Abstract
Traumatic brain injury is a common health problem affecting millions of people each year. BDNF has been investigated in the context of traumatic brain injury due to its crucial role in maintaining brain homeostasis. Val66Met is a functional single-nucleotide polymorphism that results in a valine-to-methionine amino acid substitution at codon 66 in the BDNF prodomain, which ultimately reduces secretion of BDNF. Here, we review experimental animal models as well as clinical studies investigating the role of the Val66Met single-nucleotide polymorphism in traumatic brain injury outcomes, including cognitive function, motor function, neuropsychiatric symptoms, and nociception. We also review studies investigating the role of BDNF on traumatic brain injury pathophysiology as well as circulating BDNF as a biomarker of traumatic brain injury.
Traumatic Brain Injury
Traumatic brain injury (TBI) is a growing public health concern, with approximately 69 million global cases reported each year (Dewan 2018). Clinically, TBI is classified as mild, moderate, or severe based on signs and symptoms that occur postinjury. Mild TBI (mTBI), like concussion, accounts for >80% of all TBIs and often results in various neurobehavioral and physical deficits, such as cognitive and motor impairment (Karr and others 2014; Martinia and Broglio 2018). mTBI and repetitive mTBI are commonly observed in military personnel as well as individuals who take part in contact sport (Escolas and others 2020; Finch and others 2013). Falls are identified as being the most frequent cause of mTBI in elderly populations and children aged 0 to 4 years (Eman Abdulle and van der Naalt 2020; Haarbauer-Krupa and others 2019). Other common causes of mTBI across populations include motor vehicle accidents and assaults (Hon and others 2019). While deficits typically resolve within 7 to 10 days for the majority of individuals, 10% to 30% continue to experience functional impairment at 1-year post-mTBI (McMahon and others 2014; Quinn and others 2018). The risk for developing long-term neurologic conditions, including depression and cognitive dysfunction, is significantly increased in individuals with a history of repetitive brain injury (Manley and others 2017). Although still a controversial topic within the field, there is evidence to suggest that repetitive mTBIs increase the risk for the development of neurodegenerative diseases, such as chronic traumatic encephalopathy and Alzheimer disease (Becker and others 2017; McKee and others 2009).
TBI is multifaceted and complex in that the primary, mechanically induced injuries trigger a secondary cascade of neuropathologic changes (biochemical, metabolic, and cellular) that may be delayed from the initial moment of impact and can progress over time. Although understanding of TBI pathophysiology has progressed, to date no pharmaceutical treatments have demonstrated success in TBI clinical trials (Kabadi and Faden 2014), with heterogeneity in the nature of injury and biological response to injury likely key to translational failures (Stein 2015). Certain demographic and environmental factors (e.g., age, TBI history; Dams-O’Connor and others 2013; Dhandapani and others 2012) are now known to influence functional outcomes after mTBI, but understanding how genetics and certain aspects of mTBI pathophysiology might be able to predict recovery from mTBI is still in its infancy. Genetics is widely believed to be a key factor, and while polymorphisms in some genes have been associated with poor outcomes from TBI (Davidson and others 2014; McAllister 2015), only a small number of genes have been investigated in the context of mTBI, and there is still much to be learned about their association with TBI pathophysiology. In addition, although some progress has been made with fluid biomarkers of cellular damage after mTBI (Halford and others 2017; O’Brien and others 2021), relatively few studies have investigated how markers associated with other aspects of mTBI pathophysiology may have utility to assist the diagnosis and management of mTBI (McDonald and others 2021). Herein, this literature review focuses on a protein called BDNF and its potential role in mTBI pathophysiology and outcomes. In particular, we discuss the rationale and emerging evidence for how a common single-nucleotide polymorphism (SNP) of the
Brain-Derived Neurotrophic Factor
Neurotrophins are a family of proteins primarily secreted from neuronal and glial cells (Lessmann and others 2003). They regulate the function, development, and survival of individual cells and neuronal networks throughout the brain (Huang and Reichardt 2001). BDNF, the most abundant and widely distributed neurotrophin in the brain (Tsai 2018), is particularly interesting in the context of TBI due to its crucial role in the development and maintenance of the CNS. When translated, the BDNF molecule consists of three domains: the signal peptide, the prodomain, and the mature domain (Colucci-D’Amato and others 2020). As illustrated in Figure 1, upon entering the Golgi apparatus, the signal peptide is removed, and BDNF is first synthesized as the proBDNF precursor molecule (Hempstead 2015). The proBDNF molecule may then be cleaved intracellularly by furin or extracellularly by plasmin or matrix metalloproteinase to form mature BDNF (mBDNF; Finan and others 2018). proBDNF preferentially binds to the p75-sortilin receptor complex, causing apoptosis and synaptic depression by activating the c-Jun N-terminal kinase caspase 3 pathways (Numakawa and Odaka 2021). mBDNF binds to the tropomyosin-related kinase B (TrkB) receptor with high affinity. Figure 2 shows that mBDNF-TrkB binding activates numerous pathways that promote neurotrophic functions, such as neuronal survival, synaptic plasticity, and neurogenesis (Wurzelmann and others 2017).
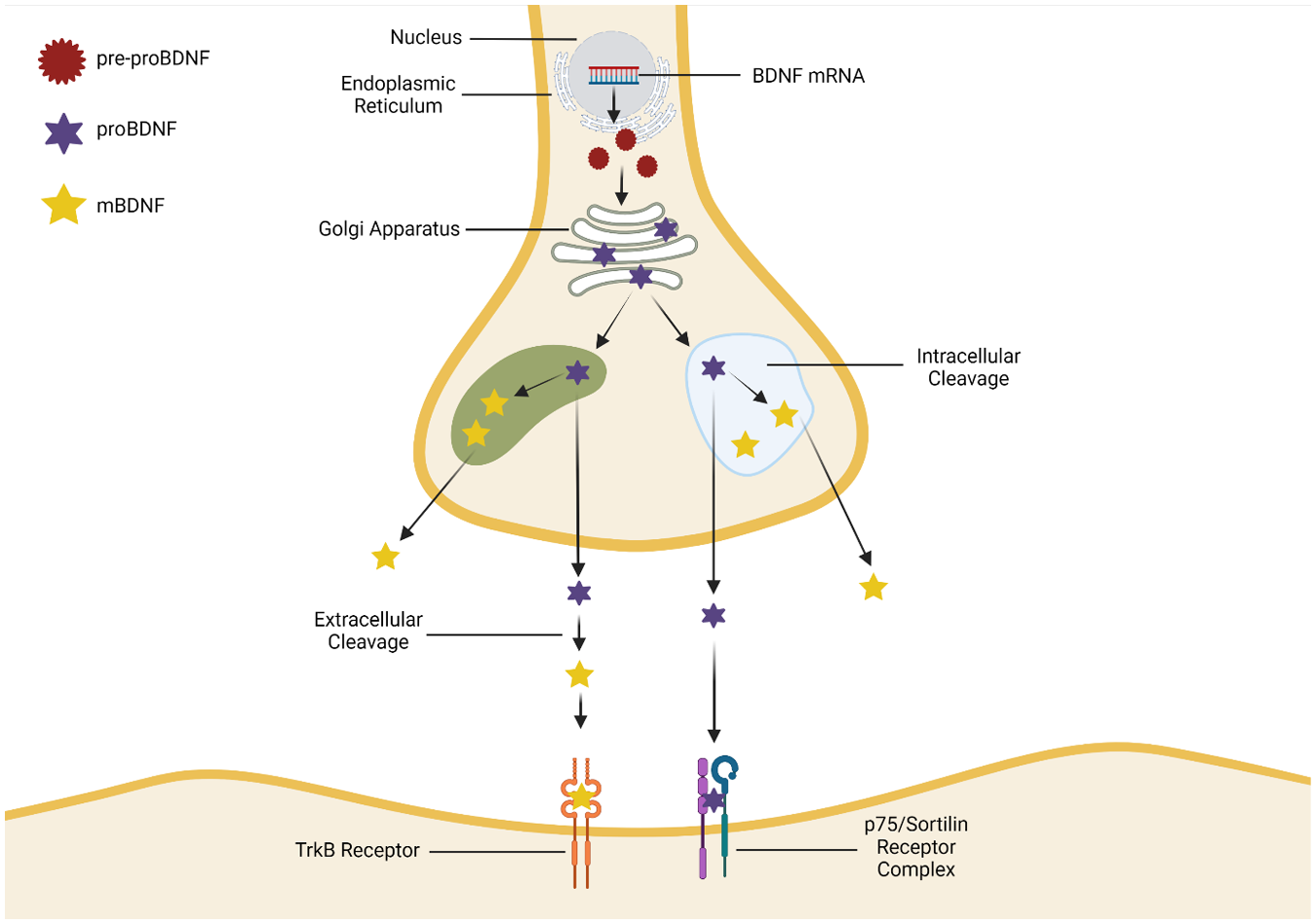
Synthesis of BDNF. Adapted from Kowiański and others (2018). The
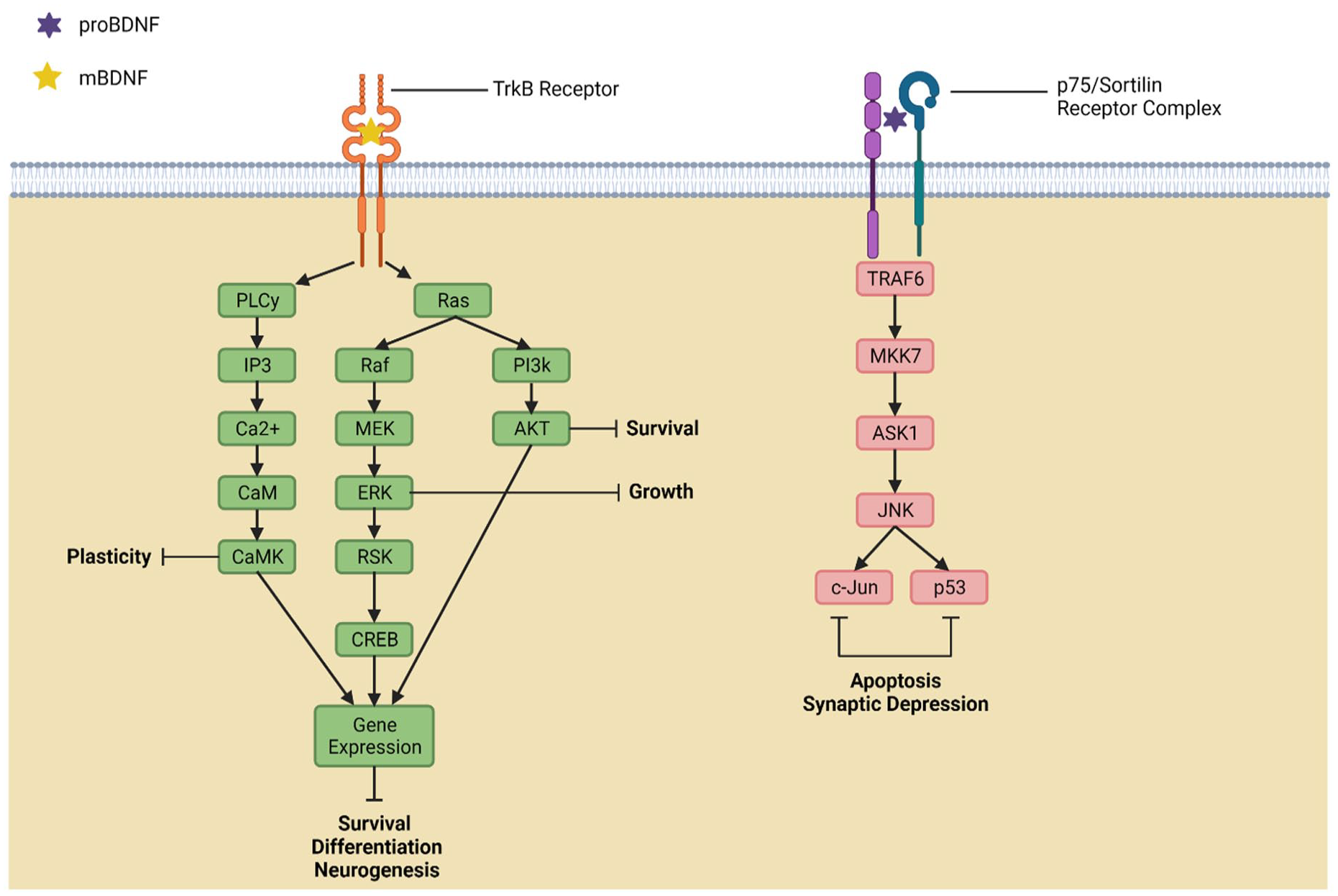
BDNF pathways. Adapted from Jin and others (2019). The proBDNF/p75/sortilin complex leads to activation of the JNK, c-Jun, and p53 signaling pathways, thus promoting deleterious processes such as apoptosis and synaptic depression. The mBDNF/TrkB receptor complex triggers activation of multiple signaling pathways, including PI3K/Akt and PLC-γ, that ultimately activate CREB, which transcribes genes associated with neuronal development and maintenance. mBDNF = mature BDNF.
Val66Met SNP
Hundreds of polymorphisms of the
Val66Met in TBI
The Val66Met SNP has been associated with poor outcomes in a number of comorbidities associated with TBI, such as major depressive disorder, stroke, and Parkinson disease (Altmann and others 2016; Liu and others 2021; Youssef and others 2018). Of relevance to the study of brain injury, BDNF has been shown to promote neural regeneration and inhibit apoptosis, inflammation, and neurotoxicity (Chen and others 2013). Research in rats demonstrated that at 1, 3, and 5 hours following a controlled lateral cortical impact, BDNF mRNA expression is significantly upregulated in the cortex and hippocampus (Wurzelmann and others 2017). Considered alongside its numerous roles in brain homeostasis discussed herein, this upregulation of BDNF after TBI may be a key neuroprotective response in the brain to trauma. As such, it is hypothesized that the Val66Met polymorphism, which disrupts BDNF expression, may alter the typical physiology of these processes, resulting in a diminished capacity for recovery and poorer outcomes following TBI.
Effect of Val66Met on TBI-Relevant Outcomes
Cognitive Function
Given its well-established role in synaptic plasticity, the effects of BDNF and the Val66Met SNP on cognition have been studied extensively. It is widely hypothesized that learning and memory impairments in Met allele carriers are due in part to differences in hippocampal morphology and function. In an initial study, Egan and others (2003) found that while genotype did not influence semantic or working memory in healthy individuals, deficits in delayed recall on the Wechsler Memory Scale–Revised suggested that episodic memory was significantly impaired in Met carriers. However, Dempster and others (2005) cited no significant difference between BDNF genotypes in episodic memory on this scale. An early fMRI study revealed that during the encoding and retrieval portions of a place recognition task, healthy Met carriers had decreased memory-related hippocampal activity when compared with healthy Val/Val participants. Of note, ~25% total variation in declarative memory performance was attributed to the interaction between the Met allele and hippocampal activity during the encoding phase (Hariri and others 2003). In addition to alterations in activity, several imaging studies have supported the findings of reduced hippocampal volume in healthy Met carriers (Bueller and others 2006; Hajek and others 2012; Pezawas and others 2004). Schofield and others (2009) utilized a computerized battery of neuropsychological tests to assess cognitive performance in healthy adults, finding that on a verbal list-learning task used to evaluate memory, Met/Met individuals had significantly higher errors than Val/Met and Val/Val individuals. These results were accompanied by lower hippocampal gray matter volume in Met carriers. Miyajima and others (2008) noted that in a healthy, elderly population, the presence of a Met allele was associated with significantly reduced processing speeds, diminished delayed recall, and lower general intelligence on a small cognitive battery; yet, no significant changes in hippocampal volume occurred.
Deficits in cognitive function associated with the Val66Met SNP have been well documented in populations with neuropsychiatric disorders. In a study examining individuals with schizophrenia, those with the Val/Val genotype had superior scores for visual working memory on the N-Back test as compared with the Val/Met and Met/Met genotypes (Rybakowski and others 2006). The same study examined the role of the SNP in individuals with bipolar disorder and revealed that Met carriers performed significantly worse than Val/Val carriers on domains of the Wisconsin Card Sorting Task, assessing working memory and executive function. This difference in cognition is in line with the hypothesis that the Val/Val genotype is associated with greater activity of the BDNF system when compared with Met carrier genotypes, thus resulting in altered neuronal function (i.e., superior cognitive performance; Rybakowski and others 2006). Similarly, Lu and others (2012) found that among antipsychotic-naive patients with schizophrenia, those with the Met allele had more persisting errors on the Wisconsin Card Sorting Task than those without the allele. These behavioral impairments are supported by neuroimaging studies that revealed decreases in gray matter volume and decreased activation within the dorsolateral prefrontal cortex of Met carriers (Pezawas and others 2004; Schofield and others 2009). Met carriers have also been reported to have significantly smaller hippocampal gray matter volumes in patients with major depression (Frodl and others 2007).
In studies evaluating the effects of the Val66Met SNP on cognitive function following TBI, the results are mixed. In an mTBI population, participants with the Val/Val genotype performed significantly better in attention, memory, executive function, and overall cognition on the Neuropsychological Assessment Battery over a 6-month time frame as compared with Met carriers (Narayanan and others 2016). McAllister and others (2012) found that Met carriers had significantly slower information-processing speeds 1 month post-mTBI. However, in a study investigating cognition at a chronic postinjury time point following a penetrating TBI, Met carriers had significantly higher scores in general intelligence, verbal comprehension, perceptual organization, working memory, and processing speed when compared with Val/Val carriers (Barbey and others 2014). Krueger and others (2011) also reported that the Met allele was associated with recovery of executive function following focal penetrating TBI. Another study demonstrated that at least 1 year after mTBI, Met carriers performed better than Val/Val carriers in the memory and executive functioning domains of a cognitive battery (Merritt and others 2020). As such, the current clinical literature, although preliminary, suggests that the Met allele may impair cognitive functioning acutely but be neuroprotective at chronic stages after TBI.
Genetically modified rodents have enabled additional insights into the role of the BDNF genotype in cognitive function and health following TBI. For example, a mouse model of the BDNF Val66Met SNP was used to evaluate the effects of genotype on a fear-conditioning paradigm targeted at measuring hippocampus- and amygdala-dependent learning (Liu and others 2021). The authors reported significant context-dependent memory impairments in Met carrier mice as compared with wild type Val/Val mice. Met carrier mice also had significant decreases in hippocampal volume, up to nearly 14%, when compared with wild type mice (Liu and others 2021). More recently, Giarratana and others (2019) conducted a study with similar genetically modified mice but examined the effects of the SNP following rmTBI with a lateral fluid percussion model. The preliminary findings of this study suggest that transgenic rodent models of this SNP replicate similar observations characteristic of the genetic variant in humans, such as deficits in learning and memory following mTBI.
While most literature to date suggests an association between the Met allele and cognitive impairments, a few studies contradict this conclusion. In healthy populations, multiple studies have reported no relationship between the Val66Met polymorphism and cognition nor related brain morphology (Karnik and others 2010; Richter-Schmidinger and others 2011). Research examining short-term memory performance suggests that the Met allele is actually protective against cognitive decline and may decrease susceptibility to white matter hyperintensities in the elderly (Huang and others 2014). A number of confounding variables could be the cause of inconsistencies in current research. For example, results could be influenced by the chosen cognitive assessments, small sample size, and sample population demographics, including age and ethnicity. The differences in brain morphology across genotypes could be the result of a combination of changes resulting from altered BDNF levels within the region, including decreased dendritic complexity, as well as increased cell death or decreased neurogenesis during development or over the life span. More targeted research is needed to determine the cause of these discrepancies.
Neuropsychiatric Symptoms
BDNF is widely considered an effective biomarker of various neuropsychiatric disorders and may play an instrumental role in the presentation and pathology of depression (Buchmann and others 2013; Lang and Borgwardt 2013), anxiety (Moreira and others 2015), and schizophrenia (Niitsu and others 2014; Notaras and others 2015). Meta-analyses evaluating depression (Molendijk and others 2014), schizophrenia (Green and others 2011), and bipolar disorder (Fernandes and others 2014) in the context of BDNF have revealed that low plasma and serum BDNF levels are associated with an increased risk of developing these disorders. Considering that Val66Met has been associated with decreased circulating BDNF levels, one can hypothesize that this SNP may modulate mood-related symptoms and pathology associated with a number of neuropsychiatric disorders.
In a population of healthy individuals, Met carriers have reported significantly stronger feelings of anxiety, nervousness, and insecurity using a visual analog scale ranging from 0 to 10 during the cold pressor test, as well as generally higher levels of social anxiety on the Liebowitz Social Anxiety Scale, as compared with Val/Val carriers (Colzato and others 2011). In addition, healthy Met/Met individuals had significantly higher rates of “anticipated worry” on the Temperament and Character Inventory when compared with healthy Val/Met and Val/Val individuals (Montag and others 2010). In a Han Chinese population, Met carriers who were schizophrenic reported significantly higher severity scores for depression and anxiety symptoms on the Positive and Negative Syndrome Scale (Sun and others 2013). Montag and others (2008) found that in a sample of healthy females, Met carriers demonstrated stronger amygdala activation while viewing pleasant and unpleasant emotional stimuli when compared with Val/Val carriers. The Met allele appears to have a similar effect on emotional processing in adolescents diagnosed with anxiety and/or depressive disorders in that Met carriers have increased activation in the amygdala as well as in the anterior hippocampus in response to emotional cues (Lau and others 2010). Soliman and others (2010) reported that when compared with healthy Val/Val individuals, healthy Met carriers demonstrated significant impairment in extinguishing a conditioned fear response. Impaired extinction learning was also accompanied by decreased activity in the ventromedial prefrontal cortex and increased activity in the amygdala, alterations commonly seen in depression and anxiety disorders. While these studies indicated significant alterations in neural activation, many reported a lack of association between genotype and brain volume in these areas (Frodl and others 2007; Kambeitz and others 2012).
In the context of TBI, Met carriers had significantly higher anxiety and depression scores at 1 week after mTBI (Wang and others 2018). Gabrys and others (2019) found that Met carriers with a history of mTBI had higher levels of brooding rumination and depressive symptoms. In addition, in children, Met carriers demonstrated a consistent trend of poorer behavioral adjustment as compared with Val/Val carriers after TBI (Treble-Barna and others 2022). Contrary to these findings, Gagner and others (2021) reported that pediatric Val/Val carriers showed significantly more internalizing behaviors 6 months post-mTBI. At 18 months postinjury, all children with mTBI presented more internalizing symptoms; however, genotype did not have an influence on behavior in this study. Adolescent Met carriers also reported having a better quality of life 6 months post-mTBI (Tuerk and others 2020). These results suggest that the Val66Met polymorphism may have a protective effect on neuropsychiatric symptoms after early childhood mTBI but exacerbate symptoms in adulthood.
Mood-related behaviors have been reported in rodent models of the Val66Met SNP. Chen and others (2006) used transgenic mice and found that when compared with wild type mice, Met/Met mice exhibited increased anxiety-like behaviors in response to environmental stressors. These anxiety-like behaviors did not normalize in response to treatment with the antidepressant fluoxetine. Met/Met carriers within the study also showed impairments in neuronal secretion of BDNF. These results were replicated by Yu and others (2012), who determined that the Met allele was associated with elevated depressive-like behaviors on the forced swim task and sucrose preference task. By using multiple tasks to assess anxiety-like behavior in Val66Met mice, stressed Met carriers spent significantly less time exploring the open arms in the elevated plus maze and demonstrated an increased latency to drink in a stressful environment on a novelty-induced hypophagia task (Dincheva and others 2017). Similarly, Soliman and others (2010) reported that Met carrier mice have significant impairments in extinction learning when compared with Val/Val controls. More recently, Hill and others (2020) reported that in an adolescent mouse model, the Met/Met genotype interacts with stress, resulting in reduced cell density and dendrite spine density in the dorsal hippocampus.
While many experts maintain that the Val66Met polymorphism is associated with a variety of mood-related symptoms comorbid with TBI, the evidence is not conclusive. The effects of this SNP on symptomatology could vary among neurologic insults and disorders owing to differences in BDNF expression in various brain regions. Similar to the current literature on cognitive function, the inconsistencies in these results may also be due to differences in study methodology and populations and attributed to small sample sizes. Further research is necessary to determine the effect of the Val66Met SNP on mood-related symptoms in preclinical and clinical samples.
Motor Function
Though few published studies exist on the topic, current literature suggests that the Val66Met SNP may influence motor function by altering sensorimotor cortex activation. Much of this research exists within the field of stroke. As compared with Val/Val patients, Met carriers have been reported to have a greater degree of disability and loss of independence (Braun and others 2020), as well as overall worse functional outcome following stroke (Kim and others 2013; van der Vliet and others 2017). A recent study used fMRI to reveal that the presence of the Met allele was associated with decreased ipsilesional primary sensorimotor cortex activation in patients after a stroke (Kim and others 2016). McHughen and others (2010) found that on a driving-based motor task, healthy Val/Val individuals demonstrated a nonsignificant trend of higher levels of short-term motor learning and retention as compared with healthy Val/Met counterparts. In the same study, Val/Val individuals had significantly greater sensorimotor cortex activation when compared with Val/Met individuals in response to index finger training. Using a complex motor learning program (i.e., a 5-day basketball-shooting exercise), Deveci and others (2020) stated that the Met allele was associated with slightly lower learning performance as compared with the Val/Val genotype in healthy adults. The SNP was also associated with a sensory-motor integration pattern in which higher afferent facilitation was shown at the early stage of motor learning after the first day of basketball exercises but not maintained to the final day of basketball exercises. These changes may be the result of altered synaptic plasticity of the primary motor cortex.
Research into the effects of the Val66Met SNP on motor function is scarce in preclinical models. Chen and others (2006) noted that locomotor activity did not differ between Met carrier and wild type mice in an open field behavioral assay. Fritsch and others (2010) found that as compared with wild type littermates, Met/Met knock-in mice demonstrated significantly impaired motor skill acquisition on an accelerating rotarod task. In a chronic stroke recovery model, Met carrier mice had significantly greater motor impairment when compared with wild type mice on the rotarod task during the acute recovery phase. However, the Met allele accelerated motor function recovery during subacute and chronic stages, with function returning to sham-related levels nearly 6 months postinjury (Qin and others 2014). In the context of TBI, rmTBI has been shown to cause a significant deficit in vestibular motor function in injured Met carrier mice relative to injured Val/Val mice (Giarratana and others 2019). While this research is still informative, further studies need to be conducted within the context of TBI to develop a better understanding of how this SNP might affect motor function postinjury.
Nociception
Posttraumatic headache, which commonly presents as a migraine- or tension-like headache, is one of the most common consequences of TBI. Posttraumatic headache is also frequently reported in conjunction with somatic, psychiatric, and cognitive symptoms following TBI (Ashina and others 2019). BDNF is widely distributed in the CNS, including key regions involved in pain regulation, such as the hippocampus, amygdala, hypothalamus, and sensory and trigeminal ganglia (Marziniak and others 2008). BDNF stimulates dopaminergic and serotonergic neurons involved in the pathogenesis of migraine and in activity-dependent plasticity in nociceptive pathways, which may lead to pain progression/persistence (Marziniak and others 2008). It has been suggested that BDNF has a role in migraine pain, due to its interaction with calcitonin gene–related peptide, a key vasodilating neuropeptide implicated in migraine pathogenesis (Terrazzino and others 2017). Therefore, it may play a role in the development and severity of posttraumatic headache.
In humans, elevated BDNF levels in serum and cerebrospinal fluid have been observed in individuals with migraine and headache (Blandini and others 2006; Fischer and others 2012; Tanure and others 2010). Serum BDNF levels are also significantly elevated during migraine attacks (Fischer and others 2012). Similar increases in CSF BDNF levels have been observed in rats with mechanical allodynia (Feliciano and others 2014). Multiple meta-analyses have revealed an association between the Val66Met polymorphism and migraine in adults, with the presence of the Met allele increasing susceptibility (Cai and others 2017; Terrazzino and others 2017). Yet, in a recent study, Koute and others (2021) noted that the presence of the SNP was associated with a decreased risk of headache and migraine in a sample of healthy pediatric participants. In spinal cord injury research, BDNF expression has been associated with the development of neuropathic pain (Garraway and Huie 2016). Elevated BDNF levels promoted functional recovery in the acute postinjury phase (Hutchinson and others 2004). However, chronic overexpression of BDNF can lead to maladaptive plasticity and hypersensitivity to pain (Boyce and others 2012; Garraway and others 2014). Although the effect of the Val66Met SNP on nociceptive response has not been directly investigated in the context of TBI, based on the role that BDNF plays in nociception, it is hypothesized that the presence of the Met allele would increase an individual’s risk for developing posttraumatic headache after TBI.
Effect of Val66Met on TBI Pathophysiology
The complex pathophysiologic cascade after TBI is becoming increasingly well understood. Although heterogenous, pathophysiologic processes such as inflammation and apoptosis can be prominent and key determinates of TBI outcome. As such, given the key role that BDNF may play in many of these processes, it has been hypothesized that the Val66Met SNP may result in altered biological and pathologic responses of the brain to trauma. Limited research has, however, focused the role of BDNF and the Val66Met SNP in TBI pathophysiology. In one recent study, injured Met carrier mice showed significant increases in hippocampal apoptosis and astrogliosis by using respective activated caspase 3 and GFAP immunostaining at 1 day post-rmTBI as compared with wild type mice (Giarratana and others 2019). The study also reported that when compared with Val/Val carriers, Met carrier mice had increases in Fluoro-Jade C+ cells, IBA1+ cells, and phosphorylated tau+ cells, indicating increased levels of neurodegeneration and activated microglia in the cortex and hippocampus following rmTBI. Met carriers had increased levels of inflammation, which was measured with MRI as higher-intensity areas relative to regular brain tissue (Giarratana and others 2019). Recent in vivo and in vitro studies of ischemic brain injury provide evidence that BDNF may protect against neuronal apoptosis after brain injury (Han and others 2000; Liu and others 2007). Regarding inflammation, in a recent study based on an epidural impact model of TBI in mice, injections of collagen-binding domain attached to BDNF decreased brain inflammatory signaling and edema (Yin and others 2020). Despite these recent findings, there remains a need for further research in animal and human models to better establish a connection between the Val66Met SNP and pathophysiologic processes after TBI.
Circulating BDNF as a TBI Biomarker
Decreased serum BDNF levels have been associated with neuropsychiatric disorders, thus highlighting BDNF as a potentially sensitive biomarker for comorbid conditions, including TBI (Klein and others 2011). However, studies on BDNF levels in patients with TBI are scarce, and their results are conflicting. Korley and others (2016) reported an association between low serum BDNF levels and poor outcomes after clinical TBI. In a study investigating the effects of exercise on persistent postconcussive symptoms in individuals with mTBI, those with persisting symptoms had significantly lower salivary BDNF levels after an exercise regimen when compared with healthy participants who had completed the same exercise program. If these peripheral circulating BDNF levels are indeed reflective of central levels, the aforementioned studies may indicate an association between reduced BDNF and increased risk of poorer functional outcomes after TBI (McGeown and others 2018). Results from a study investigating cerebral biomarkers in postmortem body fluids following fatal TBI indicate that CSF BDNF levels are significantly elevated in fatal TBI cases as compared with living TBI controls and non–head trauma cases (Ondruschka and others 2018). In a preclinical study investigating CSF BDNF levels in the acute injury phase following mTBI in rats, BDNF levels in CSF were significantly increased in rats that developed mechanical allodynia at 1 day following injury. These results suggest that CSF BDNF levels may be a prognosis for TBI-induced neuropathic pain (Do and others 2022). The current research suggests that central BDNF may increase following TBI, while peripheral BDNF decreases. Although further research is required to determine the cause of this negative correlation, it has been speculated that it may indicate blood-brain barrier disruption. This disruption may result in platelets dumping BDNF into the CNS in the acute stage following TBI (Failla and others 2016). HPA axis activation, which can reduce BDNF levels, may also explain the reduced levels of serum BDNF as compared with CSF BDNF levels (Failla and others 2016).
On the contrary, worsened TBI outcomes have been associated with increased levels of BDNF in CSF (Failla and others 2015). In a longitudinal study investigating the role of plasma biomarkers associated with deployment trauma in veterans with a history of mTBI, plasma BDNF levels were not associated with psychiatric comorbidities, such as posttraumatic stress disorder, depression, or anxiety (Pierce and others 2022). Additional studies have found no association between either serum BDNF or CSF and TBI outcomes (Simon and others 2016; Stefani and others 2017).
Limitations of Existing Literature
Pathologic and functional outcomes are incredibly heterogeneous across individuals who have sustained a TBI. These variations are seen across postinjury time points as well as injury severities and history of TBI. Researchers must consider the impact of genetics and their interactions with demographics, such as age (Biswas and others 2017), ethnicity (Arango-Lasprilla and Kreutzer 2010), and sex (Mollayeva and others 2018), when examining the effects of TBI. Given the complexity of brain injury, understanding the role that these factors play is crucial in determining an accurate prognosis and appropriate course of treatment for each patient presenting with a TBI.
Age
Age is one factor that may influence the effect of the Val66Met SNP on TBI outcomes. For example, in young adult populations, the presence of the Met allele is related to significant emotional disturbance and cognitive impairments following TBI (Gabrys and others 2019; Marziniak and others 2008; Wang and others 2018). On the contrary, in studies with elderly participants, the Met allele preserved executive functioning after injury (Barbey and others 2014; Krueger and others 2011). Consistent with these age-related findings, Failla and others (2016) reported that after sustaining a TBI, Met carriers <45 years of age had a significantly higher risk of mortality, while the opposite effect was observed in elderly Met carriers.
This interaction between genotype and age may be explained by changes in cell signaling. As previously mentioned, TrkB expression plays a crucial role in neuronal survival. In the normal aging process, TrkB expression is negatively correlated with age (Webster and others 2006). However, sortilin, which works in conjunction with the p75 receptor to initiate apoptosis and long-term depression, is upregulated in old age (Al-Shawi and others 2008). Based on these shifts in BDNF signaling, reduced secretion of BDNF associated with the Met allele may be deleterious throughout adulthood but become beneficial in the advanced stages of life.
Sex
Sex-dependent associations to the Val66Met SNP have been reported in neuropsychiatric and neurodegenerative research. For example, in a meta-analysis, Verhagen and others (2010) reported that the presence of the Met allele significantly increased the risk of major depressive disorder in males but not females. Conversely, the Met allele has been associated with an increased stress response in females but not males (Jiang and others 2017). The Met allele also significantly reduced cognitive functioning in normal aging (Laing and others 2012) and increased the risk of Alzheimer disease (Fukumoto and others 2010; Lin and others 2014) in females but not males. Although the interaction between sex and the Val66Met SNP in TBI research is very limited, the Met allele has been associated with increased depressive symptoms in females (Gabrys and others 2019) and increased anxiety in males (Wang and others 2018).
The interaction between sex and the Val66Met polymorphism can likely be attributed to sex hormones. Chan and Ye (2017) identified four mechanisms in which estrogens modulate the expression of BDNF. First, BDNF expression can be directly induced by estrogen activation of the estrogen receptor. Second, estrogen can modify the activity of
Ethnicity
One of the major limitations in studying the Val66Met polymorphism in humans is the substantial diversity of allelic frequency that exists among populations globally. In fact, according to the Allele Frequency Aggregator—a groundbreaking project recently developed by the US National Center for Biotechnology Information that provides aggregated allele frequencies from its database of genotypes and phenotypes across 12 diverse populations—the Met allele is present in approximately 19.1% of the global population (Phan and others 2020). As illustrated in Figure 3, the presence of the BDNF Met allele considerably varies across populations. The Met allele is reported to be >10 times more prevalent in East Asian populations (41.1%) when compared with African populations (4.3%; Failla and others 2016). It is possible that current studies are not accounting for this variability, leading to insufficient sample sizes when examining the SNP across multiple ethnic groups. Several meta-analyses have shown ethnicity-dependent associations to the Val66Met SNP in neurodegenerative and neuropsychiatric disorder outcomes, such as Alzheimer disease (Lin and others 2014), Parkinson disease (Lee and Song 2014; Q Wang and others 2019), obsessive-compulsive disorder (S Wang and others 2019), schizophrenia (Kheirollahi and others 2016), suicidal behavior (González-Castro and others 2017), and antidepressant response (Yan and others 2014; Zou and others 2010). However, it is important to note that meta-analyses examining these interactions have not been conducted in the context of TBI, likely due to the limited literature. Of the studies investigating the Val66Met polymorphism in TBI, the majority have included only Caucasian populations and therefore lack generalizability.
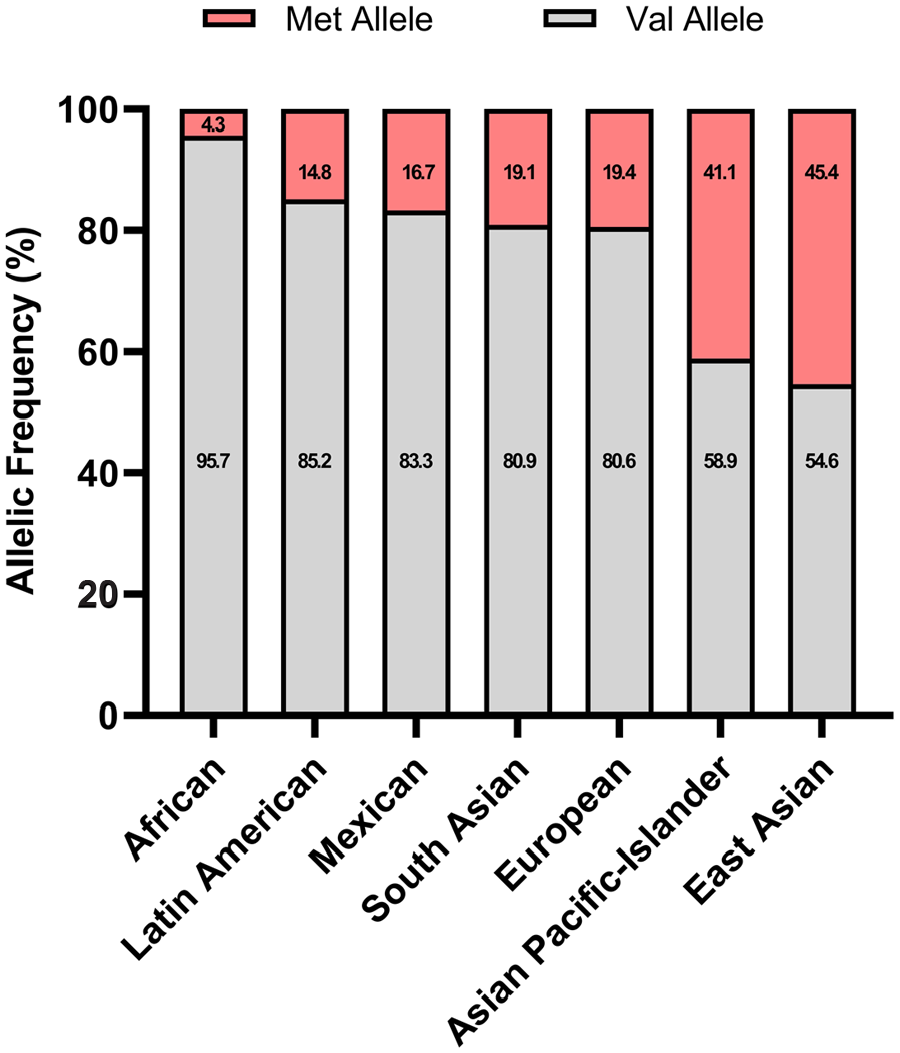
Val66Met allele frequencies by population. The presence of the BDNF Met allele considerably varies across populations. The Met allele is reported to be >10 times more prevalent in East Asian populations (41.1%) as compared with African populations (4.3%). These differences in allelic frequencies may have a role in the availability of BDNF across populations. Met = methionine; Val = valine.
To date, there are only two known studies evaluating the interaction between the Val66Met polymorphism and TBI in non-Caucasian individuals. In Asian populations, the studies show that as compared with Val/Val carriers, Met carriers demonstrate impaired cognitive functioning (Narayanan and others 2016) and increased feelings of anxiety and depression (Wang and others 2018). These results mirror findings in Caucasian samples with TBI at similar postinjury time points (Gabrys and others 2019; Marziniak and others 2008). Based on the current literature, it appears that Val66Met does not significantly influence TBI outcomes with regard to ethnicity. However, given the overall lack of literature, especially involving non-Caucasian populations, it is still unclear exactly how ethnicity may alter TBI outcomes.
Although there is currently no evidence to suggest that ethnicity interacts with the Val66Met polymorphism after TBI, there are some possible explanations for variability in genetic associations among ethnicities. For example, differences in genetic mechanisms among populations may cause one population to become more vulnerable to the molecular and behavioral dysfunction associated with the SNP while another population may be protected. It is important to consider other environmental factors that may be specific to certain ethnic groups and investigate their potential interactions with the Val66Met polymorphism.
Conclusions
TBI is a common injury but remains complicated to diagnose and treat due to the heterogeneity of pathology and behavioral outcomes. Growing evidence has emerged linking genetic factors to the sequelae following brain injury. One genetic mutation that is of particular interest is the BDNF Val66Met SNP, which leads to diminished secretion of BDNF. Research suggests that BDNF is upregulated in the acute postinjury phase and plays a crucial role in neuronal survival. However, chronic upregulation of BDNF has been suggested to have a deleterious effect. Therefore, it is hypothesized that the Met allele is the risk allele in the general population at the acute postinjury time point but becomes neuroprotective chronically. While the body of literature investigating the Val66Met polymorphism in the context of TBI is growing, future research must evaluate the effects of potential confounding factors, such as sex, age, and ethnicity, to aid in individual diagnoses and the development of personalized treatment plans.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
