Abstract
Similar to ischemic preconditioning, high-intensity exercise has been shown to decrease infarct size following myocardial infarction. In this article, we review the literature on beneficial effects of exercise, exercise requirements for cardioprotection, common methods utilized in laboratories to study this phenomenon, and discuss possible mechanisms for exercise-mediated cardioprotection.
Beneficial Effects of Exercise
Nearly 60 years have passed since Morris and Crawford first published their groundbreaking findings in the
Exercise is generally considered to be protective against the risk of myocardial events by reducing several physiological risk factors such as elevated blood pressure, obesity, hyperlipidemia, and insulin resistance. 14 Accompanying the reduction in risk factors, long-term exercise provides beneficial effects directly on the myocardium itself. Endurance exercise training has displayed the ability to positively impact the heart through beneficial hypertrophy, angiogenesis, and improved contractility. 14,15 Additionally, individuals who exercise tend to experience relative bradycardia due to autonomic modulation, allowing for longer diastolic periods and elevated myocardial perfusion at submaximal workloads. 14,16 Overall, these effects combine to improve vascular, endothelial, and myocardial health, contributing to the decreased event risk and all-cause mortality associated with exercise.
While these long-term adaptive changes in response to exercise have been associated with a decrease in the incidence of acute MI, exercise may also protect the heart in instances in which a cardiac event does take place. Although the mechanism is not fully understood, there is evidence to suggest that exercise may mimic the favorable, cardioprotective effects of ischemic preconditioning 17 and attenuate cardiomyocyte death in the setting of myocardial ischemia–reperfusion (IR). In studies examining this effect, cardioprotection is determined by preservation of cardiac function via reduction or prevention of myocardial damage. Whether an intervention has resulted in cardioprotection can be established by demonstrating attenuation of cardiomyocyte death or reduction in infarct size. The surrogate markers for cardioprotection include a decreasing trend in cardiac enzymes (such as troponin T or I and creatine kinase-MB), preserved left ventricular (LV) ejection fraction (as assessed by echocardiography, myocardial nuclear ventriculography, or cardiac magnetic resonance imaging [MRI]), and reduced myocardial infarct size (which is also assessed with myocardial nuclear scanning, cardiac MRI, and in vivo studies by evaluation of cardiac tissues).
The protection provided by exercise appears to occur in a biphasic manner, with 2 separate “windows” of protection, beginning immediately after a single episode of exertion and continuing for multiple days. 12,18 -22 The first of these 2 windows begins immediately following the event and subsides within the first few hours following preconditioning. After approximately 24 hours, a second window of protection begins, extending for days to even weeks. 22 -24 The second window of protection has been reported to disappear after cessation of activity. For example, after 4 weeks of detraining, rats were found to have relatively less cardioprotection than those that continued to exercise. However, the rats that underwent detraining still maintained some form of cardioprotection compared to the control group. 25,28 Similarly, exercise in humans has produced a cardioprotective state of increased nitric oxide and decreased endothelin 1 levels. The protective state lasted for 4 weeks after cessation of exercise, returning to baseline in 8 weeks. 24 Despite similar protective effects during each window, the relationship between the mechanisms underlying these 2 time periods has yet to be determined.
Exercise Requirements for Cardioprotection
Duration
Beginning with the inaugural animal studies of exercise and protection from IR injury, conflicting opinions on the necessary intensity and duration to achieve cardioprotection have existed. An early study by Powers et al established that a rigorous 10-week training program at 75% to 80% of maximal oxygen consumption (VO2 max) was sufficient to improve myocardial contractile performance and reduce lipid peroxidation during IR in the rat in vivo. 11 A further study established that exercise programs of 3 to 5 days at 60% to 70% VO2 max were found to be effective, improving contractile efficiency following IR in rats. 13,26 Surpassing the 3- to 5-day success, researchers strive to see if any benefit of cardioprotection can be deemed from a single episode of exercise. Several subsequent studies effectively confirmed that cardioprotective benefits can be achieved in just 1 session. 21,27 -30 Despite this agreement with regard to duration, controversy continues regarding the intensity required in each individual session.
Intensity
In 2000, Lee et al utilized data gathered from the late 1980s to early 1990s to present the concept that short high-intensity bouts of exercise may be equally effective in protecting the cardiovascular system as longer, low-intensity workouts. 31 To date, this has not been sufficiently proven in IR experiments. As mentioned previously, cardioprotective benefits have been observed from exercise at 60% to 70% VO2 max as well as 75% to 80% VO2 max, representing benefits from both moderate and vigorous intensities. 11,13,26 Controversy exists in literature about the intensity of exercise and the level of cardioprotection. An earlier study comparing moderate- and high-intensity exercise in rats showed equivalent protection against IR injury. 29 However, a study found that infarct size reduced proportionally to the intensity of the exercise in rats trained at 60% VO2 max versus rats trained at 80% VO2 max for 10 weeks. 25 While the optimal intensity threshold has not been determined for IR protection, greater intensity overall does appear to be beneficial in long-term exercise regimens. In relation to event occurrence and long-term cardioprotection, Mittleman et al established a significantly decreased risk of MI following strenuous physical exertion in those who exercise regularly. 30 Compared to individuals who exercise at an intensity of 6 metabolic equivalents of task or higher at least 5 per week, sedentary individuals have a nearly 50-fold increase in relative risk of MI following strenuous exertion. As reported by Swain et al, epidemiological studies comparing vigorous and moderate-intensity aerobic exercise suggest that vigorous exercise provides greater cardioprotective benefit due in part to risk factor modification. 31 Although high-intensity exercise appears to be better in stimulating cardioprotection, it may not be well suited for elderly patients with comorbidities such as coronary artery disease or congestive heart failure. The American College of Sports Medicine recommends “30 minutes or more of moderate-intensity physical activity such as brisk walking on most, preferably all days of the week.” 32 Moderate-intensity exercise is defined as exercise at an intensity of 40% to 60% of VO2 max. 33 This recommendation seems reasonable in terms of inducing cardioprotection due to evidence of protection against IR injury in rats exposed to exercise at 55% VO2 max. 29
Exercise: A Surrogate for Ischemic Preconditioning?
In 1986, Murry et al discovered the concept of “ischemic preconditioning,” the notion that a relatively brief cardiac ischemic event protects the heart against a subsequent more prolonged episode of ischemia. The degree of myocardial damage (i.e., infarct size) caused by prolonged ischemia and reperfusion was reduced in patients who were “preconditioned” by a preceding brief ischemic event compared to those who were not. 17
Over time, it became well established that protection provided from preconditioning did not require application of brief ischemia directly to the heart itself. Remote ischemic preconditioning (RIPC) is the phenomenon whereby brief episodes of ischemia and reperfusion applied at distant tissues or organs render the myocardium resistant to a subsequent sustained episode of ischemia. This phenomenon was first described in 1993 by Przyklenk et al who demonstrated that brief ischemia in the circumflex coronary artery bed reduced subsequent infarction in the territory of the left anterior descending (LAD) artery. 34 Interestingly, RIPC can provide a similar magnitude of protection as observed with local preconditioning. 35 Despite promising findings in animal models, the results of human studies involving RIPC have been inconsistent. In 2007, Hausenloy et al reported decreased postoperative troponin leak in 27 patients who underwent RIPC prior to coronary artery bypass graft (CABG). 36 A single-center, double-blind randomized study including 329 patients undergoing CABG also reported lower troponin leak as well as lower all-cause mortality in the RIPC group. 37 In 2015, Meybohm et al conducted a prospective, double-blind, multicenter randomized controlled trial involving adults undergoing cardiac surgery. In contrast, the study did not observe any difference in composite primary end points or other outcomes between the RIPC and the control group. 38 A recent Cochrane review including 29 studies involving 5392 patients undergoing CABG with or without valve surgery showed no evidence that RIPC has a treatment effect on clinical outcomes, such as mortality and post procedure MI. However, the Cochrane review did report moderate evidence that RIPC reduces the amount of troponin release post procedure. 39 On review of the current evidence, it appears that RIPC does have a short-term effect on decreasing troponin release and protecting the myocardium during surgery. The effects of RIPC on other outcomes have been questionable. We feel that overall RIPC to an extent does exert cardioprotection in humans and the conflicting studies prove that we still do not have a strong understanding of the RIPC mechanism in order to exploit its full potential.
The fundamental difference between remote and local preconditioning is the fact that RIPC involves the communication or transfer of the protective trigger from a peripheral site to the heart. Both release of circulating humoral factors from the remote tissue during brief IR and neuronal transfer of the protective signal have been implicated to play a role. In 2009, Shimizu et al demonstrated in rats that transient limb ischemia released hydrophobic circulating factors of <150 kD which induced cardioprotection and were transferable across species. 40 Lim et al suggested that RIPC included both a neural and a humoral component by demonstrating in mice that occlusion of either the femoral vein (humoral component) or a combined femoral and sciatic nerve resection (neural component) resulted in complete nullification of the cardioprotective effect of RIPC. 41 In addition, Pickard et al suggested that release of humoral factors in RIPC required vagus nerve activation by showing that bilateral cervical vagotomy in rats abolished cardioprotection. Hexamethonium, a ganglionic blocker, was also shown to abrogate cardioprotection, suggesting that humoral factors may induce cardioprotection via recruitment of intrinsic cardiac ganglia. 42
At present, the exact details of neuronal signal transfer in remote conditioning of the heart and their role in the release of humoral factors are not entirely clear. Despite this uncertainty, the following pathway has been suggested. Following brief cycles of ischemia and reperfusion of the upper limb, adenosine is believed to be released which not only stimulates the release of local humoral factors but also activates the sensory peripheral nerves. These nerves travel to the spinal cord and the brain stem, increasing parasympathetic activity, and possibly initiating cardioprotection via release of humoral factors from the pre- or postganglionic nerve endings of the heart. 42 As this pathway suggests, rather than existing as mutually exclusive mechanisms, neural and humoral processes may act in concert to provide cardioprotection from remote conditioning.
A substantial body of evidence suggests that exercise is an effective surrogate for ischemic preconditioning with cellular mechanisms similar to both direct and RIPC. 22 While exercise is not typically regarded as an ischemic event, physical activity stimulates the production of endogenous ligands (including, eg, adenosine) as in RIPC. One possible mechanism for this relationship is the elevated activity of anaerobic glycolysis in both strenuous exercise and RIPC. During RIPC, the remote ischemic tissue is forced to utilize anaerobic glycolysis to produce adenosine triphosphate (ATP) for cell survival. In exercise, although skeletal muscle cells preferably utilize glycogen and fatty acids for energy production, more intense activity requires cells to undergo some degree of anaerobic glycolysis. 43 Thus, despite an approximate 5-fold increase in coronary flow during exercise, elevated myocardial oxygen demand and increased compressive forces on microvasculature may provide hypoxic conditions in the myocardium 44 which could potentially act as stimuli for preconditioning. Whatever the mechanism of initiation, exercise has clearly displayed similarities to forms of ischemic preconditioning.
A common theme shared by RIPC (in particular, the humoral component of remote conditioning) and exercise is the release of circulating ligands that display the capability to interact with myocytes and trigger pathways that result in intracellular signaling associated with cardioprotection. These circulating ligands can be transmitted by the transfer of plasma or serum from a preconditioned or exercised donor to a naive acceptor participant, both within and among species. 45,46 For example, postexercise plasma from humans has been shown to decrease cardiac infarct size by 40% in a rabbit Langendorff IR model. 46 As discussed below, this property provides a significant opportunity to perform translational studies in which the mechanisms of RIPC and exercise, applied in human participants, can be investigated.
Mechanisms of Exercise Cardioprotection
Models and Methods
The established gold standard of cardioprotection by ischemic and remote preconditioning—and thus, by extrapolation, using exercise as the preconditioning stimulus—is reduction in myocardial infarct size. For preclinical models, in vivo experiments 47 can be designed to both quantify infarct size and interrogate potential mechanisms that contribute to the infarct-sparing effect of a preceding period of exercise. For investigation of the mediators and mechanisms of exercise-induced cardioprotection in humans, alternative methods are clearly required. For global pathophysiologic assessment of parameters related to cardiac function and infarct size, the Langendorff isolated perfused heart has traditionally been used. In order to explore the phenomenon of exercise-induced cardioprotection on a more molecular level, various cell models have also been utilized. Although other methods have also been used, novel insights have been provided by testing of human-derived samples in Langendorff and cell culture studies.
Langendorff isolated buffer-perfused heart
With the discovery of retrograde perfusion in the isolated heart model by Oskar Langendorff in 1895, the Langendorff isolated buffer-perfused heart method continues to provide a lasting contribution in the study of mammalian heart physiology due to the method’s ability to simulate heart function in an isolated organ system under physiological parameters. 48 The Langendorff system’s ability to eliminate systemic influences, such as hormonal factors, autonomic regulation, and the inflammatory response, enables controlled variables to be applied to a cardiac model in order to assess cardiac function. 44 Thus, the Langendorff model is extensively used in IR studies with the intent to gain insight into the mechanisms underlying IR injury. 49
The model allows for cardiac variables, such as LV developed pressure, rate of change in LV developed pressure (d
Cell models
In addition to the Langendorff model, many previously conduced IR experiments have expanded their findings with results from cardiomyocyte cell culture studies. Early cardiomyocyte cell cultures involved isolation of cardiomyocytes from adult rats. In 1983, Nag et al developed a method to remove the hearts of adult rats, enzymatically dissociate the myocardium and maintain isolated myocyte suspensions in culture for 45 days. 52 This established the feasibility of studying cardiac cells in a highly controlled environment. The basic concept of this cardiomyocyte isolation method has been expanded to include harvesting of cells from both neonatal and adult rats, with extension to multiple species including mice, guinea pig, and rabbits 53 -55 and most recently human cardiomyocytes. 56 -59 The model allows for the design of protocols measuring various aspects of cardiomyocyte function and integrity, including gene transcription and expression, protein content, cell signaling, growth, hypertrophy, and cell death. 56,60 -65 Researchers have also isolated and identified cardiomyocytes from the hearts of mice 66 and rats exposed to exercise. 67 -70 In addition to the use of primary cultures of cardiomyocytes, researchers have utilized immortalized cell lines, including H9c2 rat myofibroblasts 71 -73 and murine-derived HL-1 cardiomyocytes, the only immortal cardiomyocyte cell line. 74 The differences between primary culture and cell lines allow for varying factors to influence the environment of the IR cell model. For instance, primary cardiomyocytes begin to die within 20 to 30 minutes of ischemia; however, it takes several hours for HL-1 cells to die under ischemia. 75 The immortalized cells are highly glycolytic compared to primary cultured adult cardiomyocytes. In recent years, human induced pluripotent stem cells-derived cardiomyocytes have been successfully cultured and provide a human cardiomyocytes model in which to research exercise effects. 76 -78
However, in relation to the ischemia and reperfusion process that occurs during acute MI, the cultured cardiomyocyte model provides a reductionist approach for examining the cellular mechanisms underlying cardioprotection. Exercise preconditioning through hypoxia reoxygenation is one of the mechanisms of exercise-induced cardioprotection. Reoxygenation causes reactive oxygen species (ROS), which may not only induce oxidative damage but also provide cardioprotection through activation of cell signalings. 79 Many experiments have varied culture medium and examined the effects following exposure to hypoxia-reoxygenation. 80 -84 These in vitro conditions may not mimic the situation in vivo. For example, the death of cardiomyocytes following ischemia and reperfusion can continue up to 3 days post-reperfusion. 85 However, the cell models are usually observed for less than 24 hours. The limitation of cell models restricts the ability to generalize research findings and the application of in vivo model can provide better comparison to human conditions.
LAD artery occlusion and IR model
Myocardial injury through ligation of the LAD is an established in vivo model to mimic clinical MI. Measurement of the infarct size in LAD occlusion and IR is a validated method to evaluate the cardioprotective effect of exercise. In a study by de Waard et al, early exercise training by voluntary treadmill running started within 24 hours following a large MI had no effect on MI size but attenuated the MI-induced LV dysfunction without a detrimental effect on LV remodeling. 86 Bito et al also reported that early exercise training after MI prevents cardiac contractiles dysfunction but no effect on cardiac remodeling. 87 Exercise prior to a MI has the benefit of reducing infarct size and improving angiogenesis through activation of vascular endothelial growth factor. 88 Continuous exercise training before MI and reinitiated early after a MI blunted the trends of infarct size-dependent systolic dysfunction and LV dilation. 89 Interestingly, Vujic et al reported that exercise induces new cardiomyocyte generation in the adult mammalian heart by mouse MI model. Inhibition of miR-222, an miRNA-induced during exercise, blocked the cardiomyogenic response of exercise. 90 Exercise training also protects from IR injury. Esposito et al elucidated the relationship of training intensity and cardioprotection after IR injury in rats. 25
Mechanisms
The exact mechanism of exercise-induced cardioprotection against lethal IR injury is still not properly understood. Studies to date indicate that a number of factors and pathways are likely to be involved rather than a single mechanism. In the following paragraphs, we will highlight the factors that have thus far been suggested to play a role (Figure 1).
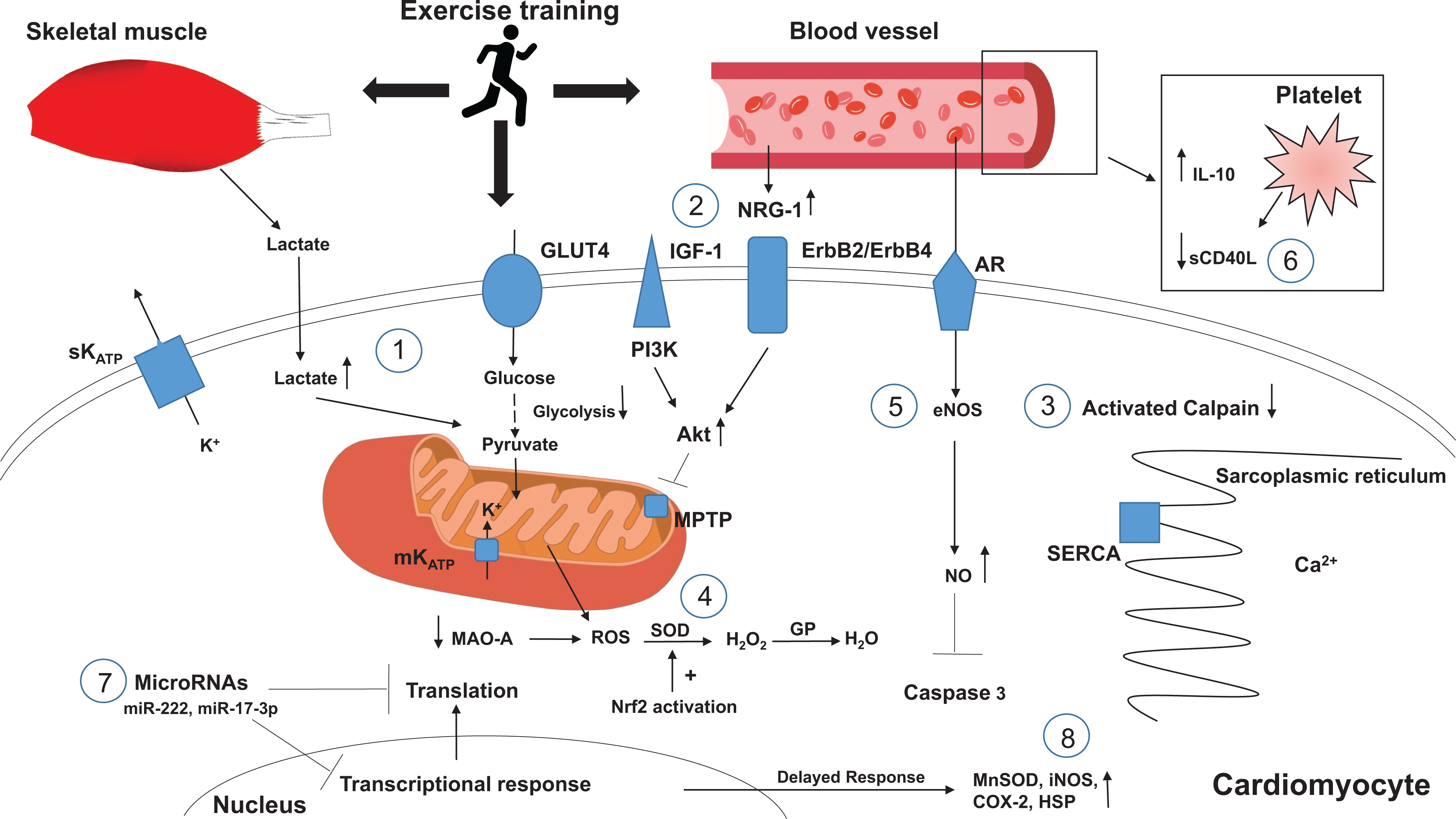
Proposed mechanisms of exercise cardioprotection.
Alteration in cardiac metabolism during ischemia
Under normal physiological conditions, fatty acid oxidation is essential for adult cardiomyocytes energy production. During an ischemic event, the decreased oxygen concentration inhibits oxidative phosphorylation in the mitochondria. To sustain ATP, cellular metabolism switches to utilizing glucose through anaerobic glycolysis, resulting in lactate formation and acidosis. The cumulative effect of decreased pH and ATP results in increased intracellular sodium and calcium concentrations, which eventually triggers the pathway of ischemic injury and, if sustained, leads to cell death. 91 Depending on the conditions, cardiomyocytes are metabolically flexible 92 and other substitute substrates, such as glucose, lactate, amino acid, and ketone bodies, can be oxidized by cardiomyocytes for generating ATP. Similarly, during exercise, the lactate produced by skeletal muscle is also oxidized by cardiomyocytes and contribute to ATP synthesis. 93,94 Endurance exercise training has been shown to decrease the rate of glycolysis in rat heart during ischemia. Although the exact mechanism is unknown, exercise training could potentially improve the efficiency of cells in producing ATP during ischemia. 95
Another potential mechanism of exercise-induced metabolic alteration is through the interaction between neuregulin (NRG) and the epidermal growth factor receptor/receptor tyrosine kinase (erbB) family of receptors. The NRGs are growth factors that are responsible for the growth and development of mammalian tissue, including skeletal and cardiac muscles. The NRG is believed to be involved in the regulation of glucose uptake in muscle cells, especially during the recruitment of glucose transporters to the cell surface and modulation of transporter expression. 96 Thus, NRG may participate in improving the availability and the efficiency of glucose utilization during periods of prolonged ischemia. Jie et al demonstrated in an in vitro study that NRG may play an additional role in determining cell fate: Specifically, NRG-1 was associated with activation of the phosphatidylinositol 3-kinase (PI3K) pathway during oxidative stress in cardiac tissue, preventing the mitochondrial permeability transition pore (MPTP) from opening and limiting myocardial damage during the ischemic event. 56 Additional studies have supported the role of NRG-1 activation of PI3K in cardiac protection and documented an elevation in plasma levels of NRG-1 in response to exercise. 97,98 Furthermore, progressive resistance training has been shown to increase the expression of erbB2 receptors, 99 and the presence of NRG β-1 in serum of humans has been established as a marker for cardiovascular fitness. 100 Furthermore, progressive resistance training has been shown to increase the expression of erbB2 receptors, 99 and the presence of NRG β-1 in human serum has been established as a marker for cardiovascular fitness. 100
Intracellular calcium homeostasis
Ischemia–reperfusion injury results in intracellular calcium overload which is responsible for a number of deleterious effects in cardiomyocytes, mainly mediated via the activation of calpain. Upregulation of this protease leads to degradation of structural proteins and activation of Bid, which causes mitochondrial dysfunction and proteolysis of sarco/endoplasmic reticulum calcium ATPase (SERCA) and ryanodine receptors, culminating in calcium dysregulation and hypercontracture of the cardiomyocytes. 101 Exercised hearts have been shown to have better calcium handling through improved diastolic pressure during IR, decreased activation of calpain, and preserved SERCA when compared to nonexercised hearts. 102 -104
Redox homeostasis
Reactive oxygen species
Another major player in IR injury is the generation of cytotoxic ROS due to depolarizations in mitochondrial membrane potential in cardiomyocytes at reperfusion 105 and calcium-medicated damage in ischemia. 106 The adverse consequences of ROS production include disruption of cell membranes via lipid peroxidation, causing damage and dysfunction of receptors and ionic channels. For example, ROS damage the sarcolemma, leading to an influx of calcium into the cytosol via sodium-calcium exchange and, as a result, intracellular calcium overload. The ROS also attracts neutrophils, contributing to further cell damage via increased inflammation. Reactive oxygen species, together with normalization of pH upon reperfusion, also contribute to the opening of the MPTP, which plays a role in triggering apoptosis via release of cytochrome C. 107 As mentioned previously, opening of MPTP triggers apoptosis by disruption of the mitochondrial membrane potential, matrix swelling, rupture of mitochondrial membranes, and release of cytochrome C in the cytoplasm. 22 Acidosis during ischemia maintains the MPTP in a closed state; however, upon reperfusion, lactate levels decrease and the pH rapidly normalizes, opening the MPTP. Exercise has been shown to delay the rapid normalization of the pH, 108,109 therefore preventing MPTP opening and providing cardioprotection. Furthermore, exercise has been shown to sustain postischemic mitochrondrial bioenergetics and redox homeostasis, which is associated with preserved mitochrondrial membrane potential and protection against ventricular arrhythmias. 69
Controlling ROS production and/or increasing the cellular concentration of antioxidant enzymes (including the superoxide dismutases [SODs], catalase, glutathione peroxidase, and glutathione reductase) have been long-standing, proposed strategies to decrease IR injury. 110 However, the proposed strategies as of yet have not been proven to be “effective” in attenuating lethal IR injury and infarct size. Nonetheless, exercise has been shown increase the expression of endogenous antioxidants such as SOD1 and SOD2 in the mitochondria 111,112 possibly via activation of the transcription factor nuclear factor erythroid-2 (Nrf2). 113 In addition, exercise training has also been suggested to increase the activity of glutathione reductase in the heart via posttranslational modifications. 114,115 Further studies have shown that exercise training decreases the production of ROS during IR from both subsarcolemmal and intermyofibrillar mitochondria and prevents the release of proapoptotic proteins from the cardiac mitochondria, hence providing cardioprotection. 112 Finally, mitochondrial monoamine oxidase A (MAO-A) catalyzes the oxidative deamination of several monoamines, thus increasing ROS production. 116 Exercise has been shown to decrease the expression of MAO-A in rat hearts. 117 In summary, exercise training may provide cardioprotection by increasing antioxidant expression and attenuating ROS-mediated cell damage.
Nitric oxide
In the initial minutes to hours following reperfusion, the endothelial cells become permeable, leading to edema, the expression of adhesion proteins, release of cytokines, and decrease in the production of nitric oxide (NO). 118 -120 Improvements in NO levels due to exercise can improve coronary hyperemia at the onset of reperfusion. 119 However, the effect of NO-mediated reduction in reperfusion hyperemia is reduced with age. 121 Nitric oxide as a mediator of exercise-induced cardioprotection also plays a role in upregulation of endothelial nitric oxide synthase (eNOS) protein and phosphorylation, inhibition of caspase 3 activity, and modification of complex I of the mitochondrial electron transport chain, resulting in decreased ROS production during IR. 122
ATP-sensitive potassium channels
Opening of ATP-sensitive potassium channels (KATP), present on both the sarcolemma (most likely in the intercalated disc in the cardiomyocytes) and the inner mitochondrial membranes, has been implicated to play a role in ischemic preconditioning cardioprotection. The sacrolemmal KATP channel is composed of 4 Kir6.x and 4 SURx subunits. 123 However, there is a debate on mitochrondrial KATP channel. 124 The proposed subunits of the mitochrondrial KATP channel are Kir1.1 (ROMK), pore-forming subunits, and additional subunits for ATP inhibition of the channel complex. Unique short-form SUR2 splice variants have been proposed as a regulatory subunit of the mitochondrial KATP channel. 123 Research has futher supported the role of these subunits in cardiac function, with Kir6.1, Kir6.2, and SUR2A found to be present in isolated ventricular myocytes. 125 Cardiac KATP channels are favorably regulated by glycolytically derived ATP. Competition for local glycolytically derived ATP may also be responsible for the known functional interaction between the KATP channel and the Na+/K+ pump in the cardiac myocyte. 123 This interaction has been suggested to be important in the protection provided by ischemic preconditioning. Transcription of KATP channel subunit mRNA, Kir6.1, SUR1, and SUR2A mRNA was upregulated in LAD rat hearts. These KATP channels open when ATP is depleted or in the setting of increased concentration of other endogenous factors such as NO, pH, fatty acids, adenosine, and acetylcholine. 126,127 Possible mechanism by which sarcolemmal KATP offers cardioprotection is by shortening the cardiac action potential duration via acceleration of phase 3 repolarization that limits calcium entry into the cell via L-type calcium channels, preventing calcium overload within the cell. 128 Sarcolemmal KATP can also trigger opening of mitochondrial KATP, 129 which have been shown to attenuate IR-induced ventricular arrhythmias. 130 Of potential interest, endurance exercise training has been shown to increase the expression of sarcolemmal KATP in cardiac myocytes. 131 -134 However, their opening appears to be more beneficial during ischemia not reperfusion. 134
Inflammation
During reperfusion, ROS, a neutrophil chemoattractant, has been implicated to contribute to further myocardial damage through increased inflammation. Moreover, the endothelial cells in the reperfused myocardium begin to express adhesion proteins, release cytokines, and reduce NO production, which promotes adhesion, accumulation, and activation of neutrophils. Thus, inflammation may exacerbate cardiomyocyte damage during IR, and exercise has been shown to play a role in decreasing inflammation 135 and providing cardioprotection. However, inflammation is a complex and controversial topic in cardioprotection, and further studies are needed to validate its role.
sCD40L
Clinical studies consistently show that the interaction between damaged endothelium, platelets, and immune cells is responsible for the development and progression of atherosclerosis. 136 Platelet activation leads to crucial integrin-mediated signaling cascades, which result in stable interactions between platelets and the endothelium as well as activation of glycoprotein IIb/IIIa receptors (primary aggregation receptors), release of α and dense granules, and circulating cytokines that activate the immune system and upregulate receptor expression on endothelial cells. 136 CD40 receptors and their ligand CD40L are one such system that is rapidly inducible upon platelet activation and has been reported to play a role in atherosclerosis as well as in ischemic preconditioning. 136 CD40 and CD40L are stored in the cytoplasm of platelets and are rapidly expressed on the platelet surface upon activation. 137 CD40L is then cleaved into a biologically active circulating form, soluble CD40L (sCD40L), which binds to CD40 on endothelial cells. The binding of sCD40L evokes a subsequent increase in proinflammatory cytokine production and adhesion molecule expression on the endothelial cell surface such as E-selectin, vascular cell adhesion molecule-1, and intercellular adhesion molecule-1. 137 Ischemic preconditioning has been shown to prevent the increase in levels of circulating sCD40L during angioplasty, suggesting that preconditioning induces anti-inflammatory effects that may, potentially, contribute to cardioprotection. 138 The association between exercise and reduced CD40 has been previously demonstrated. In mice, both moderate aerobic exercise and resistance training have been shown to decrease CD40 levels. 139,140 While in humans, high-intensity exercise has been shown to decrease CD40L levels, decrease triglycerides, and increase high-density lipoproteins, 141 whereas ultra-endurance exercise in athletes reduced sCD40L levels. 142 In addition, exercise training has been shown to significantly reduce circulating sCD40L levels in patients with chronic heart failure. 143 Therefore, exercise can contribute to cardioprotection by decreasing inflammatory mediators such as CD40 and reducing atherosclerosis, which is a major risk factor for cardiovascular disease.
Interleukins
Interleukins (IL) are a subset of cytokines secreted by white blood cells that participate in the immune response. The IL-6 and IL-10 have been shown to increase with exercise and may have a role in cardioprotection. For example, evidence obtained in exercised mice has revealed an upregulation in serum levels of IL-6 and IL-6 receptors, which, in turn, are associated with an attenuation of IR-induced necrosis and arrhythmias. These changes in IL-6 were accompanied by increased expression of the phosphorylated forms of p44/42 MAPK (Thr202/Tyr204) and p38 MAPK (Thr180/Tyr182), processes implicated as possible mechanisms for cardioprotection. 144 The IL-10 is an inhibitor of cytokine synthesis, and exogenous administration of this IL has been shown to reduce myocardial infarct size and inhibit neutrophil adhesion to vascular endothelium. 145 Further studies have shown that exercise increases serum IL-10 levels and could prevent LV remodeling by inhibiting inflammatory cytokines. 146,147
MicroRNAs
An emerging trend in the debate of exercise-induced and ischemia-induced preconditioning is the study of microRNAs (miRNAs) and their role in the various pathways involved with cardioprotection. The miRNAs are small noncoding single-stranded RNA molecules that bind to 3′ untranslated region of their mRNA targets and downregulate gene expression via degradation or translational inhibition. 148 Recent studies have suggested that families of miRNAs regulate signaling pathways involved in IR injury and may be a potential therapeutic target to reduce infarct size. Accordingly, identifying these miRNAs, their mechanism of action, and the phase of the IR injury cascade in which they are activated may provide novel insights. If miRNAs do contribute to cardioprotection, 1 potential strategy would be to create oligonucleotides with sequences similar to the identified miRNAs and use them to enhance prosurvival pathways in the setting of IR. 149
To date, increased levels of miRNA-21, miRNA-92a, miRNA-126, miRNA-133, and miRNA-144 and decreased levels of miRNA-1, miRNA-29, miRNA-199a, and miRNA-320 along with others have been shown to be associated with cardioprotection. 150 In addition, 4 microRNAs, miRNA-487b, miR-139-5p, miR-192, and miR-212, are reportedly modified by preconditioning. 149 Exercise has been shown to increase the levels of miR-222, a miRNA necessary for cardiac growth and protection against pathological hypertrophy. 66,151 Exercise also increases the levels of miR-21, miR-486, miR-378, and miR-940. 152 -155 This involvement of miRNAs and the documented ability for exercise to exert effects at an epigenetic level may provide clues that cardioprotection is regulated by more complex mechanisms than what could be explained by a simple humoral factor.
Transcription of de novo proteins
The early phase of cardioprotection after exercise is mainly believed to be due to immediate release of activated transmitters. The mechanism of the second window of protection, which comes into effect after 24 hours, is relatively more complex and involves adjustment at the genetic level and de novo synthesis of effector proteins. 156 Ischemic preconditioning is believed to produce trigger factors such as adenosine, opioids, and bradykinin. These factors activate intracellular signaling pathways that activate the transcription factors in the nucleus. Over the next 12 to 24 hours, the production of proteins (distal mediators) such as mitochondrial antioxidant manganese SOD, inducible NOS, cyclooxygenase 2, and heat shock protein ultimately activate the cardioprotective pathways. The protective pathways are usually activated within 24 to 48 hours following an ischemic insult and reperfusion injury. 157 In addition, exercise preconditioning also stimulates the production of cytokines that activates the JAK/STAT pathway and results in the transcription of distal mediators. 157
Future Direction and Clinical Implications
Despite the extensive history of research into exercise and cardioprotection, many questions remain unanswered. The possible ligands and cellular mechanisms underlying the benefits from exercise are still unclear, and further research is needed to understand what endogenous factors contribute to exercise-induced cardioprotection. Likewise, the duration and intensity of exercise that are required to induce cardioprotection, and the duration of such effects following physical activity, remain uncertain. Although long-term exercise training and single bouts have both displayed cardioprotective benefits, each may do so through different mechanisms and provide variable duration of protection—concepts that warrant future targeted investigation.
Pairing in vivo analysis with data obtained in the Langendorff isolated buffer-perfused heart and in cell culture models may contribute to the understanding of the mechanistic interactions contributing to exercise-induced cardioprotection. Translational studies with humans in moderate-intensity exercise regimens may also provide important mechanistic insights. Elucidation of these mechanisms may yield promising therapeutic targets for cardioprotection.
Footnotes
Acknowledgments
We thank Dr Karin Przyklenk of Cardiovascular Research Institute, Wayne State University School of Medicine, for sharing her valuable insight, knowledge, and suggestions. We also think Ms Hailan Hu for her excellent artistic work.
Authors’ Contribution
Liu, L contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy; Chowdhury, M and Sholl, H contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval; Sharrett, M contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and gave final approval; Haller, S contributed to conception, contributed to acquisition and interpretation, critically revised manuscript, and gave final approval; Cooper, C and Gupta, R contributed to conception, contributed to interpretation, and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received following financial support for the research, authorship, and/or publication of this article: This work was supported by University of Toledo, College of Medicine and Life Sciences, Dean’s fund.
