Abstract
Introduction
Early-onset colorectal cancer (EOCRC) is increasing worldwide, with Serbia showing a similar incidence compared to global trends. Precise mutation genotyping has gained importance following the recent approval of KRAS-specific inhibitors. Although KRAS, NRAS, and BRAF testing is routinely performed in Serbia, specific mutation subtypes in EOCRC patients have not yet been published. This retrospective cohort study aimed to investigate temporal trends in EOCRC incidence in Serbia and characterize the mutational profile of KRAS, NRAS, and BRAF in EOCRC patients.
Methods
National cancer registry data from 2016 to 2022 were analyzed to assess EOCRC incidence trends. Molecular testing for KRAS, NRAS, and BRAF was performed on 681, 420, and 67 EOCRC patients, respectively, using qPCR-based diagnostic assays, complemented by Sanger sequencing on 54 cases to characterize KRAS exon 2 and BRAF V600E mutations.
Results
Registry data revealed a consistent upward trend in EOCRC incidence, especially in the 45-49 years’ age group. In the qPCR-tested cohort, KRAS mutations were detected in 44.3% (302/681), NRAS in 6.4% (27/420), and BRAF in 8.9% (6/67). In the sequenced subset, KRAS mutations were found in 20.4%, including G12D (36.4%), G13D (27.3%), G12 C (18.1%), and G12S/G12 V (9.1%) variants. BRAF V600E was detected in 3.7%.
Conclusions
We report a rise in EOCRC in Serbia, especially in ages 45-49, and recommend policy makers to lower the screening age to 45. We present the first detailed molecular profile of Serbian EOCRC and recommend that policy makers implement routine KRAS variant testing and ensure access to KRAS G12C-targeted therapies to improve personalized care.
Keywords
Introduction
Colorectal cancer (CRC) represents a major global health challenge, ranking as the third most commonly diagnosed cancer and the second leading cause of cancer-related mortality worldwide. According to GLOBOCAN data, in 2022 there were approximately 1.93 million new cases and 904 000 deaths attributed to CRC, accounting for 9.6% of all cancer diagnoses and 9.3% of cancer deaths globally. 1 While CRC has traditionally been considered a disease of older adults, recent decades have seen a concerning rise in the incidence of early-onset CRC (EOCRC) by 1.4% annually, despite a 3.1% annual decrease in CRC incidence among individuals over 50.2-4 In the USA, about 10% of all CRC cases are diagnosed in patients under 50 years. 5 Considering recent trends, it is estimated that by 2030, the incidence of early-onset colon and rectal cancer will increase by 90% and 124%, respectively, with approximately 11% of colon cancers and 23% of rectal cancers expected to occur in individuals under 50. These projections indicate that the EOCRC is clearly becoming an critical global public health issue. 6
CRC is a significant public health challenge in Serbia as well, with recent data highlighting its substantial burden on the national healthcare system. According to 2022 data from the Serbian Cancer Registry, Institute of Public Health of Serbia “Dr Milan Jovanović Batut,” CRC was newly diagnosed in 5199 individuals (2053 females and 3146 males), accounting for 12.5% of all cancer cases. CRC ranked as the second most common cancer and the second leading cause of cancer-related morbidity in the country. 7 Serbia introduced an organized national CRC screening program in December 2012, targeting individuals aged 50-74 years with biennial invitations for fecal occult blood testing followed by colonoscopy for positive cases. 8 However, this framework excludes individuals under 50 years, leaving the younger population outside organized early detection. Notably, the proportion of EOCRC cases in Serbia (8.2%) is close to that reported in the United States (10%). 7 Given the rising burden of EOCRC in Serbia, further research is essential to elucidate its underlying risk factors, inform targeted prevention strategies, enhance early detection efforts, and guide tailored therapeutic approaches in younger populations.
Current therapeutic approach of advanced CRC largely depend on the patient and tumor characteristics, specifically tumor sidedness and molecular profiling, particularly the presence of mutations in proto-oncogenes KRAS, NRAS and BRAF, 9 which directly impact the choice of targeted therapies. KRAS mutations are identified in 35-45% of CRC cases, most commonly at codons 12 and 13 of exon 2, while mutated NRAS is present in 7.5% of the cases at codons 12, 13 or 61. 10 These mutations result in the accumulation of RAS protein in its active form, thus promoting cell proliferation, differentiation, apoptosis, and survival. Mutated KRAS is a strong independent predictor of survival in CRC patients,11-13 and is also associated with the development of lung,11,14 and liver metastases, 12 as well as poor prognosis.15,16 Therefore, accurate and sensitive assessment of RAS mutation status has become crucial for guiding personalized treatment decisions. In addition to KRAS and NRAS, mutations in the BRAF occur in approximately 11% of all CRCs,17,18 and also play a key role in tumor progression. Although there are about 30 different BRAF mutations, the V600E mutation in exon 15 is the most common, accounting for 90% of all BRAF-mutated CRCs. 19 This mutation is associated with shorter overall survival and resistance to anti-EGFR therapy, making it a recognized poor predictive and prognostic marker.20-22 Guidelines recommend routine testing for BRAF status in metastatic CRC (mCRC), especially in KRAS wild-type patients, as KRAS and BRAF mutations are generally mutually exclusive. 23 In routine clinical practice in Serbia, KRAS and NRAS mutation testing is performed, but the specific mutation subtypes are not published, while BRAF testing typically targets the V600E variant. However, knowing the exact KRAS mutation is increasingly important, as several subtype-specific KRAS inhibitors have recently been developed, with some already approved—primarily for use in non-small cell lung cancer, and, more recently, with extended application for mCRC.24-27 Given the rising burden and high incidence of EOCRC in Serbia, this study aims to characterize the epidemiological trends of EOCRC incidence and diagnostic testing in Serbia, alongside providing a detailed molecular profile of KRAS, NRAS, and BRAF mutations in EOCRC patients. By integrating incidence data and mutational analyses, we seek to inform timely screening policies and optimize personalized treatment approaches for younger CRC patients in Serbia.
Material and Methods
Study Design
This study comprised two complementary components: a. a population-based temporal analysis of EOCRC incidence in Serbia from national cancer registry data (Population 1), and b. a retrospective molecular analysis of EOCRC patients, including a large clinically tested cohort using IVD-registered quantitative PCR assays (Population 2) and a smaller sequenced population (Population 3).
EOCRC Incidence Analysis (Population 1)
Incidence rates of EOCRC were obtained by analyzing official national cancer registry data from the Institute of Public Health of Serbia “Dr Milan Jovanović Batut” for the period 2016-2022 using anonymized data and publicly available data (https://www.batut.org.rs/index.php?content=2096). EOCRC was defined as CRC diagnosed before the age of 50 years. The primary outcome for this analysis was the age-specific incidence rate of EOCRC. Calendar year of diagnosis was considered the exposure variable. The registry-based dataset included aggregated incidence data and did not contain individual-level clinical or molecular variables.
Retrospective Molecular Cohort (Population 2)
A total of 681 patients aged 18-50 years with histologically confirmed EOCRC were identified from institutional databases and included in this retrospective cohort. Patients were eligible if they had histologically confirmed primary or metastatic CRC. Both colon and rectal tumors were included. Exclusion criteria were age >50 years at diagnosis, lack of histological confirmation, or insufficient or unsuitable tumor tissue for molecular analysis. All consecutive patients meeting the inclusion and exclusion criteria during the study period were included. As this was a retrospective observational study, the sample size was determined by data availability rather than a predefined power calculation.
For 681 patients with mCRC, KRAS mutation status had been previously determined between 2008 and 2024 at the Institute for Oncology and Radiology of Serbia using IVD-registered quantitative PCR assays 9 in all patients, while NRAS was determined in 420 of those 681, and BRAF in 67 of those 681. The primary outcomes for this cohort were KRAS, NRAS and BRAF mutation status. Predictor variables included age at diagnosis, sex, and primary tumor location (colon vs rectum). Potential confounders considered in the analyses included age, sex, tumor location, and disease stage. No effect modifiers were prespecified.
Sequenced Subset (Population 3)
Due to the lack of variant-level mutation data in Population 2, a subset of 54 patients who met the same inclusion and exclusion criteria and had available tumor tissue of adequate DNA quality was selected for Sanger sequencing of KRAS exon 2 and BRAF V600E, enabling precise characterization of the mutational spectrum at the variant level.
These patients were included consecutively based on the availability of representative archival formalin-fixed paraffin-embedded (FFPE) tumor tissue suitable for sequencing. Tumor samples were obtained from the First Surgical Clinic of the University Clinical Centre of Serbia. This subset was used to enable direct sequencing-based assessment of KRAS exon 2 and BRAF V600E mutations. Potential selection bias related to tissue availability cannot be fully excluded.
To ensure comparability across patient groups, mutation status from both PCR-based testing and sequencing was classified as binary (mutant vs. wild-type), and harmonized mutation definitions were applied across all analyses.
Bias and Ethical Considerations
To minimize selection bias, only patients with histologically confirmed EOCRC and sufficient tumor material were included. Information bias was reduced by using standardized, validated molecular assays and centralized testing procedures. As this was a retrospective study, residual confounding and selection bias cannot be fully excluded. All patient data were de-identified prior to analysis.
The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024) and was approved by the Ethics Committee of the University Clinical Centre of Serbia (Ref. No.: 175/1; Date: April 27, 2021) and the Ethics Committee of the Institute for Oncology and Radiology of Serbia (Approval No.: 01-1/2023/701; Date: 30 March 30, 2023). Written informed consent was obtained from all patients prior to surgical intervention.
Genotyping of KRAS and BRAF
All formalin-fixed paraffin-embedded (FFPE) tumor samples were reviewed by a pathologist to confirm diagnosis and estimate tumor cellularity. Only samples with an estimated tumor content of ≥20% were considered suitable for molecular analysis. To isolate DNA, approximately 0.1 cm3 of paraffin-embedded tissue block was cut using a sterile scalpel. The tissue was deparaffinized by adding 1 mL of xylene (Merck, Germany), vortexing for 5 minutes (min), and centrifuging at 13 000 rpm for 5 min. After removing the supernatant, the deparaffinization step was repeated with 1 mL of xylene. The deparaffinized tissue was then washed twice by adding 1 mL of ethanol, vortexing for 1 min, centrifuging at 13 000 rpm for 5 min, and discarding the supernatant. Subsequently, the pellet was washed by 1 mL of 70% ethanol under the same conditions and after removing the supernatant, dried for 1 hour at 37°C. DNA was isolated using the QIAamp Fast DNA Tissue Kit according to the manufacturer’s instructions (Qiagen, Germany). DNA concentration and purity were assessed prior to analysis. Samples yielding insufficient DNA quantity or poor-quality amplification were re-extracted or excluded from further analysis.
For genotyping of KRAS and BRAF, DNA sequences of regions of interest (165 base pairs (bp) and 224 bp, respectively) were amplified using the following primers: KRAS Fwd: AAGGCCTGCTGAAAATGACTG; KRAS Rev: GGTCCTGCACCAGTAATATGCA; BRAF Fwd: TCATAATGCTTGCTCTGATAGGA and BRAF Rev: GGCCAAAAATTTAATCAGTGGA. PCR reactions were performed in a total volume of 50 µL, containing approximately 500 ng of DNA, 1 U of KAPA Taq DNA polymerase (Kapa Biosystems, USA), 1X KAPA Taq buffer A, 2.5 mM MgCl2, 0.2 mM of each dNTP, and 10 pmol of each primer. Cycling conditions consisted of an initial denaturation at 94°C for 5 min, followed by 35 cycles of 94°C for 1 min, 55°C for 1 min, and 72°C for 1 min, with a final extension at 72°C for 10 min. Sequencing reactions were performed using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems; ThermoFisher Scientific, Inc., USA) following the manufacturer’s instructions. Capillary electrophoresis was performed on the 3730 Genetic Analyzer (Applied Biosystems). Results were analyzed using Sequencing Analysis Software 5.2 (Applied Biosystems). Sanger sequencing was performed using a standardized and routinely validated protocol in our laboratory. PCR products were sequenced bidirectionally, and chromatograms were independently reviewed to ensure accurate base calling. Each sequencing run included appropriate positive and negative controls. Runs were considered successful when clear, high-quality electropherograms with unambiguous peak resolution were obtained. Samples yielding low-quality or ambiguous chromatograms were re-amplified and re-sequenced. This approach ensured reproducibility and reliability of mutation detection using conventional Sanger sequencing methodology.
Statistical Analysis
Quantitative variables were handled as follows: age at diagnosis was analyzed as a continuous variable and summarized using descriptive statistics. EOCRC incidence rates were calculated annually and analyzed across calendar years treated as ordered variables. Tumor location was analyzed as a categorical variable (colon vs rectum). Mutation status for KRAS, NRAS, and BRAF was analyzed as binary variables (mutant vs. wild-type). Associations between KRAS, NRAS, and BRAF mutation status and clinicopathological variables were evaluated using the χ2 test. Descriptive and comparative statistical analyses were performed using IBM SPSS Statistics (version 20; IBM Corp., Armonk, NY, USA). Linear trends in EOCRC incidence proportions across ordered calendar years were assessed using the Cochran–Armitage trend test implemented in R version 4.3.1, testing for a monotonic increase or decrease in the binary outcome. Temporal trends in molecular testing from 2008 to 2025 were evaluated using Poisson regression in R, with the number of patients tested per year modeled as a function of year of diagnosis. Rate ratios with 95% confidence intervals were used to quantify the annual change in testing counts. Absolute testing counts were analyzed since total patient counts per year were not available, so these results describe trends in observed testing numbers rather than testing probability. A two-sided significance level of 0.05 was applied throughout. The reporting of this study conforms to the STROBE guidelines. 28
Results
The overall study design, including the three study populations, is summarized in Figure 1, which illustrates sample sizes, analyses and number of cases performed for each group. Overview of study populations and analyses. The flow chart indicates the number of cases in each population and the specific analyses performed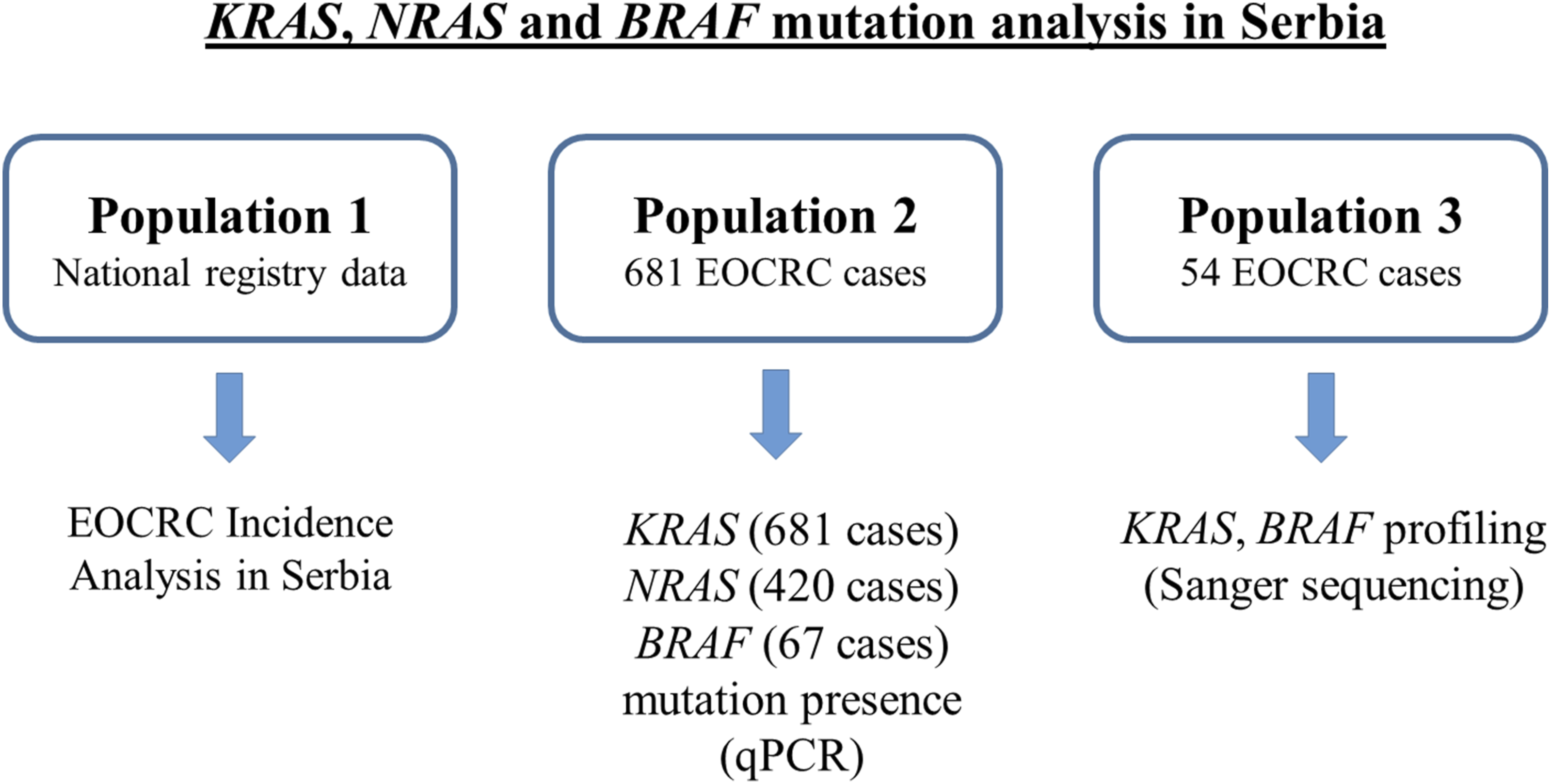
Incidence of EOCRC in Serbia
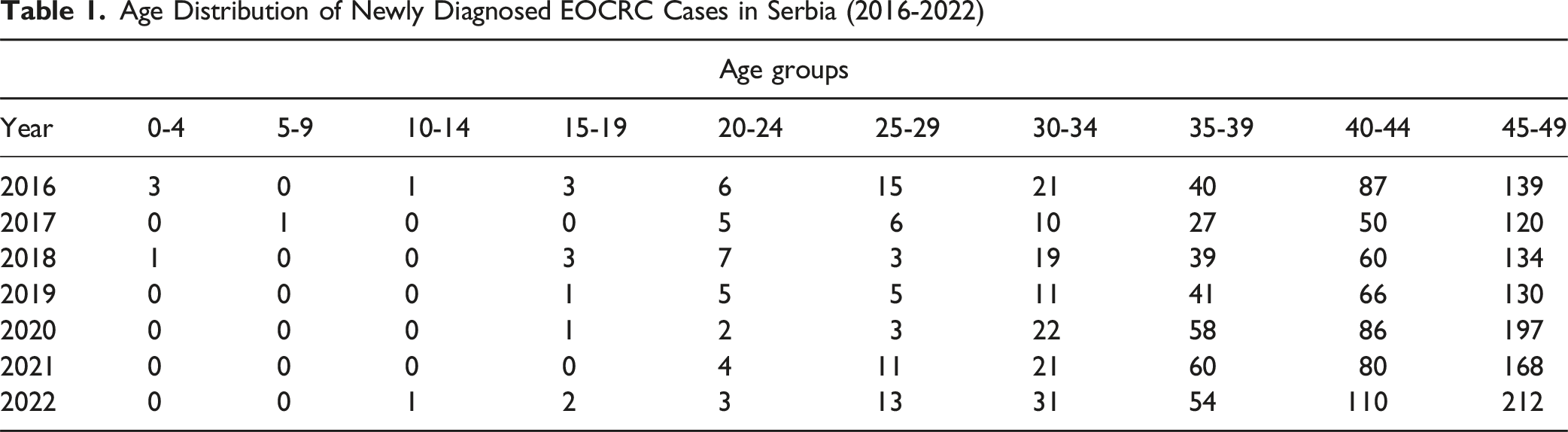
A total of 2198 EOCRC cases (Population 1) were registered in Serbia from 2016 to 2022 (Figure 2A–C). Annual case counts ranged from a minimum of 219 cases in 2017 to a maximum of 426 cases in 2022. The number of newly diagnosed EOCRC cases was consistently higher in males compared to females, with male-to-female ratios varying between 1.02 (2016) and 1.28 (2018 and 2020) (Figure 2A). When expressed relative to the total CRC burden, EOCRC accounted for 4.92% (2017) to 8.19% (2022) of all CRC cases (Figure 2C). The proportion of EOCRC demonstrated a statistically significant upward trend over the study period (Z = 4.63, P < 0.00001). Incidence and testing trends of EOCRC in Serbia (A) Annual number of newly diagnosed EOCRC cases, shown separately for males and females, (B) Annual number of newly diagnosed EOCRC cases, combined for both sexes, (C) Percentage of EOCRC cases relative to the total number CRC cases, (D) Number of EOCRC patients tested at IORS per year
Age Distribution of Newly Diagnosed EOCRC Cases in Serbia (2016-2022)
Mutational Profile of KRAS, NRAS, and BRAF in Serbian EOCRC Cases
To evaluate changes in diagnostic practices over time, we analyzed the annual number of EOCRC patients tested at IORS. Between 2008 and 2025, the number of tested patients increased over time, from just 10 individuals in 2008 to a peak of 77 in 2024 (Figure 2D), resulting in 681 patients in total (Population 2). The annual number of patients undergoing molecular testing increased significantly over time (rate ratio per year 1.09, 95% confidence interval 1.07-1.11, P < 0.001), corresponding to an approximately 8.9% increase in testing counts each year.
Clinicopathological Characteristics of EOCRC Patients (Population 2) Stratified by the KRAS, NRAS, and BRAF Mutation Status
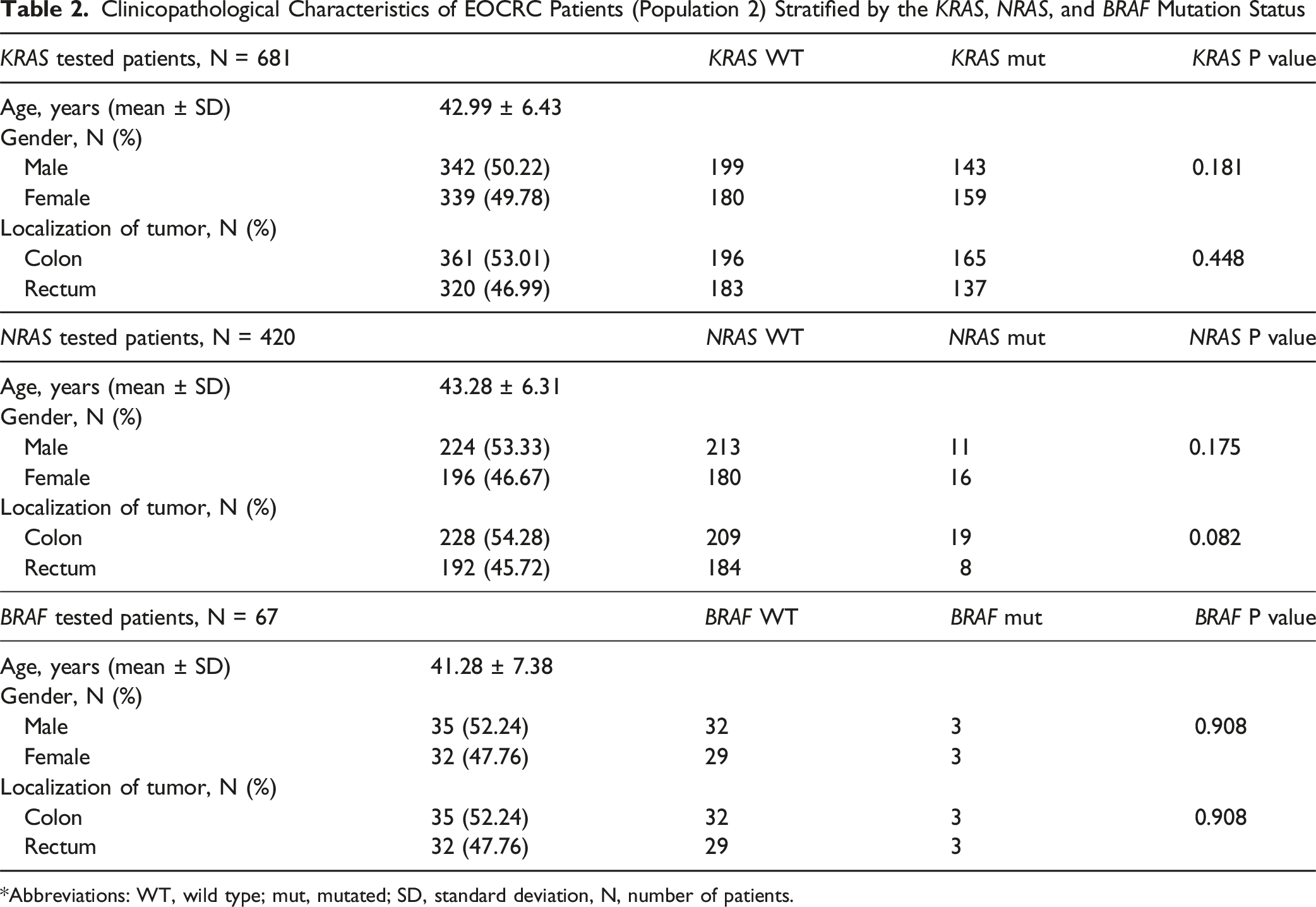
*Abbreviations: WT, wild type; mut, mutated; SD, standard deviation, N, number of patients.
Clinicopathological Characteristics of EOCRC Patients (Population 3) Stratified by the KRAS Mutation Status
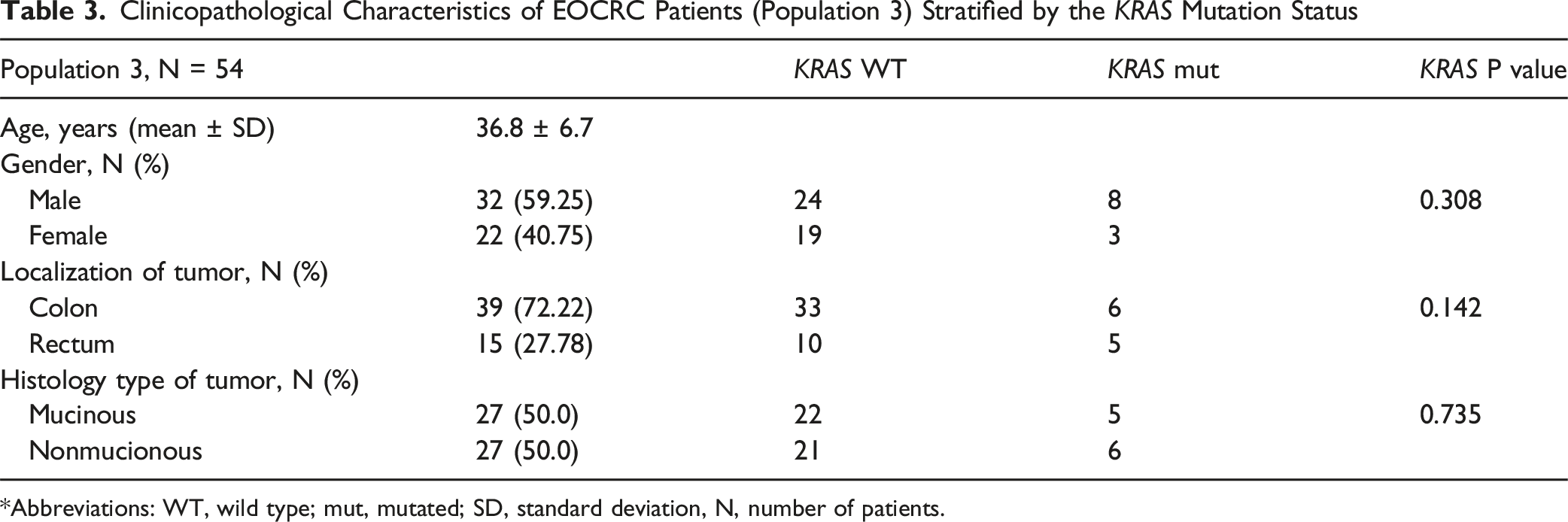
*Abbreviations: WT, wild type; mut, mutated; SD, standard deviation, N, number of patients.

Distribution of KRAS codon 12 and 13 mutations in EOCRC patients (Population 3). The bar chart illustrating KRAS status in EOCRC patients shown as percentages
Discussion
Given the evident rise in EOCRC incidence in Serbia, we aimed to investigate when this trend became apparent, to what extent KRAS, NRAS, and BRAF testing has been implemented over time, and to characterize their mutational profiles in this population. Thus, this study presents a comprehensive analysis of EOCRC in Serbia, integrating national incidence trends, institutional testing patterns, and detailed mutational profiling, which is essential for improving early detection strategies, guiding personalized treatment, and informing national healthcare policies tailored to this rising patient population.
As nationwide data became available in 2016, our analysis of EOCRC incidence was limited to the 2016-2022 period to ensure full territorial coverage. From the data, it can be observed that both the absolute number of EOCRC cases and their proportion of all CRC cases show parallel increases from 2016 to 2022, with nearly doubled cases in 2022. The consistent rise after 2020 suggests a rising contribution of EOCRC to the overall CRC burden, emphasizing the importance of problem awareness, targeted surveillance and prevention and earlier detection strategies in populations younger than 50 years. Our analysis of EOCRC incidence identified a marked increase in cases among individuals aged 45-49, an increase of over 50% within just seven years, making this age group a critical transitional zone just below the current screening threshold in Serbia. Several international guidelines, including those from the American Cancer Society and U.S. Preventive Services Task Force, have already moved to recommend CRC screening beginning at age 45 in response to rising early-onset incidence. 29 In Serbia, where screening still begins at age 50, these findings underscore the urgency of reconsidering the starting age for CRC screening. Several factors may underlie the observed rise in EOCRC incidence in Serbia. This may partly result from enhanced cancer reporting and improved diagnostic access, 30 but the persistent upward trend over several years points to a real epidemiological shift. Although the official program targets those aged 50 and above, increased public awareness may be prompting younger individuals to seek medical attention earlier, particularly when symptoms arise. This could lead to more frequent colonoscopies or follow-up investigations, even outside organized screening. Additionally, lifestyle-related risk factors such as poor diet, obesity, smoking, alcohol consumption, and sedentary behavior may also be contributing to a real rise in incidence. Hereditary syndromes, such as Lynch syndrome, and emerging evidence on the role of the gut microbiome, could further influence EOCRC risk.
In parallel with these epidemiological trends, our findings also point to evolving clinical practices, particularly in the use of molecular testing for EOCRC. Data from IORS show that EOCRC patients undergoing KRAS, NRAS, and BRAF testing increased more than seven-fold between 2008 and 2024, reaching a peak in 2024, which may reflect expanded access to molecular diagnostics, greater clinician awareness, and a broader recognition of the clinical relevance of mutation profiling in EOCRC management. As EOCRC continues to rise, ensuring equitable access to comprehensive molecular diagnostics will be essential for optimizing patient outcomes and informing public health strategies.
While KRAS, NRAS and BRAF mutations have been examined in Serbian patients with primary CRC, locally advanced and mCRC,31-35 their prevalence and clinical relevance in early-onset cases have not yet been explored. In our previously published nationwide study on CRC, which included 6369 unselected CRC patients, 9 the early-onset subgroup was not analyzed separately. In the present retrospective analysis, we identified 681 EOCRC cases (10.3%) from that cohort, enabling, for the first time, a focused examination of their mutational profile. We found that KRAS mutations were present in 44.3% (302/681) of EOCRC cases, NRAS in 6.4% (27/420), and BRAF V600E mutations in 8.9% (6/67). However, specific mutation variants were not reported. Difference in the number of tested patients for KRAS, NRAS and BRAF is due to the gradual implementation of extended molecular testing in Serbia, with NRAS testing introduced into routine clinical practice in 2017 and BRAF testing in 2018. In addition, BRAF testing was performed selectively and depended on clinician referral, further contributing to the lower number of tested cases. Furthermore, while the larger retrospective EOCRC cohort included mutation status for KRAS, NRAS, and BRAF, the present study focused exclusively on genotyping KRAS and BRAF. This decision was based on their greater clinical relevance, particularly in the context of anti-EGFR resistance and the availability of targeted therapies. Additionally, we provide the first detailed mutational profiling of a genotyped EOCRC cohort from Serbia (N = 54), revealing a KRAS mutation frequency of 20.4% and BRAF mutation frequency of 3.7%. These findings fall within the range reported in most previous studies, further supporting the distinct molecular profile of EOCRC. More specifically, a comprehensive systematic review and meta-analysis by Lawler et al (2024), encompassing 49 studies each on KRAS and BRAF in EOCRC, revealed that KRAS mutations were slightly less common, while BRAF mutations were significantly underrepresented in early-onset cases compared to late-onset CRC. These findings suggest that EOCRC follows distinct tumorigenic pathways compared to its later-onset counterpart. 36 Consistent with these meta-analytic findings, previous reports have shown a lower frequency of BRAF V600E mutation, with rates ranging from 0-8%. Similarly, most studies report a reduced prevalence of KRAS mutations in EOCRC (ranging from 4% to 27%), notable exceptions are the studies by Cercek et al and by Watson et al, which observed a notably higher KRAS mutation rate of 42% and 54%, respectively,37-43 more closely reflecting the results from our larger cohort.
In the Serbian EOCRC group, we detected five targetable nonsynonymous KRAS mutations: G12D, G13D, G12 C, G12S, and G12 V. These mutations are among the most frequently reported activating alterations in CRC. Notably, the distribution of KRAS mutations in our cohort G12D - 36.4%, G13D - 27.3%, G12 C −18.1%, G12S and G12 V - 9.1% partially aligns with global trends. The predominance of G12D is consistent with its established role as the most frequent KRAS alteration in CRC and mCRC (27.9-36.0%),44,45 although our frequency exceeds those reported in EOCRC-specific datasets (13.2-13.3%)46,47 and is closer to a more recent finding (57.1%). 47 For other mutations, we observed higher proportions of G13D and G12 C compared with typical CRC frequencies (18.8-21.8% and 1-7%, respectively),43,44 and with EOCRC data from Ponvilawan et al (4.8% and 7.1%) 47 and the UCSC Xena browser (5% and 3.3%). 46 Conversely, G12 V, usually the second most common KRAS mutation globally (∼21.8%),43,44 was underrepresented in our cohort (9.1%), though this aligns with EOCRC data from Ponvilawan et al (11.9%) 48 and UCSC Xena (8.3%). 47 These findings may reflect unique molecular characteristics of EOCRC or population-specific variation in KRAS mutation patterns, but should be verified on larger patient cohorts.
To date, no published data are available on specific KRAS variants in EOCRC patients in Serbia, likely due to molecular testing being limited to the detection of mutation presence rather than variant-level identification. KRAS mutations detected in the Serbian unselected CRC population across studies include G12D, G12 V, G13D, G12 A, G12 C, G12S, and G12 R,32,34,35 with our EOCRC cohort confirming the presence of all except G12 A and G12 R. Our comparative analysis suggests that the mutational spectrum of KRAS in EOCRC largely overlaps with that observed in unselected CRC cases in Serbia, particularly for the most common variants such as G12D, G12 V, and G13D, although with a relatively higher frequency of G13D and G12 C mutations and a lower prevalence of G12 V.
Considerable efforts have been directed toward the development of KRAS-specific inhibitors, with several agents already progressing through clinical trials. So far, two KRAS inhibitors AMG510 (sotorasib) 24 and MRTX840 (adagrasib), 25 specifically targeting the G12 C mutation have received U.S. Food and Drug Administration (FDA) approval initially for non-small cell lung cancer. Encouraged by their success, these inhibitors have since been explored in other solid tumors, including CRC. In 2024, the FDA granted accelerated approval to adagrasib plus cetuximab for KRAS G12C-mutated previously treated locally advanced or mCRC, with median response duration exceeding 6 months. 26 Most recently, in 2025 the indication for sotorasib was expanded to include its use with panitumumab for chemorefractory KRAS G12C-mutant mCRC. 27 Meanwhile, KRAS G12D-selective inhibitors, such as MRTX1133 (ClinicalTrials.gov Identifier: NCT05737706), RMC 9805 (NCT06040541), and QTX3034 (NCT06227377) are currently in early-phase clinical evaluation in CRC and other solid tumors, however, results have not yet been published. 44 In addition to KRAS-targeted therapies, BRAF inhibitors—such as encorafenib — have shown clinical benefit in mCRC patients with BRAF V600E mutations, especially when combined with EGFR inhibitors and chemotherapy. 49 In contrast, NRAS-specific inhibitors have not yet been developed. While KRAS and BRAF inhibitors are currently approved mainly for metastatic settings, their role in EOCRC and earlier disease stages remains under investigation. Although specific KRAS inhibitors are currently in early-phase clinical trials, as mentioned previously, routine testing of KRAS and BRAF mutations in EOCRC remains crucial,50,51 as it not only informs current treatment decisions but also prepares patients for potential inclusion in future targeted therapy trials, ultimately paving the way for more personalized and effective treatment strategies as these novel agents become clinically available. It is essential that Serbia remains aligned with global advancements and promptly implements new therapies, enabling more personalized treatment strategies and ensuring that EOCRC patients are not excluded from potentially beneficial targeted options due to insufficient molecular characterization. Additionally, our results imply a strong rationale to move beyond binary mutation detection and implement variant-specific KRAS testing as part of routine molecular diagnostics.
The findings of this study should be interpreted in light of several limitations. First, although molecular testing data were available for a large cohort of 681 patients (Population 2), detailed variant-level analysis was performed in a smaller sequenced subset of 54 cases (Population 3), which may limit the generalizability of specific mutation patterns. Second, NRAS and BRAF testing was not uniformly performed across the entire Population 2, reflecting historical differences in testing availability and clinical practice, and resulting in incomplete assessment of BRAF mutation prevalence. Third, data on microsatellite instability status and hereditary cancer syndromes were not available, precluding evaluation of their contribution to EOCRC in this population. Finally, the retrospective design and single-country setting may limit the generalizability of the findings to other populations. Despite these limitations, the study provides valuable insight into temporal trends and molecular characteristics of EOCRC within a national context.
Conclusion
In conclusion, this study reveals a significant and ongoing rise in EOCRC incidence in Serbia, particularly among individuals aged 45-49, highlighting the urgent need for evidence-based policy adjustments. Concurrently, we present the first comprehensive molecular characterization of KRAS, NRAS, and BRAF mutations in Serbian EOCRC patients, demonstrating a mutational profile largely consistent with global EOCRC data but distinct from late-onset cases. Notably, the lower prevalence of these mutations in younger patients supports the hypothesis of distinct oncogenic pathway in EOCRC. Based on these findings, we recommend the following actions for national healthcare decision-makers. It is essential to lower the starting age for the national CRC screening program to 45 years. This change will facilitate earlier detection and intervention in the age group most affected by the disease. The routine testing should include the identification of specific single-nucleotide substitutions in the KRAS gene, with a particular focus on the KRAS G12 C mutation. It is crucial that these tests are fully reimbursed by the Republic Health Insurance Fund. Additionally, the Fund should ensure the availability of KRAS G12C-specific inhibitors, which have demonstrated significant clinical benefits in large international trials and mark a crucial advancement in personalized therapy for EOCRC. Integrating these measures into national screening and treatment protocols will improve early detection, enable precision oncology approaches, and ultimately enhance outcomes for younger CRC patients in Serbia.
Footnotes
Acknowledgment
Artificial intelligence (ChatGPT, OpenAI) was used to assist with language editing and improvement of clarity and grammar. The authors reviewed and approved all content and take full responsibility for the accuracy and integrity of the manuscript.
Ethical Considerations
The study was conducted in accordance with the principles of the Declaration of Helsinki of 1975, as revised in 2024, and was approved by the Ethics Committee of the University Clinical Centre of Serbia (Ref. No.: 175/1; Date: April 27, 2021) and by the Ethics Committee of the Institute for Oncology and Radiology of Serbia (Approval No.: 01-1/2023/701; Date: 30 March 30, 2023).
Consent to Participate
Written informed consent was obtained from all patients prior to surgical intervention.
Author Contributions
Jovana Despotović – Conceptualization, investigation, methodology, visualization, writing – original draft, writing – review and editing.
Neda Nikolić - Data curation, investigation, methodology, resources, writing – review and editing.
Tamara Babić – Investigation, methodology, writing – review and editing.
Milena Ugrin - Investigation, methodology, writing – review and editing.
Aleksandra Đikić Rom - Data curation, investigation, resources, writing – review and editing.
Ana Damjanović - Investigation, methodology, writing – review and editing.
Aleksandra Nikolić - Funding acquisition, resources, writing – review and editing.
Sandra Dragičević – Conceptualization, formal analysis, investigation, supervision, writing – original draft, writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the strategic project of the Serbian Academy of Sciences and Arts (grant agreement No 02-2019) Molecular basis of response to chemioradiotherapy in rectal cancer MOHERATEKA, F-69, and by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (grant agreement No 451-03-136/2025-03/200042, 451-03-136/2025-03/200043 and 451-03-137/2025-03-200110).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
