Abstract
Background
The effect of neoadjuvant chemotherapy (NACT) in gallbladder cancer (GBC) patients remains controversial. The aim of this study was to assess the impact of NACT on overall survival (OS) and cancer specific survival (CSS) in patients with localized or locoregionally advanced GBC, and to explore possible protective predictors for prognosis.
Methods
Data for patients with localized or locoregionally advanced GBC (i.e., categories cTx-cT4, cN0-2, and cM0) from 2004 to 2020 were collected from the Surveillance, Epidemiology, and End Results (SEER) database. Patients in the NACT and non-NACT groups were propensity score matched (PSM) 1:3, and the Kaplan-Meier method and log-rank test were performed to analyze the impact of NACT on OS and CSS. Univariable and multivariable Cox regression models were applied to identify the possible prognostic factors. Subgroup analysis was conducted to identify patients who would benefit from NACT.
Results
Of the 2676 cases included, 78 NACT and 234 non-NACT patients remained after PSM. In localized or locoregionally advanced GBC patients, the median OS of the NACT and non-NACT was 31 and 16 months (log-rank
Conclusions
NACT may provide therapeutic benefits for localized or locoregionally advanced GBC patients, especially for those with advanced stage, node-positive, poorly differentiated or undifferentiated disease. NACT combined with radical surgery was associated with a survival advantage. Therefore, NACT combined with surgery may provide a better treatment option for resectable GBC patients.
Keywords
Introduction
Gallbladder cancer (GBC) is the most common biliary epithelial malignancy. 1 According to the Global Cancer Statistics 2020, there were 115,949 new cases of GBC (41,062 in men and 74,887 in women) and 84,695 deaths (30,265 in men and 54,430 in women) worldwide, ranking it as the sixth most common malignancy in gastrointestinal tract neoplasms, 2 and the incidence rate of GBC in China was 3.92 per 100,000. 3 Patients with GBC are mostly female, and the incidence rate is increasing with age above 60 years. 4 Radical surgery (R0 resection) is currently the mainstay treatment option of GBC, but the majority of the patients diagnosed with GBC are already at advanced stages among which surgery alone might not be sufficient enough to render long term survival benefit. 5 Moreover, relapse rates are high in GBC with approximately 60%–70% of the patients expected to experience disease recurrence. 6 Hence, surgery combined with chemoradiotherapy, targeted therapy and immunotherapy are the treatment measures for GBC patients at the present.
Multiple retrospective studies have shown that postoperative adjuvant chemotherapy improves OS in patients with GBC, especially in patients with positive and/or stage R1 lymph node resection. 6 Compared with adjuvant chemotherapy, NACT aims to achieve tumor downstaging, reduce intraoperative metastases, and improve prognosis. 7 The therapeutic effect of NACT for patients with esophageal cancer, gastric cancer, and colorectal cancer has been confirmed.8-10 Although several studies have shown encouraging results in terms of improved survival in GBC patients receiving NACT, however, most of them were single-center retrospective and non-propensity score matching studies with a limited number of patients enrolled, meaning that they were inevitably subject to inherent selection biases and/or data missing due to incomplete data entry.11-14 Therefore, it is critical to initiate appropriate studies to verify the effectiveness of NACT in GBC to assist decision-making in clinical settings. To explore if GBC patients could benefit from NACT, we analyzed data from the Surveillance, Epidemiology, and End Results (SEER) database provided information of GBC patients from multiple medical centers of different states and counties in the United States and avoided selection bias of using data from a single institution and specific population.
In summary, this study used data from the SEER database to conduct a 1:3 propensity score matching (PSM) analysis to assess the impact of NACT on overall survival (OS) and cancer specific survival (CSS) which could help identify possible protective predictors for GBC to facilitate us to achieve a better therapeutic approach for GBC patients.
Materials and methods
Data Source
The data of GBC patients were obtained from the American Surveillance, Epidemiology, and End Results Program’s (SEER) 18 Registry Research Data resources since 2004, which covered approximately 35% of the U.S. population and contained information on cancer incidence, prevalence, primary tumor location, morphology, stage, treatment, and follow-up of millions of tumor patients in multiple state and county multicenter medical institutions in the United States. 15 The SEER database is readily available with patient anonymization, and thus approval from the ethics committee was not required. In our study, the SEER*stat software (version 8.4.2) was used to identify eligible GBC patients.
Patient Selection
The selection criteria were as follows: (I) all diagnosed GBC patients based on histopathological findings, and GBC is defined using the criteria of International Classification of Diseases in Oncology, 3rd edition (ICD-O-3); (II) patients with localized or locoregionally advanced GBC stage II to III and stage IV (with no distant metastasis: cTx-cT4, cN0-2, and cM0) included, and all data had detailed TNM stage information classified according to the 8th Edition of the American Joint Cancer Commission (AJCC) cancer staging manual; (III) cases with information on neoadjuvant and adjuvant therapy; (IV) cases with complete clinical and follow-up information. The following clinic-pathological variables of GBC patients were collected: gender, age at diagnosis, race; the tumor characteristics including AJCC stage, differentiation grade, tumor size, nodal status; treatments including surgical and chemoradiotherapy data; the time from diagnosis to treatment in months, the overall survival, cancer-specific survival, and vital status.
NACT refers to the SEER designation of ‘systemic therapy before surgery’ and ‘systemic therapy both before and after surgery’, and the remaining part was categorized in this study as the non- NACT group. In consideration of clinical practicability, we converted the 7th AJCC staging to the current 8th AJCC staging. The conversion process referred to the staging guidelines. Using the maximally selected test statistics from the R package survminer, the best cutoff points for survival analysis of tumor size and month from diagnosis to treatment were determined to be 3 (cm) and 1 (month), respectively. Based on the same rule, the age at diagnosis was stratified as <70 and ≥70 years. Radiation status was recorded as ‘Yes’ and ‘No’. Histological grades of the tumors were stratified as well-differentiated, moderately differentiated and poorly differentiated/undifferentiated. Nodal Status is divided into 3 categories: no examined, positive and negative. We reviewed the SEER database based on the procedure record codes and the SEER coding manual to distinguish between the different surgery types. 16 The item ‘simple/partial surgical removal of primary site’ in the SEER data ‘RX Summ-Surg Prim Site (1998+)’ (NAACCR item 1290, code 30) corresponds to ‘simple cholecystectomy’, and the use of ‘radical surgery’ refers to the SEER designation of ‘total surgical removal of primary site’ and ‘radical surgery’ (NAACCR item 1290, codes 40 and 60). Missing data were coded as unknown. The survival outcomes of this study were overall survival (OS) and cancer-specific survival (CCS). OS was the period from the date of diagnosis to death of any cause or the last follow-up time. While the CSS was the time from the date of diagnosis to the date of gallbladder cancer-associated mortality or the last follow-up time.
A total of 12,547 localized or locoregionally advanced GBC patients were enrolled in our study. After excluding patients with missing or incomplete information, 2676 patients diagnosed with GBC since 2004 were eligible and the data was recorded for analysis (Figure 1). Flow chart of patients’ cohort selection.
Statistical Analysis
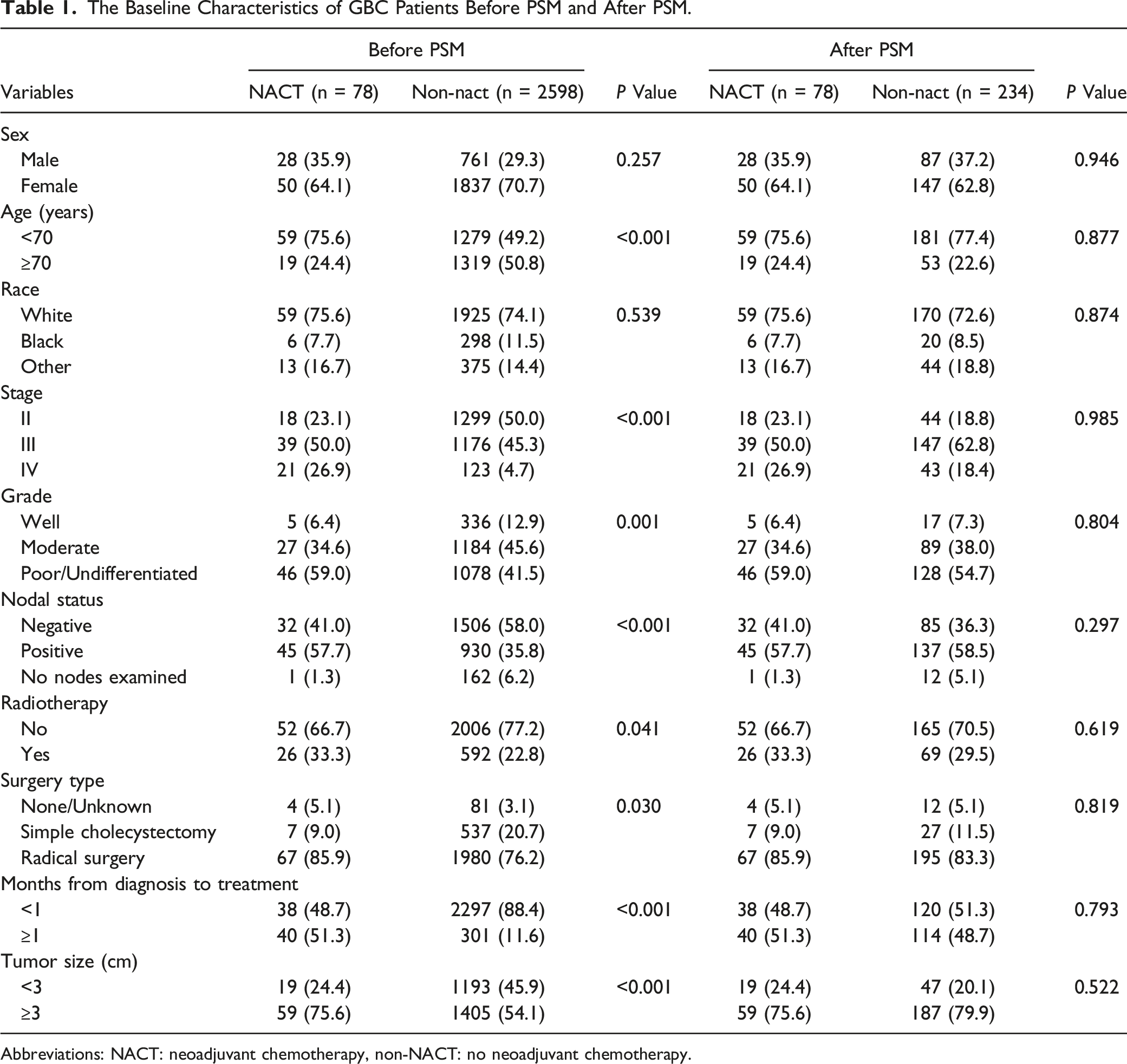
To balance the baseline characteristics of the NACT group and the non-NACT group, a 1:3 PSM method was applied, with a caliper value of 0.05, adopting the nearest neighbor matching method in which substitution was not allowed and each patient was matched only once. Information including gender, age at diagnosis, race, stage, differentiation grade, tumor size, surgical data, radiation record, nodal status, and months from diagnosis to treatment were utilized for the matching. Categorical data were displayed as counts and percentages. A Chi-squared test was performed to compare clinic-pathological variables before and after PSM.
The OS and CSS of localized or locoregionally advanced GBC patients were investigated in this study and univariate and multivariate Cox proportional hazards regression models were used to investigate the independent survival predictors. Parameters with statistical significance in the univariate analysis were further analyzed in the multivariate Cox model. The survival rates of localized or locoregionally advanced GBC patients in the NACT and non-NACT groups after matching were investigated by the Kaplan-Meier analysis with the log rank test. A Cox proportional hazard model was used in the subgroup analysis to identify patients who would potentially benefit from NACT.
IBM SPSS Statistical software (version 26.0) and R software (version 4.2.1) were utilized for statistical analysis.
Results
Baseline Clinicopathological Characteristics
In total, 2676 localized or locoregionally advanced GBC patients from the SEER database since 2004 met the inclusion criteria. Of these patients, 78 received NACT and 2598 did not. Before PSM, age, stage, grade, nodal status, radiotherapy, surgery type, months from diagnosis to treatment and tumor size showed significant differences between the NACT and non-NACT groups. A 1:3 PSM was applied to avoid potential selection bias and balance the baseline clinic-pathological characteristics of the above-mentioned 2 groups. After PSM, the NACT group consisted of 78 patients and the non-NACT group consisted of 234 patients. The standardized mean differences before and after PSM are illuminated in Figure 2. Baseline characteristics of the entire unmatched cohort and propensity score matched cohorts were summarized in Table 1. Standardized differences before and after PSM. PSM, propensity score matching. The Baseline Characteristics of GBC Patients Before PSM and After PSM. Abbreviations: NACT: neoadjuvant chemotherapy, non-NACT: no neoadjuvant chemotherapy.
Survival Analysis
Among the PSM samples, the median OS was 31 months (95% CI: 21.210-40.790 months) and 16 months (95% CI: 12.769- 19.231 months) in the NACT and non-NACT groups, respectively. After the Kaplan-Meier calculation of OS, there was a statistically significant difference between patients who received NACT and those that did not (log-rank Survival curves for OS and CSS in patients with localized or locoregionally advanced GBC receiving non-NACT and NACT by Kaplan-Meier. (A) Overall survival; (B) cancer-specific survival.
In addition, we found that compared with surgery alone, NACT was associated with a statistically significant improvement in OS and CSS. Figure 4A and B showed that the median OS was 31 months (95% CI: 19.634-42.366 months) in the NACT group and 17 months (95% CI: 13.735-20.265 months) in the upfront surgery group (log-rank Survival curves for OS and CSS in localized or locoregionally advanced GBC patients receiving NACT followed by radical surgery or upfront surgery by Kaplan-Meier. (A) Overall survival; (B) cancer-specific survival.
Prognostic Factors for Overall Survival and Cancer Specific Survival
Prognostic Factors for Overall Survival and Cancer-specific Survival.
Abbreviations: NACT: neoadjuvant chemotherapy, non-NACT: no neoadjuvant chemotherapy, CI: confidence interval.
Subgroup Analysis
Subgroup Analyses of NACT on OS in Patients With Different Features.

Abbreviations: NACT: neoadjuvant chemotherapy, non-NACT: no neoadjuvant chemotherapy, CI: confidence interval.

The forest plot of HRs comparing OS between the NACT group and non-NACT group according to different variables.
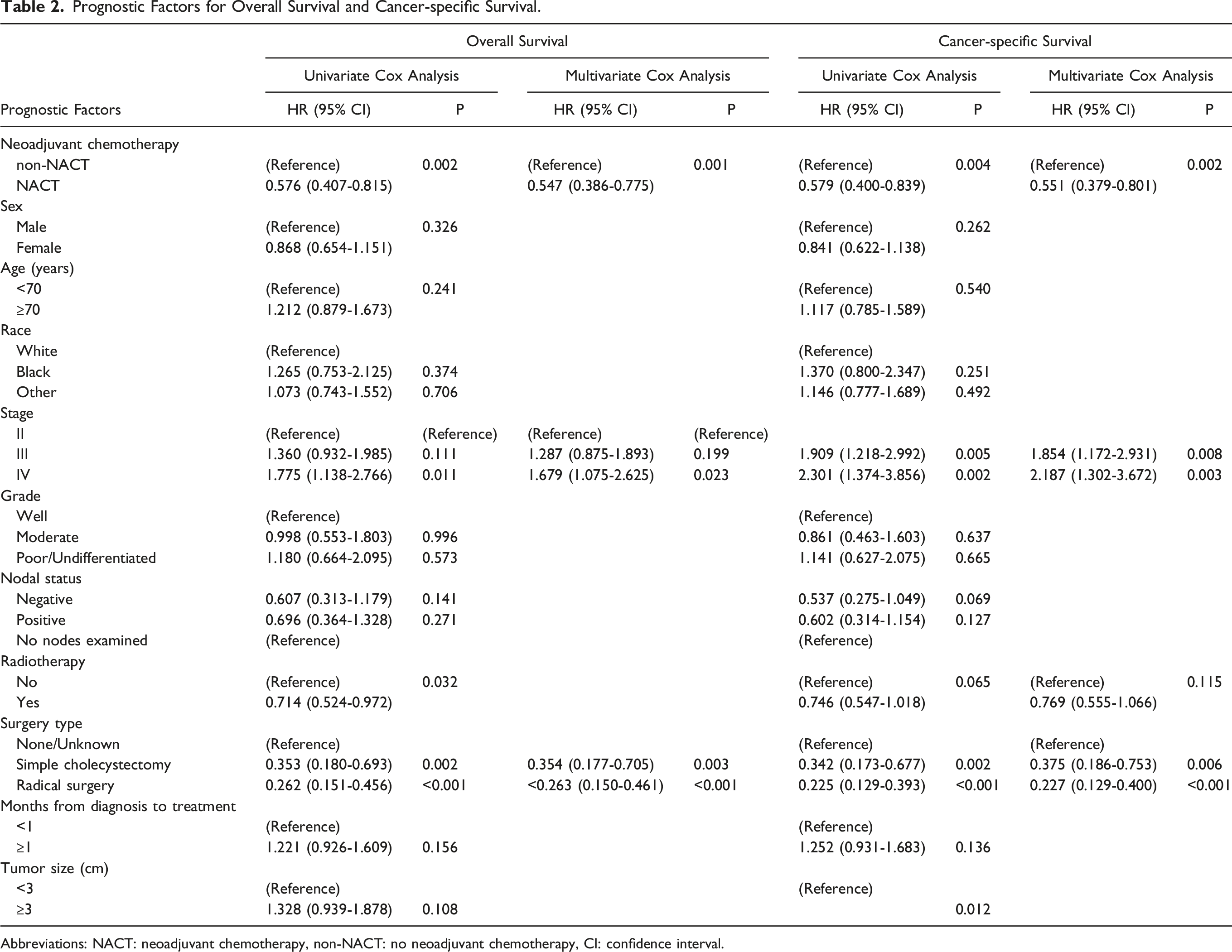
The hazard ratios (HRs) with NACT vs non-NACT for OS were significant in most subgroups. Detailedly, compared with patients without NACT, the patients with NACT presented lower HRs for OS of female (HR = 0.493, 95% CI: 0.335-0.727,
Discussion
GBC is an extremely aggressive malignant disease with the tendency of local invasion of neighboring organs and lymphatic or distant metastases. Radical surgery can provide a potential curative measure for the disease. However, due to the insidious onset and the lack of early clinical manifestations, radical surgical resection can only be achieved in about 20% of GBC patients. Most patients are diagnosed at the advanced stage with an estimated 5-year survival rate of 5%–10% and only 2% when GBC had undergone metastasis.17-19 Recent studies have shown that NACT can downstage the primary tumor and achieve a higher R0 rate and early control of systemic disease, which is the key to achieving a better overall survival for GBC patients.20,21 Accumulating evidences have shown that NACT an provide survival benefits in various malignancies including breast cancer, esophago-gastric cancer, rectal cancer, and in recent years, in 1 of the most vicious cancers that is notoriously known for its dismal prognosis, pancreatic cancer.22-25 However, the efficacy and feasibility of NACT on GBC is still elusive and debatable, and there have not been any randomized controlled trials (RCTs) comparing the effect of NACT in GBC patients. Ideally, a RCT study with a definitive result would be the best option to support the notion of NACT in GBC. However, it is difficult to conduct such RCTs in GBC patients since currently there are no highly effective chemotherapeutic regimens available and the incidence rate of GBC is relatively low in the general population. Therefore, we conducted this population-based study comparing localized or locoregionally advanced GBC patients who underwent NACT using data retrieved from the SEER database.
In our study, data of localized or locoregionally advanced GBC patients were retrieved from the SEER database and grouped with 1:3 PSM. We observed a significant improvement of OS and CSS in localized or locoregionally advanced GBC patients who underwent NACT and found that NACT had notably beneficial effects on both the OS and CSS in GBC patients undergoing radical surgery. Multivariate Cox regression analysis showed that NACT and surgery type were protective prognostic factors for localized or locoregionally advanced GBC patients, which significantly improved the OS and CSS for patients. In addition, tumor stage was a negative factor affecting OS and CSS, with a more advanced stage associated with a worse prognosis. Moreover, in subgroup analysis, our results found that for stage IV, node-positive, poorly differentiated or undifferentiated and tumor size greater than or equal to 3 cm GBC patients, the advantage of NACT for OS is significant, and thus we proposed the access to NACT for advanced patients in the clinical practice. Besides, patients without radical surgery had a worse OS, which further illustrated the importance of combining NACT with radical surgery.
Radical surgery aiming for R0 resection is particularly crucial to improve the prognosis of GBC, but for locally advanced GBC with adjacent organ or lymph node involvement, the extent and timing of primary resection remains debatable. Performing Extensive surgeries such as en bloc resection with adjacent organs involved or lymph node clearance is technically achievable but has not been associated with improved long-term survival.26,27 A meta-analysis concluded that locally advanced GBC patients receiving NACT followed by curative surgical resection had notably increased median OS compared with the non-radical surgery group and up to one-third of advanced stage GBC patients could benefit from NACT in terms of OS and disease-free survival (DFS), which is consistent with our results. 28 However, most of the studies included in the meta-analysis were retrospective studies and could not provide sufficient evidence to support the application of NACT for advanced GBC. Another retrospective analysis from American National Cancer Database (NCDB) showed that use of NACT was low in patients with surgically resected GBC, and NACT was associated with increased survival in node positive GBC. 29 The OMEGA study investigators conducted a multicenter, retrospective cohort study, showing that liver resection in T3 and T4 GBC was not associated with improved OS or recurrence-free survival (RFS), while these patients may benefit more from NACT rather than upfront surgery. 30 These positive results with NACT have also been demonstrated in our study.
At present, there is no preferred neoadjuvant treatment regimens for GBC. The ABC-02 trial established the use of gemcitabine in the addition of cisplatin as the first line chemotherapy for locally advanced GBC, 31 which currently remains as the most widely adopted regimen for the application of NACT in GBC. Some prospective studies are assessing the role of neoadjuvant treatment in BTC, trying to translate previous experience of pancreatic cancer in this setting. Several research groups including us are currently conducting clinical trials to study the efficacy and safety of novel adjuvant therapeutic measures to further improve both the resection rate and survival outcome of advanced GBC. A phase 3 randomized clinical trial (NCT02867865) of perioperative therapy in patients with locally advanced GBC designed to evaluate the survival effect of NACT alone vs chemoradiotherapy is recruiting. 32 We are currently conducting a prospective trial using gemcitabine in combination with albumin-paclitaxel for unresectable GBC (ChiCTR2200055698) and the primary data have shown promising results. 33 In the future, more high-quality prospective clinical studies should be carried out to continuously explore and optimize NACT programs to reduce early disease recurrence in GBC patients, improve the survival rate of patients, explore more drug options and combination therapy programs, and bring survival advantages to more GBC patients.
However, there are some limitations in this study. First, the SEER database contains clinical information from population-based cancer registries in the United States, which may not be representative for GBC patients as a global issue. Secondly, after PSM, the number of patients in the NACT group was small, which may not be powerful enough to strongly support the positive effect of NACT. Thirdly, the SEER database does not provide more detailed information, such as the specific regimen of NACT, the levels of tumor markers before surgery, the status of the surgical margins, and the time interval between the completion of chemotherapy and surgery, which may affect the prognosis of patients. Lastly, although PSM analysis can mitigate most of the confounding factors, as a retrospective study, selection bias still cannot be completely avoided. Hence, it is necessary to conduct prospective randomized controlled studies which might provide more powerful evidence to support the application of NACT in localized or locoregionally advanced GBC.
The application of NACT in localized or locoregionally advanced GBC is still at an early exploratory stage. We expect there will be more data from RCTs to help us determine the optimal chemotherapy regimens and establish a more comprehensive assessment system to improve the effectiveness of NACT and survival inpatients with localized or locoregionally advanced GBC.
Conclusion
NACT can improve OS and CSS inpatients with localized or locoregionally advanced GBC, and patients with advanced stage, node-positive, poorly differentiated or undifferentiated disease experienced significantly longer median survival with NACT. In combination with radical surgery, NACT increases the survival time of localized or locoregionally advanced GBC patient.
Footnotes
Author contributions
Yi-chen Xiong, Zi-yi Yang and Albie Gong participated in the study design, data analysis and wrote the manuscript. Zi-you Wu, Shi-lei Liu, Yi-di Zhu and Xiao-ling Song participated in the acquisition of the data. Bing-feng Chu, Xiang-song Wu and Wei Gong participated in the study design and critical review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (No.82172628, 81974371, 82173048); the Shanghai Scientific and Technological Innovation Plan (No.22Y11908000); Project of Health Academic Leadership from Shanghai Municipal Health Commission (No.2022XD010), Xinhua Hospital Funded Clinical Research (21XHDB10).
Ethical Statement
Data Sharing Statement
The data of this study are available from corresponding authors upon resonable request.
