Abstract
Introduction
Human papillomavirus (HPV) is a well-established cause of cervical cancer and is increasingly recognized as an important risk factor for non-cervical cancers, including oropharyngeal, anal, vulvar, vaginal, and penile cancers. This study assessed temporal trends in the incidence of HPV-related cervical and non-cervical cancers in Canada and the Netherlands by sex assigned at birth and cancer type using population-based registry data.
Methods
This study is a registry-based observational study, in which we analyzed retrospective data from the Canadian Cancer Registry (1992-2022) and the Netherlands Cancer Registry (2000-2019) for HPV-related cancers. Age-standardized incidence rates, annual percentage changes (APC), and average annual percentage changes (AAPC), with corresponding 95% confidence intervals (95% CI), were estimated using Joinpoint regression.
Results
In Canada, cervical cancer incidence rate generally declined since 1992 but has increased by 1.1% (0.5, 2.7) annually between 2013 and 2022. In the Netherlands, cervical cancer rates remained stable until 2015, followed by a 5.1% (1.9, 11.5) annual increase between 2015 and 2019. Non-cervical HPV-related cancers generally increased in both countries. In Canada, from 1992 to 2022, incidence rates increased for oropharyngeal (AAPC: 2.0% [1.6, 2.4]), anal (1.7% [1.3, 2.2]), and vulvovaginal (1.9% [1.6, 2.3]) cancers. In the Netherlands, from 2000 to 2019, incidence rates increased for anal (5.3% [4.2, 6.8]), vulvar (3.0% [2.2, 4.0]), penile cancers (2.8% [1.7, 4.0]), and oropharyngeal cancer (0.7% [0.1, 1.2]).
Conclusion
In general, we observed similar trends in both countries: that the incidence of HPV-related cervical and non-cervical cancers in Canada and the Netherlands is rising. These findings highlight the need to expand HPV vaccination programs, optimize HPV-related cancer screening programs, and enhance public health initiatives for the prevention and early detection of non-cervical HPV-related cancers.
Plain Language Summary
Human papillomavirus (HPV) is a virus linked to cervical cancer, oropharyngeal cancer (cancers of the oropharynx, or the middle of the throat), as well as anal and genital cancers. However, there are certain prevention strategies for these cancers. For instance, we have the HPV vaccine, which can prevent the virus itself. We also have screening methods, such as the Pap test, for cervical cancer. Even so, we still see rising trends in HPV-related cancers around the world. In our study, we looked at trends in HPV-related cancers in Canada and the Netherlands, two countries similar in income and latitude, but different in terms of their health systems (the way healthcare is delivered) and screening programs. For this study, we used publicly available cancer data from 1992-2022 (Canada) and 2000-2019 (the Netherlands). We found that cervical cancer rates have been rising by 1.1% per year since 2013 in Canada and by 5.1% per year since 2015 in the Netherlands. Rates for oropharyngeal, anal, and vulvovaginal cancers also increased in Canada, while rates for oropharyngeal, anal, vulvar, and penile cancers similarly increased in the Netherlands. These rising trends in HPV-related cancers reveal the need for expansion, optimization, and tailored messaging related to HPV vaccination and screening programs.
Introduction
Human papillomavirus (HPV) is one of the most common sexually transmitted infections (STIs) globally. 1 Twelve high-risk HPV types have been classified as Group 1 carcinogenic to humans by the International Agency for Research on Cancer, with two strains (HPV 16 and 18) of high-risk oncogenic HPV types responsible for most HPV-related cancers.2,3 Virtually all (>95%) cervical cancer cases can be attributed to HPV, but HPV is also causally linked to cancers of the oropharynx, anus, vulva, vagina, and penis. 4 Globally, it is estimated that 54% of these known HPV-related cancers are attributable to high-risk HPV types; however, this varies by site as well as by region. 5 Specifically, 48-58% of oropharyngeal cancers, 83-100% of anal cancers, 43-53% vulvar cancers, and 70-86% vaginal cancers are attributable to high-risk HPV subtypes.5,6 The HPV-related fraction of cancers is increasing over time and will vary between countries. 7 In Canada, HPV infection is linked with nearly all cervical cancers, 92% of anal cancers, 72.2% of vaginal and 47.4% of vulvar cancers, 39% of penile cancers, and 60.2% of oropharyngeal cancers. 8 In the Netherlands, a country with similar economic development and social order, HPV attributable fractions are largely similar to Canada: nearly all cases of cervical cancers, 88% of anal cancers, 78% of vaginal cancers, 25% of vulvar cancers, 50% of penile cancers, and 31% of oropharyngeal cancers. 9
While cervical cancer has historically been the most prominent HPV-related malignancy, advances in screening efforts, such as the Papanicolaou (Pap) test, have led to significant declines in incidence and mortality in many high-income countries.10-12 In the 1980s and 1990s, cervical cancer incidence rates decreased in both Canada and the Netherlands.13,14 These countries began vaccination programs for girls in 2008 (school-based in Canada) and 2010 (the National Immunization Programme, Netherlands).15,16 The programs were expanded to include boys in 2014 and 2022, respectively, which we expect will lead to greater increases in protection. 17 A further development in screening technology is the introduction of HPV testing, a more sensitive test than the Pap smear. 17 HPV primary screening was implemented in the Netherlands in 2017 and in some provinces in Canada (British Columbia) starting in 2023.15,16 Despite these screening measures, an increase in cervical cancer incidence has been observed in both countries in recent years, showing a 3.7% increase per year in Canada since 2015 and a 0.6% increase per year in the Netherlands since 2003.14,18
There is evidence from the US and Western European countries that the incidence of non-cervical HPV-related cancers has also been rising over the past few decades.19,20 This trend has been attributed to generational changes in sexual behaviour, increased exposure to HPV, and the absence of effective screening programs for non-cervical cancers.21,22 Of particular concern are oropharyngeal and anal cancers, which have shown the steepest increases in incidence rates over the past 30 years, particularly amongst males. 23 Strategies such as optimizing and expanding HPV vaccination programs have the potential to mitigate these trends long-term.
Understanding the trends of HPV-related cancers is critical for identifying populations at highest risk, informing public health strategies around HPV education, prevention, screening, treatment, and evaluating the effectiveness of existing HPV prevention programs. This study aims to explore temporal trends in the incidence rates of HPV-related cervical and non-cervical cancers in Canada and the Netherlands, two countries similar in terms of education, economic development, and social order. 24 Findings from our study will inform public health policies and improve efforts to prevent and manage HPV-related cancers.
Methods
Study Design and Sources of Data
This is a registry-based, observational retrospective study. For Canada, we obtained cancer data from 1992 to 2022 from the Canadian Cancer Registry (CCR) accessed through the Statistics Canada Web Data Services API. 25 The CCR is a national, population-based registry comprised of provincial and territorial cancer data submitted to Statistics Canada. 25 For this analysis, we excluded Nova Scotia and Quebec as these provinces had not reported cancer incidence data up to 2022 at the time of the study. For the Netherlands, we obtained data from 2000 to 2019 from the Dutch Cancer Registry (NCR). The different year ranges between the two countries reflect data availability. We used data from Canada and the Netherlands because the two countries are comparable in terms of economic development and latitude, but differ in their health systems structures and approaches to screening and prevention programs. In sensitivity analyses, we estimated the number of HPV-attributable cancers for each site by applying published population attributable fractions (PAFs) specific to Canada 26 and the Netherlands9,27-31 to the observed case counts (i.e., attributable cases = PAF × total cases).
We used the International Classification of Diseases in Oncology, 3rd Edition (ICD-O-3) to classify HPV-related cancers. 32 For both countries, we included cervical (C53), anal (C21), and penile (C60) as HPV-related cancer sites. In Canada, data on vulvar and vaginal cancers were not available separately; therefore, we used a combined category of other and unspecified female genital organs (C51, C52, C57.0-C58.9). For the Canadian data, oropharyngeal cancers included oropharynx (C10), tonsil (C9), and other oral cavity and pharynx cancers (C14). For the Netherlands, vulvar (C51) and vaginal (C52) cancers were analyzed separately, and oropharyngeal cancers included tongue (C1), palate (C5), tonsil (C9), and other oral cavity and pharynx (C10, excluding 10.1 epiglottis) and C14.8 (overlapping lesions of lip, oral cavity, and pharynx). The ICD codes included exclusively in the Canadian dataset were C10.1, C14.0, and C14.2, while the ICD codes included exclusively in the Dutch dataset were C1 and C5.
Since our study used retrospective, publicly available, anonymized data, ethics approval was not required. The reporting of this study conforms to the STROBE guidelines. 33
Statistical Analysis
For each HPV-related cancer site, the annual cervical cancer incidence rates between 2000-2019 for the Dutch data and between 1992-2022 for the Canadian data were calculated and age-standardized to the World Health Organization Standard Population (WHO 2000-2025). We estimated annual percent changes (APC) and average annual percent changes (AAPC) in age-standardized incidence rates with corresponding 95% confidence intervals (95% CI). We used Joinpoint Regression Program (version 5.4) to estimate APCs and AAPCs. 34 The software fits a piecewise linear regression model that assumes a constant rate of change in the logarithm of the annual incidence. Any changing point in the slope is determined as a joinpoint with a new slope and regression line parameters. This method identifies significant changes joinpoints, selected based on the Weighted Bayesian Information Criterion (BIC) method, in trends over time where the incidence trends either increased, decreased, or stabilized and estimated APCs and AAPCs. A maximum number of 5 joinpoints were used for the study to ensure study methods were consistent between the two countries.
In our supplementary analyses, we examined cervical cancer incidence rate in the Netherlands stratified by stage (using the International Federation of Gynecology and Obstetrics (FIGO) staging: IA, IB, II, III, and IV 35 ), and age based on the age distribution of the screening target population (0-29, 30-44, 45-64, 65-74, and 75+ years). Canadian trends in cervical cancer incidence stratified by age group were not included in this study, as they have been previously published by our team. 10 We conducted sex-stratified analyses for anal and oropharyngeal cancers in Canada available in our Supplementary Data. Statistics were reported by sex assigned at birth, classified according to the standards used at Statistics Canada as ‘male’ or ‘female’. FIGO staging data were not available for Canada, and sex-specific oropharyngeal and anal cancer data were not available for the Netherlands.
Results
Cervical Cancer Trends
Trends in the Age-Standardized Incidence Rates (Per 100,000) of HPV-Related Cancers, Canada, 1992-2022

APC = Annual Percentage Change, CI = Confidence Interval, AAPC = Average Annual Percentage Change. Rates standardized to the WHO Standard Population (WHO 2000-2025). Values that are significant are bolded.

Trends in age-standardized HPV-related cancer incidence rates in Canada (red) and The Netherlands (blue) by cancer sites. Orange depicts vaginal cancer (the Netherlands). Lines show the estimated trends in incidence rates based on joinpoint regression. Points show the actual incidence rates by cancer types. Population denominators for vulvovaginal, cervical, and penile cancers were sex-specific. Values that are significant are bolded and starred. Incidence rates were standardized to the World Health Organization’s standard population (WHO 2000-2025)
Trends in the Age-Standardized Incidence Rates (Per 100,000) of HPV-Related Cancers, the Netherlands, 2000-2019
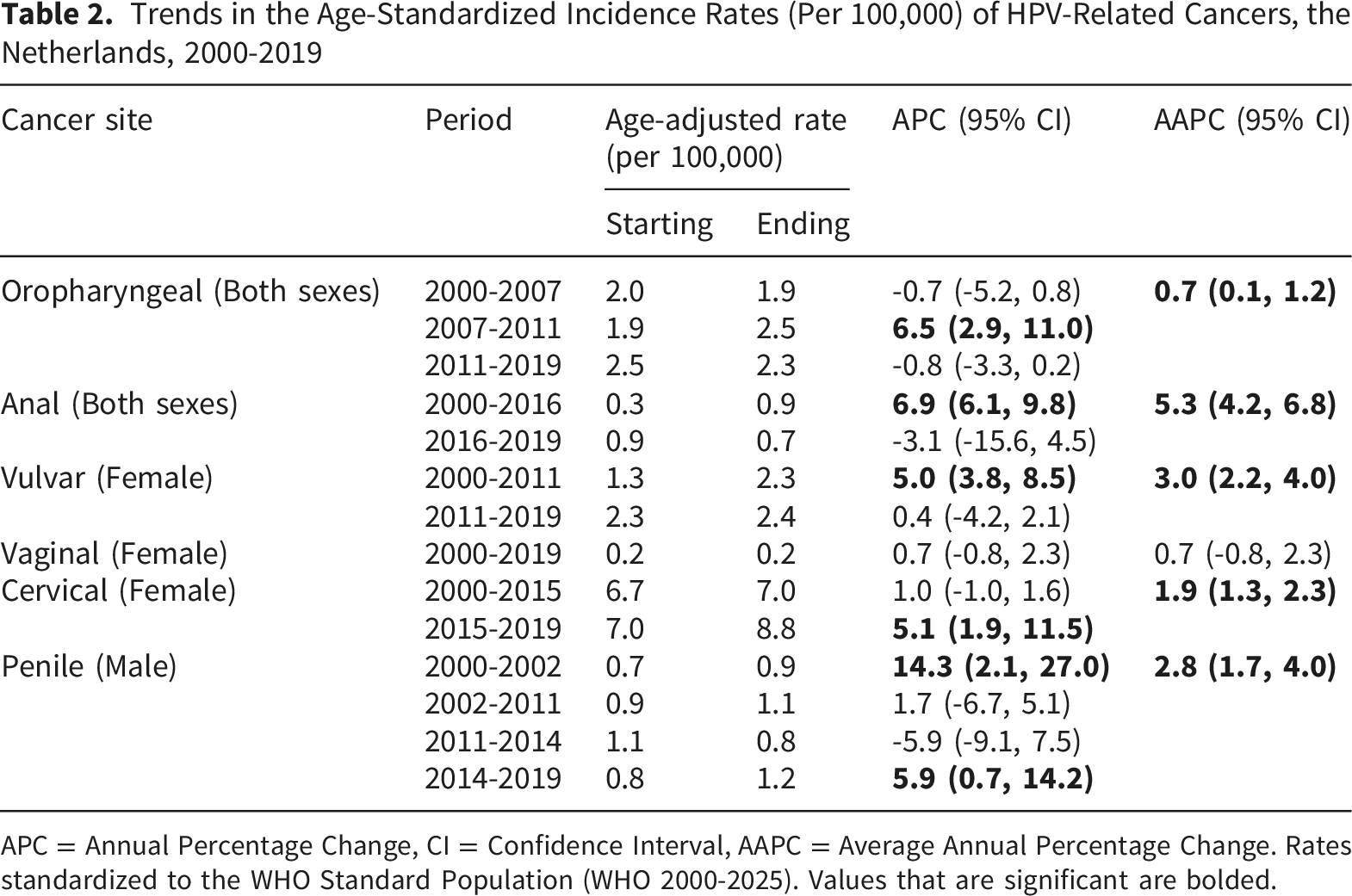
APC = Annual Percentage Change, CI = Confidence Interval, AAPC = Average Annual Percentage Change. Rates standardized to the WHO Standard Population (WHO 2000-2025). Values that are significant are bolded.
Non-Cervical HPV-Related Cancer Trends
Generally, the incidence rates of almost all non-cervical HPV-related cancers increased in Canada in recent years (Table 1, Figure 1), increasing for oropharyngeal cancers by 2.5% (2.2, 3.9) per year from 1998-2022, anal cancers by 2.4% (1.2, 6.8) per year from 2013-2022, and vulvovaginal cancers by 1.9% (1.6, 2.3) per year from 1992-2022. The incidence rate of penile cancer was relatively stable across the study period (0.4% [-0.1, 1.0]). When we stratified oropharyngeal cancers by sex, oropharyngeal cancer rates increased by 1.4% (1.2, 1.8) in females and 2.6% (2.3, 3.0) in males across the study period (Supplementary Table A3 and Figure A3). Similarly, anal cancer incidence rates increased by 3.0% (0.2, 12.2) in males from 2014 to 2022 and 4.5% (2.1, 17.0) from 1992 to 1999 in females (Supplementary Table A3 and Figure A3).
In the Netherlands, the overall long-term trends (AAPCs) showed that the incidence rates of most non-cervical HPV related cancers increased between 2000 and 2019 (Table 2 and Figure 1). Segment-specific trends (APCs) showed variations over time. The incidence rate of oropharyngeal cancers increased by 6.5% (2.9, 11.0) per year between 2007-2011 before stabilizing in the most recent study period. The incidence rates of anal cancer increased by 6.9% (6.1, 9.8) between 2000-2016, before showing slight decreases in recent years. The rate of vulvar cancer increased by 5.0% (3.8, 8.5) between 2000-2011 and stabilized after 2011. Finally, the rate of penile cancer increased by 5.9% (0.7, 14.2) since 2014. In Supplementary Tables A4 and A5, we re-estimated the number of HPV-attributable cancers for each site.
Discussion
The results of our study show increasing trends in HPV-related cancers for both Canada and the Netherlands, two Western countries working toward the goal of cervical cancer elimination. In Canada, the incidence rates increased in nearly all HPV-related cancers examined between 1992-2022, with the most pronounced recent increases observed for oropharyngeal cancers (2.5% per year), followed by anal and vulvovaginal cancers (2.4% and 1.9% per year, respectively). Of note are the large increases in oropharyngeal and anal cancer trends observed in both males and females. In the Netherlands, overall trends similarly showed increases for most HPV-related cancers, including cervical cancer incidence across almost all FIGO stages and the 30-44 and 45-64 age groups, as well as notable rises in anal and penile cancers between 2000 and 2019. The differences in penile cancer trends observed between Canada and the Netherlands may reflect variation in cancer registration practices, particularly for rarer cancers, such as penile cancer, where small case counts and differences in diagnostic coding or reporting completeness can lead to variability in the observed incidence. In contrast to Canada, oropharyngeal cancers have stayed stagnant in the Netherlands in recent years. The observed plateau in incidence may reflect a decline in HPV-negative cancers, primarily driven by reductions in smoking and alcohol use.36,37 When all HPV-positive and HPV-negative cancers are considered together, this may mask underlying increases in HPV-driven disease, resulting in an apparent plateau. As well, lack of standardization in the definition of locally advanced head and neck cancer may further contribute to the observed plateau. 38 To highlight this phenomenon, although the Netherlands Cancer Registry shows an increase in oropharyngeal cancers, standardized rates of locally advanced hypopharynx and larynx cancers have decreased, whereas the proportions of larynx cancers have increased slightly. 38
This study has several key limitations. Specifically, we used registry-based population data, which do not permit direct confirmation of HPV status for each case. Since the APCs were based on overall cancers and not HPV-induced cancer, it is not possible to directly estimate the exact numbers of cases due to HPV based on population-level data alone; we can only estimate infection-linked cancers using PAF. Since several HPV-related cancers are on the rise, the actual APCs for HPV-related cancers are likely underestimated if HPV-positive cancers are increasing while HPV-negative cancers are stable or declining, except for cervical and anal cancers, which are nearly all caused by HPV. This limitation is most relevant for cancer sites with lower HPV-attributable fractions, where inclusion of HPV-negative tumours may dilute or obscure HPV-specific trends. Our findings should be interpreted as population-level trends in cancers occurring at HPV-related anatomic sites, not direct estimates of trends in HPV-positive cancers. Similarly, the PAFs in Supplementary tables A4 and A5 are estimates and should be interpreted as such.
The rising incidence of cervical cancer among younger people in their 30s and 40s observed in the Dutch population is consistent with what we also observed in Canada, 10 highlighting an even greater need for increased HPV vaccination coverage and cervical cancer screening uptake, as well as increased awareness in both the public sphere and among providers. Furthermore, recent Dutch evidence suggested that vaccination alone may not fully explain current incidence patterns. 39 A study by Middeldorp et al. showed high reductions in cervical carcinomas among Dutch 30-year-olds screened in 2023, despite low vaccine coverage (46-63%). 39 These reductions were likely due to a combination of vaccination and screening, suggesting that vaccine uptake is unlikely to be the sole explanation for the current rise in HPV-related cancers observed in certain age groups. Instead, the increases could also reflect cohort effects, including rising HPV prevalence in specific birth cohorts, behavioural factors, limited sensitivity of cytology-based screening, and health system factors.
The increased incidence trends of non-cervical HPV related cancers observed were also consistent with other international studies in high-income countries, and these trends should especially be noted, given that cervical cancer is the only HPV-related cancer with a screening program. 7 For example, recent U.S. data showed increasing rates of oropharyngeal, anal, and vulvar cancers from 1999 to 2015, while European registry studies, such as in Norway, have also reported sustained increases in several HPV-related cancer sites.40-43 Several factors likely contribute to the rising trends observed in our study. First, many individuals aging into the higher-risk years for HPV-related cancer incidence were not eligible for early school-based vaccination programs. Canada introduced publicly funded vaccination for school-aged girls in 2007–2008 and expanded to boys only in 2014, while the Netherlands introduced vaccination for girls in 2010 and extended eligibility to boys in 2022.44,45 As such, anyone over the age of 30 would not have been vaccinated in the school-based programs during the study period. This is particularly relevant for males, who were not eligible for the school-based vaccination programs until much later and where we now see a higher burden of HPV-related cancers, e.g., penile, anal and oropharyngeal cancers. Compounded with these gaps in protection, HPV vaccine coverage rates are suboptimal in both countries, varying in Canada by province from 57-92%,46,47 and hovering between ∼46%-63% in the Netherlands. 48
Vaccination programs have a clear impact on cervical cancer incidence. For example, a large-scale study in Scotland in 2022 showed that compared with people born in 1988 who were unvaccinated, vaccinated people born between 1995-1996 showed an almost 90% reduction in the highest risk cervical intraepithelial neoplasia (CIN). 49 A study performed in Sweden in 2020 showed similar findings. 50 Studies have also shown that prophylactic HPV vaccination significantly reduced oral HPV prevalence (HPV 16, 18, 11 and 6).51-53 There is further evidence suggesting that vaccination programmes have a widespread impact on non-cervical HPV-related cancers, including head and neck cancers in males, anal cancers, and genital cancers.54-56 Even though school-based HPV vaccination programs exist, vaccination rates and series completion rates are far below the WHO 90-70-90 target by 2030 for the above countries to achieve cervical cancer elimination. 57 Strong public health policies to combat vaccine hesitancy and misinformation, removing barriers to HPV vaccination, and expansion of HPV vaccination programs may help to address these gaps. 58
In Canada, there is a shortage of access to primary care pathways and screening.59,60 In 2022, 21.8% of Canadians did not have a family physician. 61 Furthermore, lack of care coordination and fragmentation of care may lead to a lack of follow-up care, missed screening, and vaccination. Compared with Canada, the Netherlands maintains nearly universal access to primary care: more than 95% of the population has access to a primary care provider, and most of the population can reach a provider within 10 minutes. 62 That said, the Netherlands has faced its own share of workforce shortages, particularly after the COVID pandemic, with the situation projected to intensify. 63
Microsimulation studies have shown that HPV primary screening is critical to achieving the reductions in cervical cancer incidence needed to reach cervical cancer elimination.64-66 The Netherlands transitioned to primary high-risk HPV DNA testing in 2017 for their cervical cancer screening program. Canada has yet to fully transition to HPV DNA testing, with British Columbia, Prince Edward Island, and Ontario being the first provinces to transition to HPV DNA testing as the primary screening method in 2024-2025. The fact that both Canada and the Netherlands have experienced an increase in cervical cancer cases despite the availability of national cervical cancer screening and vaccination programs could be attributed to these key factors outlined, as well as suboptimal screening rates, inadequate follow-up of abnormalities detected, delays to care, and other reasons, including changes in HPV prevalence and population-level sexual behaviours. 67
Furthermore, certain subpopulations may be disproportionately affected. 68 In Canada, Indigenous peoples, immigrants, rural residents, and other underserved communities have lower vaccination and screening coverage and face barriers to healthcare access.68,69 Similar disparities in uptake are observed in the Netherlands, particularly among immigrants and those living in lower socioeconomic neighbourhoods. 70 Addressing inequities in access and acceptance of HPV prevention strategies remains essential. 71 Unless these inequities and barriers are addressed, we will likely not meet the screening and vaccination rates needed to eliminate cervical cancer in the population.70,71
This study has several notable strengths, including the use of recent, population-based national data from two countries with similar economic development and latitude, with high-quality cancer registries and near-complete (>99%) case ascertainment. Despite differences in health systems and screening programs, a shared trend in HPV-related cancer incidence rate increase may indicate that a shared epidemiological or biological reason may play a role in these trends. Stage- and age-specific analyses in the Netherlands provided additional granularity. However, we lacked morphological data, which may misclassify some HPV-negative cases, and differences in ICD-10 codes used between Canada and the Netherlands may limit comparability with other countries.
Conclusion
Our study showed increases in the incidence rates of HPV-related cancers in both Canada and the Netherlands, despite differences in healthcare system structures and prevention programs. These population-level findings reinforce the need to expand HPV vaccination programs, improve vaccine uptake and strengthen public health initiatives for prevention and early detection, particularly among younger cohorts and underserved populations.
Supplemental Material
Supplemental Material - Trends in the Incidence of Human Papillomavirus-Related Cancers in Canada and the Netherlands
Supplemental Material for Trends in the Incidence of Human Papillomavirus-Related Cancers in Canada and the Netherlands by Emily Niu, Ioana A. Nicolau, Ellen Olthof, Inge M. C. M. de Kok, Kirk Graff, Jennifer Gillis, Corinne M. Doll, Kristin A. Black, Angel Chu, Folkert van Kemenade, Darren R. Brenner in Cancer Control.
Footnotes
Acknowledgments
We would like to thank our expert advisor, Dr. Heather Bryant, MD, for her insights on the analyses and manuscript.
ORCID iDs
Ethical Considerations
This study used publicly available statistics, therefore research ethics approval was not required.
Author Contributions
All authors contributed to the study conception and design. Analysis was performed by E.N, I.A.N., E.O., and K.G. The manuscript was prepared by E.N. and I.N. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Partnership Against Cancer. I.A.N. is supported by the Canadian Institute of Health Research Fellowship (No. 530744). D.R.B. is supported by the Armstrong Investigatorship in Molecular Cancer Epidemiology.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Black has received consultancy fees and advisory board fees from GSK, unrelated to the submitted work.
Data Availability Statement
Canadian cancer data are publicly available at https://www150.statcan.gc.ca/n1/en/type/data. Dutch cancer data can be requested at ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
