Abstract
Gallbladder cancer (GBC) is a rare yet highly aggressive malignancy of the biliary tract, characterized by a five-year survival rate of less than 5%. Its asymptomatic onset and the lack of reliable early diagnostic tools contribute to delayed detection and poor clinical outcomes. Although epidemiological and genetic studies have identified numerous risk factors, the molecular mechanisms linking these factors to tumor initiation and progression remain incompletely understood. This review integrates current evidence on the multifactorial etiology of GBC—including geographic variation, genetic predisposition, environmental exposures, chronic inflammation, and infections—with emerging insights into metabolic and molecular dysregulation. Particular focus is placed on metabolic reprogramming as a central driver of carcinogenesis. Altered lipid metabolism, bile acid signaling, and redox imbalance interact with inflammatory and oncogenic pathways, fostering a permissive microenvironment for malignant transformation. Key molecular cascades include inflammation-driven NF-κB activation, bile acid–induced oxidative stress, PI3K/AKT-mediated metabolic remodeling, and DNA damage and repair defects. By consolidating diverse epidemiological and mechanistic data into a unified molecular–metabolic framework, this narrative review identifies new opportunities for biomarker discovery, metabolic imaging, early detection, and targeted therapeutic development, advancing translational research to improve outcomes in this devastating disease.
Plain Language Summary
Gallbladder cancer (GBC) is a rare but highly aggressive cancer that is often diagnosed too late for effective treatment. Many factors can increase the risk of developing GBC, including gallstones, chronic infections, lifestyle habits, and inherited genetic changes. This review explains how these different risk factors may work together to trigger cancer through changes in cell metabolism and molecular pathways. We describe how long-term inflammation, bile and lipid imbalance, and exposure to infections or toxins can damage cells and alter key biological processes such as energy use, signaling, and DNA repair. These disruptions can lead to uncontrolled growth and tumor development. By combining evidence from population studies and molecular research, our review highlights possible links between risk factors and metabolic changes that drive cancer. Understanding these connections may help identify early warning biomarkers and develop targeted treatments for GBC in the future.
Keywords
1. Introduction
Gallbladder cancer (GBC) is an aggressive malignancy associated with a dismal prognosis, frequently diagnosed at advanced stages with scarce therapeutic options. According to Globocan 2022 (https://gco.iarc.fr/), GBC ranks as the 22nd most commonly occurring cancer worldwide, with 122,491 new cases and 89,055 deaths reported in that year alone. 1 Its global five-year survival rate remains under 5%,1-3 reflecting both the asymptomatic nature of early disease and the lack of effective therapeutic strategies. 4
Nearly 90% of GBC cases are detected at stage III or beyond,5,6 due to the absence of early symptoms and limited sensitivity of non-invasive diagnostic tools.7-12 Additionally, GBC is largely resistant to chemotherapy and radiotherapy, amplifying the clinical adversity.2,3,5,6
Globally GBC incidence shows distinctive geographic and ethnic variations. It is most prevalent in parts of Asia, South and Central America, and Eastern Europe, with the highest incidence rates reported in China, India, Japan, and Bangladesh.1,2,13,14 While historically rare in developed nations, recent data indicate rising incidence in countries like the United States, highlighting an emerging global health concern.15-18
The etiology of GBC is multifactorial and complex. Risk factors span biological (gender, age), medical (gallstones, polyps, obesity, 19 diabetes, typhoid, and Helicobacter pylori infections2,20), and lifestyle aspects including dietary patterns namely, high consumption of mustard and palm oils or refined carbohydrate-rich substances.21,22 This interplay of geographic, genetic, lifestyle, and medical factors underscores the need for an integrative understanding of GBC pathogenesis.2,23-25 Such insights will be critical for the development of targeted prevention strategies, early detection biomarkers, and more effective therapies.
To support this integrative framework, a comprehensive literature search was conducted using PubMed as the primary database, with supplementary searches in Scopus and Web of Science to ensure broad coverage of relevant studies published between January 2000 and January 2025. Search terms included combinations of “gallbladder cancer,” “risk factors,” “genetics,” “metabolic reprogramming,” “bile acids,” “inflammation,” “molecular mechanisms,” “epidemiology,” “genome-wide association studies,” “single nucleotide polymorphisms,” “genetic alterations,” “tumor microenvironment,” and “biomarkers.” Abstracts were manually screened for relevance, and full-text articles were evaluated based on scientific rigor, relevance to molecular mechanisms, and contribution to the integrative framework of this review; additional studies were identified through citation tracking. Given the hypothesis-driven nature of this review, selected studies were organized thematically to synthesize current evidence and identify emerging molecular patterns and research gaps.
In this narrative review, following the Scale for the Assessment of Narrative Review Articles (SANRA), 26 we first present an overview of GBC epidemiology and established risk factors. We then explore environmental contributors—such as gallstones, bile composition, infections, diet, and chemical exposures—and their mechanistic links to GBC development. Finally, we examine genetic susceptibility, including findings from genome-wide association studies (GWAS) and candidate gene-based analyses, and discuss how these insights are shaping future directions in GBC research and clinical management.
2. Epidemiology
Global incidence and mortality rates of GBC have shown a concerning upward trend. According to GLOBOCAN 2022, there were 122,463 new cases of GBC, ranking it 22nd in global cancer incidence. Mortality figures are similarly alarming, with 89,031 deaths placing GBC 20th among cancer-related fatalities. 1 Since GLOBOCAN 2020, incidence has risen by 5.6%, while mortality has increased by 5.1%, reflecting a persistent global burden. This aligns with findings by Ouyang et al. (2021), which reported a 76% rise in gallbladder and biliary tract cancer incidence from 1990 to 2017. 27
Regionally, Asia reports the highest annual incidence rates, likely influenced by both its large population and widespread risk factors. Other high-risk regions include South-Central Asia, Eastern Asia, North Africa, South America, and Oceania (Figure 1).1,
28
Recent trends also indicate an increase in GBC incidence in developed countries, particularly the United States.2,13,15 Notably, most of these U.S. cases occur among Native American and Alaskan Native populations.2,13,29,30 For example, in New Mexico, the majority of GBC cases are reported in Native Americans, followed by Hispanic and Caucasian individuals.
30
Age-standardized incidence rates of gallbladder cancer worldwide in 2022.
GBC incidence also displays striking sex- and age-related disparities. Women are disproportionately affected, with female-to-male incidence ratios ranging from 2:1 to 5:1 in many high-incidence regions. 31 The risk increases significantly with age, particularly after the sixth decade of life, with the majority of diagnoses occurring in individuals over 60 years old.
Compounding the epidemiologic burden is the fact that GBC is often diagnosed at a late stage. More than 80–90% of cases are detected at stage III or beyond,5,6 primarily due to the asymptomatic nature of early disease and the limited sensitivity of current diagnostic tools.7-9,11,12 This contributes to the persistently poor prognosis, with a global five-year survival rate remaining below 5%.1-3
Similar patterns are observed in Chile, where a 1% increase in the proportion of the Mapuche population (an Indigenous group) is associated with a 3.7% increase in GBC risk. 32 These trends suggest a possible genetic component, further supported by studies estimating that approximately 25% of GBC cases may be familial in origin.33,34
These geographical, ethnic, age, and sex disparities underscore the need for a deeper understanding of GBC’s risk factors and etiology. Identifying vulnerable populations through epidemiological insights is critical for implementing targeted prevention and early detection strategies.
3. Risk Factors
An intricate web of environmental and genetic factors contribute to the development of GBC. While the precise mechanisms through which these factors exert their effects are still under investigation, multiple studies suggest they have a significant impact on the onset and progression of GBC. Moreover, genetic predisposition has been documented in certain populations, with several susceptibility loci identified to date. This section outlines the major environmental and genetic risk factors, highlighting how deeper understanding can illuminate the molecular pathways involved in GBC pathogenesis.
3.1. Environmental Risk Factors
Gallstones are the most well-established risk factor for GBC. Other contributing factors include a sedentary lifestyle, high-fat diet, and chronic infections of bacterial, fungal, or parasitic origin. Key environmental influences on GBC development and progression are detailed below:
3.1.1. Gallstones
Gallstones represent the most prominent and consistently reported risk factor for GBC. They can induce persistent inflammation and mechanical irritation of the gallbladder mucosa, which in turn promotes epithelial injury, oxidative stress, and tumorigenesis.31,35,36 GBC risk increases with the duration of gallstone presence. 37 Hsing et al. and Usha et al. through their study showed that 80% of GBC cases in Shanghai and India, respectively, are attributable to gallstones.37,38
Gallstone-associated GBC risk varies significantly by ethnicity and gender. Among individuals with gallstones, age-adjusted GBC risk was found to be 16.2 and 46.4 in Native American men and women, respectively; 2.3 and 3.0 in African Americans; and 4.5 and 11.5 in Swedish Whites.39,40
In a U.S.-based study, gallstones were shown to increase GBC risk across all racial groups, including white, black, and American Indian populations. Nevertheless, the risk was significantly higher among American Indians, with a relative risk of 20.9 compared to 4.4 for non-Indian groups. The cumulative risk over 20 years was calculated at 0.13% for black males and 1.5% for American Indian females. 41
Furthermore, research indicates that cholesterol stones have become more prevalent than pigmented stones in developed countries, with an occurrence rate that is 85% higher. Prevalence is intermediate in Asian and Black American populations (13.9% in women and 5.3% in men), while it remains lowest (<5%) in sub-Saharan Black Africans. 42 Given that 90–95% of GBC patients have a history of gallstones, 43 rising gallstone prevalence may help explain the increased GBC incidence observed between 1999 and 2019. 17
The progression from gallstones to GBC involves a cascade of molecular and pathological changes (Figure 2). As previously noted, gallstones can cause frequent inflammation and irritation of the gallbladder lining, and this chronic inflammation is a significant driver of cancer development.
31
Key pathways implicated in this process include the nuclear factor kappa B (NF-κB) and COX-2/PGE2 pathways (Figure 3). NF-κB is activated by inflammatory signals and promotes the production of cytokines such as IL-1, IL-6, and TNF-α, which foster an inflammatory environment conducive to cancer.44,45 Additionally, Jiang et al. have identified the NF-κB pathway’s role in gallbladder cancer invasion and lymphangiogenesis.
46
Meanwhile, Cyclooxygenase-2 (COX-2) and prostaglandin E2 (PGE2) contribute to inflammation and cancer progression by promoting cell proliferation and inhibiting apoptosis.
47
Schematic representation of gallstone-induced molecular events in GBC progression. Altered bile acid composition promotes gallbladder cancer progression and metastasis.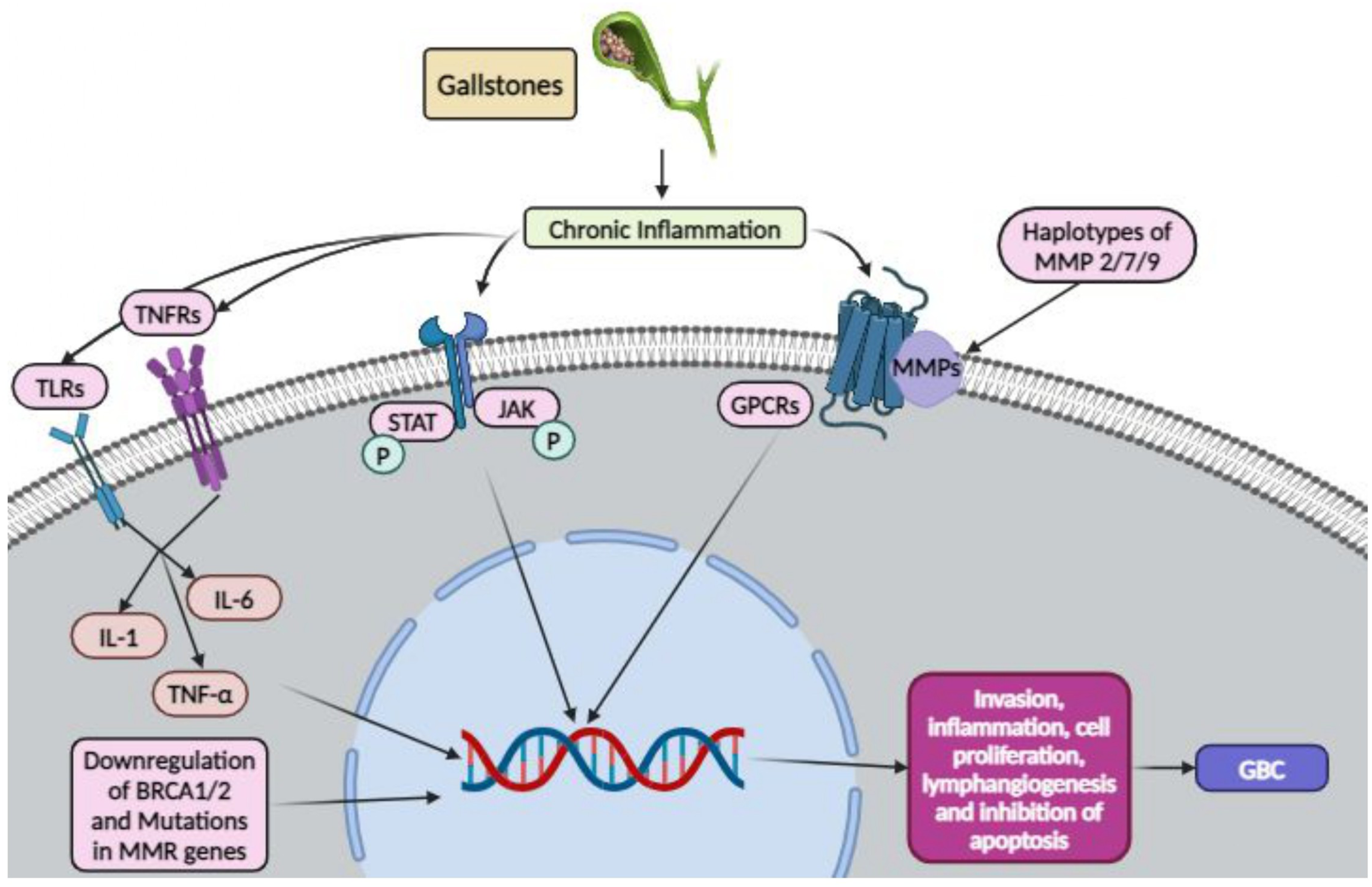
Beyond inflammation, gallstones can also inflict mechanical damage on the gallbladder epithelium, resulting in heightened cell turnover, possible DNA damage and oxidative stress 48 and the activation of DNA damage pathways. Several genes associated with DNA repair are found to be mutated in GBC patients. 49 Deficiencies in DNA repair mechanisms, such as those involving BRCA1/2 or mismatch repair genes, can further contribute to genomic instability. 50 The altered inflammatory microenvironment also promotes extracellular matrix degradation through matrix metalloproteinases (MMPs), particularly MMP-2, -7, and -9. Haplotypes of these MMP genes have been associated with increased GBC risk in gallstone patients.51,52
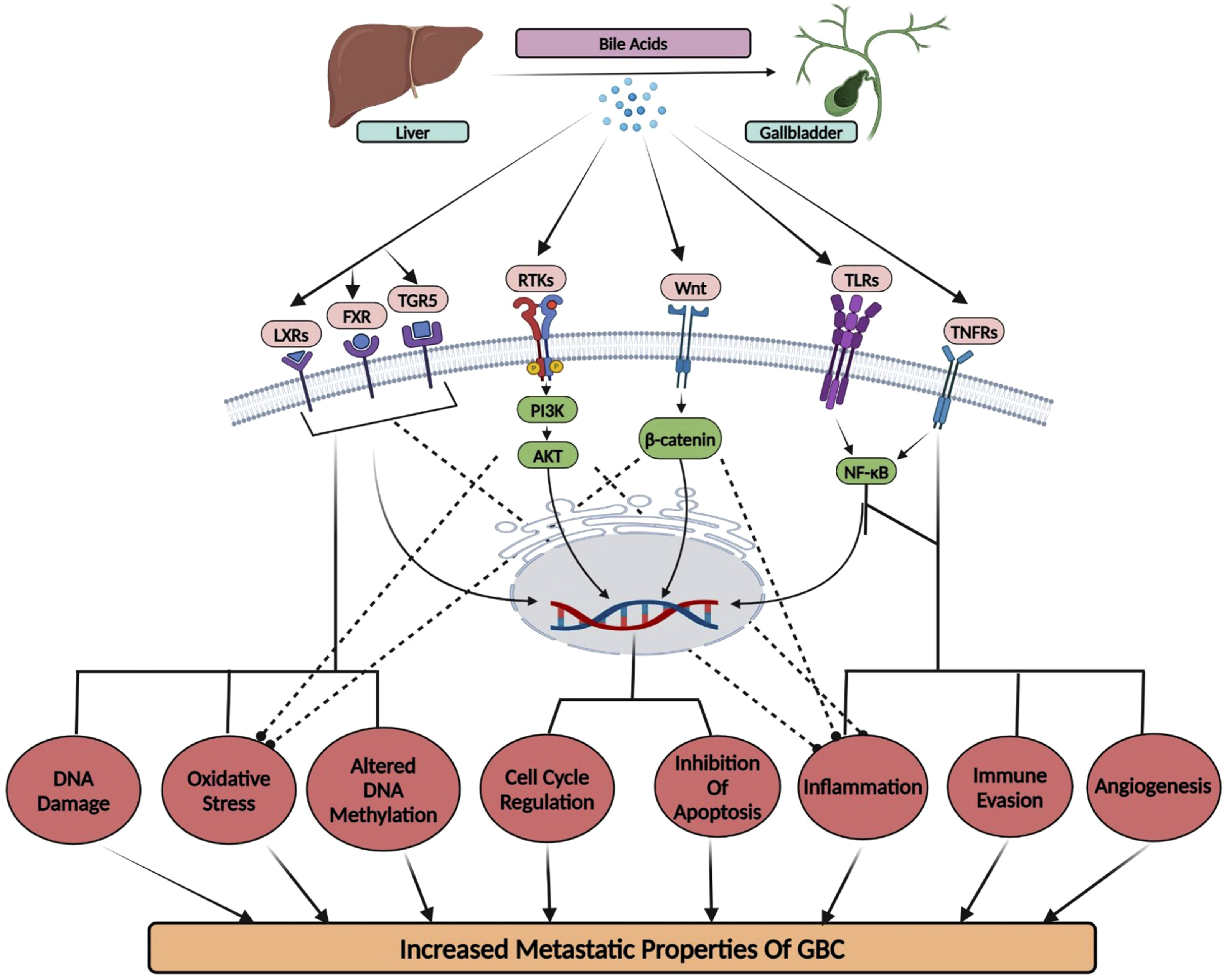
3.1.2. Bile Composition
Bile, a yellow-green fluid produced by the liver and stored in the gallbladder, plays a critical role in lipid digestion. It is composed of bile salts, phospholipids, cholesterol, conjugated bilirubin, electrolytes, and water. In humans, typical bile composition includes approximately 200 mEq/L of inorganic salts, 0.7% bile salts, 0.2% bilirubin, 0.51% fats (fatty acids, cholesterol, lecithin), and 97–98% water. The liver synthesizes primary bile acids such as cholic acid and chenodeoxycholic acid, which are then converted by intestinal microbiota into secondary bile acids. 53
Alterations in bile composition have been noted in malignancies of the biliary system. These changes may either contribute to carcinogenesis or result from tumorigenic processes. Numerous studies have documented increase in secondary bile acids, such as glycodeoxycholic acid, taurodeoxycholic acid, glycoursodeoxycholic acid, and tauroursodeoxycholic acid, in patients with GBC.53-56 In contrast, some research suggests that individuals with gallstones alone exhibit a greater rise in primary bile acids compared to those with GBC.53-56
Despite these observations, data on individual bile acid profiles in GBC patients remain limited, often due to small sample sizes. For instance, Park et al. reported significantly lower concentrations of deoxycholic acid (a secondary bile acid) and total bile acids in GBC patients, yet their study included only six GBC patients. 53 Consequently, it remains uncertain whether the rise in secondary bile acids are a cause or consequence of GBC, and whether these shifts reflect enhanced bacterial conversion of primary bile acids or diminished hepatic synthesis.
At the molecular level, sustained exposure to irregular concentrations of specific bile acids may trigger oncogenic pathways (Figure 3). This occurs through their interaction with nuclear receptors such as the Farnesoid X receptor (FXR) and the G-protein-coupled bile acid receptor (TGR5). Altered FXR signaling can disrupt cell cycle regulation and apoptosis, facilitating cancer development. These interactions may also lead to oxidative stress, DNA damage, and epigenetic alterations, including abnormal DNA methylation and histone modifications. 57 Conversely, TGR5 receptors, activated particularly by lithogenic diets and deoxycholic acid, have been shown to increase gallbladder volume and reduce biliary motility. Nonetheless, the exact role of TGR5 in GBC remains unclear. 58
Disruptions in bile composition can also significantly affect signaling pathways such as Wnt/β-catenin, which is essential for cell proliferation and differentiation, thus promoting cancer growth. 59 Additionally, alterations in bile composition particularly ursodeoxycholic acid can influence the PI3K/Akt pathway, which is involved in cell survival and metabolism.60-62 Abnormal activation of this pathway due to changes in bile composition may encourage cancer cell proliferation and resistance to apoptosis. Moreover, an abnormal bile composition can instigate inflammatory responses and oxidative stress, resulting in cellular damage that promotes carcinogenesis.63,64 Chronic inflammation driven by altered bile composition can activate the NF-κB pathway, an important regulator of inflammation and cell survival, and lead to the release of inflammatory cytokines such as IL-6 and TNF-alpha.65-67 Furthermore, increased oxidative stress due to toxic bile acid accumulation generates reactive oxygen species (ROS), resulting in DNA damage and genomic instability. 68 This pro-inflammatory, oxidative microenvironment promotes tumor initiation, progression, angiogenesis, and immune evasion.
In summary, prolonged exposure to altered bile composition can lead to metaplasia and dysplasia of the gallbladder epithelium, increasing the risk of malignant transformation. A deeper understanding of the complex interplay between bile acids, inflammation, oxidative stress, and signaling dysregulation is essential for identifying novel therapeutic targets and preventive strategies for gallbladder cancer.
3.1.3. Abnormal Lipid Profile
Emerging evidence suggests that altered lipid metabolism contributes to tumor progression in various cancers, including GBC. Tumor cells often reprogram lipid metabolic pathways, leading to the accumulation of specific lipids and their derivatives within the tumor microenvironment. These changes can enhance cancer cell migration, invasion, and resistance to apoptosis.
A study conducted by Sun et al., showed higher levels of total cholesterol (TC), high-density lipoprotein (HDL), apolipoprotein B (ApoB), and lipoproteins were associated with poor outcomes in patients with malignant GBC and biliary tract tumors. 69 Population-based studies from Shanghai have reported strong associations between dysregulated lipid profiles and gallbladder cancer risk, with elevated triglyceride levels associated with approximately two-fold increased risk and markedly reduced HDL levels linked to over ten-fold higher susceptibility. These findings suggest that lipid metabolic imbalance may represent an important population-translatable risk mechanism warranting further investigation. Both low and high concentrations of LDL, ApoA, ApoB, and total cholesterol were also associated with increased risk of GBC and other biliary tract cancers 70 .
Metabolic conditions often associated with dyslipidemia—including obesity, insulin resistance, and metabolic syndrome—have also been linked to an elevated risk of GBC.
At the molecular level, abnormal lipid profiles marked by elevated cholesterol, triglycerides, and sphingolipids (e.g., ceramide and sphingosine-1-phosphate) can influence several key signaling pathways, such as PI3K/Akt and mTOR, which regulate cell proliferation, metabolism, and survival (Figure 4). Disruption of cholesterol balance can alter lipid rafts—cholesterol-rich microdomains within the cell membrane—affecting the function of receptors and signaling proteins involved in oncogenesis. Schematic representation of the potential link between altered lipid profiles and gallbladder cancer (GBC) development.
Additionally, a high intake of saturated fats, combined with low consumption of beneficial unsaturated fats, contributes to chronic inflammation, a hallmark of cancer.71,72 This pro-inflammatory state activates pathways such as NF-κB, which promote cell proliferation and survival. 73 Furthermore, elevated lipid levels—particularly oxidized low-density lipoproteins (oxLDL)—can generate reactive oxygen species (ROS), leading to oxidative stress and DNA damage, thereby fostering genomic instability and carcinogenesis. 74
In summary, abnormal lipid metabolism contributes to GBC development through its effects on cellular signaling, inflammation, oxidative stress, and potentially epigenetic regulation. A better understanding of these interrelated processes could lead to novel preventive and therapeutic strategies.
3.1.4. Porcelain Gallbladder
A considerably higher risk of GBC has been associated with porcelain gallbladder, a condition in which the gallbladder becomes calcified, frequently as a result of long-standing gallstones. 74 This association is thought to be largely explained by the chronic inflammatory response in the lining of the gallbladder. 75 However, the risk of GBC due to porcelain gallbladder alone is considerably small (∼6%) and is more likely attributable to the gallstones and chronic inflammation that have approximately 95 percent association with porcelain gallbladder.75,76 Two main molecular pathways have been linked to GBC in patients with porcelain gallbladder, making their understanding vital for future research and therapeutic strategies. 77
The first pathway involves alterations in the TP53 gene, which is essential for cell cycle regulation. 78 The second significant pathway is chronic inflammation, which contributes to DNA damage and induces changes in cellular environments that may promote cancer development. Ongoing irritation from gallstones can result in dysplasia, creating a cancer-prone environment. This persistent inflammation can also lead to genetic and epigenetic changes that facilitate gallbladder cancer progression.77,79 In conclusion, the molecular mechanisms affected by porcelain gallbladder in gallbladder cancer primarily involve TP53 gene alterations and chronic inflammatory processes. These insights underscore the relationship between porcelain gallbladder and gallbladder cancer, indicating a need for further research to identify precise interactions and potential therapeutic targets within these pathways.
3.1.5. Long-Term Inflammation Due to Diseases
Long-term inflammation of the gallbladder and biliary tract—particularly due to conditions such as primary sclerosing cholangitis (PSC) and chronic cholecystitis—is a well-established risk factor for GBC. 80 Persistent inflammation in these settings exerts pathological effects that promote carcinogenesis through both genetic and microenvironmental changes. Chronic inflammatory conditions increase the risk of mutations in tumor suppressor genes such as TP53, which is critical for regulating DNA repair, cell cycle arrest, and apoptosis.81,82 Inflammatory signaling also leads to repeated cycles of epithelial injury and regeneration, promoting excessive cell proliferation, DNA damage, and the accumulation of mutations in key oncogenes, including K-ras.80,81 Inflammation not only affects intracellular signaling but also remodels the tumor microenvironment. The release of pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6), plays a pivotal role in sustaining chronic inflammation and facilitating a tumor-supportive niche.81,82 These cytokines collectively contribute to increased angiogenesis—providing essential nutrients and oxygen to the tumor—enhanced tumor cell survival, immune evasion, and activation of transcription factors like NF-κB, which perpetuate a self-sustaining inflammatory, pro-carcinogenic loop. One well-characterized pathway linking chronic inflammation to malignancy in GBC is the metaplasia–dysplasia–carcinoma sequence. Chronic irritation of the gallbladder epithelium leads to metaplastic changes, which progress to dysplasia and eventually to invasive carcinoma. This sequence is commonly observed in gallbladder specimens from patients with a history of chronic cholecystitis or gallstones and understanding these mechanisms is essential for improving diagnostic and therapeutic strategies against GBC.
3.1.6. Diet and Lifestyle
Gallstones, obesity, and a sedentary lifestyle have all been linked to GBC. Studies on a variety of dietary practices and components have been carried out to clarify the roles of various food components in GBC, as both obesity and gallstones can be influenced by an individual’s diet and lifestyle (Figure 5). Impact of lifestyle and dietary habits on gallbladder cancer risk: a molecular perspective.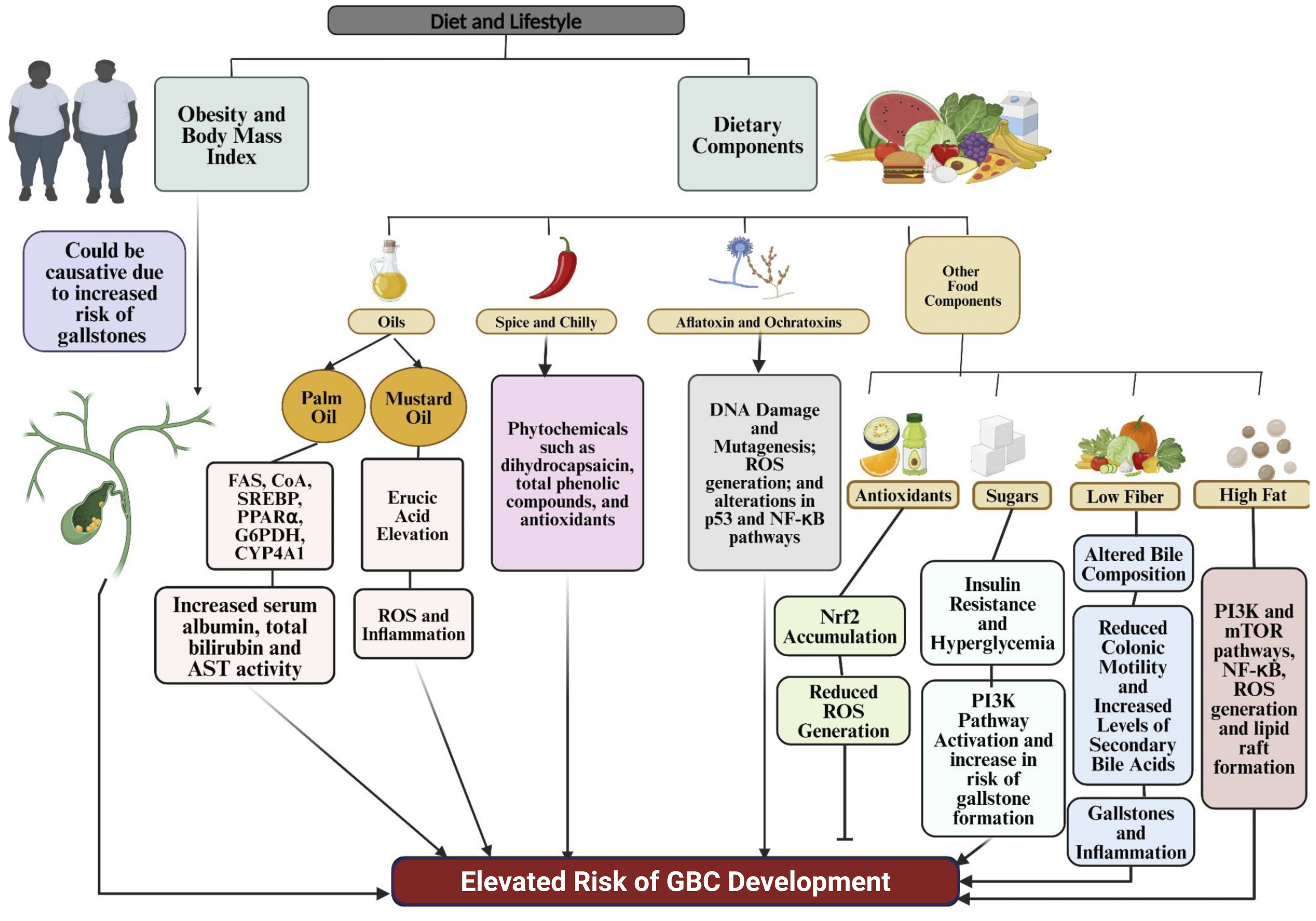
3.1.6.1 Obesity and Body-mass Index
Excess body weight and obesity have been global health concerns leading to several disorders. Between 1980 and 2013, global obesity rates increased by 27.5%, with notable rises in Asia, a region with high GBC incidence. Given that the highest number of GBC incidences are also reported from this region and the fact that obesity has been attributed to the risk of gallstone formation, which in turn is a risk factor for GBC, researchers began looking into the correlation between the two.83,84
Population-based studies have reported that overweight individuals exhibit modest increases in gallbladder cancer risk, while obesity is associated with markedly higher susceptibility, with risk elevations approaching 50–60% in some cohorts. Meta-analyses further demonstrate a dose-dependent increase in GBC risk with rising BMI. Despite these strong epidemiological associations, the molecular mechanisms underlying obesity-driven gallbladder carcinogenesis remain to be fully elucidated. 85 Although these findings established a link between the two epidemiologically, the underlying molecular mechanisms remain to be fully understood.
3.1.6.2 Dietary factors
Dietary components influence GBC risk through effects on obesity, inflammation, and cellular damage. Key elements include: Chili peppers contain several phytochemicals, such as dihydrocapsaicin, total phenolic compounds, and antioxidants, with capsaicin (trans-8-methyl-vanillyl-6-nonenamide) being the most potent active component in spicy foods like kimchi, chili, and pepper.22,93,94 Experimental studies have produced mixed findings regarding their carcinogenic potential. While some animal models suggest that high dietary chili exposure may induce hepatic and biliary neoplastic changes,
95
other long-term feeding studies report no significant carcinogenic effects.
96
These inconsistencies highlight the complexity of dose-dependent, context-specific biological responses and underscore the need for well-controlled mechanistic studies to clarify whether chili-derived compounds contribute to gallbladder carcinogenesis and through which molecular pathways. Ochratoxin A, another mycotoxin potentially linked to gallbladder cancer, is produced by Aspergillus and Penicillium species. It can be found in a variety of foods such as cocoa, coffee, beer, cereals, spices, and nuts.
102
Research has indicated that ochratoxin A levels in dried red chili peppers from Chile, Bolivia, and Peru are significantly higher than aflatoxin levels in the same samples. These findings suggest that there may be a stronger link between ochratoxin A contamination in red chili peppers and the onset of gallbladder cancer, compared to the effects of aflatoxin exposure.99-101 Aflatoxins cause DNA damage and mutations by binding to cellular macromolecules, which activates carcinogenic pathways. This occurs through the generation of reactive oxygen species (ROS) and the alteration of signaling pathways like p53 and NF-κB. As a result, there is enhanced cellular proliferation and decreased apoptosis, which facilitates cancer development.
99
In addition to dietary risks, emerging research suggests a link between oxidative stress regulation and chemoresistance in GBC. A 2019 study demonstrated that the protein aPKCι competes with Nrf2 for binding to Keap1, resulting in Nrf2 accumulation in the nucleus and reduced intracellular ROS levels.
117
This shift in redox balance may enable cancer cells to resist apoptosis and survive chemotherapy. Although antioxidant supplementation has shown benefits in other cancers, such as bladder and colorectal, direct evidence for its role in GBC is lacking.
118
Nevertheless, targeting redox pathways may offer a novel therapeutic strategy for overcoming drug resistance in GBC. Collectively, these findings highlight the complex interplay between diet, metabolism, inflammation, and oxidative stress in GBC development and treatment, offering potential avenues for prevention and personalized therapy.
3.1.7. Chemicals and Heavy Metals Exposure
Chemicals and Heavy Metals Linked to Gallbladder Cancer Risk and Their Associated Molecular Pathways

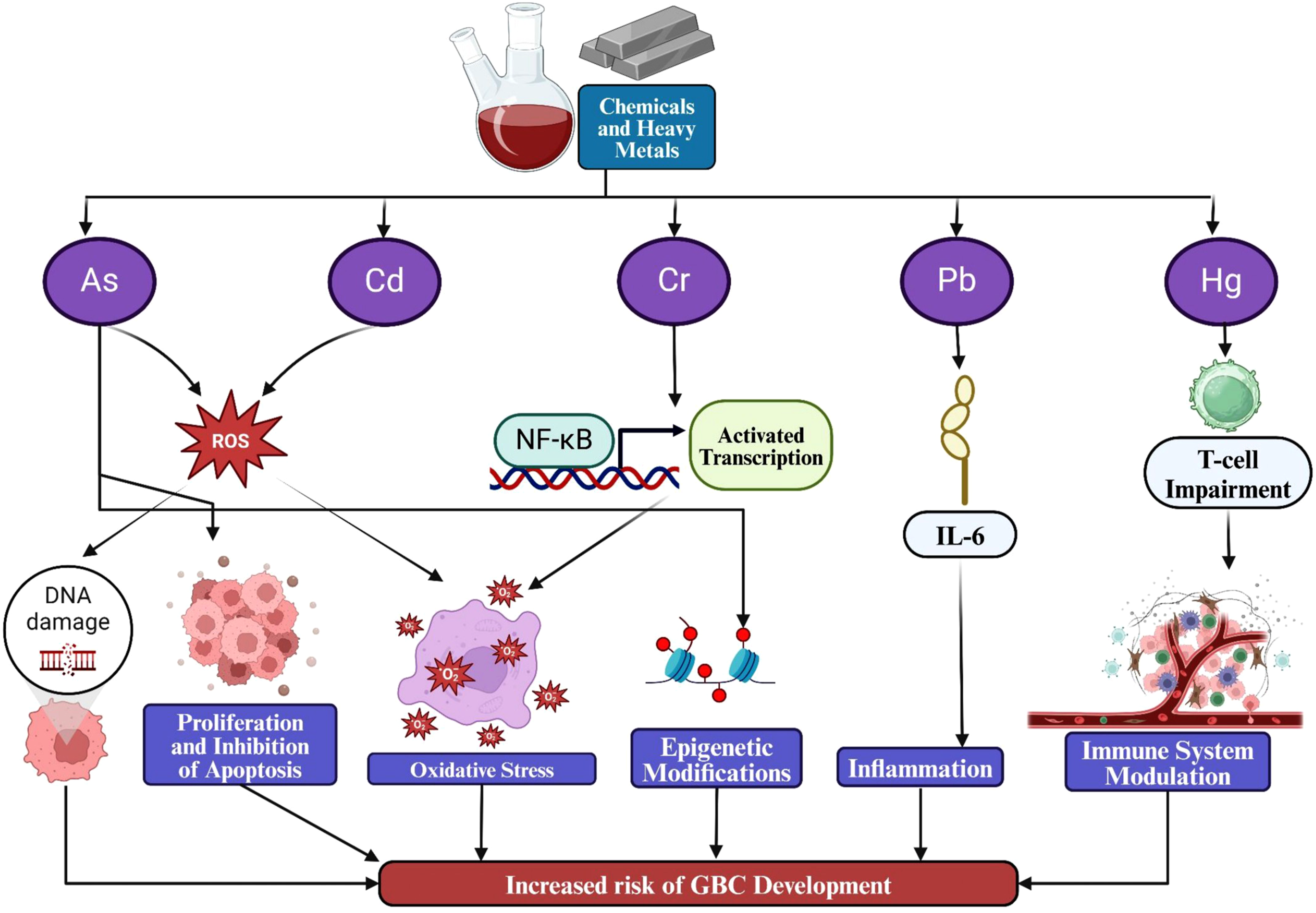
Impact of metal exposure on gallbladder cancer development.
3.1.8. Infections
Chronic infections with Salmonella typhi and Helicobacter species have been explored as potential risk factors for GBC. These infections might contribute to GBC development either by directly affecting gallbladder cells or by causing ongoing inflammation.
3.1.8.1. Chronic Salmonella Typhi Infection
Chronic infection with Salmonella Typhi—the causative agent of typhoid fever—is a recognized risk factor for GBC. This bacterium can persist in the gallbladder, especially in individuals with gallstones, leading to long-standing inflammation.
145
Several studies, including those by Shukla et al. and Tewari et al., have reported S. typhi detection rates of 22–33% in GBC patients.146,147 Molecularly, S. typhi promotes carcinogenesis by inducing immune responses involving cytokines like IL-6 and TNF-α, which contribute to a chronic inflammatory environment. This inflammation produces reactive oxygen species (ROS), causing oxidative stress and DNA damage that can drive mutations.148,149 Moreover, S. typhi activates cell survival and inflammation-related pathways, such as NF-κB and MAPK, which enhance cell proliferation, inhibit apoptosis, and foster tumor development.150-152 The bacteria may also induce epigenetic changes and impair immune surveillance, further enabling malignant transformation. Thus, S. typhi contributes to GBC through inflammation, oxidative stress, and disruption of key cellular pathways (Figure 7). Salmonella Typhi, Helicobacter pylori and Liver fluke infection as a microbial trigger in gallbladder carcinogenesis.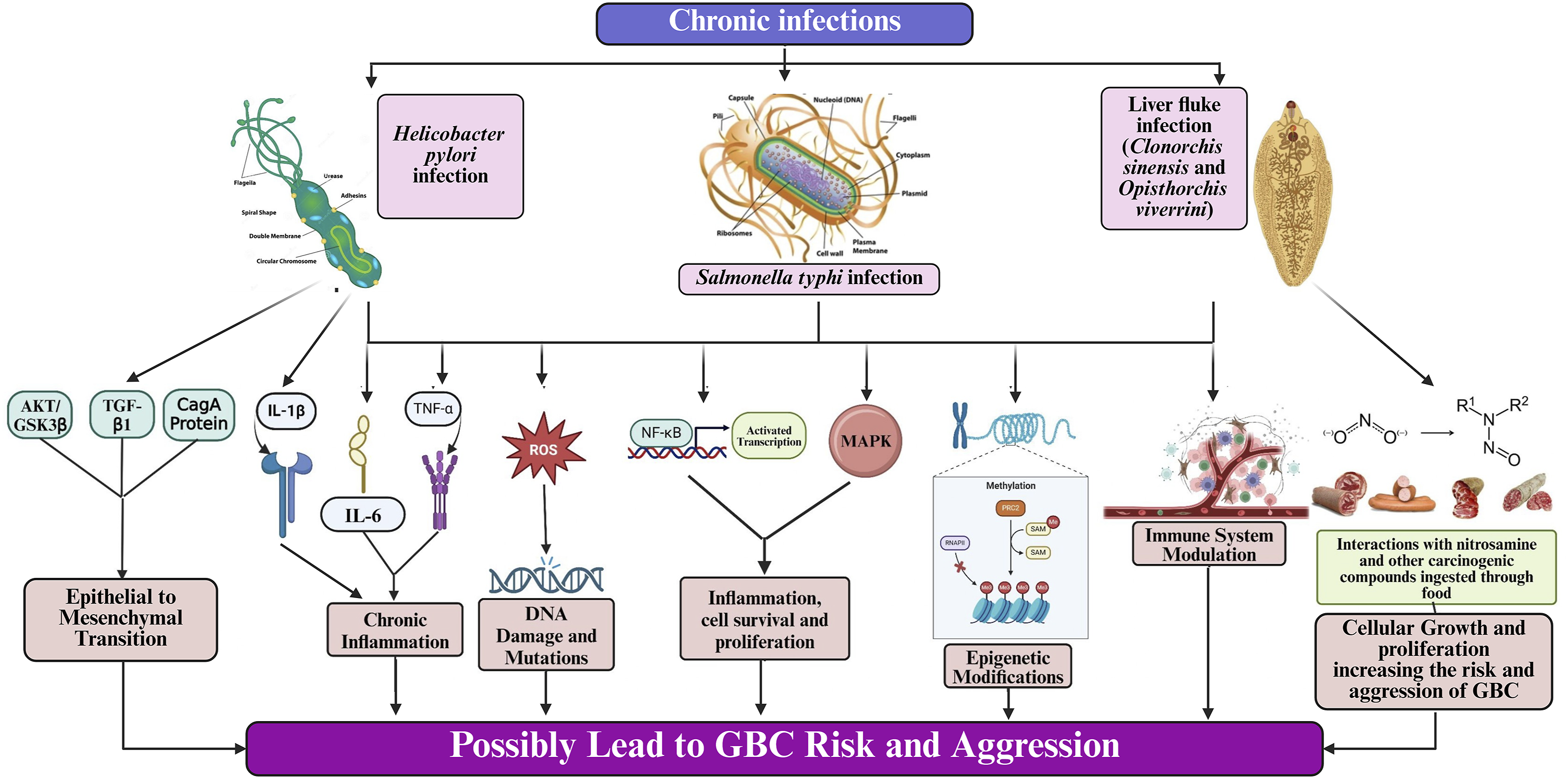
3.1.8.2. Helicobacter Species
Helicobacter pylori (H. pylori) is a gram-negative, spiral-shaped, microaerophilic bacterium that colonizes the gastrointestinal tract and infects over half of the global population. While it is best known for its role in gastric disorders, accumulating evidence suggests a potential link between H. pylori infection and GBC. Studies have associated it with cholelithiasis, chronic cholecystitis, biliary tract cancers, and autoimmune cholangiopathies such as primary sclerosing and biliary cholangitis. 45 A case-control study by Hassan et al. demonstrated a significant increase in mucosal hyperplasia (p = 0.028), metaplasia, and dysplasia (p = 0.049) in gallbladders infected with H. pylori, suggesting a role in epithelial transformation. 153
At the molecular level, H. pylori contributes to carcinogenesis primarily through chronic inflammation. It stimulates the release of pro-inflammatory cytokines like IL-1β and TNF-α, creating a microenvironment conducive to cell proliferation and mutagenesis.45,154,155 The infection also generates reactive oxygen species (ROS), which induce oxidative damage to DNA, lipids, and proteins, increasing mutation risk.154,156 Additionally, H. pylori activates key signaling pathways such as NF-κB and MAPK, disrupting the regulation of inflammation, cell survival, and apoptosis. Emerging evidence also implicates oncogenic mechanisms observed in gastric cancer, including activation of AKT/GSK3β and TGF-β pathways, and the CagA-mediated promotion of epithelial–mesenchymal transition (EMT) via TWIST and vimentin upregulation and E-cadherin downregulation.155,157
Overall, H. pylori may contribute to GBC by promoting chronic inflammation, oxidative stress, and signaling dysregulation (Figure 7). Further investigation is warranted to clarify its role and identify potential preventive and therapeutic targets in GBC pathogenesis.
3.1.8.3. Liver Fluke Infections
Chronic infection with liver flukes such as Opisthorchis viverrini, Clonorchis sinensis, and Schistosoma japonicum is a well-recognized risk factor for GBC, especially in endemic regions of Southeast Asia. These parasites induce chronic inflammation by attaching to the bile duct wall, causing repeated mechanical injury, ulceration, and subsequent healing. For instance, the larger size of C. sinensis can lead to partial obstruction, bile stasis, and increased intrabiliary pressure, thereby exacerbating inflammation and epithelial damage. Over time, these cycles contribute to DNA damage and neoplastic transformation. 158
Persistent liver fluke infection promotes a tumor-supportive microenvironment through sustained immune activation and the release of inflammatory cytokines, such as IL-6 and TNF-α.159,160 These factors enhance epithelial proliferation and increase the risk of mutations. Concurrent oxidative stress further contributes to genomic instability and epigenetic modifications, potentially disrupting genes involved in DNA repair, apoptosis, and cell cycle control.161,162 Although much of this mechanistic insight is derived from studies on cholangiocarcinoma, the pathways are likely relevant to GBC as well.163,164
Moreover, liver fluke infections can impair immune surveillance by altering immune signaling in the biliary epithelium, facilitating immune evasion by emerging malignant cells. The oncogenic potential may be amplified in regions where parasite exposure coincides with nitrosamine-rich diets, suggesting synergistic effects between infection and environmental carcinogens.163,164
Altogether, liver fluke infections contribute to GBC pathogenesis through chronic inflammation, oxidative DNA damage, and immune modulation (Figure 7). Understanding these pathways is crucial for developing region-specific prevention and treatment strategies.
3.2. Genetic Susceptibility Studies for Gallbladder Cancer
Apart from the environmental risk factors, genetic risk factors play a crucial role in altering the risk for cancer by influencing susceptibility to the disease through inherited or acquired genetic mutations, interactions with environmental factors, and the cumulative effects of multiple genetic variants. Understanding these genetic factors is essential for developing targeted prevention, screening, and treatment strategies for cancer.165,166
3.2.1. Familial Risk and Heritability
A family history of cancer is associated with an increased risk of certain cancers, including GBC. Hereditary syndromes contribute to this susceptibility. For instance, Peutz-Jeghers syndrome, characterized by mucocutaneous pigmentation and gastrointestinal polyps, has been linked to GBC, as seen in a reported case of a 39-year-old woman. 167 Studies report gallbladder polyps in 4.1% of individuals with Peutz-Jeghers syndrome, with a similar familial incidence of GBC noted in the Swedish population.33,168
Moreover, Lynch syndrome carriers face a higher lifetime risk of GBC. 169 Familial Adenomatous Polyposis (FAP), an autosomal dominant disorder caused by mutations in the APC gene on chromosome 5q21, is also associated with biliary tumors and gallbladder polyps.169,170
3.2.2. Genetic Association Studies
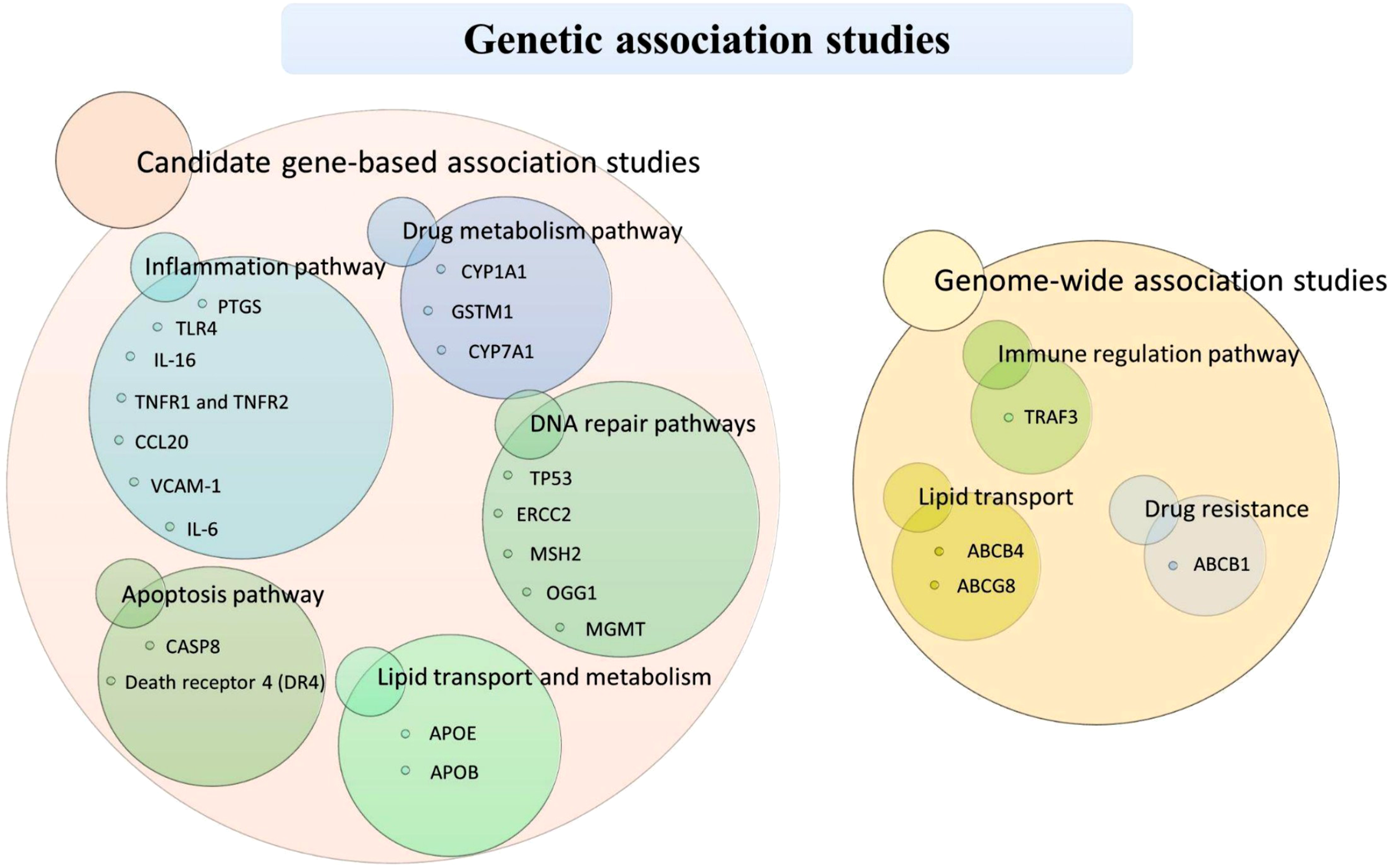
Genetic associations with GBC have been explored using two main approaches: candidate gene-based studies, which focus on preselected genes with known or suspected roles in disease, and genome-wide association studies (GWAS), which scan the entire genome to identify novel genetic variants linked to disease risk. The following sections detail these approaches in the context of GBC.
3.2.2.1. Candidate Gene-Based Association Studies
The candidate gene identification approach involves a systematic screening and evaluating effort to identify, prioritize, and validate genes that play a role in a particular biological process or disease phenotype. The analysis of candidate genes typically involves a case-control study, where allele frequencies in cancer patients and healthy controls are compared, and the resulting data are subjected to statistical analysis. Many of the identified candidate genes for GBC are associated with classical rate-limiting enzymes and proteins involved in lipid metabolism, steroidogenesis, lipid transport, bile acid synthesis, bile canalicular transport, gallbladder contractility, cell cycle regulation, DNA repair, and inflammatory pathways (Figure 8). Cytochrome P450 enzymes play pivotal roles in synthesizing steroid hormones, bile acids, certain fats, and metabolizing medications and toxins.171,172 Enzyme encoded by the CYP1A1 gene, is an integral part of xenobiotic metabolism, which involves processing exogenous compounds like drugs, tobacco, agricultural chemicals,
173
and carcinogenic intermediates.
174
Individuals with variation in CYP1A1 gene could activate or detoxify environmental carcinogens that can significantly impact susceptibility to GBC.175,176 This aspect has been studied in the North Indian population, revealing that genotypes CYP1A1-MspI [CC] (p = 0.006) and CYP1A1-Ile462Val (rs1048943)(p = 0.03) are significantly associated with GBC.
177
Also, rs4646903 CC genotype of CYP1A1 gene is associated with a 2.3-fold increase in GBC risk compared to TT genotype (95% CI: 1.1 – 4.5, P=0.026).
178
A candidate gene-based study conducted in a high-risk region of Bolivia examined genetic predisposition to GBC by analyzing key genes, including CYP1A1 (cytochrome P450), GSTM1 (glutathione S-transferase mu 1), GSTT1 (theta 1), and TP53 (tumor suppressor protein p53). The study found a significantly higher prevalence of the GSTM1 null genotype in GBC patients compared to healthy individuals (OR: 2.35; 95% CI: 1.03–5.37). However, no significant associations were observed for CYP1A1 (rs1048943), GSTT1 (deletion polymorphisms), or TP53 (rs1042522) polymorphisms. These findings suggest that the GSTM1 null genotype may be a potential risk factor for GBC in the Bolivian population.
179
In contrast, a similar study in the Chilean population showed no significant association with GBC.
180
A study conducted in the Japanese population reported that the rs1048943 variant of CYP1A1, which results in the substitution of isoleucine with valine at position 462, is linked to an increased risk of GBC. Individuals carrying the Ile/Val genotype have a 2.7-fold higher risk of developing GBC compared to those with the Ile/Ile genotype (95% CI: 1.1–6.4, P < 0.05). Similarly, in the Chilean population, the T allele of the CYP1A1 variant rs2606345 was associated with twice the risk of GBC compared to the G allele (OR: 2.0; 95% CI: 1.3–3.0).
181
Overall, genetic variations in CYP1A1 and GSTM1 influence gallbladder cancer susceptibility, with associations varying across populations. While the GSTM1 null genotype is a potential risk factor in Bolivia, CYP1A1 variants contribute to GBC risk in Japan and Chile. Further research is needed to confirm these findings across diverse populations. Variant rs708272 in cholesteryl ester transfer protein (CETP), is associated with an increased risk of developing GBC. For example, CETP - rs708272 polymorphisms particularly the T/T genotype may be related to the risk of developing GBC in Chilean women.
190
Additionally, other genetic variants related to DNA repair pathways have been associated with an elevated risk of GBC, including Asp312Asn in the ERCC2 gene, IVS1 + 9G>C in the MSH2 gene, Ser326Cys in the OGG1 gene, and EX5-25C>T in the MGMT gene. However, the most notable among these mutations are the TP53 polymorphisms that significantly contribute to the susceptibility to gallbladder cancer.194,198 Accordingly, the considerable body of research demonstrating the involvement of genetic variants in DNA repair pathway genes in GBC risk provides a foundation for future studies aimed at developing personalized medicine tailored for patients affected by this disease. Research also showed the influence of low penetrance variants in the caspase 8 (CASP8) gene on susceptibility to GBC. This was supported by an evaluation of CASP8 variant genotypes and haplotypes (rs3834129, rs1045485, and rs3769818) within the North Indian population. The study found that the “del” allele of the rs3834129 variant was associated with a 0.61-fold reduced risk for GBC (95% CI: 0.42–0.88, p = 0.005).
200
Although existing studies highlight the involvement of genetic variants from apoptosis pathway genes in GBC, the precise roles and molecular mechanisms underlying these associations remain to be clarified. Candidate genes and associated molecular pathways with therapeutic potential in gallbladder cancer.
3.2.2.2. Genome-Wide Association Studies
Genome-wide association studies (GWAS) analyze the entire genomes of patients and controls to identify numerous genetic variants associated with disease risk. This method is based on the premise that common variants across multiple genes contribute incrementally to the risk of developing diseases.
One of the first GWAS for GBC involved 41 patients and 866 controls from a Japanese population, identifying 130 SNPs linked to GBC. 201 A validation cohort of 30 GBC cases and 898 controls confirmed a strong association with the SNP rs7504990 in the deleted in colorectal cancer (DCC) region, showing an odds ratio (OR) of 6.95 (95% CI: 3.43–14.08, p = 7.46 × 10−8). 201 Additionally, a GWAS involving 1,042 GBC cases and 1,709 controls from North and Northeastern India found significant associations with genetic variants in the 7q21.12 region, particularly near the ABCB1 and ABCB4 genes. 202
A GWAS identified significant associations between variations in the ABCG8 and TRAF3 genes and GBC in the Chilean population. The initial study involved 529 gallstone disease cases and 566 controls, identifying ten candidate variants which were further validated in an independent cohort of 1,643 individuals to assess their link to GBC. 203 This association was subsequently confirmed by analyzing single nucleotide polymorphisms (SNPs) in 397 GBC patients, with ABCG8 (rs11887534, OR = 1.59) and TRAF3 (rs12882491, OR = 1.30) showing significant correlations. 204 These results underscore the critical role of ABCG8 and TRAF3 genetic variants in influencing susceptibility to gallbladder cancer.
In summary, GWAS have significantly advanced our understanding of genetic factors associated with GBC across different populations. The collective evidence from these GWAS underscores the incremental contribution of common genetic variants to GBC susceptibility, paving the way for improved risk assessment and potential therapeutic strategies in the future.
3.2.3 Limitations of Genetic Association Studies
GBC is a complex disorder influenced by numerous genetic alterations across various biological pathways. 205 The current understanding of the genetic and molecular changes in GBC, especially in high-risk populations, remains limited. Most studies have focused on candidate modifier genes encoding proteins involved in carcinogenesis, such as those related to apoptosis, cell-cycle regulation, DNA repair, xenobiotic metabolism, hormonal pathways, inflammatory pathways, and other risk factors. However, these known genes account for only a small portion of the heritability of gallbladder cancer, indicating that many genes with modest effects are yet to be discovered.78,206 An alternative approach is to conduct genome-wide association studies (GWAS) to explore the contribution of genes with varying levels of risk to GBC pathogenesis. 206
Genetic association studies also face several limitations. They often require large sample sizes to detect associations, especially for genes with small effect sizes. Additionally, these studies may not capture rare genetic variants or interactions between genes and environmental factors. The complex nature of GBC, influenced by multiple genetic and environmental factors, further complicates the identification of specific genetic contributors. Furthermore, findings from one population may not necessarily apply to others due to genetic diversity, highlighting the need for studies across different populations to gain a comprehensive understanding of GBC genetics.
4. Translational Implications, Emerging Biomarkers, and Future Therapeutic Directions
An improved understanding of the complex interplay between environmental exposures, genetic susceptibility, and metabolic dysregulation in GBC has important implications for both clinical management and population-level prevention strategies. The molecular insights synthesized in this review provide a conceptual framework for advancing biomarker-guided risk stratification, enhancing early detection, and identifying rational therapeutic targets. By integrating epidemiological evidence with mechanistic and molecular data, these findings support a shift toward precision-oriented approaches in GBC risk assessment and clinical intervention.
Based on current evidence, candidate biomarkers for GBC can be broadly stratified into three tiers according to their level of epidemiological support and biological relevance. First, genetic susceptibility markers supported by population-based studies—such as polymorphisms in detoxification-related genes including CYP1A1 and GSTM1—may help identify individuals at elevated risk due to impaired carcinogen metabolism. Second, mechanistically supported pathway-based biomarkers, including components of bile acid signaling, inflammatory mediators, lipid metabolism regulators, and oxidative stress markers, reflect core biological processes implicated in gallbladder carcinogenesis. Third, emerging circulating biomarkers—such as circulating tumor DNA, extracellular vesicles, and metabolomic signatures derived from plasma or bile—represent promising tools for early detection and disease monitoring, although their clinical utility remains contingent upon validation in well-powered prospective cohorts. This tiered framework emphasizes the need to prioritize biomarkers that demonstrate both biological plausibility and reproducible population-level associations.
The clinical translation of these candidate biomarkers will require rigorously designed prospective studies incorporating standardized biospecimen collection, longitudinal follow-up, and harmonized analytical pipelines. Bile-based metabolomic profiling, serum inflammatory markers, and circulating nucleic acid assays are particularly attractive due to their minimally invasive nature and potential applicability in high-risk populations, where conventional imaging-based screening has limited sensitivity. Importantly, integrating molecular biomarkers with imaging modalities and established clinical risk factors—such as gallstone disease and chronic biliary inflammation—may enhance early detection accuracy and enable personalized surveillance strategies.
From a therapeutic standpoint, the molecular pathways highlighted in this review suggest several potential intervention opportunities. Inflammation-associated signaling pathways, including PTGS2 (COX-2)–mediated prostaglandin signaling, represent biologically plausible targets given their established roles in gallstone-associated inflammation and biliary carcinogenesis. However, direct clinical evidence supporting COX-2 inhibition in GBC prevention or treatment remains limited, underscoring the need for disease-specific preclinical evaluation and safety validation. Targeting metabolic dysregulation—such as aberrant bile acid signaling, altered lipid metabolism, and oxidative stress responses—may offer additional therapeutic avenues, particularly in genetically susceptible subgroups. Moreover, dysregulation of apoptosis-related genes, including CASP8 and death receptor signaling components such as DR4 haplotypes, highlights opportunities for therapeutic strategies aimed at restoring programmed cell death and enhancing tumor sensitivity to treatment.
At the population level, integrating genetic susceptibility markers with environmental and lifestyle risk factors—including gallstone disease, chronic inflammation, dietary exposures, and persistent infections—supports the development of targeted prevention and early intervention strategies, particularly in high-incidence regions. Nonetheless, several challenges continue to limit rapid translational progress, including the low global incidence of GBC, substantial population heterogeneity in genetic and metabolic profiles, limited availability of large prospective cohorts, and the lack of standardized in vitro and in vivo disease models. Overcoming these barriers will require coordinated multicenter collaborations, harmonized biobanking efforts, and the development of robust, disease-specific experimental platforms.
5. Conclusion
Gallbladder cancer remains a highly lethal malignancy marked by striking geographic and demographic variation. This review underscores the complex interplay among environmental exposures, chronic infections, dietary factors, and metabolic disorders that converge to drive tumorigenesis through chronic inflammation, bile acid dysregulation, and oxidative stress. In parallel, advances in genetic and genomic research have identified susceptibility loci and functional variants involved in lipid metabolism, DNA repair, immune modulation, and oncogenic signaling. Together, these findings illuminate molecular pathways that promote genetic instability, aberrant signaling, and immune evasion, linking epidemiological risks to mechanistic drivers of disease (Figure 9). Gallbladder cancer risk factors and associated molecular mechanisms.
Integrating molecular, metabolic, genetic, and epidemiological data provides a cohesive framework for understanding gallbladder cancer pathogenesis and identifying novel biomarkers and therapeutic targets. However, significant gaps remain, particularly the limited representation of high-risk populations and the incomplete characterization of gene–environment interactions. Future research should prioritize multi-omic, population-based studies that capture global diversity and elucidate molecular mechanisms underlying disease heterogeneity. Translating these insights into early detection tools and targeted interventions will be essential for improving prevention strategies and patient outcomes worldwide.
Footnotes
Acknowledgement
Deeptima Jaiswar, Soham Choudhury and Vinay Jeeyar received a fellowship from National Institute of Science Education and Research (NISER), Department of Atomic Energy (DAE), Govt. of India.
Author Contributions
Dr. Manjusha Dixit (MD), Deeptima Jaiswar (DJ), Dr. Vinay Jeeyar (VJ) and Soham Choudhury (SC) were involved in writing of the original draft. MD and DJ were involved in reviewing and editing the article. DJ, MD, VJ and SC were involved in data curation. DJ was involved in visualization and diagrammatic representation. MD was involved in the conceptualization of the article.
Funding
We did not receive any funding for this work.
Declaration of Conflicting Interests
Authors have no conflict of interest.
Data Availability Statement
All the data is available from the first or corresponding authors on request.
Additional Information
All diagrams were generated using BioRender: Scientific Image and Illustration Software. Permission for use of data from Globocan 2022 was procured from the respective organization following the appropriate protocols.
