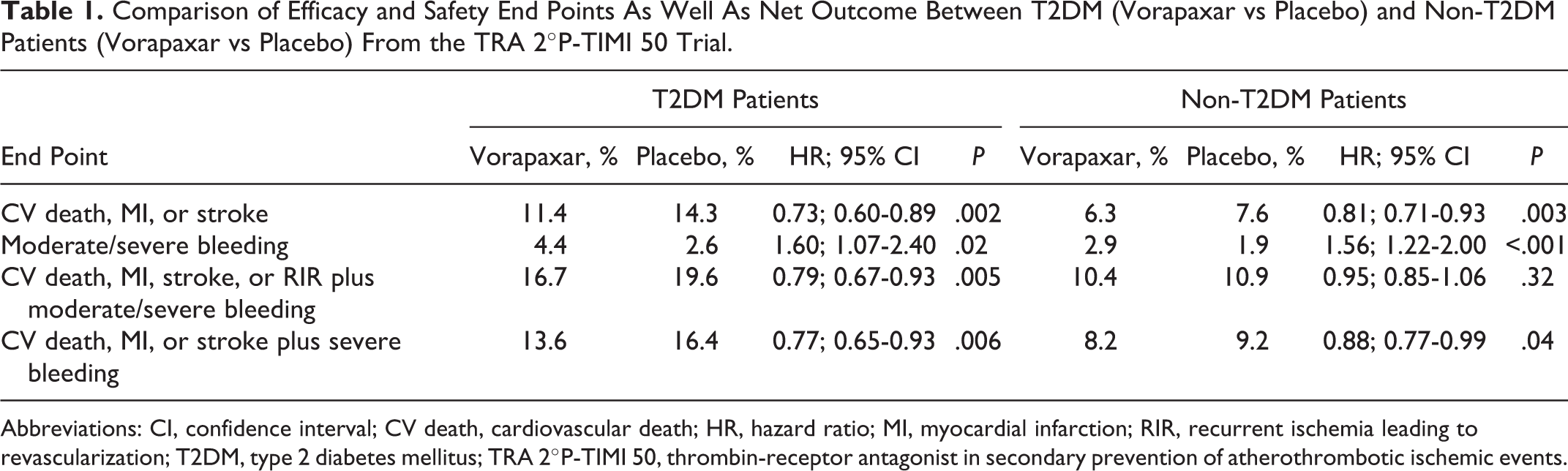
Abbreviations: CI, confidence interval; CV death, cardiovascular death; HR, hazard ratio; MI, myocardial infarction; RIR, recurrent ischemia leading to revascularization; T2DM, type 2 diabetes mellitus; TRA 2°P-TIMI 50, thrombin-receptor antagonist in secondary prevention of atherothrombotic ischemic events.
Mechanisms Underlying the Beneficial Effect of Vorapaxar in T2DM
As reported earlier, DAPT with aspirin and clopidogrel (which was the baseline regimen in the TRA 2°P-TIMI 50 trial) was underpowered to efficiently prevent recurrent events in patients with T2DM. 61 -63 Current knowledge is not enough to permit a definite conclusion on the mechanism(s) underlying the high cardiovascular event rate in patients with T2DM under DAPT therapy. However, this high rate may be explained, at least partially, by the low efficacy of either aspirin or P2Y12 antagonists, including clopidogrel, to inhibit the increased platelet activation through platelet receptors other than the TxA2 and ADP receptors in diabetic patients. For example, both aspirin and P2Y12 antagonists are weak inhibitors of platelet activation induced by von Willebrand factor (through its GP Ibα receptor), which is highly expressed on endothelium in the microcirculation of patients with T2DM. 61,64 -66 Nonetheless, the use of vorapaxar on top of aspirin and clopidogrel in patients with T2DM who participated in the TRA 2°P-TIMI 50 trial offered a more pronounced net clinical benefit compared with nondiabetic patients. 60 This beneficial effect of vorapaxar may at least partially be explained by its effectiveness in preventing the induction of multiple signaling pathways by thrombin through its PAR-1 receptor, which is greater in platelets of patients with T2DM compared with nondiabetic patients (Figure 1B). Vorapaxar may also diminish the high incidence of HTPR to aspirin or clopidogrel observed in patients with T2DM. Indeed, until to date, there is no evidence for the existence of high on-vorapaxar platelet reactivity in any patient population. The fact that vorapaxar does not require metabolic modification in order to be active, in concert with its long plasma half-life, 14 lowers the possibility of the existence of HTPR to vorapaxar. Therefore, vorapaxar could be a suitable antiplatelet agent to overcome HTPR to aspirin or clopidogrel.
Conclusions and Future Perspectives
Patients with T2DM with previous MI who participated in the TRA 2°P-TIMI 50 trial exhibited substantially higher thrombotic risk as compared with nondiabetic patients, despite the fact that in addition to common therapy routinely administered in T2DM, such as hypoglycemic, antihypertensive, and hypolipidemic regimens, almost all of them were receiving aspirin and the majority were also under clopidogrel treatment. 60,67 -69 The use of vorapaxar on top of aspirin and clopidogrel in these patients resulted in a greater absolute reduction in recurrent events compared with nondiabetic patients, thus offering a more pronounced net clinical benefit in patients with T2DM. 60 However, an important limitation of the clinical use of vorapaxar on top of DAPT is the high bleeding rates, as it was demonstrated in both clinical trials (TRA-CER and TRA 2°P-TIMI 50). 56,58 Consequently, future clinical studies in which vorapaxar will be used either on top of DAPT with aspirin and clopidogrel or in combination with a single antiplatelet or even as monotherapy in patients with T2DM are required to further support the usefulness of this new potent antiplatelet drug. This elucidation will definitely show whether vorapaxar will be the antiplatelet of choice for long-term treatment of patients with T2DM for the secondary prevention of an atherothrombotic event, minimizing at the same time the bleeding adverse events.
We should emphasize that the results presented and discussed in the present review article concern only patients with T2DM; therefore, more data are necessary to show whether vorapaxar could be also useful in patients with type 1 diabetes mellitus.
Considering the high rates of thrombin generation in T2DM, another issue that remains to be addressed in future studies is the possible involvement of the other thrombin receptors, particularly PAR-4, in the vascular complications observed in T2DM. Indeed, diabetes is associated with vascular remodeling and human as well as animal studies in vivo and in vitro 70,71 have suggested that PAR-4 inhibition attenuates both thrombin-stimulated migration following high glucose and exaggerated intimal thickening in diabetes mellitus. Furthermore, thrombin promotes vascular smooth muscle cell (SMC) mitogenesis and migration via activation of PARs, while high glucose pretreatment enhances SMC responsiveness to thrombin through transcriptional upregulation of PAR-4 but not PAR-1. 70 In this regard, more recent data showed that PAR-4 expression was increased in carotid atherectomies and saphenous vein specimens from diabetic versus nondiabetic patients and in aorta and carotid arteries from streptozotocin-diabetic versus nondiabetic C57BL/6 mice. 71 The aforementioned data suggest that PAR-4 may play a role in diabetic vasculopathy; therefore, the development of PAR-4 inhibitors might serve to limit mainly proliferative processes in restenosis-prone diabetic patients, particularly in patients whom severe bleeding is attributed to selective PAR-1 blockade. 71
Footnotes
Author Contributions
I. C. Moschonas contributed to acquisition, analysis, and interpretation, drafted manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. A. D. Tselepis contributed to conception and design, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
