Abstract
Introduction
Immune checkpoint inhibitors plus chemotherapy has become the new standard of care for advanced biliary tract cancer (BTC). However, the real-world effectiveness and safety of this approach, and its association with molecular alterations, remains uncertain.
Methods
In this single institutional retrospective cohort study, patients with advanced BTC treated with systemic chemotherapy with or without immunotherapy from July 2020 to June 2025 were included. Primary endpoint was overall survival (OS) in patients treated with first-line chemoimmunotherapy; secondary endpoints included progression-free survival (PFS), objective response rate (ORR), safety, prognostic factors, and molecular profiling.
Results
49 of 52 patients (94.2%) received chemoimmunotherapy. Median OS and PFS were 10.2 months and 5.4 months, respectively; the ORR was 24.5%. Poor Eastern Cooperative Oncology Group (ECOG) performance status and hypoalbuminemia were independent predictors of poor OS. Among the 52 patients, molecular profiling identified IDH1 mutations (9.6%), FGFR2 fusions (3.9%), HER2 amplification (5.8%), dMMR (5.8%), and BRCA mutations (5.8%). HER2 amplification was associated with worse OS; dMMR and BRCA mutations showed a trend toward improved OS.
Conclusion
Real world first-line chemoimmunotherapy for advanced BTC achieved outcomes comparable to those in pivotal trials. The ECOG performance status, serum albumin level, and HER2 status were prognostic for OS, supporting the importance of baseline patient selection and molecular profiling in treatment planning.
Keywords
Introduction
Biliary tract cancers (BTCs), comprising intrahepatic cholangiocarcinoma (iCCA), extrahepatic cholangiocarcinoma (eCCA), and gallbladder carcinoma (GBC), are aggressive malignancies with poor prognoses and limited therapeutic options.1-3 Although rare in Western countries, their incidence has been increasing worldwide, particularly iCCA.4,5 Most patients present with advanced-stage disease, for which systemic chemotherapy remains the mainstay of treatment. However, outcomes remain suboptimal, with median overall survival (OS) rarely exceeding 1 year.6-8
For over a decade, gemcitabine combined with cisplatin has been the standard first-line therapy for advanced BTC. 6 Recent phase III trials, including TOPAZ-1 and KEYNOTE-966, have demonstrated the clinical benefit of adding immune checkpoint inhibitors to standard chemotherapy in the first-line setting.7,8 These studies showed modest but statistically significant improvement in survival with durvalumab and pembrolizumab, respectively, leading to the establishment of chemotherapy plus immunotherapy as the new standard of care for advanced BTC.
Concurrently, the increasing use of molecular profiling has identified a spectrum of potentially targetable genomic alterations in BTC, including fibroblast growth factor receptor 2 (FGFR2) fusions, isocitrate dehydrogenase 1 (IDH1) and B-Raf (BRAF) mutations, and human epidermal growth factor receptor 2 (HER2) amplification.5,9-12 These biomarkers may be prognostic and are of increasing predictive significance,13-19 although their clinical utility in the context of chemoimmunotherapy remains to be fully defined. Despite the paradigm shift in treatment, evidence for the real-world effectiveness of chemotherapy combined with immunotherapy remains scarce. Clinical trials typically enroll highly selected patients under controlled conditions, and their findings may not fully reflect outcomes in routine oncology practice.
This retrospective study aimed to evaluate the real-world effectiveness of chemotherapy combined with immunotherapy in patients with advanced BTC and to describe the molecular alteration landscape in this population. Our findings may complement clinical trial evidence, support decision-making in clinical practice, and provide insights into the potential role of molecular profiling in the contemporary management of advanced BTC.
Materials and Methods
Study Design and Participants
This was a retrospective cohort study conducted at the Peter MacCallum Cancer Centre, Melbourne, Australia. Medical records of patients diagnosed with BTC between July 2020 and June 2025 were reviewed. Eligible patients were required to meet the following criteria: (1) histologically confirmed iCCA, eCCA, or GBC; (2) advanced disease at the time of treatment, defined as unresectable or metastatic; (3) receipt of first-line palliative systemic chemotherapy (gemcitabine plus cisplatin or carboplatin) with or without immune checkpoint inhibitors (durvalumab or pembrolizumab); and (4) age ≥18 years. Patients were excluded if they had (1) histological subtypes other than adenocarcinoma, or (2) received only best supportive care without systemic treatment. Clinical information was extracted from the institution’s electronic medical record system. All eligible patients were included in this study to provide a real-word perspective.
The study protocol was approved by the Human Research Ethics Committee and Governance Office of the Peter MacCallum Cancer Centre (HREC: QA/121051/PMCC/September 3, 2025). Owing to the retrospective study design, the requirement for written informed consent was waived. All data were de-identified to maintain patient confidentiality and adhere to privacy regulations. Patients were enrolled until June 2025, and data were censored at the institutional cut-off (July 30, 2025). The reporting of this study conformed to STROBE guidelines. 20
Outcome Measures
The primary outcome was OS in patients with advanced BTC who received first-line palliative chemotherapy in combination with immunotherapy. Secondary outcomes included progression-free survival (PFS), objective response rate (ORR), safety, and identification of prognostic factors for OS in this treatment setting. Additionally, biomarker analyses from all available patients to characterize molecular alterations were conducted as exploratory analyses.
OS was defined as the time from the initiation of systemic therapy to death from any cause. PFS was measured from the start of systemic therapy until either radiologically confirmed disease progression or death, whichever occurred first. Tumor assessments were performed every 2-3 months using chest and abdominal computed tomography. Treatment response was evaluated in accordance with the Response Evaluation Criteria in Solid Tumors version 1.1 using official institutional radiology reports; no retrospective central re-review of imaging was undertaken. Adverse events were graded by the treating oncologists according to the Common Terminology Criteria for Adverse Events version 5.0, using clinical, biochemical, and radiographic features consistent with the observed toxicities.
The treatment regimen consisted of durvalumab or pembrolizumab in combination with gemcitabine and either cisplatin or carboplatin, administered intravenously in 21-day cycles for up to 8 cycles. Durvalumab was administered at 1500 mg on day 1 of each cycle, in combination with gemcitabine (1000 mg/m2) and cisplatin (25 mg/m2) on days 1 and 8. After completion of the chemotherapy, durvalumab monotherapy (1500 mg every 4 weeks) was continued until disease progression, unacceptable toxicity, patient withdrawal, or death. When cisplatin was substituted with carboplatin, it was administered at an AUC of 1.5 on days 1 and 8. For patients receiving pembrolizumab instead of durvalumab, the dose was 200 mg on day 1 of each cycle, with administration every 3 weeks thereafter. Treatment selection was determined by the treating oncologists in agreement with the patients, based on baseline characteristics, drug accessibility, and reimbursement policies during each period. This could introduce potential selection bias in both treatment allocation and molecular testing. However, the primary aim of this study was to describe real-world outcomes.
Statistical Analysis
Baseline demographic and clinical characteristics were summarized using descriptive statistics. Continuous variables are presented as mean ± standard deviation (SD) for normally distributed data, or median with interquartile range (IQR) for non-normally distributed data. Categorical variables are reported as absolute frequencies and percentages. Survival outcomes, including OS and PFS, were estimated using the Kaplan–Meier method with censoring applied for patients alive at the data cut-off, and median survival times with corresponding 95% confidence intervals (CIs) were reported. Differences between survival curves were assessed using the log-rank test. Univariable Cox proportional hazards regression was used to assess factors associated with OS, and variables deemed potentially significant (P < 0.10 in univariable analysis) were subsequently included in a multivariable Cox regression to identify independent prognostic factors, reported as hazard ratios (HRs) with 95% CIs. The proportional hazards assumption was evaluated using Schoenfeld residuals. Model stability was assessed by the events-per-variable (EPV) criterion for the final multivariable model, with EPV ≥10 considered acceptable. Analyses were conducted on an available-case basis without imputation. All statistical analyses were conducted using R software, version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided P-value of <0.05 was considered statistically significant.
Results
Baseline Characteristics
Baseline Characteristics of the Patients

BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; PS, performance status; CA19-9, cancer antigen 19-9; IQR, interquartile range; iCCA, intrahepatic cholangiocarcinoma; eCCA, extrahepatic cholangiocarcinoma; GBC, gallbladder carcinoma.
Biomarker Assessment
Biomarker Assessment (N = 52)

BRAF, b-Raf; BRCA, BReast CAncer gene; CPS, combined positive score; FGFR2, fibroblast growth factor receptor 2; FISH, fluorescence in situ hybridization; HER2, human epidermal growth factor receptor 2; IDH1, isocitrate dehydrogenase 1; IHC, immunohistochemical testing; KRAS, K-Ras; MMR, mismatch repair; NGS, next-generation sequencing; NTRK, neurotrophic tyrosine receptor kinase; PD-L1, programmed death-ligand 1; TMB, tumor mutational burden.
Of all 52 patients, deficient mismatch repair (dMMR) was identified in 5.8%, and HER2 overexpression/amplification in 5.8%. NGS revealed targetable molecular alterations including IDH1 mutation (9.6%), FGFR2 fusion (3.9%), and high tumor mutational burden (TMB-high, 1.9%). No cases were identified with neurotrophic tyrosine receptor kinase (NTRK) fusions, BRAF V600E mutations, K-Ras (KRAS) G12 C mutations, or RET fusions. Baseline characteristics of patients by molecular alterations is presented in Supplemental Table S1. The complete spectrum of molecular alterations detected by NGS is shown in Figure 1 and detailed in Supplemental Table S2. Molecular Alterations Findings From Next-Generation Sequencing Testing (N = 29)
Real-World Effectiveness of First-Line Chemotherapy Plus Immunotherapy
A total of 49 patients treated with first-line chemotherapy plus immunotherapy were included in the effectiveness analysis, which evaluated OS, PFS, ORR, safety, and prognostic factors for OS.
Treatment Information
Treatment Information for Patients Who Received First-Line Chemotherapy Plus Immunotherapy (N = 49)

FOLFOX, 5-fluorouracil plus leucovorin and oxaliplatin; FOLFIRI, 5-fluorouracil plus leucovorin and irinotecan; ECOG, Eastern Cooperative Oncology Group; PS, performance status.
OS
The median follow-up duration was 8.4 months (range, 0.5-53.5 months). At the data cut-off, 35 patients (71.4%) had died. The median OS for the entire cohort was 10.2 months (95% CI, 6.7-15.3), with 1-year and 2-year OS rates of 35.6% and 11.9%, respectively (Figure 2A). Overall Survival (A) and Progression-free Survival (B) in Patients Who Received First-Line Chemotherapy Plus Immunotherapy
When stratified by first-line regimen, the median OS was 10.2 months for cisplatin plus gemcitabine with durvalumab, 7.4 months for carboplatin plus gemcitabine with durvalumab (HR, 1.05; 95% CI, 0.31-3.60; P = 0.940), and 14.3 months for cisplatin plus gemcitabine with pembrolizumab (HR, 0.56; 95% CI, 0.17-1.87; P = 0.347) (Supplemental Figure S1).
When stratified by primary tumor location, the median OS was 10.6 months for iCCA, 10.2 months for eCCA (HR, 1.20; 95% CI, 0.52-2.76; P = 0.673), and 3.7 months for GBC (HR, 1.83; 95% CI, 0.81-4.14; P = 0.148) (Supplemental Figure S2).
The PS at baseline was strongly associated with survival. Patients with ECOG PS 0 had a median OS of 15.5 months, compared with 6.0 months for ECOG PS 1 (HR, 4.06; 95% CI, 1.83-8.99; P < 0.001) and 2.2 months for ECOG PS 2 (HR, 22.32; 95% CI, 7.23-68.90; P < 0.001) (Supplemental Figure S3).
PFS
The median PFS for the overall cohort was 5.4 months (95% CI, 3.8-8.4; Figure 2B). When stratified by first-line regimen, the median PFS was 5.4 months for cisplatin plus gemcitabine with durvalumab, 3.0 months for carboplatin plus gemcitabine with durvalumab (HR, 2.01; 95% CI, 0.60-6.70; P = 0.255), and 6.9 months for cisplatin plus gemcitabine with pembrolizumab (HR, 0.56; 95% CI, 0.17-1.86; P = 0.342) (Supplemental Figure S4).
When stratified by primary tumor location, the median PFS was 5.8 months for iCCA, 4.4 months for eCCA (HR, 0.91; 95% CI, 0.42-1.98; P = 0.813), and 3.7 months for GBC (HR, 1.50; 95% CI, 0.70-3.21; P = 0.293) (Supplemental Figure S5).
Baseline ECOG PS was strongly associated with PFS. Median PFS was 9.7 months for ECOG PS 0, 3.8 months for ECOG PS 1 (HR, 4.47; 95% CI, 2.08-9.63; P < 0.001), and 1.7 months for ECOG PS 2 (HR, 19.51; 95% CI, 6.56-58.06; P < 0.001) (Supplemental Figure S6).
Response rates
Best Response to First-Line Chemotherapy Plus Immunotherapy

Safety
Adverse Events in Patients Receiving First-Line Chemotherapy Plus Immunotherapy (N = 49)

Prognostic factors for OS
Prognostic Factors for OS
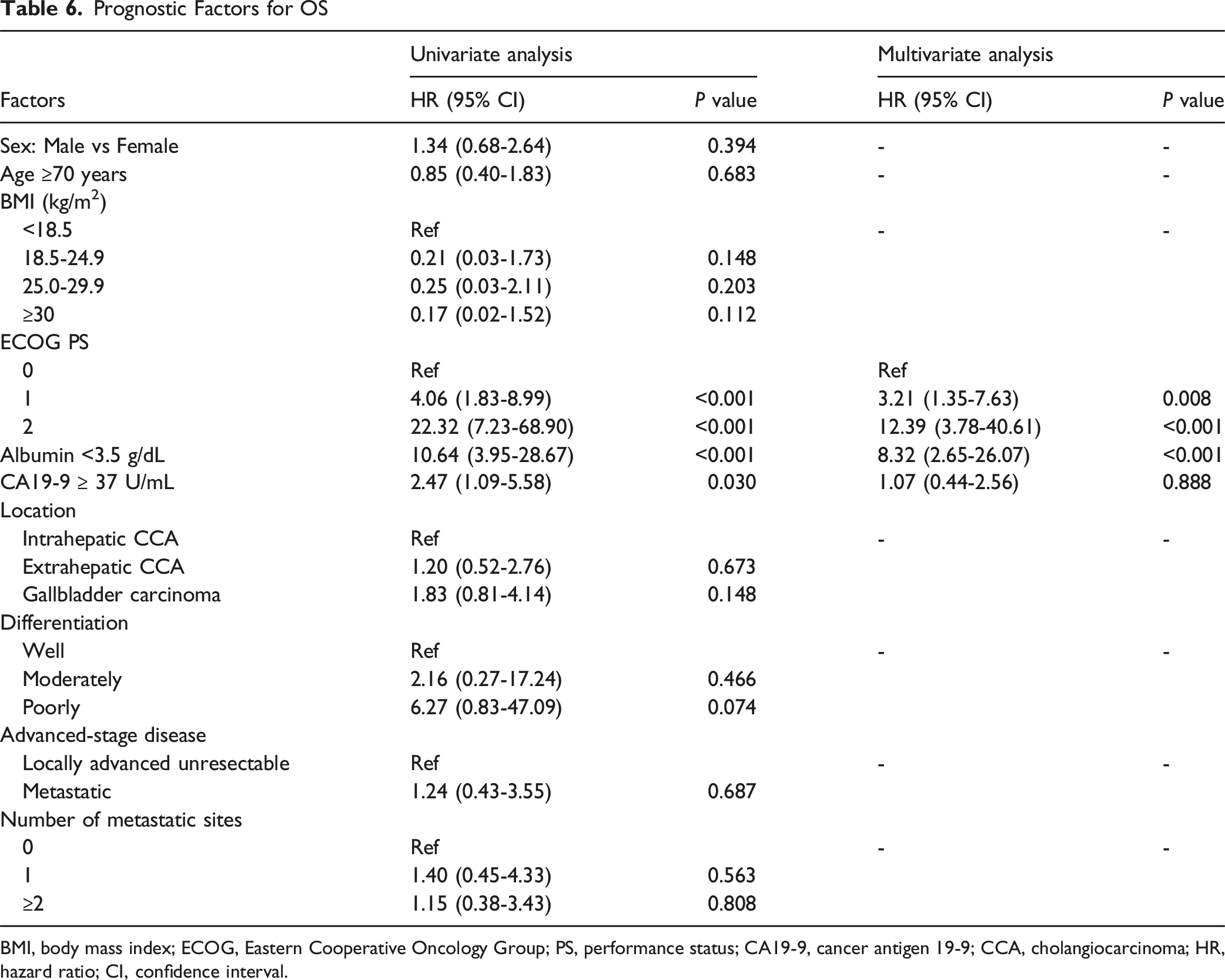
BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; PS, performance status; CA19-9, cancer antigen 19-9; CCA, cholangiocarcinoma; HR, hazard ratio; CI, confidence interval.
An exploratory analysis was performed to assess the prognostic impact of molecular alterations (Supplemental Table S6). HER2 overexpression/amplification was associated with markedly worse OS (HR, 14.09; 95% CI, 2.31-85.74; P = 0.004), whereas dMMR and BRCA mutation showed a trend toward improved OS, although not statistically significant. For FGFR2 fusion and TMB-high, only 1 patient was present in each subgroup, both of whom were alive at the data cut-off, precluding HR estimation. A swimmer plot demonstrating molecular alteration in each patient, and time to progression and death, is presented in Figure 3. A Swimmer Plot Demonstrating Time to Disease Progression and Death for Patients Who Received Chemotherapy Plus Immunotherapy. Each bar Represents One Patient in the Study and Their Length of Survival in Months (N = 49)
Discussion
This real-world study demonstrated the effectiveness of first-line chemotherapy combined with immunotherapy in patients with BTC. In our cohort, the median OS was 10.2 months, median PFS was 5.4 months, and ORR was 24.5%. These outcomes are broadly comparable to those reported in pivotal phase III trials such as TOPAZ-1 (median OS 12.8 months, PFS 7.2 months) and KEYNOTE-966 (median OS 12.7 months, PFS 6.5 months),7,8 although both OS and PFS were slightly lower in our cohort. The most likely explanation lies in the differences in patient selection. Our study included a real-world population with 15.4% ECOG PS 2, a group typically excluded from clinical trials. Consistent with this, survival outcomes differed markedly by baseline PS, with a median OS of 15.5, 6.0, and 2.2 months and median PFS of 9.7, 3.8, and 1.7 months for ECOG PS 0, 1, and 2, respectively. These results emphasize that while chemoimmunotherapy is effective, its benefit is maximized in patients with good baseline functional status.
Our findings are in line with a real-world Taiwanese study series (median PFS 5.6 months) that also included ECOG PS 2 patients, 21 but their longer OS (median OS 15.8 months) may be attributable to younger age, more locally advanced rather than metastatic disease, and a lower proportion of GBC, which is generally associated with poorer outcomes.6,22,23
Another factor potentially influencing OS is the lower rate of subsequent systemic therapy in our study (38.8%) compared to that in TOPAZ-1 (54.8%). In TOPAZ-1, median OS was 16.6 months in patients who received subsequent treatment vs 7.9 months in those who did not, underscoring the prognostic value of post-progression therapy. 7 The shorter OS in our cohort may partly reflect this gap in access to second-line options. Ensuring timely access to effective subsequent therapies could extend survival in the BTC population.
Baseline ECOG PS and serum albumin level were independent prognostic factors in our cohort, confirming prior studies that identified poor functional status and hypoalbuminemia as markers of inferior survival in BTC.24-28 While cancer antigen 19-9 level was not independently associated with OS in our multivariable model, it showed significance in univariate analysis, suggesting potential prognostic relevance depending on the cutoff thresholds used in future studies. Altogether, baseline functional and nutritional assessments should be integral to treatment planning in BTC.
The ORR of 24.5% in our cohort was consistent with the 26%-29% reported in the pivotal trials,7,8,29 despite nearly one-quarter of patients lacking a response evaluation. A response was observed across all primary tumor sites, although GBC maintained a poorer survival prognosis despite reported higher response rates in some earlier studies.6,30,31 These findings reinforce the aggressive biology of GBC and the need for site-specific research, especially focusing on molecular alterations. In our cohort, patients with ECOG PS 2 and those with GBC appeared to have the poorest OS and PFS. However, these findings should be interpreted with caution given the small sample size and single-center setting. While chemoimmunotherapy may still be a treatment option in this subgroup, it should be considered with careful discussion, close monitoring, and efforts to optimize functional status.
The safety profile was generally consistent with clinical trial data, with anemia, neutropenia, thrombocytopenia, elevated aspartate aminotransferase/ALT levels, nausea, and fatigue being the most common adverse events. irAEs occurred in 28.6% of patients, with 12.3% being grades 3-4, and leading to discontinuation of immunotherapy in 11.9%. This rate is higher than the 2%-7% reported in TOPAZ-1 and KEYNOTE-966,7,8,29 with hepatitis being the most frequent severe irAE. Overall, safety profile is manageable but vigilance for irAEs is crucial, reinforcing the need for close monitoring and rapid management of irAEs.
Molecular profiling, performed in 55.8% of patients, identified potentially targetable alterations: IDH1 mutations (9.6%), FGFR2 fusions (3.9%), dMMR (5.8%), TMB-high (1.9%), HER2 amplification (5.8%), and BRCA mutations (5.8%). When restricted to patients tested using NGS, IDH1 and FGFR2 alterations were found in 17.2% and 6.9%, respectively, suggesting an underestimation because of incomplete testing. This incomplete testing rate reflects real-world practice, where tissue availability and test accessibility are variable, and may introduce selection bias in biomarker prevalence estimates. These findings are consistent with previous reports for IDH1 mutations (10%-20%) but slightly lower for FGFR2 fusions (10%-15%) and HER2 amplification (typically 10%-20% in eCCA and GBC).11,32,33 IDH1 mutation and FGFR2 fusion were observed almost exclusively in iCCA, whereas HER2 overexpression/amplification occurred mainly in eCCA and GBC. Although the prevalence of actionable alterations is relatively low, comprehensive molecular profiling, preferably using NGS to cover the full spectrum of targets, 10 is essential to identify patients who could benefit from targeted therapy.
In exploratory biomarker analyses, HER2 overexpression/amplification was associated with significantly poorer OS, whereas dMMR and BRCA mutations showed a non-significant trend toward better survival. These findings align with previous studies reporting HER2 as a negative prognostic marker in BTC 34 and support the integration of comprehensive molecular testing into standard care. For HER2-positive BTC, evidence from gastric cancer suggests potential benefit from HER2-targeted therapy in combination with chemotherapy and immunotherapy, 35 an approach that warrants evaluation in prospective BTC trials. Taken together, routine comprehensive genomic testing can identify high-risk subgroups and guide targeted therapy strategies. Because molecular testing was not universally performed, biomarker missingness may be non-random and could bias alteration prevalence and reduce precision of exploratory subgroup analyses; accordingly, these findings should be interpreted with caution and results should be viewed as hypothesis-generating.
Although no predictive biomarkers have been validated for chemoimmunotherapy benefit in BTC, molecular testing remains essential to guide second-line therapy choices, particularly for IDH1 mutation, FGFR2 fusion, and other tumor-agnostic targets such as BRAF V600E mutation, RET fusion, KRAS G12C mutation, NTRK fusion, dMMR, and TMB-high.5,32 In other tumor types, the presence of certain mutations may be associated with reduced benefit from chemoimmunotherapy compared with targeted therapy,36,37 suggesting the need to explore combination or sequencing strategies in BTC.
This study is limited by its retrospective, single-center design, which introduces the potential for selection bias in both treatment allocation and molecular testing. The modest sample size limits the statistical power for subgroup, biomarker, and multivariable Cox regression analyses. Incomplete and heterogeneity in NGS testing may have influenced alteration detection rates. Consequently, the findings should be interpreted as exploratory and hypothesis-generating rather than definitive. Despite these limitations, this study provides a pragmatic real-world benchmark that complements prospective trial evidence and highlights the importance of patient selection, toxicity monitoring, and genomic profiling in BTC management.
Conclusions
In a real-world setting, this study provides real-world evidence supporting the use of chemotherapy plus immunotherapy as first-line treatment for advanced BTC, with outcomes and safety broadly consistent with clinical trial findings. ECOG PS, hypoalbuminemia, and HER2 status emerged as key prognostic factors. These findings underscore the need for careful baseline patient assessment, proactive management of toxicity, and universal access to molecular profiling to optimize treatment selection. Future multicenter prospective studies should validate these results and explore personalized strategies for molecularly defined subgroups, particularly those with HER2-positive disease.
Supplemental Material
Supplemental Material - Effectiveness of Chemotherapy Plus Immunotherapy and Molecular Alterations in Advanced Biliary Tract Cancer: Real-World Evidence From a Single-Center Australian Cohort
Supplemental Material for Effectiveness of Chemotherapy Plus Immunotherapy and Molecular Alterations in Advanced Biliary Tract Cancer: Real-World Evidence From a Single-Center Australian Cohort by Jirapat Wonglhow, Hui-Li Wong, Michael Michael in Cancer Control
Footnotes
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Human Research Ethics and Governance of Peter MacCallum Cancer Centre, Australia (HREC: QA/121051/PMCC/September 3, 2025).
Consent to Participate
Patient consent was waived because of the retrospective nature of the study and approved by the Human Research Ethics and Governance of Peter MacCallum Cancer Centre. Patient information was anonymized for confidentiality.
Author Contributions
Conceptualization, J.W. and M.M.; methodology, J.W. and M.M.; software, J.W.; validation, J.W.; formal analysis, J.W; investigation, J.W.; resources, J.W., H.W., and M.M.; data curation, J.W.; writing—original draft preparation, J.W.; writing—review and editing, J.W., H.W., and M.M.; visualization, J.W.; supervision, J.W. and M.M.; project administration, J.W. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
