Abstract
Background
Diffuse large B-cell lymphoma (DLBCL) is the most common aggressive non-Hodgkin lymphoma and a leading cause of cancer-related mortality worldwide. Evidence on long-term survival and prognostic determinants remains limited, particularly among people living with HIV. This study evaluated survival outcomes and predictors of mortality among adults with DLBCL in Northwest Ethiopia.
Methods
We conducted a multicenter retrospective cohort study of consecutively enrolled adults with newly histologically confirmed DLBCL diagnosed between August 1, 2020, and July 31, 2025, at three referral hospitals in Northwest Ethiopia. Patients were followed from diagnosis until death, loss to follow-up, or study end. Overall survival (OS) was estimated using the Kaplan–Meier method. Cox proportional hazard regression was used to identify independent predictors of mortality. Subgroup analyses were performed depending on HIV status. Statistical significance was set at P ≤0.05.
Results
A total of 175 patients with DLBCL were included, of whom 31 (17.7%) were HIV-positive. The median survival time was 36 months (IQR: 21–49). The estimated 5-year OS was 25%. Advanced disease (stage IV; AHR = 2.7, 95% CI: 1.1–6.5), HIV-positive status (AHR = 2.3, 95% CI: 1.1–5.0), elevated serum lactate dehydrogenase (LDH) (≥2× upper limit of normal; AHR = 2.2, 95% CI: 1.0–4.9), and delayed initiation of chemotherapy (≥2 months; AHR = 2.6, 95% CI: 1.2–5.8) were independently associated with increased mortality. Among HIV-positive patients, neutropenic fever was the sole independent predictor of death (AHR = 4.3, 95% CI: 1.1–17.4).
Conclusions
Survival among patients with DLBCL in Northwest Ethiopia remains poor, particularly among those presenting with advanced disease, those living with HIV, those that have delayed treatment initiation, and those that have elevated serum LDH – all these parameters were independently associated with increased mortality. Strengthening diagnostic capacity, ensuring equitable access to immunochemotherapy, integrating HIV–oncology services, and prioritizing early treatment initiation are essential to improving outcome.
Plain Language Summary
Diffuse large B-cell lymphoma (DLBCL) is an aggressive type of blood cancer that can be deadly if not treated promptly. This study looked at how long adults with DLBCL in Northwest Ethiopia survive and what factors increase their risk of death, especially among people living with HIV. We reviewed records of 175 adults diagnosed with DLBCL between 2020 and 2025 at three major hospitals. About 18% of these patients were HIV-positive. Overall, only 25% of patients were alive five years after diagnosis, and the median survival time was three years. Patients were more likely to die if they had advanced cancer, high levels of a blood marker called LDH, delayed chemotherapy, or HIV infection. Among HIV-positive patients, developing a serious infection called neutropenic fever greatly increased the risk of death. These findings highlight the need to diagnose DLBCL early, start treatment without delay, improve access to chemotherapy, and provide coordinated care for people with HIV to improve survival in Ethiopia.
Introduction
Diffuse large B-cell lymphoma (DLBCL) is the most common aggressive subtype of non-Hodgkin lymphoma, accounting for more than 40% of cases worldwide. 1 In high-income countries (HICs), the introduction of novel immunotherapies and expanded oncology services has led to substantial improvements in survival, with 5-year overall survival (OS) now exceeding 60-70%.2,3 Nevertheless, DLBCL remains a major contributor to lymphoma-related mortality globally, with outcomes varying widely across health-care systems. 4
Survival outcomes for DLBCL are markedly poorer in low- and middle-income countries (LMICs). Evidence from sub-Saharan Africa (SSA) and parts of Asia indicates that 5-year OS is frequently below 40%, with median survival in some cohorts under two years.5-7 These disparities are largely attributable to late presentation, limited availability of rituximab and radiotherapy, suboptimal supportive care, and constrained oncology infrastructure.8,9 Although the World Health Organization (WHO) has set a target of reducing cancer mortality by 25%, the mortality-to-incidence ratio for lymphoma in SSA has surpassed 61%, highlighting persistent inequities in cancer outcomes.2,3,10
HIV infection is a major driver of DLBCL in SSA.8,11 People living with HIV have a 10-to-20-fold higher risk of developing DLBCL than the general population. 12 Despite widespread antiretroviral therapy (ART) that has reduced the incidence of some AIDS-related malignancies. HIV-associated DLBCL continues to be aggressive and is associated with poorer survival.13-15 Multiple studies report lower 5-year OS among HIV-positive patients with DLBCL. 16 Moreover, in SSA, lymphoma is a leading cause of cancer-related death among people living with HIV. 17 In regions with high HIV burden, the occurrence of HIV and DLBCL represents a substantial and persistent barrier to improving lymphoma survival. 9
Established prognostic determinants for DLBCL include age at diagnosis, disease stage, performance status, and serum lactate dehydrogenase (LDH).12,18,19 However, in resource-limiting settings, additional predictors, such as HIV status, prior ART exposure, treatment delays, chemotherapy completion, and access to supportive care, significantly influence survival.20-24 These context-specific factors are often underrepresented in prognostic models derived from HICs, limiting their applicability for cancer control planning.
In Ethiopia, lymphomas constitute approximately 8.7% of all newly diagnosed cancers. 25 Despite this burden, robust data on long-term survival for DLBCL are limited; existing studies were hampered by short follow-up and incomplete assessment of HIV-related prognostic factors.7,26-28 Addressing this evidence gap is crucial for strengthening cancer control, informing clinical practice, and guiding resource allocation. Therefore, this multicenter cohort study aimed to estimate 5-year OS among adults with DLBCL treated in Northwest Ethiopia and examine the impact of HIV infection and treatment-related factors on survival.
Methods and Materials
Study Setting, Design, and Period
This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (STROBE) for cohort studies. 29 We performed a multi-center, hospital-based retrospective cohort study across three comprehensive specialized hospitals in Northwest Ethiopia: the University of Gondar Comprehensive Specialized Hospital, Tibebe Ghion Comprehensive Specialized Hospital (TGCSH), and Felege Hiwot Comprehensive Specialized Hospital (FHCSH). These centers provide most public-sector oncology care in the region.
Eligible patients were identified from oncology registries and medical records covering 1 August 1, 2020, and July 31, 2025, allowing for the evaluation of five OS. Data abstraction was conducted between September 1 and October 20, 2025. To maximize outcome ascertainment, structured telephone follow-up was performed for patients with incomplete survival documentation.
Study Populations and Eligibility Criteria
We included consecutive adult patients (≥18 years) with a first-time histopathological diagnosis of DLBCL who initiated chemotherapy at a study hospital during the study period. Inclusion required a documented HIV serostatus at diagnosis. Patients were excluded if outcome data were missing or insufficient to calculate the survival time.
Lymphoma Classification and Staging
Histopathological diagnoses were made by institutional pathologists using available classification systems (Working Formulation and REAL/WHO frameworks).30,31 Limited immunohistochemistry (IHC) capacity meant routine molecular subtyping and CD20 staining were not consistently available; accordingly, treatment decisions were guided primarily by histopathology and clinical features, assuming most DLBCL cases are CD20-positive. 32 Epstein–Barr virus (EBV) testing was not routinely performed. Disease staging used the Ann Arbor system, 33 based mainly on clinical assessment supplemented by available radiology (X-ray, ultrasound, and CT). Bone marrow aspiration was performed selectively; trephine biopsy and advanced imaging (MRI/PET) were not routinely available.
HIV-Related Clinical Assessment
HIV status was ascertained at lymphoma diagnosis through routine hospital testing. HIV-positive patients were staged using the WHO clinical staging system (excluding lymphoma-related manifestations). Baseline CD4 counts were recorded when available; plasma viral load testing was inconsistently accessible. ART exposure was defined as ART initiation before or at lymphoma diagnosis with continuation during lymphoma therapy. Adherence data were not routinely documented and thus were not systematically analyzed. Opportunistic infections were managed according to national protocols.
Treatment Exposure and Care Delivery
First-line therapy followed national and institutional protocols, typically CHOP every 21 days for 4–8 cycles, with rituximab added when available. 34 Rituximab use varied by availability and affordability; CD20 testing was not consistently documented. Predefined laboratory safety thresholds determined full-dose chemotherapy (hemoglobin ≥7 g/dL, platelets ≥30,000/µL, liver enzymes ≤2.5× upper normal limit (ULN)).
Dose reductions were applied for severe weight loss or, in HIV-positive patients, CD4 <200 cells/µL; under these circumstances, approximate dose intensities were cyclophosphamide 75%, doxorubicin 50%, and prednisone 60% of standard doses. Patients were followed monthly for the first six months and every 1–3 months after that for up to two years. Granulocyte colony-stimulating factor (G-CSF) was used selectively for severe neutropenia when available.
Study Variables and Definitions
The primary outcome was OS, defined as the time from DLBCL diagnosis to death from any cause; death was the event. Loss to follow-up was defined as no clinical contact for ≥3 months; phone tracing was used, and the last successful contact date was used for censoring. Cause of death was triangulated from hospital records, physician notes, and family reports; uncertain cases were classified as all-cause mortality.
Independent variables included sociodemographic factors (age, residence, insurance), baseline clinical features (Eastern Cooperative Oncology Group (ECOG) performance status, Ann Arbor stage, B symptoms, extranodal involvement, neutropenic fever, serum LDH), HIV-related factors (HIV status, CD4 count, ART exposure), and treatment parameters (chemotherapy regimen, cycles completed, dose reduction, treatment delays). Comorbidity was assessed using the Charlson Comorbidity Index (CCI).
35
Performance status was assessed using ECOG and categorized as 0–1 versus ≥2.
36
Data Collection, Quality Assurance, and Procedures
A structured data abstraction tool was developed based on variables identified through an extensive literature review.26,39-42 The tool was designed to ensure comprehensive capture of clinically relevant variables aligned with the study objectives. Data were extracted using a pretested abstraction form that captured sociodemographic, clinical, pathological, and treatment-related characteristics.
For patients with incomplete outcome data, telephone interviews were conducted with patients or their primary caregivers (parents, spouses, or adult children). The interview questionnaire was translated into Amharic and back-translated into English to ensure linguistic consistency. A pretest was conducted on 5% of the sample, and necessary modifications were made before data collection to enhance the clarity and consistency of responses.
To ensure data quality during telephone follow-up, data collectors received training on standardized interviewing techniques, confidentiality, and neutral probing. Training also emphasized minimizing interviewer bias and maintaining consistency in outcome ascertainment. At least three call attempts were made at different times and on different days before classifying a participant as lost to follow-up. A call log was maintained documenting the date, time, respondent, and interview outcome. Where feasible, survival status obtained via telephone was cross-checked against available hospital records to minimize recall bias and outcome misclassification.
Three trained senior oncology nurses conducted data collection under the supervision of three physicians. Completed forms were reviewed daily for completeness and internal consistency. Primary investigators conducted periodic random audits of selected records to verify abstraction accuracy. Random cross-checks and verification were performed before data entry into EpiData Association version 3.1 to ensure data accuracy and reliability. Data entry was performed using double-entry verification procedures to reduce transcription errors.
Statistical Analysis
Analyses were performed using StataCorp (Stata) version 17. Descriptive statistics summarize baseline characteristics. Categorical variables were compared with chi-square or Fisher’s exact tests and continuous variables with the Wilcoxon rank-sum test. Survival probabilities were estimated using the Kaplan–Meier method, with differences assessed by the log-rank test. Cox proportional hazard regression identified determinants of survival; variables with P<0.20 in bivariable analysis were candidates for multivariate models. Proportional hazards assumptions were evaluated by log(−log) plots and Schoenfeld residuals; model fit was assessed using Cox–Snell residuals. Statistical significance was defined as P≤0.05.
All centers were in the same geographic region and had similar structural/resource constraints; exploratory center-level analyses found no significant differences (P > 0.05) in all-cause mortality or rituximab use across hospitals. Given the retrospective design, randomization was not feasible and matching was not performed due to limited HIV-positive cases and risk of overfitting; potential confounding was addressed with multivariable Cox regression adjusting for a priori selected prognostic variables.
Management of Missing Variables
Missingness was assessed for all variables. Variables with <10% missingness were imputed by single imputation (median for continuous, mode for categorical), assuming missing at random. Variables with ≥10% missingness were analyzed using available case analysis. Sensitivity analyses assessed robustness to missing data handling.
Ethics Approval and Informed Consent
Ethical approval was obtained from the Ethical Review Committee of the School of Pharmacy, College of Medicine and Health Sciences, University of Gondar (Ref: SOPS/281/2024). Permission to access records was obtained from participating hospitals. Informed verbal consent was obtained for telephone interviews. Confidentiality was maintained by anonymization and restricted access in following the Declaration of Helsinki.
Results
Cohort Description and Follow-Up
Of the 206 patients identified, 175 met the predefined eligibility criteria and were included in the analytic cohort. All included cases were histologically confirmed with DLBCL according to the 5th edition WHO classification. Among these patients, 144 (82.3%) were HIV-negative and 31 (17.7%) were HIV-positive at diagnosis. Patients were followed retrospectively from the date of diagnosis for up to five years.
Baseline characteristics of included versus excluded patients are presented in Supplemental Table S1. Excluded patients were slightly older, had marginally poorer ECOG performance status, a higher comorbidity burden, and modestly higher mortality; however, none of these differences reached statistical significance, indicating minimal selection bias.
During follow-up, 67 patients (38.3%) died. The median survival time was 36 months (interquartile range [IQR]: 21–49 months). Of the deaths, 48 were confirmed through hospital records (36 HIV-negative and 12 HIV-positive), and 19 were identified through structured telephone tracing (14 HIV-negative and 5 HIV-positive). All confirmed deaths were included as events in the survival analysis.
At the end of follow-up, 70 patients (40.0%) remained under active follow-up. Thirty-eight patients were initially classified as lost to follow-up; among these, 19 (10.8%) were subsequently confirmed deceased through telephone tracing. Twelve patients (6.9%) remained untraceable despite repeated attempts and were censored at their last documented clinical contact. Follow-up duration and censoring patterns did not significantly differ by HIV status (p = 0.23).
Sociodemographic and Clinical Characteristics of the Study Participants
Among the 175 patients, 73.2% were aged ≤60 years and 56.0% were male. There were no significant differences in age (p = 0.50) or sex distribution (p = 0.60) according to HIV status.
Baseline Clinical and Demographic Characteristics of the Study Participants Depending on HIV Status

CCI: Charlson comorbidity index, DLBCL: diffuse large B-cell lymphoma, ECOG: Eastern Cooperative Oncology Group, UNL: upper normal limit, LDH: lactate dehydrogenase. P-values were calculated using chi-square tests for categorical variables and the Wilcoxon rank-sum test for continuous variables. Significant values are marked with an asterisk (*). B-Symptoms: presence of fever (>38°C), drenching night sweats, or unintentional weight loss.
aPercentages are based on available data; denominators vary due to missing values. Staging information was available for a subset of patients.
Treatment Characteristics of the Study Participants
Distribution of Chemotherapy Type, Cycle Characteristics, and Supportive Interventions of Study Participants Depending on HIV Status (N = 175)

CHOP: cyclophosphamide, doxorubicin, vincristine, and prednisone; R-CHOP = rituximab plus CHOP, G-CSF: granulocyte-colony stimulating factor. P-values were calculated using chi-square tests. Statistically significant values are marked with an asterisk (*).
HIV-Related Clinical and Immunologic Characteristics of the Study Participants
HIV Clinical and Immunologic Characteristics of HIV-Positive DLBCL Patients (N = 31)

ART: antiretroviral therapy; TDF: tenofovir disoproxil fumarate; ABC: abacavir. OIs: Opportunistic infections.
Comparison of Baseline Characteristics by Rituximab Exposure
Chi-square analysis demonstrated significant differences between the rituximab and non-rituximab groups regarding age, health insurance status, and relapse. No significant differences were observed for sex, residence, treatment-related toxicity, stage at diagnosis, or extranodal involvement (Supplemental Table S2).
Among the 175 patients, 36 (20.6%) underwent CD20 IHC testing, of whom 29 (16.6%) were CD20-positive. Rituximab was administered to 17 CD20-positive patients (9.7%) and 54 patients without documented CD20 testing (30.9%). Twelve CD20-positive patients (6.9%) did not receive rituximab (Supplemental Table S3).
Overall Survival Rate of the Study Participants
The 5-year overall survival (OS) rate was 25% (95% CI: 12.7%–39.8%). Estimated cumulative OS at 1, 2, 3, and 4 years was 82.3%, 67.9%, 44.7%, and 36.9%, respectively. Survival probability declined progressively over time, with the steepest decrease occurring after the second year of follow-up (Figure 1). Kaplan- Meier Plot for overall survival function in months of study participants (n = 175)
Kaplan–Meier Curve for Overall Survival Among Variable Subgroups
Kaplan–Meier analyses demonstrated that patients diagnosed at early stages (I–II) had the highest survival probabilities, whereas those with stage IV disease had the poorest outcomes. HIV-negative patients exhibited consistently higher survival probabilities than HIV-positive patients throughout follow-up. Survival declined stepwise with increasing LDH levels, with the ≥2× UNL group demonstrating the lowest survival probabilities. Longer intervals between diagnosis and treatment initiation were associated with progressively poorer survival. Patients treated with R-CHOP showed higher survival probabilities than those receiving CHOP alone (Figure 2). Furthermore, the survival curves separated early, suggesting an early benefit associated with R-CHOP, and the difference persisted over time, although the magnitude of separation narrowed during later follow-up (Supplemental Figure S1). Kaplan-Meier plots comparing OS for patients with DLBCL according to: (A) disease stage at diagnosis, (B) HIV status, (C) serum LDH level, and (D) diagnosis to treatment interval (in month)
Predictors of All-Cause Mortality Among Patients with DLBCL
Cox Proportional Hazards Analysis of Factors Associated With Mortality Among Study Participants
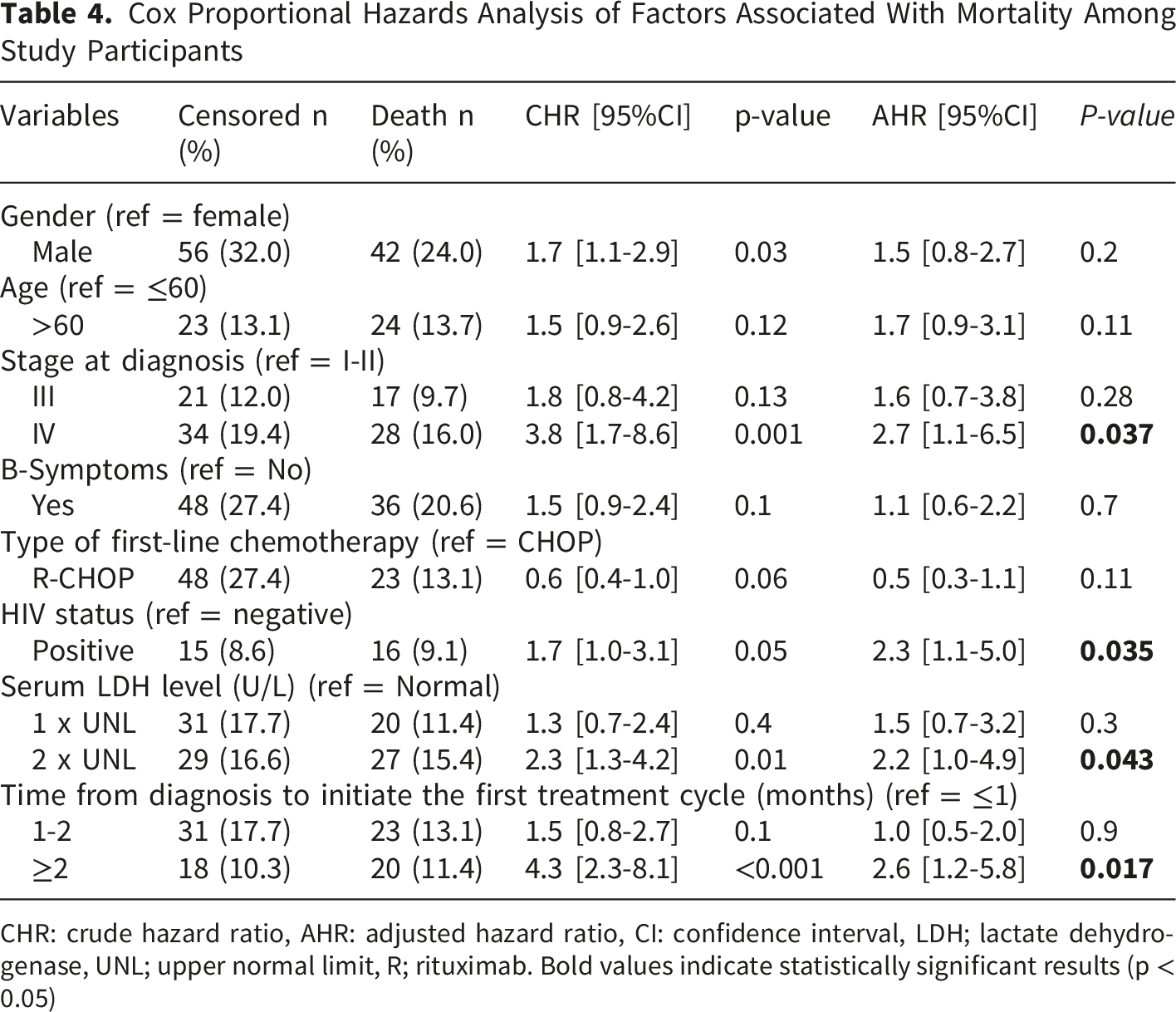
CHR: crude hazard ratio, AHR: adjusted hazard ratio, CI: confidence interval, LDH; lactate dehydrogenase, UNL; upper normal limit, R; rituximab. Bold values indicate statistically significant results (p < 0.05)
Predictors of Study Participants with HIV-Positive Status
Cox Proportional Hazards Analysis of Mortality in HIV-Positive Participants (n = 31)

Neutropenic fever: A single oral temperature ≥38.3°C or ≥38.0°C sustained for ≥1 hour, occurring with an absolute neutrophil count (ANC) <500 cells/mm3, or <1000 cells/mm3 with an expected decline to ≤5.
HR: hazard Ratio; AHR = adjusted hazard ratio; CI = confidence interval; ART = antiretroviral therapy; Ref = reference category. Bold values indicate statistically significant results (p < 0.05)

Kaplan–Meier estimates of overall survival according to neutropenic fever status in HIV-positive patients
Discussion
This multicenter cohort study provides the first comprehensive evaluation of 5-year OS and its prognostic determinants among patients with DLBCL, including those living with HIV, in Northwest Ethiopia. We observed markedly poor long-term survival, with a 5-year OS of 25%, highlighting the substantial mortality burden associated with aggressive lymphomas in this setting. Advanced disease stage, HIV infection, elevated LDH, and delayed initiation of chemotherapy were independently associated with increased mortality. Among HIV-positive patients, outcomes were particularly poor in those who developed neutropenic fever, underscoring the combined impact of immunosuppression and treatment-related toxicity.
The observed 5-year OS in our cohort remains substantially lower and consistent with data from other regions in SSA,7,28 as well as findings from cohorts of aggressive lymphoma in Asia. 43 In SSA, survival for aggressive lymphomas remains limited. For instance, a study from Zimbabwe among HIV-infected patients with high-grade lymphoma reported an 18-month OS of 30.6%. 27 In contrast, five-year OS for aggressive lymphomas in HICs ranges from 60% to 75%.44,45 For example, data from the Surveillance, Epidemiology, and End Results Program in the United States report five-year OS of approximately 61–64%.46,47 Similarly, a nationwide study from the Netherlands demonstrated five-year relative survival of around 60%. 48 This disparity reflects inequities in timely diagnosis, pathology services, and access to standardized treatment. In SSA, delayed and incomplete diagnostic pathways further contribute to advanced stage at presentation and poorer survival outcomes. 9
Advanced stage at diagnosis emerged as a dominant contributor to poor survival. More than two-thirds of patients presented with Ann-Arbor stage III-IV disease, a pattern consistent with other reports from SSA.38,49 This contrasts sharply with data from HICs, 50 where earlier-stage disease is more common and associated with substantially better long-term outcomes. 51 In our cohort, stage IV diseases independently predicted mortality. This aligns with global evidence showing advanced stage as a key adverse prognostic factor.39,44,45 This possibly advanced stage is often associated with higher tumor burden, greater organ involvement, and reduced responsiveness to therapy. Late presentation in this setting is probably multifactorial, including delayed health-seeking behavior, limited public awareness, financial barriers, and constrained diagnostic infrastructure. Shortages of oncology specialists and fragmented referral pathways further contribute to advanced disease at presentation.
Therapeutic limitations also appear to have influenced survival outcomes. Limited access to CD20 IHC testing and rituximab is a major treatment gap. Some eligible patients did not receive rituximab, whereas others received it empirically without documented CD20 status, reflecting diagnostic and supply constraints. Although rituximab did not retain independent statistical significance in multivariate analysis, its use was associated with lower relapse rates and a trend toward improved early and intermediate survival. Evidence from LMICs indicates that adding rituximab to CHOP improves outcomes even in advanced-stage disease. 52 The absence of radiotherapy services in the participating centers may signify an additional limitation, as consolidation radiotherapy has been shown to improve survival in early-stage and selected advanced-stage lymphoma patient. 53 Furthermore, limited access to advanced staging modalities and comprehensive immunophenotyping restricts accurate risk stratification and individualized treatment planning. These findings emphasize the need to expand diagnostic capacity and access to evidence-based therapies.
Age-related differences in treatment selection were also observed, with younger patients more likely to receive R-CHOP. This may reflect clinician concerns regarding treatment tolerance in older adults. However, evidence indicated that rituximab improves outcomes in older patients without substantially increasing toxicity. 54 In our cohort, toxicity profiles were comparable between CHOP and R-CHOP, whereas relapse rates were lower among patients receiving rituximab. These findings support the broader use of R-CHOP across age groups when clinically feasible.
Elevated serum LDH, a recognized marker of tumor burden and aggressive disease biology, was independently associated with mortality. 55 Similar findings have been reported in other African cohorts. 18 In resource-limited settings where advanced molecular diagnostics are not routinely available, LDH represents a practical and accessible tool for baseline risk stratification. Incorporating LDH into routine assessment may help identify high-risk patients who require closer monitoring and optimized supportive care.
Treatment delay was another critical and potentially modifiable determinant of survival. Initiation of chemotherapy 2 months or more after diagnosis was associated with significantly increased mortality. This finding is consistent with previous studies.24,56 In Ethiopia, delays in initiating cancer therapy are driven by recurrent drug stock-outs, financial barriers, and referral or scheduling bottlenecks within the limited oncology infrastructure. Prolonged baseline investigations, infection screening requirements prior to chemotherapy, and patient-level factors, including fear, stigma, and reliance on traditional remedies, further contribute to delayed treatment initiation. 57 While treatment deferral may be acceptable in selected indolent lymphomas, such delays are inappropriate in high-grade lymphomas, where rapid disease progression can occur. 58 Strengthening diagnostic pathways and reducing the time to treatment initiation should therefore be prioritized.
Survival disparities between HIV-positive and HIV-negative patients were substantial. Unlike reports from resource-rich settings, where optimized ART and integrated care have narrowed survival gaps, 59 HIV infection in our cohort remained an independent predictor of mortality, consistent with findings from other settings.60,61 ART alone did not eliminate excess mortality. Potential contributors include incomplete viral suppression, suboptimal ART adherence, pharmacologic interactions between ART and chemotherapy, delayed regimen optimization, and gaps in supportive care, which collectively may have increased vulnerability to treatment-related toxicity and mortality. 62 Moreover, HIV-positive patients in our cohort also exhibited higher LDH levels and more frequent treatment interruptions and dose reductions. 62 These findings highlight the importance of integrated HIV–oncology care models that address both immunologic vulnerability and systemic barriers.
Among HIV-positive patients, neutropenic fever was the only independent predictor of mortality in multivariate analysis. 63 This likely reflects the combined effects of baseline immune dysfunction and chemotherapy-induced myelosuppression. 64 In our setting, the limited use of G-CSF, delayed empiric antibiotic therapy, and resource constraints in infection management may have contributed to early deaths. 65 Establishing standardized neutropenic fever protocols and improving access to supportive care could substantially reduce infection-related mortality.
Implications and Recommendations
Improving DLBCL outcomes in Ethiopia requires coordinated expansion of diagnostic and treatment capacity. Priorities include ensuring access to CD20 testing, expanding the availability of rituximab, reducing delays in treatment initiation, and strengthening supportive care systems. Integration of HIV and oncology services is essential to address the complex needs of co-infected patients.
At the health-system level, strengthening referral networks, improving laboratory infrastructure, and enhancing workforce training are critical. Community-level interventions aimed at increasing awareness, reducing stigma, and facilitating early presentation may also improve the stage at diagnosis. Alignment with Ethiopia’s National Cancer Control Plan provides a strategic framework for sustainable progress.
Future research should include prospective studies evaluating early detection strategies, decentralized diagnostic models, and supportive care interventions. Establishing population-based lymphoma registries will improve epidemiologic surveillance and inform policy. Operational research addressing supply chains, workforce development, and financial protection mechanisms is also essential to strengthen health-system resilience.
Strengths and Limitations
This study has several strengths, including its multicenter design, 5-year follow-up period, and inclusion of HIV-specific clinical variables. These features enhance its relevance to real-world oncology practice in resource-limited settings. However, limitations should be acknowledged. The retrospective design and modest sample size may limit generalizability. Incomplete staging data and exclusion of records with missing variables could have introduced selection bias. Outcome ascertainment through telephone follow-up may have resulted in misclassification, although efforts were made to verify reported deaths. Residual confounding cannot be excluded despite multivariate adjustment. Limited access to advanced imaging and bone marrow trephine biopsy may have affected staging accuracy. Additionally, incomplete data on ART adherence and the absence of molecular biomarkers reflect routine practice constraints but limit more detailed prognostic characterization.
Conclusion
DLBCL in Northwest Ethiopia is associated with poor long-term survival. Advanced stage at diagnosis, elevated serum LDH, delayed treatment initiation, and HIV infection were key determinants of mortality. Among HIV-positive patients, neutropenic fever was strongly associated with an early death. These findings underscore the importance of timely diagnosis, prompt initiation of immunochemotherapy, and strengthened supportive care systems. Expanding access to rituximab, improving diagnostic infrastructure, and integrating HIV and oncology services are essential steps toward reducing disparities and improving lymphoma outcomes in resource-constrained settings.
Supplemental Material
Supplemental Material -Five-Year Survival and Determinants of Diffuse Large B-cell Lymphoma in Resource-Limited Settings: The Impact of HIV/AIDS in a Multi-Center Cohort Study
Supplemental Material for Five-Year Survival and Determinants of Diffuse Large B-cell Lymphoma in Resource-Limited Settings: The Impact of HIV/AIDS in a Multi-Center Cohort Study by Abel Temeche Kassaw, Ephrem Tafesse Teferi, Getachew Yitayew Tarekegn, Tigabu Eskeziya Zerihun, Samuel Agegnew Wondm, Tilaye Arega Moges
Footnotes
Acknowledgments
I would like to express my heartfelt thanks to the oncology staff at the University of Gondar, Tibebe Ghion, and Felege-Hiwot hospitals for their cooperation and assistance. Additionally, I extend special gratitude to all the patients and their relatives who participated in the phone interviews.
Ethical Considerations
Ethical approval was obtained from the Ethical Review Committee of the School of Pharmacy, College of Medicine and Health Sciences, University of Gondar (Ref: SOPS/281/2024).
Consent to Participate
Permission to access records was obtained from participating hospitals. Informed verbal consent was obtained for telephone interviews. Confidentiality was maintained by anonymization and restricted access in following the Declaration of Helsinki.
Consent for Publication
All authors have read and approved the manuscript and consent to its submission and publication
Authors Contribution
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be disclosed on a reasonable request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
