Abstract
Introduction
One of the key mechanisms in cardiovascular disease is an acceleration of vascular pathology particularly in patients with cardiometabolic disorders such as hypertension, diabetes mellitus, and dyslipidemia. 1–4 The metabolic syndrome consists of risk factors including diabetes, prediabetes, abdominal obesity, high cholesterol, and high blood pressure. 5,6 Patients with these disorders are twice as likely to die from and 3 times as likely to have a myocardial infarction or stroke. 6,7
Development of hypertension, diabetes mellitus, and other cardiovascular diseases is associated with activation of the renin-angiotensin system (RAS) and endothelial dysfunction. The major bioactive product of the RAS, angiotensin II (ATII), affects plaque buildup in the arteries. 8–10 Evidence suggests that hypertension, obesity, impaired glucose tolerance, and increased insulin resistance are linked to stimulation of the RAS and endothelial dysfunction in multiple tissue types. 11 Agents that inhibit the RAS such as angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) show considerable benefit in hypertension, atherosclerosis, and heart failure. 12–14 Studies have shown that ARBs and ACEIs improve endothelium-dependent vasomotor responses as they indirectly promote the presence of endothelial nitric oxide, thereby improving endothelial function. 15–18 Also, ACEIs and ARBs exert protective effects by limiting ATII-mediated inflammatory and vasoconstricting responses. 19,20 Angiotensin-converting enzyme inhibitors work by inhibiting ACE, thereby limiting the production of ATII. Angiotensin-converting enzyme inhibitors also inhibit kinase II, thereby indirectly resulting in increased nitric oxide release. 21,22
α-Lipoic acid (ALA), also known as thioctic acid, is a naturally occurring dithiol compound synthesized in the mitochondria. The compound is both water and fat soluble; therefore, it is distributed in cellular membranes and cytosol in both plants and animals. Both ALA and its reduced form (dihydrolipoic acid) exhibit antioxidant properties including quenching of reactive oxygen species, regeneration of antioxidants, chelation of metal ions, and repair of oxidized proteins. 23 α-Lipoic acid is abundant in the human diet mostly from animal sources, but also in small quantities from fruits and vegetables. 24 In Europe, ALA has been prescribed for decades for the treatment of diabetic neuropathy; whereas in the United States, it is available as a dietary supplement. Therapeutic doses of ALA in humans range from 200 to 1800 mg/d, with no significant adverse effects reported 25 at doses as high as 2400 mg/d. Studies have demonstrated that supplementation with ALA reduces blood pressure, reduces plasma triglycerides, and increases low-density lipoprotein (LDL) oxidation lag time. 26–29 In diabetics, ALA has been shown to be beneficial in preventing damage to the endothelium and controlling the progression of diabetic nephropathy. 30,31 Additionally, it has been observed that ALA improves glucose metabolism and insulin sensitivity in diabetic patients. 32,33 We have previously reported that in conjunction with irbesartan (a RAS inhibitor), ALA decreases vascular inflammation and improves endothelial function in patients with the cardiometabolic syndrome. 34–36 Studies have demonstrated that supplementation with ALA has cardiovascular risk reduction benefits in terms of LDL oxidation lag time, reduction in plasma triglycerides, and control of blood pressure. 37–40 In diabetic patients, ALA has been shown to be beneficial in preventing endothelial damage and controlling the progression of diabetic nephropathy as measured by urinary albumin levels. 41 Additionally, clinical studies suggest that ALA improves glucose metabolism and insulin sensitivity in diabetic patients. 42–44 In the ISLAND study, our group evaluated the effects of the combination of ALA and irbesartan, a RAS inhibitor. Our results suggest that ALA and irbesartan decreased inflammation as measured by serum interleukin-6 levels and improved endothelial-dependent flow-mediated vasodilation. 26
Testing for urinary albumin is a simple and inexpensive way to assess early stage diabetic nephropathy. Previous data indicate that renal failure may be attenuated by RAS inhibitors, which limit the progression of microalbuminuria and proteinuria. Use of ARBs results in improved arterial compliance and reduced cardiac overload. 27 Angiotensin receptor blockers, used alone or with ACEIs, also preclude left ventricular remodeling after a myocardial infarction. 28,29
The purpose of this study is to determine potential associations of the ACEI Quinapril (QUI) in the presence or absence of ALA, in participants with both diabetes and hypertension. Associations examined are in respect to blood pressure regulation, proteinuria, and endothelial function.
Methods
Participants
Men and women aged 18 years or older with type II diabetes mellitus (fasting blood glucose level ≥126 mg/dL) and a history of hypertension (defined as on medical therapy or having systolic blood pressure >125 mm Hg at the time of screening) were enrolled in the study. Participants were excluded if they had any of the following: systolic blood pressure >180 mm Hg; hemoglobin A1C, > 7.5%; serum creatinine, >2.5 mg/dL; clinical history of coronary artery disease or congestive heart failure; coronary angiography planned prior to baseline sampling; chronic autoimmune disease; chronic inflammatory disease or known cancer in evolution; life expectancy less than a year; pregnant or nursing women; on immunosuppressives; or treatment with another investigational drug within the last 30 days. Patients on lipid-lowering therapy at the time of enrollment continued with the therapy without change throughout study. Written informed consent was obtained from all participants.
Study Design
Participants were randomized in a double-blinded, crossover manner to either QUI (40 mg/d; Pfizer, Groton, Connecticut) or QUI + ALA (QUI 40 mg/d; Pfizer, and ALA 600 mg/d; Greenstone Pharmaceutical, Peapack, New Jersey) for 8 weeks. A placebo pill was given to the QUI group, and all participants received 2 tablets throughout the treatment periods. After the first treatment period, patients underwent a 4-week washout period. Patients were then randomized in a crossover fashion to the alternate pharmacological regimen. Allocation concealment was maintained until the end of the study. Pill counts were obtained at the end of treatment period to determine compliance. Participants were advised to self-administer half the QUI dose during the initial week of therapy, after which they took the full dose at the same time each morning. The total study period was 22 weeks.
After 2 weeks of treatment, blood pressure was rechecked, and blood was drawn to measure serum creatinine and potassium. Blood pressure was checked on at least 3 separate measurements taken approximately 5 minutes apart using an Omron sphygmomanometer. All vascular measurements were conducted in the morning by a single trained and experienced investigator. The patients were asked to fast prior to blood draw. The study protocol was approved by the Institutional Review Board prior to its implementation.
Measurement of 24-Hour Collection of Urinary Albumin
To determine the effect of therapy on albuminuria in type II diabetic patients with hypertension, participants provided a 24-hour collection of urine at the beginning and end of study period for each treatment period. The urine was analyzed, and protein analysis was performed via chemical analysis (Quest Laboratories, Scranton, Pennsylvania).
Measurement of Endothelial Function by Flow-Mediated Dilation
Evaluation of endothelial function was made noninvasively using brachial artery reactivity testing (BART) to evaluate endothelium-dependent flow-mediated dilation (FMD) in the brachial artery. 30 The test was performed using a Philips 5500 (11/3 MHz) ultrasound machine. Patients were positioned briefly in the supine position with the arm in a comfortable position for brachial artery imaging. A blood pressure cuff was placed on forearm, after which a baseline rest image was acquired. The brachial artery was imaged above the antecubital fossa along the longitudinal plane. A segment with clear anterior and posterior intimal interfaces between the lumen and the vessel wall was selected for continuous two-dimensional (2-D) gray-scale imaging. Blood flow velocity was estimated by time averaging the pulsed Doppler velocity signal obtained from a mid-artery sample volume. The cuff was then inflated to ≥50 mm Hg above systolic blood pressure to occlude arterial flow for 5 minutes. After cuff deflation, the longitudinal image of the artery was recorded continuously from 30 seconds before to 2 minutes following cuff deflation. A mid-artery pulsed Doppler signal was obtained on immediate cuff release, no later than 15 seconds after cuff deflation to assess hyperemic velocity. After 15 minutes, nitroglycerin, 0.4 mg, was given sublingually, and additional images were obtained to determine endothelium-independent vasodilation.
The diameter of the brachial artery was measured from longitudinal images in which the lumen-intima interface was visualized on both the near (anterior) and far (posterior) walls. Upon selecting image for analysis, boundaries for diameter measurements are identified manually with electronic calipers (Vascular Tools 5, Medical Imaging Applications, Coralville, Iowa); average diameter was calculated from at least 3 different diameter measurements determined along a segment of the vessel. Brachial artery diameter was measured concurrently within cardiac cycle, using electrocardiograph (ECG) gating during image acquisition. Flow-mediated dilation was typically measured as the change in poststimulus diameter as a percentage of the baseline diameter. In accordance with the guidelines, baseline diameter, absolute change, and percentage change in diameter were measured and reported. 30
Statistical Analysis
All values are presented as a mean ± standard deviation for continuous variables, and a percentage of total patients for categorical variables. The paired t test and chi-square test were used for comparison of continuous and categorical variables, respectively. A P value of <.05 was considered statistically significant, and all P values were 2-sided. Calculations were performed with SPSS software (version 10.0, Statistical Package for the Social Sciences, Chicago, Illinois).
Results
Study Demographics
In total, 40 participants (18 men and 22 women) were enrolled in the study and followed for 22 weeks. A total of 28 patients completed the study. Baseline characteristics are summarized in Table 1. Noted parameters had no significant change before, during, or after study. At baseline, 30% of the study population was on antihypertensive therapy, 25% on aspirin, 32.5% on statin, 37.5% on metformin, 27.5% on sulfonylurea, 15.0% on thiazolidinedione (TZD), and 2.5% on insulin.
Patient Demographics and Baseline Characteristics
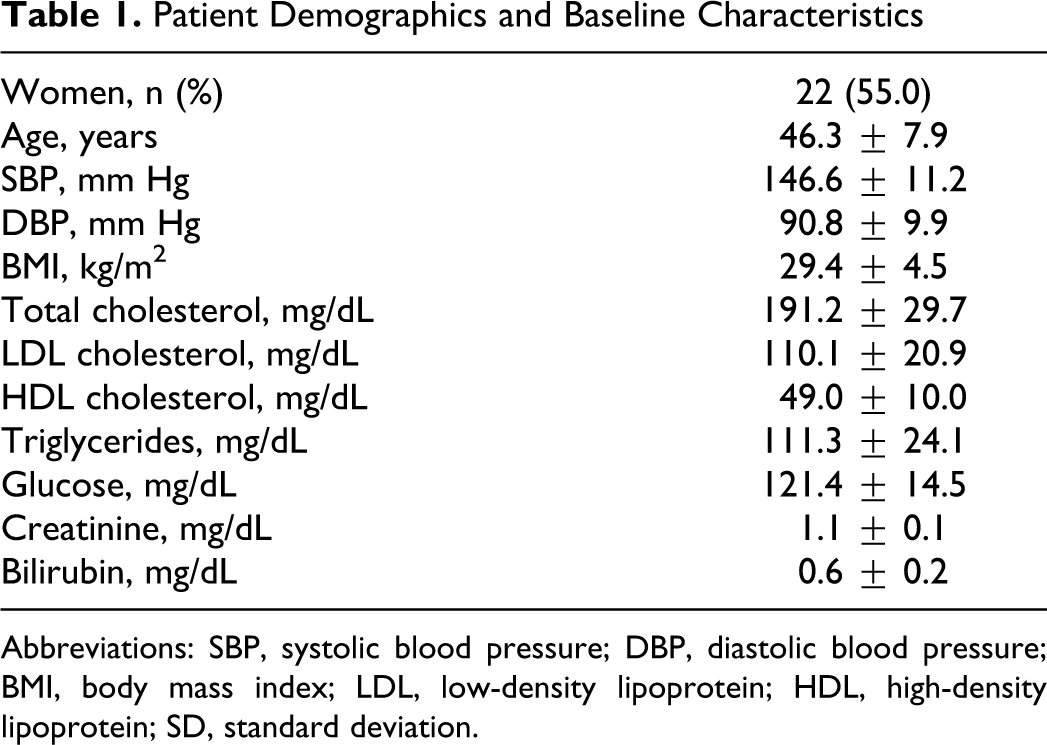
Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; LDL, low-density lipoprotein; HDL, high-density lipoprotein; SD, standard deviation.
a Data are means ± SD or n (%).
Side Effects and Blood Pressure Lowering in Response to QUI and QUI + ALA
There was a similar incidence of cough in QUI and QUI + ALA groups (QUI: 14% and QUI + ALA: 13%). Nevertheless, enrolled participants were compliant with study medication. Specific adverse effects are summarized in Table 2. No angioedema was noted throughout the study. There was a rise in serum potassium or creatinine of >20% in 1 of 40 participants in the QUI group. A significant reduction in systolic (10%) and diastolic blood pressure (10%) was also observed in QUI and QUI + ALA groups. There was no additional blood pressure-lowering effect of ALA (Figure 1). No participant in either group experienced hypotension (ie, systolic BP < 100 mm Hg) during the study.
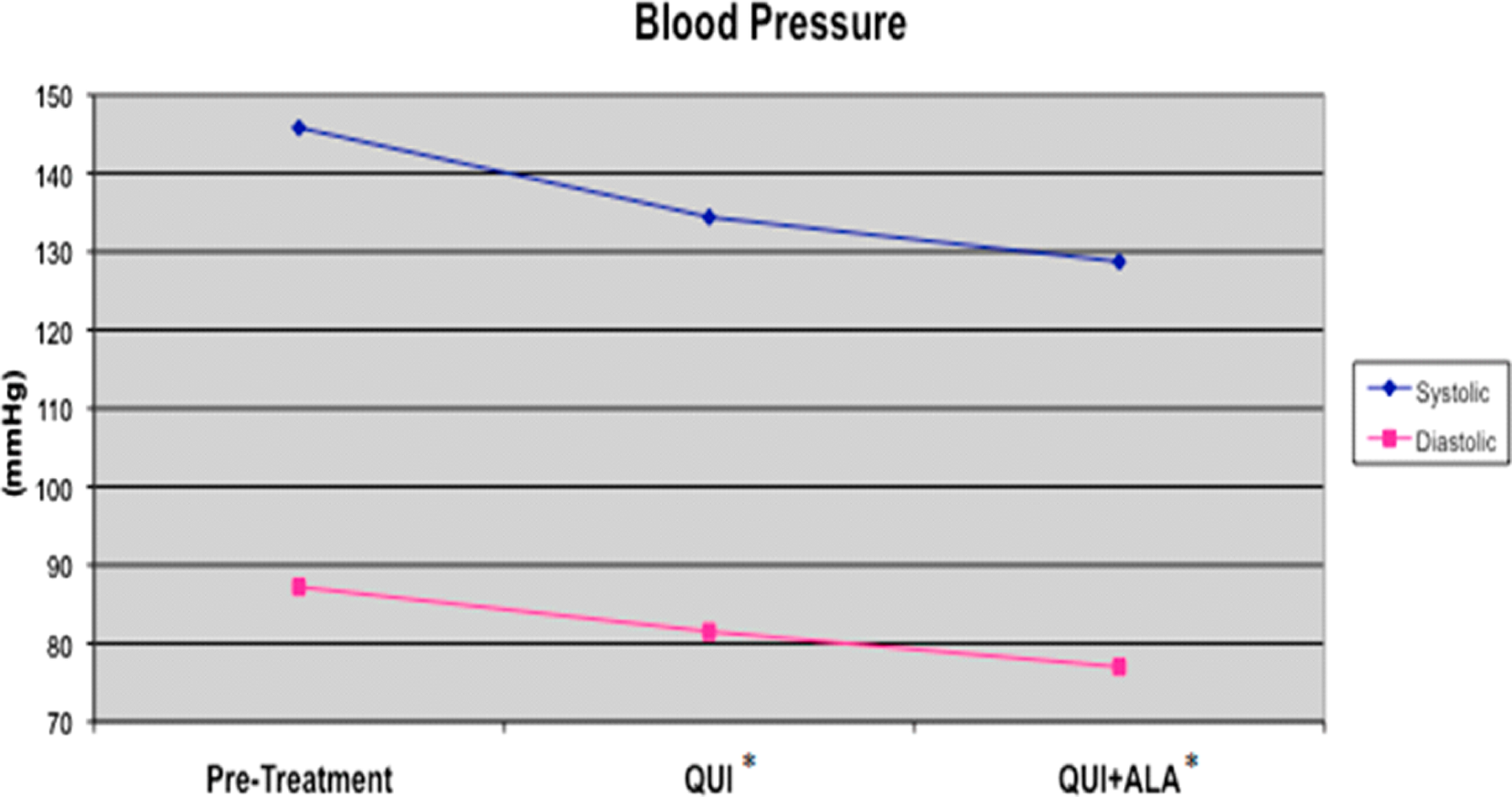
Effect of quinapril (QUI) and α-lipoic acid (ALA) on systolic and diastolic blood pressure. Participants were placed in a double-blinded, crossover fashion to QUI for 8 weeks or QUI + ALA for an 8-week period. Blood pressure was measured, as described in the Methods section. * indicates value differs (P < .05) from pretreatment.
Adverse Effects and Tolerability of Therapy a

aValues are percentage of patients enrolled in the study.
Changes in Urinary Albumin in Response to QUI and QUI + ALA
Patients treated with QUI exhibited a 30% decrease in the 24-hour urinary albumin with respect to baseline (pretreatment: 61.4 ± 17.6 mg/24 h; QUI: 42.7 ± 9.0 mg/24 h; P = .018 QUI vs pretreatment). QUI + ALA patients had a 53% decrease in urinary albumin (pretreatment: 61.4 ± 17.6 mg/24 h; QUI + ALA: 28.6 ± 6.5 mg/24 h; P < .005 QUI + ALA versus pretreatment and QUI + ALA versus QUI; Figure 2). It should be noted that treatment with QUI + ALA resulted in a reduction in urinary albumin and no change in serum creatinine.

Effect of quinapril (QUI) and α-lipoic acid (ALA) on 24-hour urinary albumin excretion in diabetes and hypertension. Participants were placed in a double-blinded, crossover fashion to QUI for 8 weeks or QUI + ALA for an 8-week period. Briefly, 24-hour collection of urine was obtained and the albumin levels were measured, as described in the Methods section. * indicates value differs (P < .05) from pretreatment; Δ, value differs (P < .05) from QUI-treatment group.
Changes in Endothelial Function in Response to QUI and QUI + ALA
Quinapril treatment arm had a significant increase of 58% in FMD of the brachial artery at 24 weeks in comparison to baseline (pretreatment: 3.8% ± 0.6%; QUI: 6.0% ± 0.9%; P < .005 QUI vs pretreatment), suggesting an improvement in endothelial function (Figure 3). Moreover, patients treated with QUI + ALA exhibited a substantial increase in FMD by 116% at the end of the 8-week treatment period (pretreatment: 3.8% ± 0.6%; QUI + ALA: 8.2% ± 1.1%; P < .005 QUI + ALA vs pretreatment and QUI + ALA vs QUI).

Effect of quinapril (QUI) and α-lipoic acid (ALA) on brachial artery reactivity/flow-mediated dilation. Participants were placed in a double-blinded, crossover fashion to QUI for 8 weeks or QUI + ALA for an 8-week period. Flow-mediated dilation of the brachial artery was performed, as described in the Methods section. * indicates value differs (P < .05) from pretreatment; Δ, value differs (P < .05) from QUI treatment group.
Discussion
The combination of ALA and the ACEI QUI demonstrated reduction in proteinuria, and increased FMD suggesting improved endothelial function. These findings were observed in participants exhibiting hypertension and diabetes (as well as other cardiometabolic factors such as dyslipidemia and obesity). Additionally, QUI was shown to lower blood pressure. Between QUI and QUI + ALA groups, no significant difference in blood pressure was present during the study.
Endothelial dysfunction is thought to be a hallmark of the atherogenic process. 1,2,31,32 The RAS plays a central role in the pathogenesis of atherosclerosis-related diseases. 8–10 Angiotensin II, the central molecule in the RAS, has multiple effects on atherosclerotic plaque initiation and progression. 8–10 On a molecular and cellular level, blockade of the RAS reduces the extent of vascular lesions in atherosclerosis, and it appears that these effects may be independent of blood pressure reduction. 33,34,45,46 Mechanisms of actions include improved endothelial function, plaque stabilization, nitric oxide bioactivity, pulse pressure, and a reduction of pro-oxidative species. Our study highlights the concept that an improvement by the addition of ALA may benefit participants with these cardiometabolic risk factors.
α-Lipoic acid is a uniquely multifaceted bioactive agent and adjunctive therapy using an ACEI, with ALA exhibiting greater benefit in contrast to ACEIs/ARBs monotherapy. Urinary albumin as well as the urine albumin-creatinine ratio significantly decreased in the QUI + ALA group, as has also been previously shown. 47 The results suggest that ALA limits angiotensin II production, thereby further stabilizing RAS inhibition.
The significant increase in FMD due to ALA suggests potential improvement in endothelial function. This beneficial effect of ALA on endothelial function has been reported previously. 26 The effects of ALA on endothelial function may be due to the anti-inflammatory and antioxidant effects of ALA. 25,26
Limitations of the Study
The investigation is short term (ie, approximately 22 weeks) and examines potential associations by which ALA and ACEIs may be effective in patients with diabetes mellitus and hypertension. Due to a small study population (n = 40), the evaluation of differences among various subsets was not feasible. The response to therapy with QUI may have been more pronounced if a higher dose had been used initially in the study. There was no group consisting of patients treated with solely lipoic acid as it was previously observed that lipoic acid alone had a modest effect on parameters of vascular function in patients with the cardiometabolic syndrome. 26 Finally, many of our study participants had cardiometabolic risk factors such as obesity and dyslipidemia, but this was not a requirement of the study. Therefore, these results may not necessarily be applicable to patients with the metabolic syndrome. However, the growing problem of obesity worldwide is a major factor in the increased incidence and prevalence of cardiometabolic diseases such as diabetes mellitus, atherosclerosis, and hypertension. There are ongoing investigations regarding the mechanisms of action associating obesity and vascular disease. It does appear that the prevalence of obesity in part plays a direct role in the initiation of vascular dysfunction. 48,49
Conclusion
The incidence of cardiometabolic factors, including hypertension and diabetes mellitus, is increasing throughout the world, 48,49 with a concomitant increase in the risk of atherosclerosis-related diseases. Proinflammatory and pro-oxidative mechanisms are important in the pathogenesis of atherosclerosis. 31,32 The study suggests the nutritional supplement ALA potentiates the vascular benefits in preventing atherosclerosis-related diseases in these patients.
The pharmacological addition of ALA and QUI has powerful effects on endothelial function. Long-term outcome studies should be considered to determine the utility of these agents and lifestyle modification in the primary prevention of atherosclerosis-related diseases in participants with the growing problem of a combination of diabetes mellitus and hypertension.
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Bobby Khan and Dr Nadya Merchant are consultants to InVasc Therapeutics.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: InVasc Therapeutics sponsored this study.
