Abstract
Aims:
The diabetic heart is resistant to the myocardial infarct-limiting effects of ischemic preconditioning (IPC). This may be in part due to the downregulation of the phosphatidylinositol 3′-kinase-Akt pathway, an essential component of IPC protection. We hypothesized that treating the diabetic heart with the sulfonylurea, glimepiride, which has been reported to activate Akt, may lower the threshold required to protect the diabetic heart by IPC.
Methods:
Goto-Kakizaki rats (a type II lean model of diabetes) received glimepiride (20 mg/kg per d, by oral gavage) or vehicle for (a) 3 months (chronic treatment) or (b) 24 hours (subacute treatment). In the third group, glimepiride (10 μmol/L) was administered only to the isolated hearts on the Langendorff apparatus (acute treatment). All hearts were subjected to 35 minutes ischemia and 120 minutes reperfusion ex vivo, at the end of which infarct size was determined by tetrazolium staining. Preconditioning treatment comprised 1 (IPC-1) or 3 (IPC-3) cycles of 5 minutes global ischemia and 10 minutes reperfusion.
Results:
The diabetic heart was found to be resistant to IPC such that 3-IPC cycles, instead of the usual 1-IPC cycle, were required for cardioprotection. However, pretreatment with glimepiride lowered the threshold for IPC such that both 1 and 3 cycles of IPC elicited cardioprotection: chronic glimepiride treatment (IPC-1 31.9% ± 3.8% and IPC-3 33.5% ± 2.4% vs 43.9% ± 1.4% control, P < .05; N > 6 per group); subacute glimepiride treatment (IPC-1 31.1% ± 3.0% and IPC-3 29.3% ± 3.3% vs 42.2% ± 2.3% control, P < .05 N > 6 per group); and acute glimepiride treatment (IPC-1 28.2% ± 3.7% and IPC-3 24.6% ± 5.4% vs 41.9% ± 5.4% control, P < .05; N > 6 per group). This effect of glimepiride was independent of changes in blood glucose.
Conclusions:
We report for the first time that glimepiride treatment facilitates the cardioprotective effect elicited by IPC in the diabetic heart.
Introduction
Coronary heart disease (CHD) is a leading cause of death in diabetic patients. The number of diabetic patients is increasing at an alarming rate and is estimated to reach 300 million worldwide according to the World Health Organization. Diabetic patients are 2 to 3 times more likely to develop CHD, and experience worse clinical outcomes following an acute myocardial infarction, 1 –3 coronary angioplasty, 4 and cardiac bypass surgery. 5 –7 The reason for the worsened cardiovascular outcomes in diabetic patients is currently unclear. Preclinical animal studies suggest that there may be specific defects in diabetic cardiomyocytes which may be responsible including increased mitochondrial generation of reactive oxygen species, downregulation of the PI3-Akt survival kinase pathway, and reduced rates of adenosine triphosphate (ATP) synthesis (reviewed in refs 8 –10).
One potential endogenous strategy for protecting the diabetic heart against acute ischemia-reperfusion (I/R) injury is ischemic preconditioning (IPC), a phenomenon which was first described in 1986 by Murry and colleagues, 11 as the reduction in myocardial infarct size observed in canine hearts pretreated with short bouts of nonlethal myocardial ischemia and reperfusion. Whether the diabetic myocardium is amenable to the cardioprotective effects of IPC has been debated with reports of cardioprotection in some experimental studies. 12 –16 However, the vast majority of studies find that the diabetic heart is resistant to the cardioprotection elicited by IPC. 17 –33 The fact that the diabetic heart may be resistant to IPC may impact on the translation of novel cardioprotective strategies into the clinical setting where about 15% to 20% of the patients with CHD are also diabetic. 34 –36 Studies from our laboratory have also found that the diabetic heart is resistant to IPC, but this resistance can be overcome when a stronger IPC stimulus is used to elicit cardioprotection. 16,37 Initial findings have suggested that this abnormal response of the diabetic heart to IPC may be due to impaired phosphatidylinositol 3′-kinase (PI3K)-Akt signaling. 16 Previous studies have clearly established a role for the PI3K-Akt pathway in the signal transduction pathway underlying IPC-induced cardioprotection in the nondiabetic heart. 38,39
Importantly, the antidiabetic sulfonylurea, glibenclamide, which is used less frequently nowadays, is known to block the cardioprotective effects of IPC by acting as an antagonist of the mitochondrial ATP-dependent potassium channel. 40,41 Interestingly, the sulfonylurea, glimepiride, which does not block IPC cardioprotection, 42 has been reported to activate Akt when administered to the endothelial cells, 43 human umbilical vein endothelial cells, 44 and adipocytes. 45 In the present study, we investigate whether pretreatment with glimepiride can facilitate IPC in the diabetic heart.
Materials and Methods
Animals and Materials
Adult Goto-Kakizaki rats, a rat model of type II diabetes mellitus, were purchased from Denmark (Taconic, Denmark). 46,47 Animals received humane care in accordance with the United Kingdom Animal (Scientific Procedures) Act of 1986. For oral gavage, glimepiride (20 mg/kg/d) was dissolved in methylcellulose. For the Langendorff perfusion, glimepiride (10 µmol/L, Sigma, UK) dissolved in dimethyl sulfoxide (DMSO) was further dissolved in Krebs-Henseleit buffer, giving a final DMSO concentration of <0.01%. All other reagents were of standard analytical grade.
Glimepiride Pretreatment Protocols
Animals were randomized to receive 1 of the following 3 glimepiride treatment protocols:
Chronic glimepiride treatment
Rats received either glimepiride (20 mg/kg per d) dissolved in methylcellulose or methylcellulose vehicle control by oral gavage for 3 months, following which the hearts were excised and mounted on the Langendorff apparatus and subjected to the treatment protocols outlined in Figure 1.
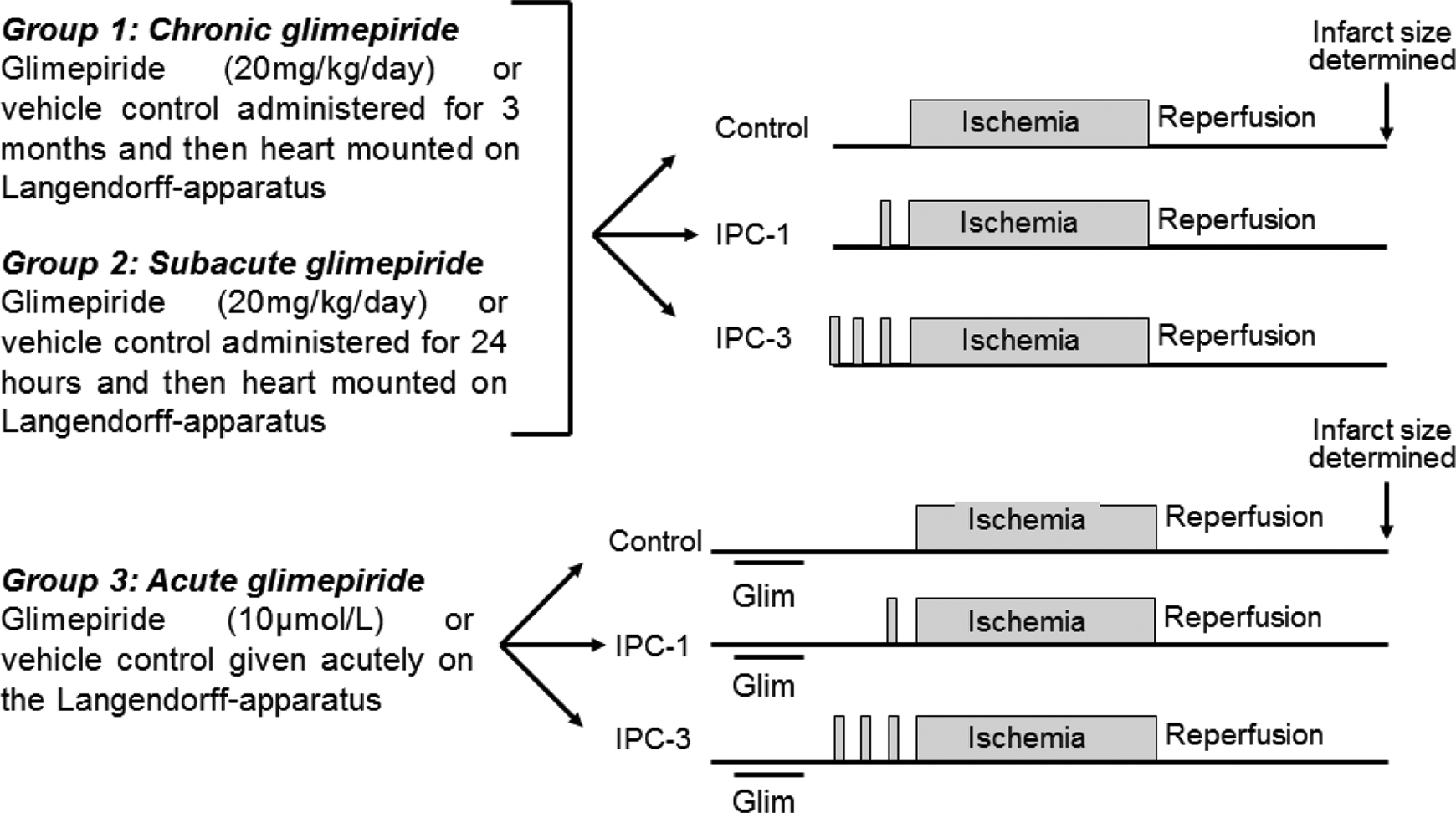
Experiment protocols. Hearts were randomized to receive 1 of the 3 different glimepiride treatment regimens depicted in the scheme. Following pretreatment with glimepiride, all hearts were randomized to receive either control, 1 cycle of IPC (IPC-1) or 3 cycles of IPC (IPC-1), with each cycle of IPC comprising 5 minutes of global myocardial ischemia followed by 10 minutes of myocardial reperfusion. IPC indicates ischemic preconditioning.
Subacute glimepiride treatment
Rats received either glimepiride (20 mg/kg per d) dissolved in methylcellulose or methylcellulose vehicle control by oral gavage for 24 hours, following which the hearts were excised and mounted on the Langendorrf-apparatus and subjected to the treatment protocols outlined in Figure 1.
Acute glimepiride treatment
Hearts were excised from untreated animals and mounted on the Langendorff apparatus, and were perfused for 15 minutes with either glimepiride (10 µmol/L) or vehicle control (DMSO, <0.01%) prior to the treatment protocols outlined in Figure 1.
Myocardial Infarction Protocols
All hearts were subjected to 35 minutes of regional ischemia followed by 120 minutes of reperfusion at the end of which infarct size was determined by tetrazolium staining. Hearts were randomized to the following treatment groups (see Figure 1):
Control. Hearts were subjected to the standard I/R protocol as above.
IPC-1. Hearts were preconditioned with one 5-minute episode of global myocardial ischemia followed by 10 minutes of reperfusion prior to the index ischemic period.
IPC-3. Hearts were preconditioned with three 5-minute episodes of global myocardial ischemia with 10 minutes of intervening reperfusion prior to the index ischemic period.
Langendorff Perfusion of Rat Hearts
Rats were anesthetized with sodium pentobarbital (55 mg/kg intraperitoneally) and heparin (300 IU). The hearts were rapidly excised into ice-cold buffer and mounted on a constant pressure (80 mm Hg) Langendorff perfusion apparatus and perfused with modified Krebs-Henseleit bicarbonate buffer in mmol/L: NaCl 118.5, NaHCO3 25.0, KCl 4.8, MgSO4 1.2, KH2PO4 1.2, CaCl2 1.7, and glucose 11.0. The buffer was gassed with 95%O2/5%CO2 and pH maintained at 7.35 to 7.45 at 37.0°C. A suture was placed around the left main coronary artery and the ends were inserted into a pipette tip to form a snare. A latex, fluid-filled balloon was placed in the left ventricle through an incision in the left atrial appendage and inflated to a pressure of 8 to 10 mm Hg. Left ventricular developed pressure, heart rate, and coronary flow were monitored at regular intervals. Temperature was constantly measured via a thermoprobe inserted into the pulmonary artery and maintained at 37.0°C ± 0.2°C.
Regional myocardial ischemia was induced by tightening the suture placed around the left main coronary artery and reperfusion initiated by releasing the snare. At the end of the reperfusion period, the suture was tied and the heart was perfused with 0.25% Evans Blue in saline to delineate the area at risk. Hearts were then frozen at −20°C for several hours before being sliced into 2-mm thick transverse sections and incubated in triphenyltetrazolium chloride solution (TTC; 1% in phosphate buffer). The TTC reacts with intracellular dehydrogenases to stain viable risk zone tissue red leaving the infarcted areas off-white. The slices were then transferred and fixed in 10% formalin overnight. The slices were drawn onto acetate and computerized planimetry (Summa Sketch III, Summagraphics, Seymour, Connecticut) was used to assess the percentage of infarcted tissue in the myocardium risk zone area (I/R%). 48
Blood Glucose and Glycated Hemoglobin Assessment
Samples for nonfasting blood glucose and glycated hemoglobin (HbA1c) were taken at baseline and after 3 months treatment with either glimepiride or methylcellulose control. Blood glucose measurements (mmol/L) were determined using an ABL 700 series blood gas analyzer (Radiometer, Copenhagen) and HbA1c measurements (%) were determined by an antibody-colorimetric assay using a Cobas Mira Plus analyzer (Roche Diagnostic Systems, Roche, UK).
Statistical Analysis
All values are expressed as mean ± standard error of the mean. Myocardial infarct size was analyzed by 1-way analysis of variance and Fisher protected least significant difference test for multiple comparisons. Differences were considered significant when P < .05.
Results
The Threshold for IPC Is Elevated in Untreated Diabetic Hearts
Diabetic untreated hearts were resistant to the cardioprotection elicited by 1 cycle of IPC and required 3 cycles of IPC before a reduction in myocardial infarct size was observed, whether the heart had been subjected to pretreatment with chronic vehicle control (IPC-3 27.1% ± 3.5% vs control 38.4% ± 5.1%, P < .05; IPC-1 33.1% ± 4.7% vs control 38.4% ± 5.1%, P = not significant [NS]), subacute vehicle control (IPC-3 26.0% ± 5.9% vs control 42.3% ± 2.8%, P < .05; IPC-1 43.0% ± 4.7% vs control 42.3% ± 2.8%, P = NS) or acute vehicle control (IPC-3 28.6% ± 6.0% vs control 42.6% ± 5.3%, P < .05; IPC-1 36.8% ± 4.5% vs control 42.6% ± 5.3%, P = NS; see Figures 2 to 4).

Myocardial infarct size (expressed as a percentage of the volume of myocardium at risk) in hearts excised from rats pretreated with 3 months of either glimepiride (20 mg/kg/d) or methylcellulose vehicle by oral gavage. The results demonstrate that diabetic hearts are resistant to IPC such that 3 cycles (IPC-3) instead of the usual 1 cycle (IPC-1) of IPC are required to reduce myocardial infarct size. However, glimepiride pretreatment facilitates IPC such that both 1 and 3 cycles of IPC elicit cardioprotection. Values are mean ± SEM. *P < .05. N > 6 per group. IPC indicates ischemic preconditioning; SEM, standard error of the mean.
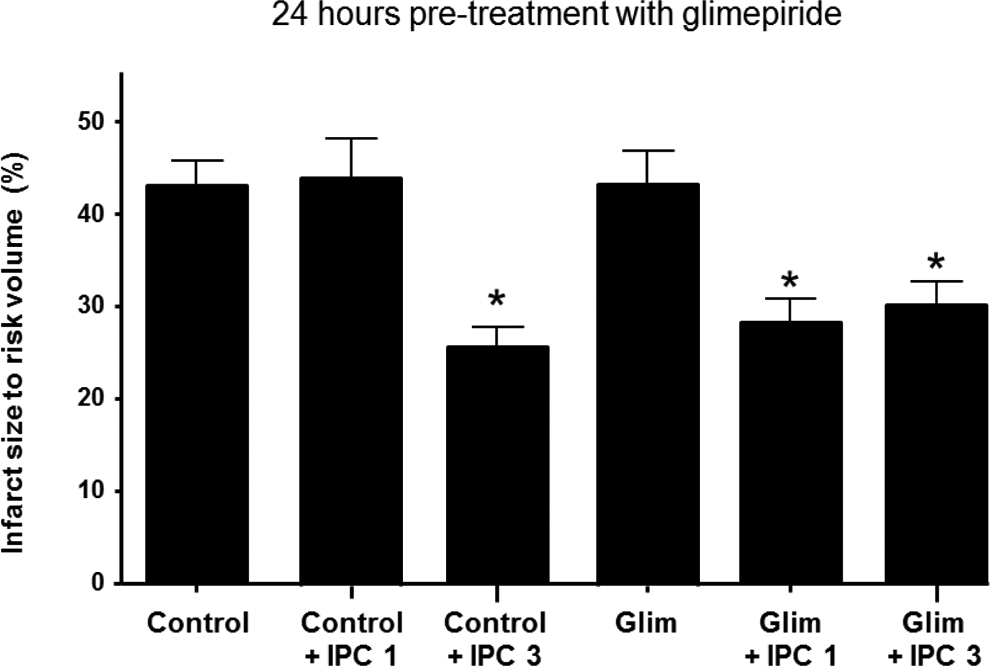
Myocardial infarct size (expressed as a percentage of the volume of myocardium at risk) in hearts excised from rats pretreated with 24 hours of either glimepiride (20 mg/kg/d) or methylcellulose vehicle by oral gavage. The results demonstrate that diabetic hearts are resistant to IPC such that 2 cycles (IPC-3) instead of the usual 1 cycle (IPC-1) of IPC are required to reduce myocardial infarct size. However, glimepiride pretreatment facilitates IPC such that both 1 and 3 cycles of IPC elicit cardioprotection. Values are mean ± SEM. P < .05. N > 6 per group. IPC indicates ischemic preconditioning; SEM, standard error of the mean.
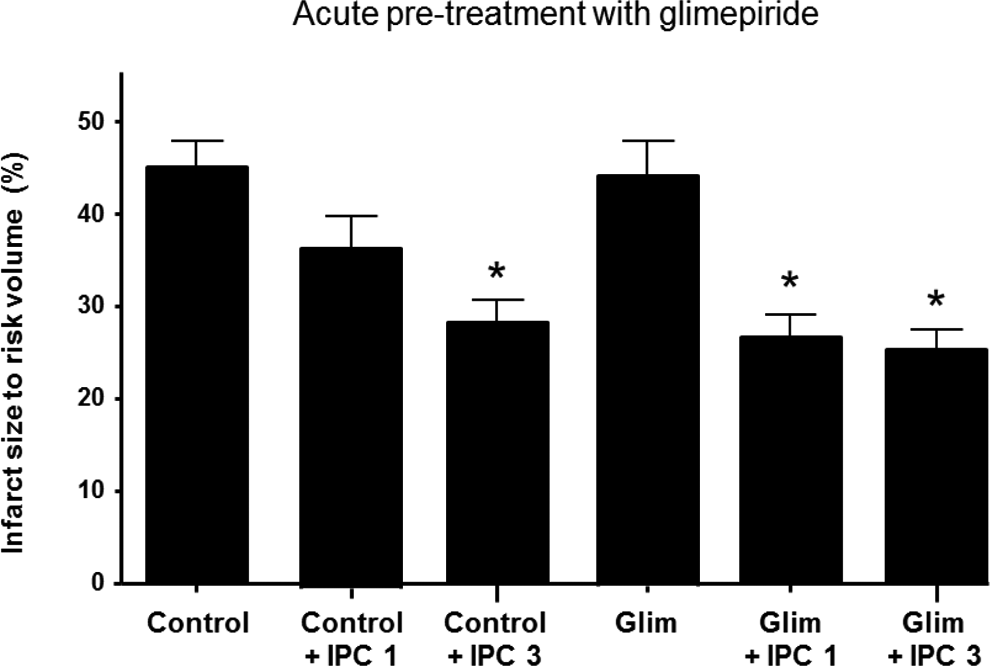
Myocardial infarct size (expressed as a percentage of the volume of myocardium at risk) in rat hearts pretreated with either glimepiride (10 µmol/L) or vehicle control (<0.01% DMSO) on the Langendorff apparatus. The results demonstrate that diabetic hearts are resistant to IPC such that 2 cycles (IPC-3) instead of the usual 1 cycle (IPC-1) of IPC are required to reduce myocardial infarct size. However, glimepiride pretreatment facilitates IPC such that both 1 and 3 cycles of IPC elicit cardioprotection. Values are mean ± SEM. P < .05. N > 6 per group. DMSO indicates dimethyl sulfoxide; IPC, ischemic preconditioning; SEM, standard error of the mean.
Glimepiride Pretreatment Facilitates IPC in the Diabetic Heart
Interestingly, pretreatment with glimepiride facilitated IPC such that both 1 and 3 cycles of the IPC were able to elicit cardioprotection, irrespective of whether the heart had been subjected to pretreatment with chronic glimepiride (IPC-1 31.9% ± 3.8% and IPC-3 33.5% ± 2.4% vs 43.9% ± 1.4% control, P < .05), subacute glimepiride (IPC-1 31.1% ± 3.0% and IPC-3 29.3% ± 3.3% vs 42.2% ± 2.3% control, P < .05), or acute glimepiride (IPC-1 28.2% ± 3.7% and IPC-3 24.6% ± 5.4% vs 41.9% ± 5.4% control, P < .05; see Figures 2 to 4).
Blood Glucose and HbA1c
Three months treatment of glimepiride or methylcellulose did not affect nonfasting blood glucose (in mmol/L: 7.1 ± 0.8 in glimepiride-treated animals at baseline vs 5.9 ± 0.9 glimepiride-treated animals after 3 months; 7.0 ± 1.0 in methylcellulose-treated animals at baseline vs 6.1 ± 0.7 methylcellulose-treated animals after 3 months; P > .05). Nor was there any change in HbA1c (from 4.0 ± 0.2 at baseline to 4.1% ± 0.2% in glimepiride-treated animals after 3 months vs 4.1 ± 0.2 in methylcellulose-treated animals after 3 months; P > .05). The HbA1c values were considered diabetic when >3.45% using our assay.
Discussion
The main findings from the present study are as follows: (a) we confirm that the diabetic heart is resistant to IPC such that 3 cycles of IPC instead of the usual 1 cycle are required to elicit cardioprotection; (b) the resistance to IPC observed in the diabetic heart can be overcome by pretreatment with the sulfonylurea, glimepiride, such that both 1 and 3 cycles of IPC confer cardioprotection in the diabetic heart; (c) this effect of glimepiride in facilitating IPC appears to be independent of its glucose-lowering effects, as evidenced by the lack of change in fasting glucose levels of HbA1c over 3 months glimepiride therapy.
The majority of previous experimental studies have demonstrated the diabetic heart to be resistant to the cardioprotection elicited by IPC, 17 –33 attributing the inability to precondition the diabetic myocardium to hyperglycemia, 19 impaired mitochondrial function, 32 and sarcolemmal KATP function. 23,28 In the current study, we too find that the diabetic heart is resistant to IPC, but that this can be overcome by increasing the IPC stimulus or treating with glimepiride. Our previous study 15 demonstrated that the diabetic heart was resistant to IPC, although this could be overcome by applying 3 cycles of the standard 1 IPC cycle. In the nondiabetic Wistar control rat, a single cycle of IPC was sufficient to achieve cardioprotection. In that study, the resistance to IPC was attributed to impaired signaling through the PI3K-Akt pathway, a key mediator of IPC-induced cardioprotection. 16 It has been well-established that PI3K-Akt signaling is impaired in diabetic animals, 8 and that PI3K-Akt signaling in both the preischemic and the postischemic reperfusion phases underlies the cardioprotection elicited by IPC in nondiabetic animal hearts. 38,39,49 However, in some large animal studies the contribution of PI3K-Akt signaling to reperfusion protection has been questioned with the suggestion that the Survival Activator Factor Enhancement pathway being more important in this setting. 50 –53 It would be interesting to determine whether glimepiride facilitates IPC by activating the PI3K-Akt pathway in the diabetic heart. In this regard, glimepiride has been reported to activate Akt in both coronary endothelial cells, 43 human umbilical vein endothelial cells, 44 and adipocytes. 45 Furthermore, it has been reported that the peroxisome proliferator-activated receptor (PPAR)-γ agonist, rosiglitazone, also has the ability to cardioprotect the diabetic rat heart through the activation of Akt. 54 Whether rosiglitazone would also overcome the resistance for IPC in the diabetic heart remains to be determined. One limitation of the present study is that we did not investigate whether the effect of glimepiride pretreatment on the threshold for IPC was mediated through the activation of the PI3K-Akt pathway. Whether antidiabetic therapy with glimepiride has the same effect on the cardioprotective efficacy of IPC in the diabetic patient is unknown given that the findings in our experimental study were undertaken in the ex vivo perfused rat heart.
With respect to IPC, previous studies by our laboratory and others have reported that the sulfonylurea glibenclamide but not glimepiride 42,55,56 or gliclazide 57 has the ability to abolish the cardioprotective effect elicited by IPC from its antagonistic actions on the ATP-dependent potassium channels within cardiac mitochondria, which are recognized to be pivotal to IPC-induced cardioprotection. 58 In the current study, we demonstrate that the antidiabetic sulfonylurea, glimepiride, is able to modify the response of the diabetic heart to IPC, but does not elicit cardioprotection itself suggesting that glimepiride is somehow potentiating the IPC signal. The mechanism through which glimepiride actually lowers the threshold for IPC in the diabetic heart is unclear, especially considering the different regimens in which it was shown to be effective. We speculate that glimepiride potentiates the PI3K-Akt signaling pathway in the diabetic heart which upregulates downstream signaling mechanistic pathways important in preconditioning. Whether the effect of glimepiride in overcoming the resistance of IPC in the diabetic heart is specific to this antidiabetic medication is unknown. Interestingly, the other classes of antidiabetic mediation such as PPAR-γ agonists 59 and metformin 60 have been demonstrated to confer cardioprotection in their own right.
In conclusion, we demonstrate that the treatment with the sulfonylurea glimepiride facilitates IPC such that the resistance to IPC observed in the diabetic heart is overcome. Therefore, certain types of antidiabetic medication may facilitate IPC and permit diabetic patients with CHD to benefit from this endogenous form of cardioprotection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the British Heart Foundation grant number FS/06/023. This work was undertaken at University College London who received a proportion of funding from the Department of Health’s National Institute of Health Research Biomedical Research Centres funding scheme.
