Abstract
Purpose
To examine disparities in receipt of the first course of treatment modality used among individuals diagnosed with early-onset colorectal cancer, focusing on sex, race/ethnicity, and rurality differences.
Methods
We conducted a cross-sectional analysis utilizing national data from the 2006-2020 Surveillance, Epidemiology, and End Results Program among adults aged 20-49. Key factors included sex, race/ethnicity, and rurality. Our main outcomes were whether patients started treatment, and which types they received. Multivariable logistic regression models were performed.
Results
Of total 82,427 patients, males (54.9%, p=0.097), racial minorities (0.8%-23.3%, p<0.001), and patients in all urban areas (70.7%; p<0.001) had higher rates of no treatment. Adjusted analysis showed that male patients had16%-19% lower odds of receiving surgery regardless of rurality (p<0.05) compared with female patients. In all/mostly urban areas, Black patients were found to had 17%-41% lower odds of receiving any treatment modalities (p<0.05); Hispanic patients had 11%-24% lower odds of receiving any treatment modalities (mostly urban: OR, 0.76; 95% CI, 0.60-0.95) or surgery alone (all urban: OR, 0.89; 95% CI, 0.84-0.95) compared with White patients. In rural areas, 33% and 34% lower odds of receiving radiation treatment were found among American Indian (AI)/Alaska Native (AN) (OR, 0.67; 95% CI, 0.46-0.98) and Asian/Pacific Islander (PI) patients (OR, 0.66; 95% CI, 0.44-0.99) compared with White patients, respectively.
Conclusions
Disparities in receipt of surgery treatment were observed in males regardless of rurality. Black and Hispanic patients in urban areas had lower treatment use, while AI/AN and Asian/PI patients in rural areas were less likely to start radiation. Targeted approaches for specific groups are needed.
Plain Language Summary
Younger adults with colorectal cancer often experience diagnostic delays and are more frequently diagnosed at later stages than those diagnosed at age 50 or older, likely reflecting challenges in accessing timely and appropriate care. Evaluating whether patients receive treatment after diagnosis is therefore critical for understanding access to care in this population. In our study, we observed several disparities in receipt of treatment. Male patients were consistently less likely to receive surgery, regardless of whether they lived in urban or rural areas. Black and Hispanic patients in all or mostly urban settings had lower odds of receiving treatment, while American Indian/Alaska Native (AI/AN) and Asian/Pacific Islander (PI) patients in rural areas showed reduced receipt of radiation therapy. These patterns underscore the need for targeted interventions that improve provider awareness and strengthen cultural competency. Addressing barriers faced by Black and Hispanic patients in urban areas and enhancing patient navigation and referral pathways for AI/AN and Asian/PI patients in rural communities are essential steps toward equitable treatment access.
Introduction
The incidence of early-onset colorectal cancer (EOCRC), defined as colorectal cancer diagnosed in individuals <50 years old, has been increasing in the United States (US), 1 and is projected to rise by 90% among individuals aged 20-34 years by 2030. 2 Moreover, EOCRC incidence rates are higher in some racial/ethnic groups (e.g., Hispanic, Black people) and males.3-6 Younger adults with EOCRC are more likely to experience diagnostic delay 7 and to be diagnosed at later stages of the disease than those diagnosed at age 50 years or older,8-10 a disparity that may stem from limited access to timely and appropriate care. Therefore, evaluating the receipt of treatment after a CRC diagnosis is essential, as it sheds light on healthcare access for individuals with EOCRC.
However, sex and racial differences in receipt of healthcare have been reported. For example, members of racial/ethnic groups (e.g., Hispanic, Black people) often live in areas that are more socioeconomically disadvantaged and experience disparities in health outcomes due to a lack of access to high quality of care.11-13 Black and Hispanic patients are less likely to receive surgery or chemotherapy.14,15 In addition, evidence indicates that among older adults with advanced CRC, females are less likely than men to receive systemic treatments (e.g., adjuvant or neoadjuvant therapy) and are more likely to receive only supportive care or to die without receiving any treatment.16-18 Importantly, residents of rural areas often face systemic challenges to healthcare access, including lack of transportation, lack of healthcare insurance, other financial barriers, and disinvestment in local services and hospital closures.19-21 Conversely, residents of smaller metropolitan areas may also face challenges, partly because healthcare systems in these regions can be strained by high patient volumes and longer wait times, potentially limiting timely access to treatment.22,23
Despite existing evidence, the role that sex and racial/ethnic differences play in influencing the receipt of treatment for EOCRC, particularly when accounting for geographic disparities using area-level measures, remains understudied. Thus, our study addresses this gap by examining disparities in receipt of any treatment and modality specifically among individuals diagnosed with EOCRC (including surgery, chemotherapy, and radiation), with a specific focus on sex, race/ethnicity, and a three-level rurality classification (all urban, mostly urban, and rural). We hypothesize that young adults may encounter distinct barriers to getting treatment based on where they live. This understanding is critical for informing healthcare delivery strategies aimed at enhancing equitable access to cancer care for groups disproportionately affected by EOCRC.
Methods
Study Design
We conducted a cross-sectional analysis using data from the 2006-2020 Incidence Data with Census Tract Attributes from the Surveillance, Epidemiology, and End Results (SEER) Program (November 2022 submission, https://seer.cancer.gov/), which are sources for comprehensive population-based information in the U.S. These data include patient demographics, primary tumor site, tumor morphology and stage at diagnosis, first course of treatment, and follow-up for vital status as well as information on census tract-level socioeconomic status [Urban Rural Indicator Code (URIC) and socioeconomic status (SES) quintile]. The eligible study population included patients diagnosed with CRC defined by the SEER Site Recode ICD-O-3/WHO 2008 definition of colon cancer (C180–C189), rectosigmoid junction cancer (C199), and rectal cancer (C209). 24 The cancer of the appendix was excluded following the recent reclassification of CRC definitions. 25 Data extracted for this study were publicly available and de-identified and thus considered exempt from institutional review board (IRB) review at Augusta University.
Study Samples
A total of 835,907 CRC patients were included in the 2006-2020 SEER program. We first excluded duplicate records for the same patients (n=11,338). Further, we excluded CRC diagnosis found in the appendix (n=24,199), unknown primary site (n=609), unknown age (n=327), unknown survival status (n=17), unknown survival time (n=9,585), unknown SES quintile (n=15,384), and unknown rurality (n=1). To obtain the eligible study sample for EOCRC patients, we excluded CRC patients aged < 20 years and aged 50 years or older (n=690,363), and those with missing race/ethnicity (n=651), or missing information on receipt of any treatment (n=1,006). We restricted the analytic sample to adults older than 20 years because SEER reports age at diagnosis in categorical form, and EOCRC is defined as occurring between ages 18 and 49. As a result, 82,427 eligible EOCRC patients diagnosed with CRC were included in full analysis (Supplementary Figure 1).
Measures: Outcome, Exposure, and Covariates
Receipt of any first course of treatment (also termed as receipt of treatment, e.g., surgery, chemotherapy, radiation) as well as receipt of each mentioned individual treatment modality were outcomes of interest. 26 Receipt of treatment in this study serves as a proxy for whether treatment was initiated, rather than for the timeliness of treatment initiation, which was the primary focus of our analysis. All were classified as yes or no. Sex (male or female), race/ethnicity (White, Black, Hispanic, American Indian/Alaskan Native [AI/AN], Asian/Pacific Islanders [PI]), and three level rurality were exposures of interest. The definition of rurality is based on the Urban Rural Indicator Code (URIC), and takes into account the U.S. Census Bureau’s percentage of the population living in non-urban areas defined by four categories: 100% urban (all urban), ≥50% but <100% urban (mostly urban), >0% but <50% urban (mostly rural), and 100% rural (all rural) tracts. We further classified four-level rurality into a three-level variable: 1) all urban, 2) mostly urban, and 3) rural (mostly rural and all rural). This revised classification helps reveal disparities in access to care that may be overlooked in mostly urban areas, particularly given that such existing research has focused primarily on rural health.
Covariates of interest include age at diagnosis (20-29, 30-39, 40-49 years), marital status (married, unmarried, or unknown), and SES level (low, medium, high). The SEER SES level is a census tract–based socioeconomic status index derived from multiple American Community Survey (ACS) indicators and categorized to reflect the socioeconomic environment of patients’ communities. For tumor characteristics, we included tumor grade (well differentiated, moderately differentiated, poorly differentiated, undifferentiated, or unknown), stage at diagnosis (localized, regionalized, distant, or unknown), and primary site (right or left). Finally, we included the five-year diagnosis interval (2006-2010, 2011-2015, 2016-2020) as one of the covariates.
Statistical Analysis
Descriptive statistics were used to describe the distribution of EOCRC patients according to sex, race/ethnicity, rurality, sociodemographic characteristics, tumor characteristics, and five-year diagnosis interval. Bivariate differences in the relationship of receipt of any treatment modality with sex, race/ethnicity, rurality, sociodemographic characteristics, tumor characteristics, and five-year diagnosis interval across three level of rurality were examined using chi-square tests. We further examined bivariate differences in the relationship of receipt of each treatment modality using chi-square tests. Further, we performed multivariable logistic regression to examine the association between sex, race/ethnicity, rurality, and receipt of any treatment as well as each treatment modality. For radiation therapy, we included only patients with rectal cancer, as radiation is used primarily for rectal—not colon—cancer in standard clinical practice. 27 Stratified analyses were also conducted to examine sex and racial/ethnic differences on receipt of any treatment and each treatment modality by three-level rurality. We additionally conducted a sensitivity analysis by excluding individuals diagnosed at a late stage, as metastatic tumors are generally not expected to undergo surgical treatment. All multivariable models were adjusted for sociodemographic characteristics, tumor characteristics, and five-year diagnosis interval. We calculated adjusted odds ratios (ORs) along with their corresponding 95% confidence interval. The level of statistical significance was set at an alpha level of 0.05 and the p-values were based on two-sided probability tests. We used SAS version 9.4, SAS Institute Inc., Cary, North Carolina, to perform all the analyses. The reporting of this study conforms to RECORD guidelines. 28
Results
Sample Characteristics and Receipt of First Course Treatment
Characteristics of EOCRC Patients by Receipt of Treatment

Abbreviations: EOCRC, early-onset colorectal cancer; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaska Native; PI, pacific islander.
Notes: To maintain consistency with the total column, column percentages were reported.
Characteristics of EOCRC Patients by Receipt of Surgery, Chemotherapy, and Radiation
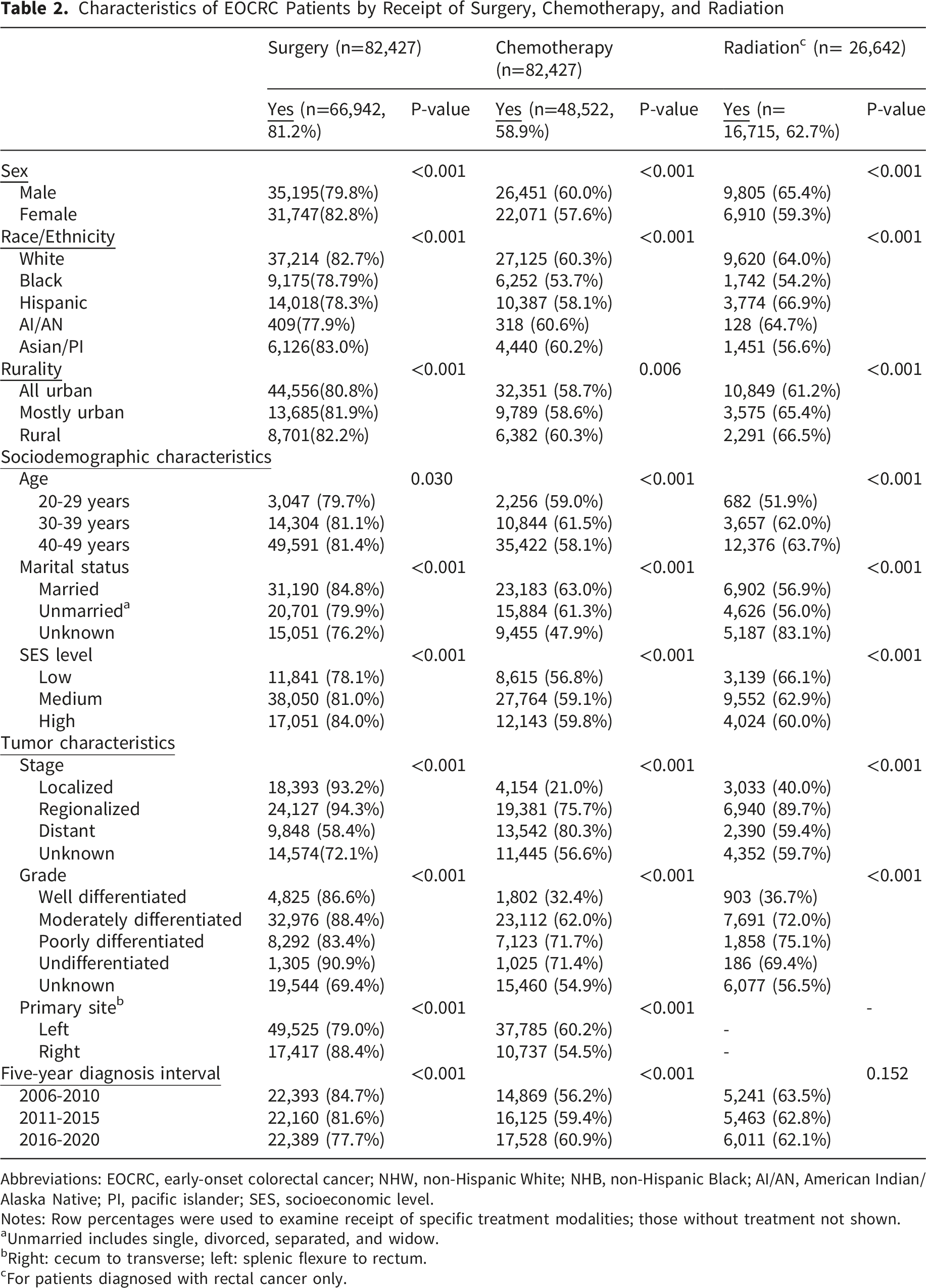
Abbreviations: EOCRC, early-onset colorectal cancer; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaska Native; PI, pacific islander; SES, socioeconomic level.
Notes: Row percentages were used to examine receipt of specific treatment modalities; those without treatment not shown.
aUnmarried includes single, divorced, separated, and widow.
bRight: cecum to transverse; left: splenic flexure to rectum.
cFor patients diagnosed with rectal cancer only.
Multivariable Analysis
Association Between Sex, Race/Ethnicity, Rurality, and Receipt of Treatment

Abbreviations: EOCRC, early-onset colorectal cancer; OR, Odd ratio; CI, confidence interval; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaska Native; PI, Pacific Islander.
Italicized text indicates statistically significant results.
aAdjusted models were adjusted for sociodemographic characteristics, tumor characteristics, and five-year diagnosis interval.
bFor patients diagnosed with rectal cancer only.
Association Between Sex, Race/Ethnicity, and Receipt of any Treatment/ Surgery by Rurality

Abbreviations: EOCRC, early-onset colorectal cancer; OR, Odd ratio; CI, confidence interval; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaska Native; PI, Pacific Islander.
Italicized text indicates statistically significant results.
aAdjusted models were adjusted for sociodemographic characteristics, tumor characteristics, and five-year diagnosis interval.
Association Between Sex, Race/Ethnicity, and Receipt of Chemotherapy/Radiation Therapy by Rurality

Abbreviations: OR, Odd ratio; CI, confidence interval; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaska Native; PI, Pacific Islander.
Italicized text indicates statistically significant results.
aAdjusted models were adjusted for sociodemographic characteristics, clinical related factors, and five-year diagnosis interval.
bFor patients diagnosed with rectal cancer only.
As showed in Supplementary Table 2, male patients living in all urban and mostly urban areas had lower odds of receiving surgery by 20% (95% CI, 0.72-0.88) and 17% (95% CI, 0.69-0.99), respectively, compared with female patients. Compared with White patients, Black and Hispanic patients living in all urban areas had 20%-26% lower odds of receiving surgery (all p-values <0.05). Hispanic patients living in mostly urban areas had 27% lower odds of receiving surgery (95% CI, 0.58-0.93), and Asian/PI patients living in rural areas had 59% lower odds of receiving surgery (95% CI, 0.17-0.99).
Discussion
To our knowledge, this is the first comprehensive study to examine demographic differences in receipt of treatment access while considering geographic locations using three-level rurality classification for EOCRC among national representative sample of US. Our findings showed that compared to White patients: (1) male patients had 16%–19% lower odds of receiving surgery, regardless of where they lived; (2) Black patients in all or mostly urban areas had 17%–41% lower odds of receiving any treatment modality; (3) Hispanic patients in all or mostly urban areas had a 11%-24% lower odds of receiving any treatment or surgery alone; and (4) AI/AN and Asian/PI patients in rural areas had 33%–34% lower odds of receiving radiation therapy among those with rectal cancer. These patterns can be understood through the Healthy People 2030 social determinants of health (SDoH) framework, which emphasizes how social and structural conditions shape access to appropriate care. 29 They also align with healthcare access models such as Andersen’s Behavioral Model, 30 which highlights how predisposing, enabling, and system-level factors influence treatment receipt. Together, these frameworks underscore the need for tailored strategies to improve equitable access to care.
First, we found that male patients had 16%-19% lower odds of receiving surgery regardless of rurality. These findings suggest that sex may have a stronger influence on receipt of treatment than geographic location. Our findings are partially aligned with existing literature on the timeliness of EOCRC treatment, although research specifically focused on EOCRC remains limited. For example, Popp and colleagues found that male EOCRC patients were more likely to delay treatment, including surgery. 31 To explain men’s reluctance to engage with healthcare services, studies have often cited cultural norms of stoicism and self-reliance as contributing factors,32-34 which is aligned with the SDoH domain of social and community context. This is concerning because EOCRC incidence and mortality rates in the U.S. have risen significantly in men compared with women. 35 Moreover, EOCRC survival is generally worse in men than in women. 36 Non-adherence to treatment access could contribute to poorer outcomes and warrants further investigation.
In addition, we found that Black patients living in all or mostly urban areas had 17% to 41% lower odds of receiving any treatment modality, which is consistent with prior research. Leech and colleagues reported that Black patients had 31% lower odds of receiving CRC-related surgery in North Carolina (NC). 37 Results from our sensitive analysis also align with this NC study, which found that Black patients living in all urban areas were 20% less likely to receive surgical treatment for those diagnosed at an earlier stage. Black Medicare beneficiaries were also 8% less likely to receive any cancer-related treatment compared with White patients. 13 Similar patterns were also observed in Hispanic patients in our analysis. Notably, an intersecting relationship between race/ethnicity and radiation treatment was identified, highlighting the nuanced ways in which social and geographic factors shape access to radiation therapy. Consistent with this intersection, we found that Hispanic patients living in mostly urban areas had 11%-24% lower odds of receiving any treatment modality or surgery alone. In particular, our sensitivity analysis showed that Hispanic patients living in all/mostly urban areas were less likely to receive surgical treatment by 26%-27% for those diagnosed at an earlier stage. In line with prior study, Obrochta and colleagues found that Hispanic patients in California were more likely to be undertreated. 12 Several factors may explain why Black and Hispanic patients living in urban areas continued to experience limited access to care, including systemic racism, fragmented insurance coverage, potential provider bias, and strain within the healthcare system.38,39 These factors align with the mentioned SDoH and healthcare access frameworks. Although prior studies of EOCRC have not identified disparities in receipt of treatment among individuals residing in all or mostly urban areas, most research has centered on racial minorities living in rural or persistent poverty areas facing lack of access to treatment resources. 13 Only one study focusing on Medicare beneficiaries found that people living in a metropolitan area were less likely to undergo surgical resection for CRC but without examining racial differences. 13 Differences from our study may reflect the fact that prior studies predominantly focused on older populations, potentially overlooking the unique barriers faced by younger racial/ethnic minorities affected by EOCRC.
Another important finding is that AI/AN and Asian/PI patients living in rural areas had 33% - 34% lower odds of receiving radiation therapy among those diagnosed with rectal cancer only. Our sensitivity analysis also showed that Asian/PI living in rural areas were also had lower odds of receiving surgical treatment for those diagnosed at an earlier stage by 59%. However, there is a notable lack of literature addressing racial minorities beyond Black and Hispanic populations, making it difficult to contextualize our findings within existing research. Although the literature is limited, prior research consistently demonstrated that cancer patients living in rural or poverty areas are more likely to experience limited access to cancer-related surgery.37,40 In particular, AI/AN cancer patients living in nonmetropolitan areas usually experience substantial travel disparities in accessing radiation therapy. This is particularly true for racial minorities because they are more likely to reside in socioeconomically disadvantaged areas (e.g., rural or poverty areas) experience not receiving appropriate treatment.12,15,41,42 Additional explanations related to several SDoH factors may include a lack of healthcare insurance, lower health literacy, and the limited healthcare facilities and shortage of professionals in rural settings.43,44 Cultural, linguistic, and systemic barriers—such as trust in healthcare systems and the availability of culturally competent providers—may also help explain disparities in access to care among racial minority groups, including AI/AN and Asian/PI populations. 38 These factors correspond to SDoH and healthcare access frameworks noted at the beginning of discussion. Delivering radiation therapy services in rural areas can also be particularly challenging due to the substantial infrastructure and financial investment required to build and maintain linear accelerators, along with ensuring adequate radiation safety protocols.45,46 Despite well-established social disadvantage among racial minority groups, research examining barriers to care remains limited for some populations, including AI/AN and Asian/PI patients. Future research that incorporate more diverse data and account for unique barriers faced by these groups may further elucidate the disparities in receipt of radiation among AI/AN and Asian/PI EOCRC patients.
Finally, an unexpected finding was that patients living in rural areas had higher odds of receiving treatment in the non-stratified analysis (Table 3), particularly for surgery and radiation therapy. This finding differs from prior literature that frequently highlights persistent rural health disparities.19-21 Several factors may help explain this pattern. For rural patients, sample selection and referral pathways bias may partly contribute to the observed differences, as SEER may primarily capture individuals who are already connected to oncology services—often through referred to consolidated tertiary or regional cancer centers. 47 This interpretation is supported, but not proven, by prior work showing shorter travel times to care in rural SEER regions compared with rural non-SEER regions, as well as by broader concerns regarding the generalizability of SEER for rural cancer surveillance. 48 Future research gathering the actual barriers faced by patients living in specific geographic areas may help identify context-specific factors that influence receipt of treatment and guide targeted interventions.
Strength and Limitations
A major strength of this study is the use of specialized dataset including census tract-level rurality classified into three distinct categories, offering a more nuanced approach compared to the traditional binary definition of rurality. This refined classification helps illuminate potential barriers experienced by individuals residing in areas that may not be considered fully rural yet still differ structurally from fully urban environments. In particular, residents of mostly urban areas may face unique system-level or structural obstacles that are not captured when rurality is dichotomized. For example, healthcare resources in mostly urban areas may differ from those in fully urban settings, potentially reducing timely access to treatment among younger patients. Additionally, healthcare systems may prioritize resource allocation toward older adults with more complex medical needs, which could further limit the availability of timely treatment for younger populations. 39
Despite its strengths, there were some limitations that should be noted. First, a key limitation is the SEER program’s aggregation of AI/AN individuals into a single category and similar grouping of Asian/PI populations. This lack of disaggregation substantially restricts our capacity to examine subgroup-specific experiences and may obscure important differences in treatment barriers. Future research incorporating fully disaggregated data will be essential to validate and deepen our understanding of the disparities identified in this study. Second, structural barriers faced in urban areas (e.g., healthcare strain) and rural areas (e.g., transportation barriers, limited treatment facilities) were not available for evaluation as cancer registries usually do not collect data on SDoH. In addition, individual-level SDoH (e.g., income, education, healthcare insurance), the presence of comorbidities, and information on patients’ knowledge of cancer risk and treatment options were not available, limiting our ability to evaluate and adjust for potential confounding effects. Further, incomplete or inconsistent reporting regarding receipt of treatment across facilities can lead to underestimation of disparities. While cancer registries may document receipt of treatment, they often do not capture whether treatment was delayed or declined, nor do they provide actual treatment dates. Moreover, SEER does not clearly specify whether treatment records reflect only those administered within defined timeframes, which may introduce uncertainty in the interpretation of first-course treatment data. The lack of detailed timeframes and information on the reasons for not receiving treatment may lead to misclassification, hinder comparisons across groups, limit the ability to identify actual barriers to care, and ultimately affect the interpretation of treatment patterns. Finally, although the rural–urban difference in receipt of treatment was statistically significant, the absolute difference was small (approximately 1%). For example, the odds of receiving surgery were modestly higher among individuals living in mostly urban areas compared with those in rural areas (approximately 12% vs 11% higher odds, respectively), reflecting minimal absolute differences. Even so, small disparities can matter at the population level, particularly for young patients, where delays or gaps in care may affect long-term outcomes. At the same time, we acknowledge that the modest magnitude of this difference limits its clinical implications and should be interpreted with caution.
Conclusions
Findings from our study revealed that disparities in receipt of surgery treatment were observed in males regardless of residential rurality. Black and Hispanic patients living in all or mostly urban areas were less likely to receive treatment, while AI/AN and Asian/PI patients living in rural areas also faced lower rates of receipt of radiation. This underscores the need for targeted interventions focusing on improving provider awareness and enhancing cultural competency to mitigate these disparities for young males. Identifying the barriers faced by Black/Hispanic patients living in urban areas and allocating resources to improve equitable access to treatment remain essential. Actionable supports—such as strengthening patient navigation and improving referral pathways—may be particularly beneficial for AI/AN and Asian/PI patients in rural settings who face unique challenges in accessing radiation treatment.
Supplemental Material
Supplemental Material - Geographic and Demographic Disparities in Receipt of Treatment for Early-Onset Colorectal Cancer
Supplemental Material for Geographic and Demographic Disparities in Receipt of Treatment for Early-Onset Colorectal Cancer by Meng-Han Tsai, Steven S. Coughlin, Jorge Cortes in Cancer Control.
Footnotes
Ethical Considerations
This research does not involve human and/or animal experimentation by any of the authors. Data extracted for this study was publicly available and de-identified and thus considered exempt from IRB review at Augusta University.
Author Contributions
Dr. Tsai had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Tsai. Acquisition, analysis, or interpretation of data: Tsai. Drafting of the manuscript: Tsai, Coughlin, Cortes. Critical revision of the manuscript for important intellectual content: Tsai, Coughlin, Cortes. Statistical analysis: Tsai. Obtained funding: Tsai. Administrative, technical, or material support: Coughlin, Cortes. Supervision: Cortes.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Meng-Han Tsai was supported at least in part through Augusta ROAR SCORE Career Enhancement Core (U54HL169191). The sponsor did not contribute to study design, data collection/analysis/interpretation, manuscript drafting, or manuscript submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
