Abstract
Background:
Hypertension is a major cause of death and morbidity worldwide and is increasing in prevalence. The Renin–angiotensin system (RAS) is the most common mechanism involved in the pathophysiology of hypertension. Understanding the mechanism of the pathophysiologic processes will help direct potential therapeutic strategies to treat hypertension and improve cardiac function. Recently, a novel drug LCZ696 containing both an angiotensin receptor blocker valsartan and a neprilysin inhibitor (AHU377) has shown a promising effect on the treatment of hypertension. However, the effects of LCZ696 on the expression of main components of RAS, namely, angiotensin-converting enzyme (ACE), angiotensin-converting enzyme 2 (ACE2), angiotensin II type 1 receptor (AT1 R), angiotensin II type 2 receptor (AT2 R), and angiotensin (1-7) receptor/Mas receptor (MasR) remain unclear. The aim of the present study was to evaluate the effects of LCZ696 on the protective arms of RAS in the cardiac tissue when compared with valsartan under the equal inhibition of AT1 R. We hypothesized that the superior effects of LCZ696 may contribute to its greater effect on the RAS than valsartan.
Materials and Methods:
Sixteen-week-old female spontaneously hypertensive rats (SHRs) were used in this study. Wistar-Kyoto (WKY) rats were used as controls. All rats were randomly divided into LCZ696 (n = 10), valsartan (n = 10), SHR (n = 10), and WKY (n = 10) groups under a 12-hour dark and 12-hour light cycle and provided with regular chow diet and water. The tail-cuff method was performed to measure blood pressure. Cardiac function was assessed by echocardiography.
Results:
The blood pressure value was lower in LCZ696 than valsartan in SHR after 12 weeks of treatment. Further, LCZ696 inhibits the ACE and AT1 R protein expression in the cardiac of SHR and significantly upregulate the protective axis of RAS components, including ACE2, MasR, and AT2 R. Left ventricular AT2 R messenger RNA (mRNA) expression was higher in the LCZ696+SHR group compared with valsartan. In addition, real-time polymerase chain reaction analysis revealed that LCZ696 enhanced the mRNA expression of antihypertensive components AT2 R, ACE2, and MasR and decreased the expression of AT1 R. However, only AT2 R and ACE2 mRNA expressions have a statistical difference between the LCZ696 and valsartan groups. No difference was observed in the mRNA expression of ACE and MasR. The stronger positive signal of transforming growth factor β in the left ventricle was inhibited in each administrated group compared with SHR groups.
Conclusions:
LCZ696 ameliorates the vasoconstrictor axis of the RAS AT1 R and stimulate the protective arm effectors, ACE2 and AT2 R, as well as reverses the compensatory upregulation of neuronal nitric oxide synthase and endothelial nitric oxide synthase in SHR. These findings suggest the mechanistic insight of the cardiac-protective and greater hypotensive effects of LCZ696.
Introduction
Hypertension is the leading global risk factor for cardiovascular disease with increasing prevalence 1 –3 and has become a global health and economic problem. 4 It is very common in patients with heart failure (HF) to have a long-standing history of hypertension, and more than 70% of HF cases had a history of hypertension. 5,6 Hypertension treatment in clinical practice aims to maintain goal blood pressure (BP). 7 It has been demonstrated that an increase of 20 mm Hg in systolic BP (SBP) or 10 mm Hg in diastolic BP doubles the risk of cardiovascular disease. 8,9 According to the Joint National Committee (JNC8) guidelines, thiazide diuretics, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), and calcium channel blockers are the first-line treatment for hypertension. 10 Angiotensin-converting enzyme inhibitors, ARBs, and thiazide diuretics have equal efficacy in the nonblack hypertensive population. 11 These drugs have been demonstrated to reduce BP, prevent cardiovascular endpoints in hypertensive patients, and reduce ventricular remolding in patients with coronary artery disease. 12
LCZ696 combines a moiety of the ARB valsartan and the neprilysin (NEP) inhibitor prodrug sacubitril (AHU377). After oral administration, LCZ696 is delivered into valsartan and inactive AHU377, which then cleaves into the active NEP inhibitor LBQ657. 13,14 Recently, the clinical trial of Prospective Comparison of ARNI with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure showed that LCZ696 significantly reduced overall mortality and HF hospitalizations. 15 LCZ696 was also evaluated in hypertensive patients. 16 -18 The NEP inhibitor component of LCZ696 (LBQ657) works by reducing natriuretic peptides (NPs) degradation, which results in decreased blood volume and systemic vascular resistance, leading to lower arterial pressure. Natriuretic peptides counteract the renin–angiotensin system (RAS) by promoting the vasodilation, natriuresis, and inhibition of fibrosis and hypertrophy. 19 Angiotensin receptor blocker exerts its effect by blocking angiotensin II type 1 receptor (AT1 R) blockers on blood vessels and the heart or other tissues, thereby preventing the deleterious cardiovascular effects of this compound. The mechanism underlying the superiority of LCZ696 might be a result of the synergistic effects of the NEP inhibitor and the AT1 R blocker in patients with hypertension or HF.
von Lueder et al demonstrated in animal experiments that LCZ696 can ameliorate cardiac remolding and improve cardiac function after myocardial infarction. 20 Another study showed that LCZ696 improved cardiac remolding, probably by ameliorating cardiac fibrosis, as reflected by the reduction of transforming growth factor-β (TGF-β). 21 LCZ696 also ameliorates cardiac hypertrophy compared with either stand-alone NEP-inhibitor or ARB. 20 However, the underlying mechanism of LCZ696 on RAS components and cardiac oxidative stress is unclear. The present study is designed to determine the extent to which targets of the RAS pathway such as AT1 R, AT2 R, and MasR, and the downstream targets TGF-β will be affected by LCZ696 and valsartan treatment by measuring the expression of genes and proteins of the RAS pathway in cardiac.
Materials and Methods
Animals and Experimental Design
All the experiments were performed according to the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals (8th edition National Academy Press, revised 2011). Thirty female spontaneously hypertensive rats (SHRs) and 10 Wistar-Kyoto (WKY) rats were purchased from Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). Each of the rats was housed in a 22°C ± 2°C room with a 12-hour light and 12-hour dark cycle and given free access to rat chow and tap water. The SHRs were randomly divided into 3 groups at 12 weeks of age: (1) SHR+LCZ696 (AHU377 [30 mg/kg/d] + valsartan [30 mg/kg/d]; n = 10), (2) SHR+valsartan (30 mg/kg/d; n = 10), and (3) SHR (n = 10); WKY (n = 10) was used as the control and received the same volume of sterile water. The purity of AHU377 was more than 99% (Sigma, MO, USA). Blood pressure and heart rate were measured in acclimated, conscious rats by tail-cuff plethysmography (AD Instruments Pty Ltd, Castle Hill, New South Wales, Australia) after 12 weeks of treatment and averaged from 5 readings. Cardiac function was performed with echocardiography on isoflurane-anesthetized rats using a Vevo 2100 Imaging System equipped with a 400-MHz liner probe at the end of the experiment. Body temperature was maintained at 37°C to 38°C. Rats were sacrificed at 24 weeks of age, after 12 weeks of treatment. Then cardiac tissue was harvested immediately from 5 rats in each group, snap-frozen in liquid nitrogen, and stored immediately in −80°C for Western blot.
Real-Time Polymerase Chain Reaction
After 12 weeks of treatment, the rats were euthanized with an overdose of pentobarbital. The heart was harvested immediately and frozen in liquid nitrogen. The total RNA was extracted by TRIzol reagent Thermo Scientific™, MA, USA), and reverse transcribed into complementary DNA with GoScript Reverse Transcription System following the manufacturer’s instruction. The messenger RNA (mRNA) expression levels were quantified from the total RNA using real-time polymerase chain reaction (RT-PCR) with SYBR Green. The mRNA expression of RAS components (ACE, ACE2, AT1 R, AT2 R, MasR) and its downstream target TGF-β were analyzed with SYBR Green RT-PCR. Glyceraldehyde-3-phosphate dehydrogenase (GADPH) was used as an internal control for the mRNAs. Primer sequences for RT-PCR amplification are shown in Table 1. Real-time PCR was performed with the CFX96 Touch Real-Time PCR Detection System (Thermo Scientific™, MA, USA) . The mRNA expression values are shown in folds by using the formula x = 2− ΔΔCt, where x refers to the fold difference relative to the control group.
Sequence for Primers.

Abbreviations: ACE, angiotensin converting enzyme; ACE2, angiotensin converting enzyme 2; AT1 R, angiotensin II type 1 receptor; AT2 R, angiotensin II type 2 receptor; GADPH, Glyceraldehyde-3-phosphate dehydrogenase.
Western Blot Analysis
The cardiac tissue was homogenized and lysed with RIPA Lysis and Extraction buffer (Thermo Scientific™, MA, USA). The supernatant was collected after centrifugation at 14 000 rpm for 15 minutes at 4°C. The proteins were then quantified by the Bradford assay with a protein assay kit. Portions of protein (100 μg) per sample were separated through a 10% sodium deodecyl sulfate-polyacrylamide gel and transferred to Polyvinylidene difluoride (PVDF) membranes at 80°C for 2 hours. Subsequently, the membrane was blocked with 5% bovine serum albumin (BSA) in 0.1% Tris-buffered saline-Tween (TBST) and then probed with antibodies ACE (1:500; Abcam), ACE2 (1:5000; Abcam), AT1 R (1:100; Abcam, USA), AT2 R (1:10 000; Abcam), MasR (1:10 000; Abcam), and GADPH (1:1000; Abcam) at 4°C overnight. The membranes were washed 3 times in TBST (5 minute each wash), then incubated with secondary antibody goat anti-rabbit/rat IgG (H+L)-Horseradish Peroxidase (HRP)-conjugated (1:10 000; Bio-Rad lab, CA, USA) for 2 hours at room temperature. After the last wash, signals were detected using Enhanced chemiluminescence (ECL) reagents. All the data were analyzed by Image J software (National Institute of Health, USA).
Histological Studies
Heart tissue was collected, fixed with 4% paraformaldehyde and processed for paraffin sections. Paraffin tissue sections (5 μm) were cut with a microtome, then deparaffinized, rehydrated, and stained with hematoxylin–eosin for histological analysis. For immunohistochemistry, endogenous peroxidase activity in the cardiac section was removed by hydrogen peroxide; the sections were then blocked with 5% BSA for 1 hour to avoid nonspecific protein binding sites. Afterward, the slides were incubated with anti-β1 primary antibody (1: 250 ab64715; Abcam) for 10 hours and then washed 3 times and incubated with goat anti-rabbit IgG H&L (Biotin, TX, USA) overnight at 4°C, secondary antibody (1:10 000, ab6720; Abcam) and goat anti-rat IgG 2 hours at room temperature. Sections were analyzed under a fluorescent microscope (BX61; Olympus,Tokyo, Japan).
Statistical Analysis
All the data were presented as mean difference ± standard deviation and then analyzed by one-way analysis of variance with Bonferroni post hoc analysis. Paired t test was used for comparisons between treated groups using the GraphPad Prism version 6.0 for Windows (GraphPad Software, San Diego, California). Correlations were evaluated by the Pearson correlation analysis. A value of P < .05 was considered significant.
Results
Blood Pressure and Body Weight
Blood pressure in the SHRs was lowered in both administrated groups. LCZ696 showed a significantly greater reduction in SBP than valsartan. As shown in Table 2, there was no significant difference in heart rate between the SHR and WKY groups. The body weight (BW) in the SHRs were similar but significantly lighter when compared with the WKY rats. The heart weight (HW) were significantly heavier in the vehicle (SHR+sterile water) compared with the SHRs+LCZ696 and SHRs+valsartan groups. Similarly, both LCZ696 and valsartan induced a significantly lower HW to BW ratio compared with the vehicle group. However, there were no significant differences in the HW, BW, or HW/BW between the LCZ696 and valsartan groups.
Tissue Weight and Blood Pressure at 24 Week (After 12 Weeks Treatment).a
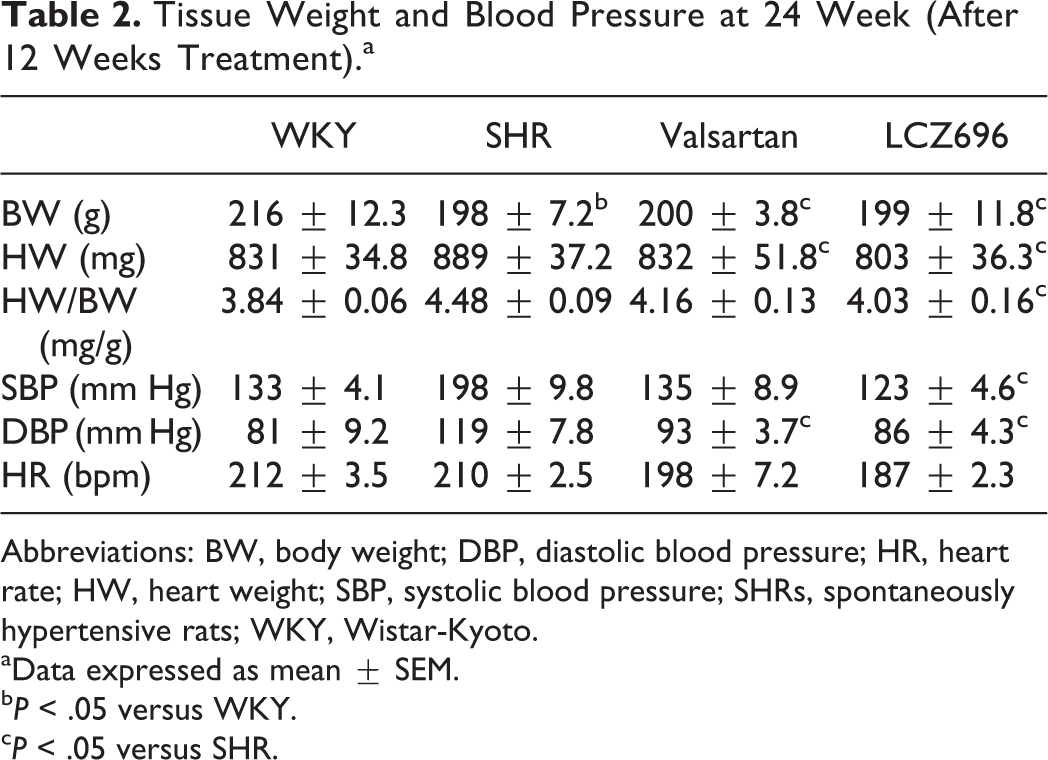
Abbreviations: BW, body weight; DBP, diastolic blood pressure; HR, heart rate; HW, heart weight; SBP, systolic blood pressure; SHRs, spontaneously hypertensive rats; WKY, Wistar-Kyoto.
aData expressed as mean ± SEM.
b P < .05 versus WKY.
c P < .05 versus SHR.
Echocardiography Parameters
The vehicle group showed significantly reduced left ventricular ejection fraction (LVEF) and fractional shortening (FS) compared with the WKY group (Figure 1A-D). Both administrated groups showed improved cardiac function when compared with the vehicle (LCZ696 vs vehicle: EF% 48.39% ± 1.364% vs 41.61% ± 0.722%, P < .05; valsartan vs vehicle: EF% 44.34% ± 1.017% vs 41.61% ± 0.722%, P < .05). LCZ696 significantly increased LVEF compared with valsartan (48.39% ± 1.364% vs 44.34% ± 1.017%, P = .0289). LCZ696 also significantly increased LV FS compared with valsartan (25.45% ± 0.726% vs 22.40% ± 0.638%, P < .05) and to the vehicle (25.45% ± 0.726% vs 20.67% ± 0.766%, P < .05). Left ventricular posterior wall thickness and LV mass (Figure 1E) were lower in both LCZ696 and valsartan groups compared with vehicle (P < .05, Figure 1F). LCZ696 significantly decreased LV mass compared with valsartan (833.7 ± 14.76 mg vs 795.0 ± 10.11 mg, P < .05). There were no significant differences between LCZ696 and valsartan in LV posterior wall thickness (1.639 ± 0.065 mm vs 1.720 ± 0.045 mm, P = .2962).
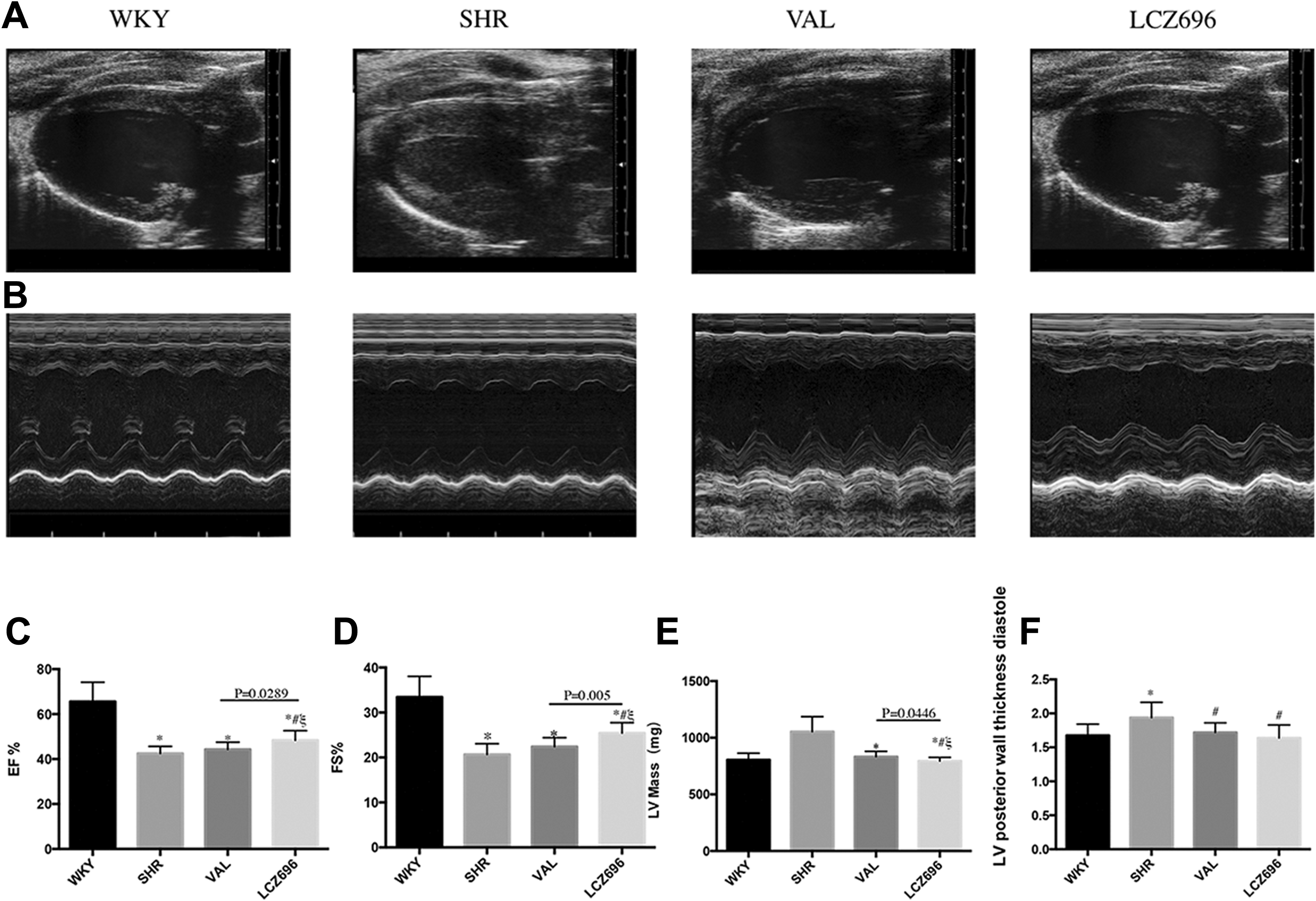
(A and B) M-mode; (C) EF, ejection fraction; (D) FS, fractional shortening; (E) LV mass, left ventricle mass; (F) left posterior wall thickness diastole.
Effects of LCZ696 on Cardiac RAS mRNA Expression
To demonstrate the effects of LCZ696 on the cardiac RAS, we investigated the mRNA expression of AT1 R, AT2 R, MasR, ACE, and ACE2 (Figure 2A-E) in the cardiac tissue of each group. The AT1 R and ACE mRNA expressions were upregulated in the vehicle but were downregulated by LCZ696 and valsartan. No significant difference was detected in the AT1 R mRNA expression between LCZ696 and valsartan (Figure 2A). LCZ696 significantly upregulated the AT2 R mRNA expression compared with valsartan (Figure 2B, #P < .05). LCZ696 also significantly upregulated the ACE2 mRNA expression compared with valsartan (Figure 2E, *P < .05).

(A to E) Determination of RAS mRNA expression; (A) AT1 R, angiotensin II type 1 receptor; (B) AT2 R, angiotensin II type 2 receptor; (C) MasR, angiotensin-(1-7) receptor; (D) ACE, angiotensin-converting enzyme; (E) ACE2, angiotensin-converting enzyme 2. mRNA indicates messenger RNA; RAS, renin–angiotensin system.
Effects of LCZ696 on RAS Components Protein Expression in the LV
To determine whether LCZ696 modulated the components of RAS, we examined the protein levels of ACE, AT1 R, AT2 R, ACE2, and MasR in the LV. As shown in Figure 3A and B, the ACE and AT1 R protein expressions in the LV were significantly upregulated in the vehicle but downregulated by LCZ696 and valsartan. However, no significant differences were detected in A between LCZ696 and valsartan. The protein expressions of ACE2 (Figure 3C) and MasR (Figure 3E) were significantly lower in SHR compared with WKY. Western blot analysis confirmed that LCZ696 significantly increased the protein expression of AT2 R (Figure 3D) and MasR compared with valsartan. LCZ696 significantly inhibited the protein expression of TGF-β (Figure 3F) compared with the valsartan and vehicle groups.

(A-F) Determination of renin–angiotensin system (RAS) protein expression; (A) ACE; (B) AT1 R; (C) ACE2; (D) AT2 R; (E) MasR; (F) TGF-β. ACE indicates angiotensin-converting enzyme; ACE2, angiotensin-converting enzyme 2; AT1 R, angiotensin II type 1 receptor; AT2 R, angiotensin II type 2 receptor; TGF-β, transforming growth factor β.
Effects of LCZ696 on Oxidative Stress in the LV
The neuronal nitric oxide synthase (nNOS; Figure 4A) and endothelial nitric oxide synthase (eNOS; Figure 4B) protein expressions were higher in the vehicle group but were lowered by LCZ696 and valsartan. However, no significant difference was detected in the nNOS and eNOS protein expressions between LCZ696 and valsartan.
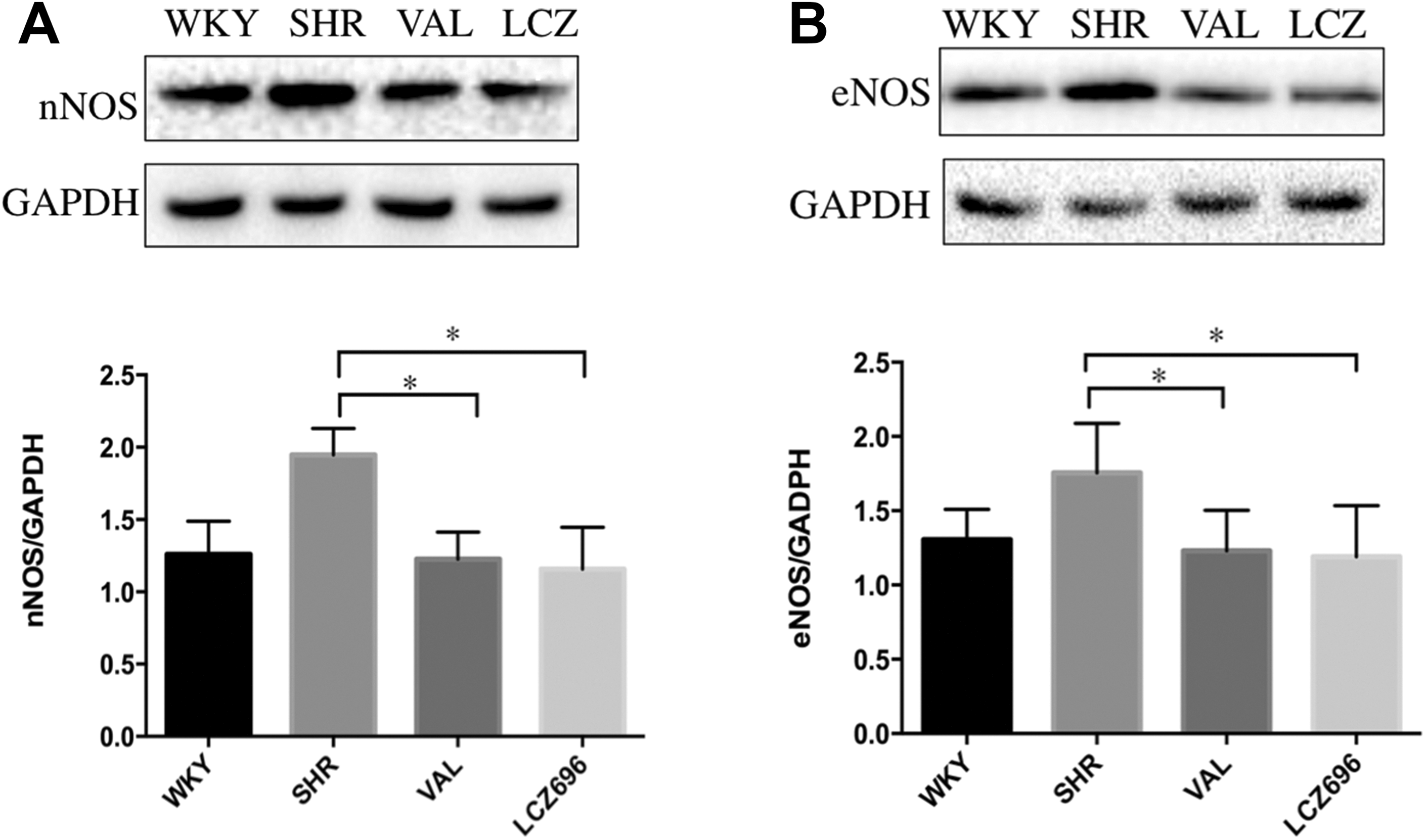
(A-B) nNOS and eNOS protein expression; (A) nNOS, neuronal nitric oxide synthase; (B) eNOS, endothelial nitric oxide synthase.
Effects of LCZ696 Versus Valsartan on Cardiac Fibrosis
Hematoxylin–eosin staining was performed to measure the cardiomyocyte area. As shown in Figure 5E-H, heart sections from the WKY group (Figure 5E) showed regular cell distribution, clearly crossed striation, and normal myocardium architecture, illustrating the viable diameter of the fibers and the central position of their nuclei. Sections from SHR exhibited mild multifocal myocyte necrosis, and most of the myofibrils lose their striations (Figure 5F). This disorganization of cardiomyocytes was partly improved by LCZ696 and valsartan. No significantly different effects were observed between LCZ696 and valsartan. We performed immunohistochemistry on the heart tissue with TGF-β antibodies from 4 groups (Figure 5A-D, Bar = 50 μm). The positive signal of TGF-β in the left ventricle was stronger in the vehicle group and weaker in the LCZ696 and valsartan groups.

(A-D) Immunohistochemistry on heart tissue with TGF-β antibodies from 4 groups. (E-H) Hematoxylin-eosin staining on heart tissue. TGF-β indicates transforming growth factor β.
Discussion
Although the pathophysiology of hypertension has been extensively investigated, the RAS has not been elucidated with respect to its important role in contributing to hypertension and HF. The present study showed that the greater benefits of LCZ696 compared with valsartan might be due to the stimulating expression of AT2 R and MasR, key elements of the protective arm of the RAS, and decreased downstream protein expression of TGF-β, improving the imbalance of renin–angiotensin–aldosterone systems in SHRs. To the best of our knowledge, this is the first study to show that the greater benefits of LCZ696 in patients with hypertension or HF than conventional RAS blockers might be due to its greater influence on the renin–angiotensin–aldosterone system. The greater expression of AT2 R and MasR, key elements of the protection of the RAS, and decreased protein TGF-β expression in LCZ696 compared with valsartan were showed in our present study. Renin–angiotensin system blockers are the first-line drug for the treatment of hypertension. 22 For a long time, it was thought that the more RAS suppression, the better. However, dual RAS blockade trials have now demonstrated that excess RAS inhibition may limit the benefits of this approach. 6 In the present study, the doses of LCZ696 and valsartan were given to maintain the same degree of AT1 R inhibition. We found that the AT2 R and MasR expressions were higher in SHR+LCZ696 than SHR+valsartan, suggesting that the greater effects of LCZ696 might involve greater AT2 R expression and ACE2-Ang(1-7)-MasR axis stimulation rather than AT1 R inhibition. Santos et al found that in MasR-deficient mice, the FS and posterior wall thickness were significantly decreased in systole and left end-diastolic dimension. 23 A clinical study also reported that the resistance vessels of patients with high risk have a higher expression of AT2 R during treatment with selective AT1 R blockade for 1 year. 24 It is believed that AT2 R contributes to maintaining BP by controlling vascular tone through vasodilation. 25 With this phenomenon also reflected by the BP data in our study, we found that BP was significantly lower in the LCZ696 group with an increased expression of AT2 R. Moreover, Ang-(1-7), mainly produced from Ang II by ACE2, was found strongly increased by NEP inhibition. 26 It has been reported that Ang-(1-7) binds and activates the MasR. 27 Since the LCZ696 combines NEP inhibitor and AT1 R blocker, we speculate that the enhanced expression of MasR might be explained by the elevated level of Ang-(1-7) mediated by LCZ696. Meanwhile, the fibrosis of heart tissue was significantly improved treated with LCZ696 when compared with valsartan. It has been demonstrated that AT2 receptors may become AT1 receptor-like in the SHR. 28,29 This tendency might be weakened by LCZ696.
Angiotensin II type 2 receptor has been suggested to exert its cardioprotective effects via NOS, 30,31 and AT2 R activation reduces BP, caused NOS initiates its vasorelaxation, which may balance the constrictor effects of AT1 R stimulation. 32 The family of NOS is known to be critical in a variety of physiological mechanisms and also involves many pathological progressions. 33 The main isoform related to mediating the vascular function is eNOS. However, nNOS in the vascular endothelium is substantially considered to be the major contributor of maintaining the homeostasis of the cardiovascular system. 34,35 In fact, the expression of the different NOS isoforms in SHR shows controversial results. Seko et al found that LCZ696 (60 mg/kg/d) did not show any influence on NO-mediated vasorelaxation. Some studies reported that the expression of inducible nitric oxide synthase (iNOS) and eNOS was increased in 8- and 12-week-old SHR. 36,37 Other studies found decreased expression of eNOS and iNOS in cardiomyocyte and vascular smooth muscle of SHR. 38,39 A study from Caniffi et al found greater protein expression of the 3 isoforms of NOS in the left ventricle and the aorta from SHR than normotensive rats. Furthermore, the increase in NOS activity is not associated with an increase in NOS protein expression in SHR. 40 Our results also showed that nNOS and eNOS protein expressions were higher in the vehicle group and lowered when treated with LCZ696 and valsartan. Phosphorylation of eNOS and nitric oxide release can be increased by Mas receptor stimulation. 41 Also, increased BP stimulates nitric oxide synthesization, which is a counterregulatory mechanism activated to compensate for the increase of BP. 42
Note that several studies have provided evidence that Reactive oxygen species (ROS) uncouple NOS under pathological conditions, thereby diminishing NO production and potentially promoting ROS formation. 43,44 Our previous meta-analysis concluded that LCZ696 has better antihypertensive efficacy in hypertensive patients without increased risk of adverse effects when compared with ARB. 45 Suematsu et al reported that the mechanism of LCZ696 improved cardiac function is associated with the reduction of fibrosis in an Heart failure with reduced ejection fraction (HFrEF) model by suppressing TGF-β. 21 The PARAMOUNT (Prospective Comparison of ARNI with ARB on Management of Heart Failure with Preserved Ejection Fraction) study, a randomized, double-blind clinical trial comparing LCZ696 with valsartan, found that the patients who received LCZ696 had lower levels of NT-proBNP at 12 weeks and then improved the New York Heart Association class and left atrial reverse remodeling at 36 weeks. 46
The present study confirms that LCZ696 containing both an RAS blocker (valsartan) and an NEP inhibitor (AHU377) improves cardiac function. It also provides evidence that the improvement is due to stimulating the protective arms of the RAS components AT2 R and MasR and debilitating the tendency of AT2 Rs to become AT1 R-like in the SHR model.
Study Limitations
First, this study only investigated the effects of LCZ696 in female SHRs. Therefore, it is possible that LCZ696 has different effects on RAS expression when it is performed in male SHRs. Second, we treated the rats with 60 mg/kg of LCZ696 and 30 mg/kg of valsartan. Other doses of LCZ696 or valsartan might have variant effects on cardiac RAS and oxidative stress. Third, although the phenomenon that AT2 R becomes AT1 R-like is debilitated by LCZ696 in female SHR, which level of AT2 R expression exerts maximum benefits is unknown. Meanwhile, we cannot rule out that the increased expression on AT2 R and MasR on LCZ696 group might be caused by its greater effects on BP. Further clinical and basic studies will be needed to resolve this issue.
Footnotes
Acknowledgment
The authors thank Martha Walmsley for proofreading and language assistance.
Author Contributions
Yang, Z contributed to conception or design, acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ruixin, M and Ningying, L contributed to conception or design and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Xiaorong, Y contributed to acquisition, analysis, or interpretation; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Xu, Z contributed to acquisition, analysis, or interpretation. Jing, Y contributed to conception or design, acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (NSFC-81670385).
