Abstract
Introduction
Urological cancers—primarily prostate, bladder, and kidney cancers—pose a growing global public health challenge, particularly affecting men. While high-income countries have achieved early detection and better treatment outcomes, low- and middle-income countries (LMICs) continue to face late diagnoses and healthcare resource limitations.
Methods
This study leveraged Global Burden of Disease Study 2021 (GBD 2021) data to assess trends in age-standardized incidence rates (ASIRs), age-standardized mortality rates (ASMRs), and disability-adjusted life years (DALYs) for the three major urological cancers among men across 204 countries from 1990 to 2021. The ASIRs, ASMRs, and DALYs experienced the same age standardization. Estimated Annual Percentage Change (EAPC) was used to evaluate temporal trends. Risk attribution was examined using GBD’s comparative risk assessment framework and stratified by Sociodemographic Index (SDI) levels. Uncertainty intervals (UIs) were based on 1000 posterior simulations.
Results
Globally, prostate cancer ASIRs showed minimal change (EAPC −0.06), while ASMRs declined significantly (EAPC −1.05). North America had the highest incidence but notable mortality reduction, whereas Eastern Europe and sub-Saharan Africa experienced rising mortality despite lower incidence. Bladder cancer incidence declined worldwide (EAPC −0.35) but increased in Central Europe and Southeast Asia. Kidney cancer ASIRs rose globally (EAPC 0.72), with Mongolia showing the fastest increase (EAPC 16.22). Major risk factors included tobacco use, occupational exposures (eg, trichloroethylene), and metabolic conditions like obesity and hypertension. High SDI regions benefited from early screening and targeted therapies, while LMICs bore a heavier DALYs burden due to late-stage diagnoses and limited care.
Conclusion
Global progress in urological cancer detection and treatment is offset by widening disparities, particularly in LMICs. Addressing these inequities requires policies promoting equitable access to screening, risk factor reduction, and stronger cancer surveillance. Integrated, patient-centered strategies are crucial for achieving cancer control goals and reducing preventable deaths.
Plain Language Summary
Prostate, bladder, and kidney cancers are becoming increasingly common among men worldwide, posing a major health challenge—especially in low- and middle-income countries where access to early diagnosis and quality care is limited. Using global data from 1990 to 2021, this study found that while prostate cancer cases rose slightly, deaths have decreased in wealthier countries due to better screening and treatment, whereas mortality is rising in poorer regions. Bladder cancer has generally declined, though rates are increasing in parts of Europe and Asia, and kidney cancer is growing globally, with Mongolia seeing the fastest rise. Key risk factors include smoking, workplace chemicals, obesity, and high blood pressure. Countries with stronger healthcare systems have made progress, but the gap between rich and poor regions continues to widen. To reduce preventable deaths, there is an urgent need for fair access to testing, education, and healthcare improvements worldwide.
Keywords
Highlights
1. This study presents the first three-decade global comparison of prostate, bladder, and kidney cancer burdens in men across 204 countries. 2. Prostate cancer incidence is highest in high-SDI regions, but mortality is increasingly concentrated in low-SDI countries. 3. Bladder cancer shows rising incidence in Central Europe and Southeast Asia despite global declines. 4. Kidney cancer incidence has surged in Mongolia, linked to obesity and environmental exposures. 5. Tobacco, occupational chemicals, and metabolic risks remain leading contributors to urological cancer disparities.
Introduction
Urological cancers—including prostate, bladder, and kidney cancers—are major non-communicable diseases contributing substantially to the global cancer burden among men. Prostate cancer is the second most frequently diagnosed cancer and a leading cause of cancer-related mortality in men worldwide. 1 Bladder cancer, which occurs more commonly in men than women, ranks fourth in cancer incidence and eighth in cancer-related deaths among men. 2 Kidney cancer is the sixth most commonly diagnosed cancer in the male population. The global burden of these cancers demonstrates marked geographical heterogeneity, largely driven by variations in population aging, lifestyle-associated risk factors (such as tobacco use and obesity), and environmental exposures, including occupational carcinogens. 3
High-income countries have significantly reduced prostate cancer mortality through widespread implementation of early detection strategies, such as prostate-specific antigen (PSA) screening, and access to advanced therapeutic interventions. In contrast, LMICs face persistent challenges related to delayed diagnosis, limited screening coverage, and inadequate treatment infrastructure. 4 For bladder and kidney cancers, etiological factors such as tobacco consumption, occupational exposure to industrial chemicals, and components of metabolic syndrome play central roles. However, the global and regional disparities in their burden have not been systematically quantified across different levels of socioeconomic development. 5
Despite the critical insights provided by the GBD studies for cancer epidemiology, recent literature utilizing GBD 2021 data on urological cancers exhibits notable limitations. For instance, the analysis by Zi Hao et al. (2024) encompassed both benign and malignant urological conditions, failing to provide an in-depth investigation of the specific burden patterns, risk factor attribution, and systemic disparities across SDI strata for the principal urological malignancies (prostate, bladder, and kidney cancers) within the male population. 6 Similarly, Zhang Xinyu et al. (2025) focused exclusively on testicular and prostate cancers among adolescent and young adult males (aged 15-49 years), precluding a comprehensive assessment of the full disease spectrum, long-term trends, and SDI-associated dynamic inequalities for all three major urological cancers across the entire adult male lifespan. 7 Crucially, existing research lacks a systematic, integrated analysis of the burden data for these three cancers to elucidate their shared global challenges (eg, healthcare resource accessibility) and distinct epidemiological characteristics (eg, risk factor profiles, divergent temporal trends). This gap impedes the formulation of evidence-based, integrated prevention and control strategies tailored to SDI-stratified regions.
This study aims to characterize the evolving global, regional, and national burden of prostate, bladder, and kidney cancers in men from 1990 to 2021. Using data from the GBD 2021 study, we assess temporal trends, estimate attributable risk factors, and examine disparities across SDI strata. These findings are intended to support the development of differentiated, equity-focused cancer control strategies and to contribute to achieving the United Nations Sustainable Development Goal 3.4 of reducing premature mortality from non-communicable diseases. 8
Results
Prostate Cancer
The global burden of prostate cancer, as reflected in the GBD 2021 data, demonstrates significant variations in incidence, mortality, and DALYs across different SDI regions and geographic locations from 1990 to 2021. Globally, the ASIRs for males increased marginally from 32.64 (95% UI: 30.86, 33.86) in 1990 to 34.05 (31.27, 36.00) in 2021, with an estimated annual percentage change (EAPC) of −0.06 (−0.21, 0.08). Mortality rates declined from 16.35 (15.02, 17.28) to 12.63 (11.16, 13.55), with an EAPC of −1.05 (−1.14, −0.95), while DALYs decreased from 275.30 (251.66, 292.14) to 217.83 (192.65, 235.53), with an EAPC of −0.96 (−1.05, −0.87).
High SDI regions exhibited the highest ASIRs in 2021 at 70.92 (66.29, 74.22), despite a decline from 73.74 (70.21, 75.85) in 1990 (EAPC: −0.36 [-0.53, −0.20]). Mortality and DALYs in these regions also showed substantial reductions, with EAPCs of −2.02 (−2.14, −1.91) and −1.83 (−1.93, −1.73), respectively. Conversely, low SDI regions experienced an increase in ASIR from 14.50 (9.59, 18.27) to 18.14 (11.84, 22.34) (EAPC: 0.69 [0.61, 0.76]), alongside rising mortality (EAPC: 0.36 [0.29, 0.42]) and DALYs (EAPC: 0.24 [0.19, 0.30]).
Age-Standardized Incidence, Mortality, and DALYs of Prostate Cancer in 1990 and 2021
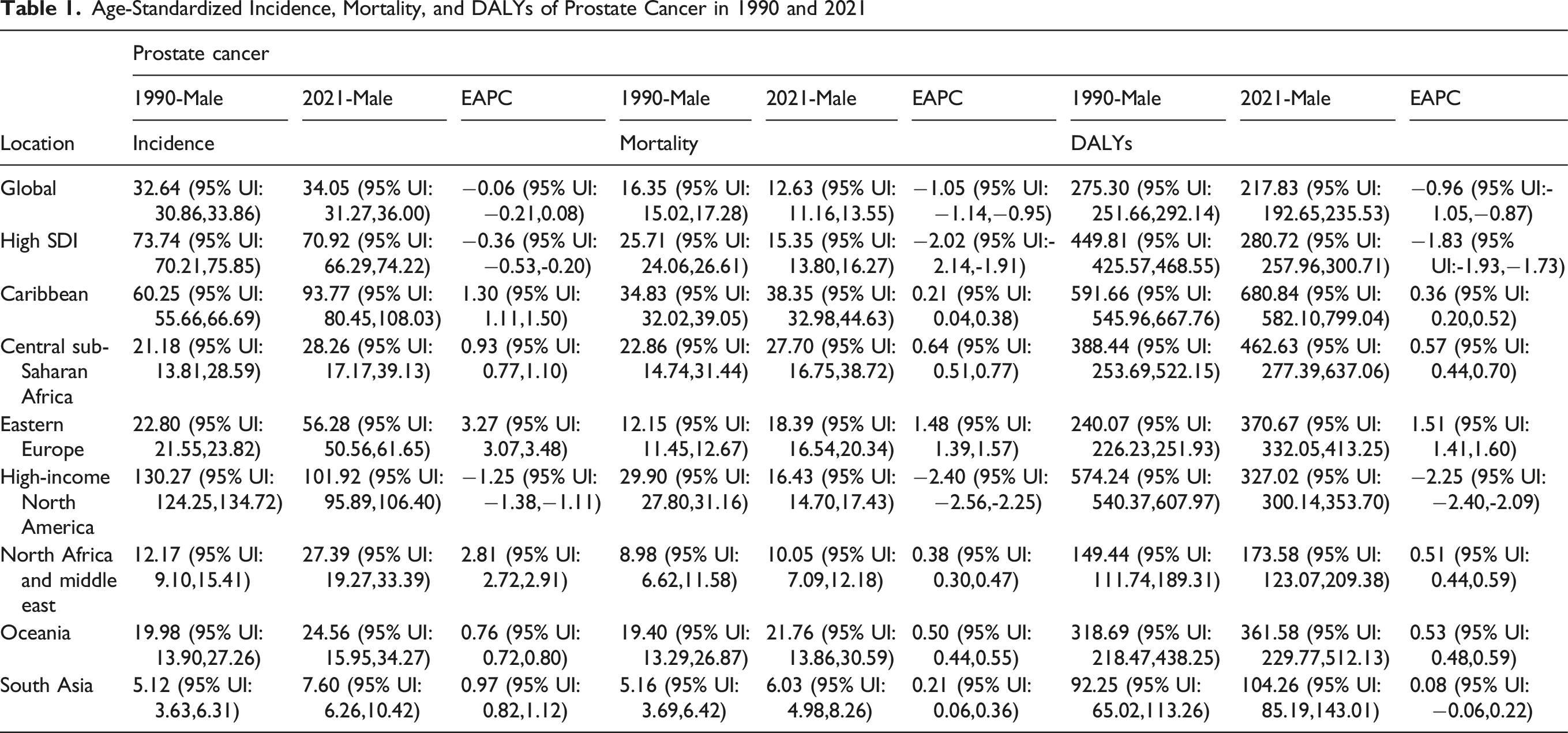
Analysis of country-level EAPC from 1990 to 2021 revealed that South Korea exhibited the fastest-growing prostate cancer incidence (EAPC 4.35; 95% UI: 3.74 to 4.96), followed by Georgia (4.30; 95% UI: 3.55 to 5.06) and Estonia (4.14; 95% UI: 3.40 to 4.88) (Figure 1A). Mortality increased most in Georgia (EAPC 4.07; 95% UI: 3.18 to 4.97), Egypt (2.58; 95% UI: 2.24 to 2.93), and Latvia (2.36; 95% UI: 2.06 to 2.65) (Figure 1C). DALYs rose most sharply in Georgia (3.89; 95% UI: 3.10 to 4.68), Zambia (2.58; 95% UI: 2.23 to 2.93), and Egypt (2.47; 95% UI: 2.18 to 2.77) (Figure 1E). Notably, Georgia ranked among the top three countries for incidence, mortality, and DALYs growth, underscoring the country’s rapidly worsening prostate cancer burden. EAPC of prostate cancer incidence, mortality, and DALYs rates, 1990-2021. Prostate cancer incidence, mortality, and DALYs rates in 2021. (A) EAPC for prostate cancer incidence, 1990-2021. (B) Incidence of prostate cancer in 2021. (C) EAPC for prostate cancer mortality, 1990-2021. (D) Mortality from prostate cancer in 2021. (E) 1990-2021 EAPC for prostate cancer DALYs. (F) DALYs for prostate cancer in 2021
Bladder Cancer
The GBD 2021 data reveals significant variations in the incidence, mortality, and DALYs associated with bladder cancer among males across different SDI regions and geographic locations from 1990 to 2021. Globally, the ASIRs declined from 11.89 (95% UI: 10.92, 12.57) in 1990 to 10.92 (10.04, 11.91) in 2021, with an EAPC of −0.35 (−0.40, −0.29). Mortality rates decreased from 6.10 (5.48, 6.53) to 4.67 (4.22, 5.16) (EAPC: −0.97 [−1.01, −0.92]), while DALYs fell from 119.24 (104.92, 128.18) to 86.31 (78.87, 95.94) (EAPC: −1.18 [−1.23, −1.13]).
High SDI regions exhibited the highest ASIR in 2021 (17.64 [16.44, 18.46]), despite a decline from 19.99 (19.14, 20.61) in 1990 (EAPC: −0.42 [−0.49, −0.34]). Mortality and DALYs also decreased significantly, with EAPCs of −1.05 (−1.09, −1.01) and −1.22 (−1.25, −1.19), respectively. In contrast, low SDI regions showed minimal change in ASIRs (1990: 3.94 [2.91, 4.85], 2021: 4.11 [3.44, 5.03]; EAPC: −0.01 [−0.13, 0.12]), with modest declines in mortality (EAPC: −0.28 [-0.39, −0.17]) and DALYs (EAPC: −0.38 [-0.48, −0.28]).
Age-Standardized Incidence, Mortality, and DALYs of Bladder Cancer in 1990 and 2021

At the national level, Cape Verde recorded the fastest-growing incidence of bladder cancer in men (EAPC 6.29; 95% UI: 4.67 to 7.92), followed by the Northern Mariana Islands (EAPC 2.52; 95% UI: 2.09 to 2.96) and Bulgaria (EAPC 1.96; 95% UI: 1.84 to 2.09) (Figure 2A). Cape Verde also led in mortality growth (EAPC 5.77; 95% UI: 4.11 to 7.46), followed by the Northern Mariana Islands (EAPC 2.25; 95% UI: 1.83 to 2.68) and Georgia (EAPC 1.60; 95% UI: 0.96 to 2.24) (Figure 2C). In terms of DALYs, the top three countries were Cape Verde (EAPC 5.54; 95% UI: 3.90 to 7.20), the Northern Mariana Islands (2.21; 95% UI: 1.80 to 2.62), and Kenya (1.48; 95% UI: 1.24 to 1.73) (Figure 2E). Notably, Cape Verde and the Northern Mariana Islands ranked in the top two for all three indicators, with non-overlapping 95% UIs, indicating a consistent upward trend in disease burden in these island regions. EAPC of bladder cancer incidence, mortality, and DALYs in men, 1990-2021. Incidence, mortality, and DALYs of bladder cancer in men in 2021. (A) EAPC of bladder cancer incidence in men, 1990-2021. (B) Incidence of bladder cancer in men in 2021. (C) EAPC for bladder cancer mortality in men, 1990-2021. (D) Mortality rate from bladder cancer in men in 2021. (E) EAPC of bladder cancer DALYs in men, 1990-2021. (F) DALYs for bladder cancer in men in 2021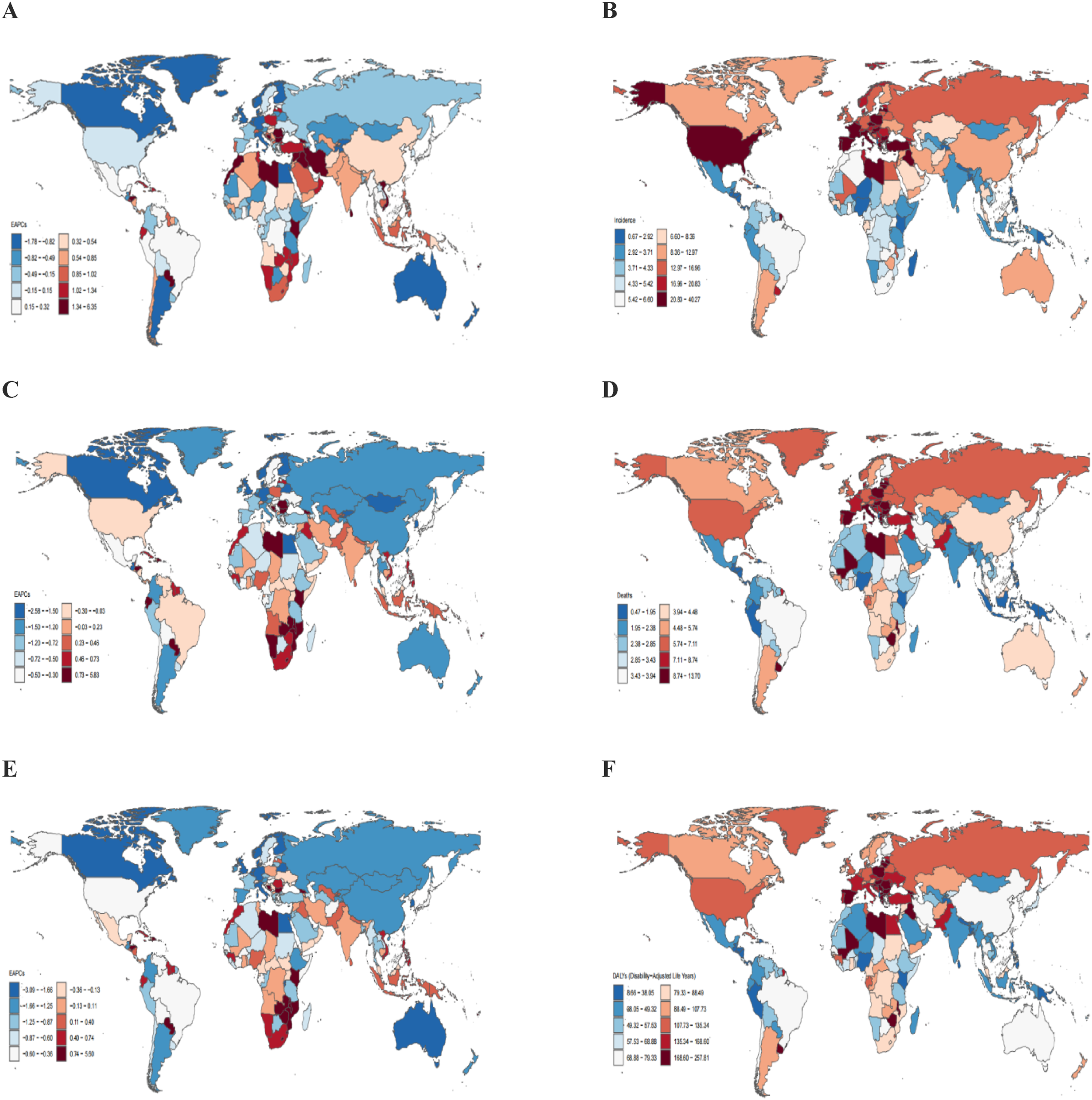
In 2021, countries with the highest bladder cancer incidence rates among men (20.83-40.27 per 100 000) included the United States, Portugal, Spain, France, Italy, Czech Republic, Poland, Hungary, Croatia, Bulgaria, Greece, Turkey, and Iran (Figure 2B). High mortality rates (8.74-13.70 per 100 000) were clustered in Portugal, Italy, Latvia, Lithuania, Poland, Croatia, Hungary, Romania, Bulgaria, Greece, Libya, Mali, Zimbabwe, Malawi, Uruguay, Georgia, and Azerbaijan (Figure 2D). Countries with the highest DALYs rates (168.60-257.81 per 100 000) were located in Eastern Europe (eg, Latvia, Romania), Western Asia (eg, Iraq, Georgia), and Sub-Saharan Africa (eg, Mali, Zimbabwe, Malawi) (Figure 2F), reflecting the compounded effects of delayed diagnosis, environmental risk exposure, and limited treatment infrastructure.
Kidney Cancer
The GBD 2021 data demonstrates significant global and regional variations in kidney cancer incidence, mortality, and DALYs among males between 1990 and 2021. Globally, the ASIRs increased from 5.09 (95% UI: 4.93, 5.23) to 6.26 (5.88, 6.64), with an estimated annual percentage change (EAPC) of 0.72 (0.59, 0.86). Mortality remained stable (1990: 2.75 [2.66, 2.84], 2021: 2.79 [2.61, 2.95]; EAPC: 0.05 [−0.02, 0.12]), while DALYs showed a slight decline (EAPC: −0.14 [-0.21, −0.06]).
High SDI regions reported the highest ASIRs in 2021 (12.46 [11.88, 12.92]), up from 10.80 (10.49, 11.09) in 1990 (EAPC: 0.52 [0.33, 0.72]), alongside decreasing mortality (EAPC: −0.31 [−0.39, −0.22]) and DALYs (EAPC: −0.56 [-0.67, −0.46]). Conversely, low-middle SDI regions exhibited the most pronounced ASIR increase (1990: 0.99 [0.90, 1.08], 2021: 1.74 [1.58, 1.90]; EAPC: 1.88 [1.86, 1.91]), with rising mortality (EAPC: 1.33 [1.29, 1.36]) and DALYs (EAPC: 1.25 [1.22, 1.27]).
Age-Standardized Incidence, Mortality, and DALYs of Kidney Cancer in 1990 and 2021

East Asia recorded the highest ASIRs increase (EAPC: 2.82 [2.62, 3.01]), while Western Europe demonstrated moderate growth (EAPC: 0.84 [0.67, 1.01]) with declining mortality (EAPC: −0.26 [−0.33, −0.20]). These findings highlight divergent epidemiological patterns, with low- and middle-income regions experiencing rising kidney cancer burdens, whereas high-income regions show stabilization or improvement in outcomes. The data underscore the need for targeted interventions to address these disparities.
Country-level analysis from 1990 to 2021 identified Mongolia as the global leader in the rate of increase for all three indicators. Its incidence rose at an EAPC of 16.22 (95% UI: 14.05 to 18.44) (Figure 3A), mortality at 15.42 (95% UI: 13.27 to 17.60) (Figure 3C), and DALYs at 15.20 (95% UI: 13.06 to 17.39) (Figure 3E). Cape Verde and Turkmenistan followed closely across all indicators. These sustained increases may be linked to escalating tobacco use, obesity prevalence, inadequate diagnostic capacity, and environmental exposures in these settings. EAPC of incidence, mortality, and DALYs of kidney cancer in men, 1990-2021. Incidence, mortality, and DALYs of kidney cancer in men, 2021. (A) EAPC of kidney cancer incidence in males, 1990-2021. (B) Incidence of kidney cancer in men in 2021. (C) EAPC for kidney cancer mortality in men, 1990-2021. (D) Mortality rate of kidney cancer in men in 2021. (E) EAPC of the DALYs of kidney cancer in men, 1990-2021. (F) DALYs for kidney cancer in men in 2021
In 2021, the highest incidence rates of kidney cancer in men (13.08-24.03 per 100 000) were reported in North America (eg, United States, Canada), Western Europe (eg, Denmark, France, Germany), and Eastern Europe (eg, Czech Republic, Hungary, Baltic states) (Figure 3B). Elevated ASMRs (5.43-10.37 per 100 000) and DALYs (130.44-264.23 per 100 000) were concentrated in Eastern Europe (eg, Poland, Ukraine), South America (eg, Chile, Argentina), and Turkey (Figure 3D-F). Eastern European countries such as Russia and Belarus ranked among the highest across all three metrics, potentially due to overlapping risk factors, including high smoking rates, red meat consumption, and occupational carcinogen exposure. By contrast, despite high incidence, North American regions maintained relatively moderate mortality and DALYs, likely reflecting widespread access to early detection and modern therapies.
Global and Regional Trends Across SDI Regions
Between 1990 and 2020, the global burden of prostate, bladder, and kidney cancers in men demonstrated marked heterogeneity across SDI regions. High-SDI regions consistently exhibited the highest ASIRs across all three cancers (Figure 4A-C). However, by the end of the study period, low-SDI regions surpassed high-SDI regions in both prostate cancer mortality and DALYs, underscoring substantial inequities in late-stage diagnosis and treatment (Figure 4D and G). Medium-SDI regions bore the highest burden of bladder cancer-related mortality and DALYs throughout the study period, although they also showed the steepest rates of decline over time (Figure 4E and H). For kidney cancer, incidence in high-SDI regions initially increased and then plateaued, with steadily declining mortality rates. In contrast, low-SDI regions experienced persistently low incidence but relatively stable mortality and DALYs, suggesting limited access to early detection and advanced care (Figure 4C, F and I). Age-standardized incidence, mortality, and DALYs for prostate, bladder, and kidney cancer in men globally and across all five SDI regions, 1990– 2020. (A) Age-standardized incidence of prostate cancer. (B) Age-standardized incidence of bladder cancer. (C) Age-standardized incidence of kidney cancer. (D) Age-standardized mortality rate for prostate cancer. (E) Age-standardized mortality rate for bladder cancer in men. (F) Age-standardized mortality rate for kidney cancer in male. (G) Age-standardized DALYs for prostate cancer. (H) Age-standardized DALYs for bladder cancer in men. (I) Age-standardized DALYs for kidney cancer in men
Burden Stratified by SDI in 2021
In 2021, prostate cancer incidence was strongly and positively associated with the national SDI level. Countries in high-SDI regions—such as Bermuda and several Caribbean nations—reported ASIRs exceeding 150 per 100 000 (Figure 5A). However, prostate cancer mortality declined steadily beyond an SDI threshold of 0.7, while countries in low-SDI regions (eg, St Vincent and Antigua) continued to experience ASMR exceeding 75 per 100 000 (Figure 5D). For bladder cancer, incidence rose steeply at SDI ∼0.6, reaching over 30 per 100 000 in countries like Lebanon and Italy (Figure 5B). Mortality peaked around the same SDI level, with exceptionally high rates in Poland and Mali, but improved substantially in countries with SDI ≥0.85 (Figure 5E). Kidney cancer incidence also increased with SDI, with ASIRs surpassing 20 per 100 000 in countries such as Argentina and the Czech Republic (Figure 5C). Both mortality and DALYs declined after SDI ≥0.8. However, select countries with advanced-stage case concentrations, such as Uruguay and Lithuania, continued to report ASMRs over 100 per 100 000 and DALYs above 2000 per 100 000 (Figure 5F and I). Notably, the Caribbean (eg, Grenada) and Eastern Europe (eg, Georgia) had the highest DALYs burdens for prostate cancer (>1500 per 100 000) and bladder cancer (>250 per 100 000), respectively (Figure 5G and H), reflecting the combined effects of delayed diagnosis, environmental exposures, and limited therapeutic access. Age-standardized incidence, mortality, and DALYs for prostate, bladder, and kidney cancer in men in 204 countries with different SDI levels in 2021. (A) Age-standardized incidence of prostate cancer. (B) Age-standardized incidence of bladder cancer. (C) Age-standardized incidence of kidney cancer. (D) Age-standardized mortality rate for prostate cancer. (E) Age-standardized mortality rate for bladder cancer in men. (F) Age-standardized mortality rate for kidney cancer in male. (G) Age-standardized DALYs for prostate cancer. (H) Age-standardized DALYs for bladder cancer in men. (I) Age-standardized DALYs for kidney cancer in men
Age Burden of Three Urological Cancers
In 2021, the global age distribution of urological cancers in men revealed distinct patterns across prostate, bladder, and kidney cancers. For prostate cancer, incidence peaked at ages 70-74 (∼270 000 cases), followed closely by ages 65-69 (∼250 000 cases) (Figure 6A). However, peak mortality occurred later, at ages 80-84 (∼81 000 deaths) (Figure 6D), while the highest burden of DALYs was concentrated at ages 70-74 (∼1.5 million DALYs) (Figure 6G). This lag suggests that while diagnosis commonly occurs earlier, mortality follows after several years, with DALYs indicating substantial health loss beginning in mid-elderly populations. Global incidence of prostate, bladder, and kidney cancer in men by age group, number of deaths, and DALYs in 2021. (A) Number of cases of prostate cancer. (B) Number of bladder cancers. (C) Incidence of kidney cancer. (D) Number of deaths from prostate cancer. (E) Number of deaths from bladder cancer in men. (F) Number of deaths from kidney cancer in men. (G) DALYs for prostate cancer. (H) DALYs for bladder cancer in men. (I) DALYs for kidney cancer in men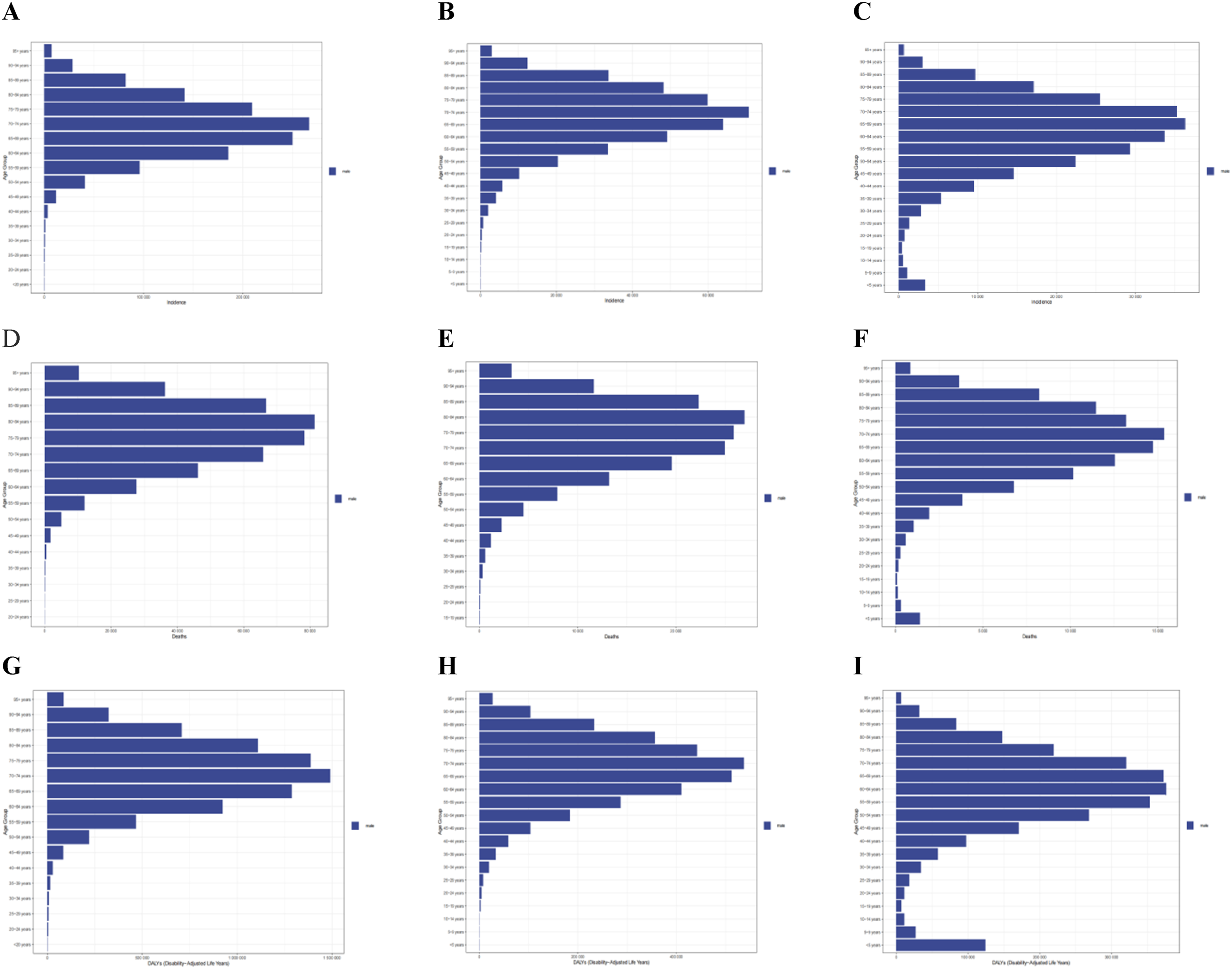
Bladder cancer showed a similar temporal shift. Incidence peaked at ages 70-74 (∼71 000 cases) (Figure 6B), whereas deaths peaked a decade later at ages 80-84 (∼27 500 deaths) (Figure 6E). DALYs were most concentrated at ages 70-74 (∼530 000 DALYs) (Figure 6H), reflecting the extended duration of disease-related disability and mortality following initial diagnosis.
Kidney cancer demonstrated a more complex pattern. Incidence peaked at ages 65-69 (∼36 000 cases) (Figure 6C), with mortality highest at ages 70-74 (∼15 500 deaths) (Figure 6F), and DALYs concentrated at 60-64 years (>350 000 DALYs) (Figure 6I), suggesting an earlier and more aggressive disease impact in middle-aged populations. Notably, kidney cancer DALYs were anomalously high in children aged ≤5 years (∼125 000 DALYs), indicating the burden of early-onset disease in this age group.
Overall, incidence for all three cancers peaked between ages 65-75, with mortality following 5-10 years later. DALYs consistently showed that the greatest health losses occurred within the first decade after diagnosis. Among the three, prostate cancer contributed the highest DALYs burden (∼1.5 million at ages 70-74), followed by bladder cancer (∼550 000), while kidney cancer displayed unique age-burden patterns due to both early-onset and middle-aged case accumulation.
Trends in Risk Factors by SDI Region
From 1990 to 2020, global and regional trends in modifiable risk factors for urological cancers demonstrated divergent trajectories across SDI regions. Smoking’s contribution to mortality and DALYs declined significantly in high and high-middle SDI regions for all three cancers but remained unchanged or declined minimally in low and middle SDI regions (Figure 7A-F). This disparity highlights the uneven effectiveness of tobacco control policies globally. Trends in mortality and DALYs due to risk factors in men from prostate, bladder, and kidney cancer, 1990-2020, globally and in the five SDI regions. (A) Prostate cancer mortality due to risk factors. (B) Risk factor-induced mortality from bladder cancer in men. (C) Mortality from kidney cancer in men due to risk factors. (D) Rate of DALYs for prostate cancer due to risk factors. (E) Rate of bladder cancer DALYs in men due to risk factors. (F) Risk factor-induced DALYs of kidney cancer in men
Dietary risks, particularly low calcium or milk intake—potentially protective factors—showed increased attributable burdens in lower-SDI regions, suggesting deteriorating dietary patterns and nutritional imbalances (Figure 7A and D).
Occupational exposure to trichloroethylene, a known carcinogen for kidney cancer, showed no appreciable improvement over the study period, particularly in lower SDI regions, indicating persistent deficiencies in workplace safety and regulatory enforcement. Collectively, these findings indicate that while high-SDI regions have curbed tobacco-related cancer risks, they now face growing metabolic threats. In contrast, low and middle SDI regions contend with dual burdens from traditional and emerging risk factors.
Discussion
This comprehensive global analysis of GBD 1990-2021 data reveals striking geographic and socioeconomic disparities in the burden of prostate, bladder, and kidney cancers among men. This study’s novelty lies in its integrated, cross-cancer analysis of prostate, bladder, and kidney cancers using harmonized GBD 2021 estimates, providing SDI-stratified burden profiles and aligned risk-attribution that directly inform differentiated policy responses. Prostate cancer remains the most prevalent and burdensome male urological malignancy, with a marginal global increase in age-standardized incidence but divergent regional trends. High-SDI countries such as North America and Oceania reported significant declines in incidence and mortality, largely attributable to widespread PSA screening and access to targeted therapies. 9 Conversely, incidence and DALYs surged in regions such as Eastern Europe,10-12 North Africa, and Latin America—reflecting delayed diagnosis, aging populations, and constrained health systems.13-15 Notably, Georgia consistently ranked among the top three globally for incidence, mortality, and DALYs growth, underscoring systemic weaknesses in cancer prevention and care. 16
Bladder cancer exhibited an overall decline in global incidence and mortality, but hotspots of rising burden emerged in medium-SDI regions, including Central Europe, Southeast Asia, and the Caribbean.17,18 Industrial exposure and smoking remain key drivers in these regions. Cape Verde and the Northern Mariana Islands show simultaneous increases in all burden indicators. The ASMRs and DALYs in Eastern Europe have increased significantly.
Kidney cancer showed the fastest increase in global burden, with incidence rising most rapidly in medium-SDI countries. Mongolia led the world in EAPC for incidence, mortality. While high-SDI regions achieved reductions in mortality through early imaging and modern therapies,19,20 regions such as Central Europe and the Caribbean demonstrated rising trends, 21 Especially in areas and countries with low SDI, driven by late-stage diagnosis and weak health infrastructure.22,23 Regional increases in mortality that are not accompanied by large incidence changes likely reflect health-system and data factors rather than only changes in disease occurrence. Contributing explanations include later stage at diagnosis, limited access to curative therapies, differences in screening intensity, and heterogeneity in registry completeness and cause-of-death certification. Because GBD does not consistently capture stage or treatment access, these interpretations are hypothesis-generating and require targeted country-level analyses to confirm.
Age-stratified analyses revealed a lag between peak incidence (65-74 years) and peak mortality (75-84 years), with DALY losses concentrated in the first 5-15 years after diagnosis. We also observed unexpectedly high DALYs for kidney cancer among children aged ≤5 years. While early-onset nephroblastoma and delays in access to pediatric oncology care may contribute to this pattern, this attribution is tentative; direct, country-level evidence is limited and we therefore present this interpretation cautiously and refer to the relevant literature.24-26
Globally, The burden of prostate cancer caused by smoking has been alleviated, 27 the burden of smoking-related bladder and kidney cancer has declined in high-SDI settings, while the metabolic risk-attributable burden (high BMI, hyperglycemia) has increased across all regions, This is similar to previous research results,28,29 This is similar to previous research. 30 Occupational exposures (eg, trichloroethylene) and environmental pollutants (eg, arsenic) continue to exacerbate kidney cancer risk in low-income regions, where occupational and environmental protections are limited.31-33
Limitations
This study has several limitations. First, data quality and cancer registry coverage vary substantially across countries, and coverage is particularly limited in some low- and middle-income regions such as sub-Saharan Africa; consequently, estimates for these locations rely more heavily on model-based inference and therefore carry greater uncertainty and potential for systematic bias. Second, the GBD data aggregation and local reporting practices are heterogeneous, and these differences can introduce additional variation that statistical modelling cannot fully eliminate; to promote transparency we report country estimates together with their uncertainty intervals and conduct sensitivity analyses (see Methods and Supplemental Table 1). Third, the additive attribution framework used by GBD does not capture interaction effects between co-occurring risk factors (for example, potential synergistic effects of smoking and occupational exposures), which could lead to underestimation of multifactorial risk burdens. Fourth, unmeasured genetic heterogeneity (for example, population differences in TP53 mutation prevalence) and the presence of early-onset cancer subtypes were not explicitly modelled, limiting our ability to explain etiologic diversity and region-specific vulnerabilities. Taken together, while numerous secondary analyses of GBD data yield broadly consistent macro-level findings, country-level estimates—particularly in low-coverage settings—should be interpreted cautiously, and strengthening local registry systems and targeted molecular epidemiology studies remain priorities to improve the precision and explanatory power of future assessments.
Conclusions
This study quantifies substantial disparities in the global burden and temporal trends of prostate, bladder, and kidney cancers among men between 1990 and 2021. Major findings include: (1) pronounced health inequities, with declining burdens in high-SDI regions contrasted by persistent or increasing burdens in many low-to-middle-SDI settings (for example, parts of Eastern Europe and sub-Saharan Africa); (2) cancer-specific trajectories, namely high prostate cancer incidence with declining mortality, a general global decline in bladder cancer incidence and mortality, and the most rapid increases in kidney cancer incidence; and (3) principal limitations stemming from data gaps in resource-constrained settings and from the simplified treatment of complex risk-factor interactions in current modelling frameworks. These results highlight priority areas for targeted surveillance and context-specific prevention strategies.
Policy Recommendations and Future Directions
Our findings point to targeted, context-sensitive actions that derive directly from observed burden patterns and risk attributions. We recommend the following priority measures:
Low-SDI settings (priority: basic diagnostic & treatment capacity): strengthen primary cancer registration, pathology and imaging capacity (basic ultrasound/ultrasound-guided biopsy), and ensure timely referral pathways to improve stage at diagnosis; invest in essential surgical and perioperative services for curative care, and integrate reproductive and urologic cancer management into primary NCD programs.
Middle-SDI settings (priority: scale-up plus targeted screening pilots): implement pilot, risk-stratified screening programs (eg, targeted PSA strategies for high-risk men) alongside public education; expand capacity for endoscopic diagnostics (cystoscopy) and timely urologic oncology treatment; roll out occupational exposure controls where bladder and kidney cancer burdens correlate with industrial exposures.
High-SDI settings (priority: prevention, optimization): intensify prevention of metabolic and tobacco-related risk factors through population-wide NCD measures; refine screening protocols to reduce overdiagnosis and focus on high-value interventions; strengthen survivorship and secondary prevention services.
Cross-cutting recommendations (all SDI levels): (1) prioritize strengthening cancer registries and routine data systems to monitor the impact of interventions; (2) invest in workforce training (pathology, surgical oncology, urology, radiology) and referral networks; (3) tailor interventions to the dominant local drivers identified in our risk-attribution analysis—e.g., tobacco control and occupational regulation where these risks are major contributors, metabolic control programs where kidney-cancer-attributable fractions are rising; (4) evaluate the cost-effectiveness and feasibility of proposed measures using local data before broad implementation.
Methods
Ethical Approval and Consent to Participate
This study utilized publicly available, de-identified data from the GBD database. As such, it did not involve direct contact with human participants or the collection of personal health information. Therefore, no ethical approval or informed consent was required. The study adhered to the principles outlined in the Declaration of Helsinki to ensure ethical behavior when using secondary data in public health research. The reporting of this study conforms to GATHER guidelines. 34
Data Sources and Case Definitions
We obtained male-specific estimates of age-standardized incidence rates (ASIRs), age-standardized mortality rates (ASMRs) and disability-adjusted life-years (DALYs) for prostate cancer (ICD-10: C61), bladder cancer (ICD-10: C67) and kidney cancer (ICD-10: C64–C66) from the Global Burden of Disease (GBD) 2021 database for the period 1990-2021 (ICD code in Supplemental Table 4). GBD standard cause-mapping protocols were applied and benign neoplasms and carcinoma in situ were excluded. Included data sources comprised population-based cancer registries, vital registration systems, autopsy series, health insurance claims and published registry reports. For locations with insufficient continuous high-quality registry data (defined here as <5 consecutive years of high-quality coverage), missing values were estimated using the GBD hierarchical spatio-temporal modelling framework that borrows strength from neighboring locations and incorporates covariates such as the Healthcare Access and Quality (HAQ) Index and the Socio-demographic Index (SDI). Uncertainty was propagated using the GBD posterior-draw approach (1000 draws), which captures sampling variability, model specification uncertainty and additional uncertainty due to data sparsity. DALYs were computed as the sum of years of life lost (YLLs) and years lived with disability (YLDs); YLDs were estimated by multiplying cause-, age-, sex-, location- and year-specific incidence of sequelae by their corresponding disability weights, while YLLs were calculated as deaths multiplied by the standard life expectancy at the age of death. See Supplemental Table 1 for ICD mappings and a data-source inventory. 35
Statistical Analysis
To account for differences in population structure, crude rates were age-standardized using the GBD standard population. Countries were stratified into five groups according to their socio-demographic index (SDI): low, low–middle, middle, middle–high, and high SDI. We calculated mean values and 95% uncertainty intervals (UIs) for each SDI category and outcome indicator. UIs represent the 95% probability that the true value lies within the specified interval and were estimated using hierarchical Bayesian models and stepwise Monte Carlo simulations (Figure 8). Methodological workflow of data acquisition, cleansing, conversion, and analysis based on the GBD 2021 database for prostate, bladder, and kidney cancer burden estimation across 204 countries (1990-2021)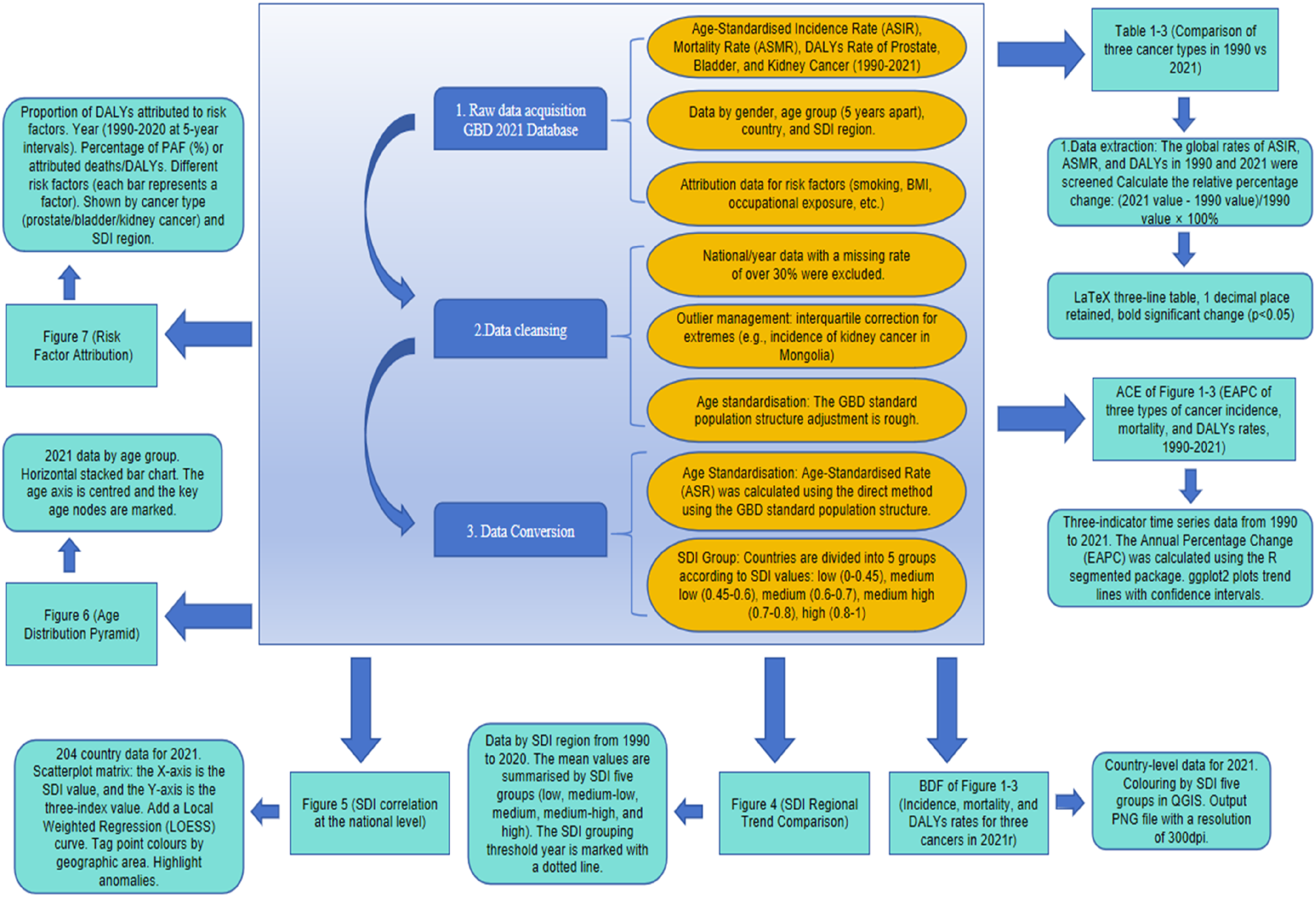
Data completeness, defined as the percentage of health-related indicators (eg, morbidity, mortality, risk factor prevalence) available in a given region and period relative to what is theoretically required, was used to assess regional coverage and reliability. Standardized analytical tools were applied using R software (version 4.2.2) for statistical computation.
Risk attribution followed the GBD Comparative Risk Assessment (CRA) framework. Exposure distributions were estimated at the age–sex–location–year level using the Tobacco Exposure Database 2.0 (TED 2.0) for tobacco, the ILO Occupational Exposure Matrix combined with national industry workforce distributions within the O-RAP tool for occupational trichloroethylene, and NCD-RisC outputs for metabolic exposures (BMI and fasting plasma glucose). Exposure–response functions (relative risks) and theoretical minimum risk exposure levels (TMRELs) were drawn from the GBD evidence base and primary literature. Population attributable fractions (PAFs) were computed by age–sex–location–year using standard continuous or categorical CRA formulas, and PAFs were multiplied by cause-specific rates to estimate attributable incidence, mortality and DALYs. Uncertainty from exposure estimates and relative risks was propagated using the GBD posterior-draw approach (1000 draws), and 95% uncertainty intervals are reported. The EAPC was employed to assess temporal incidence, mortality, and DALYs trends. EAPC was derived by fitting a log-linear regression model:
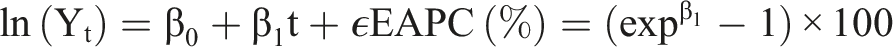
Statistical significance was defined as a non-zero
The selection of EAPC as the core trend indicator is based on the following rationale: GBD Standard Analytical Requirement: EAPC is the designated metric within the Global Burden of Disease (GBD) framework for assessing temporal trends in disease burden across decades, facilitating global comparability. Applicability of the Log-Linear Model: The log-linear regression model underpinning EAPC calculation is well-suited for modeling long-term trends in cancer burden. Clinical Interpretability: EAPC provides a single, integrated measure of overall trend, offering superior clinical interpretability by avoiding the complex interpretation required with segmented regression approaches.
Supplemental Material
Supplemental Material - Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021)
Supplemental Material for Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021) by Zidian Hu, Ding Zeng, Xu Zhao, Yuanyuan Luo, Zhengnan Li, Wanxiang You, Junjie Zhao, Shiwei Xiao, Bo Yu, Hao Su, Dongbo Yuan, Jianguo Zhu, and Bo Yan in Cancer Control
Supplemental Material
Supplemental Material - Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021)
Supplemental Material for Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021) by Zidian Hu, Ding Zeng, Xu Zhao, Yuanyuan Luo, Zhengnan Li, Wanxiang You, Junjie Zhao, Shiwei Xiao, Bo Yu, Hao Su, Dongbo Yuan, Jianguo Zhu, and Bo Yan in Cancer Control
Supplemental Material
Supplemental Material - Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021)
Supplemental Material for Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021) by Zidian Hu, Ding Zeng, Xu Zhao, Yuanyuan Luo, Zhengnan Li, Wanxiang You, Junjie Zhao, Shiwei Xiao, Bo Yu, Hao Su, Dongbo Yuan, Jianguo Zhu, and Bo Yan in Cancer Control
Supplemental Material
Supplemental Material - Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021)
Supplemental Material for Global Disparities and Temporal Trends in Prostate, Bladder, and Kidney Cancer Burden Among Men: A Systematic Analysis of 204 Countries (1990-2021) by Zidian Hu, Ding Zeng, Xu Zhao, Yuanyuan Luo, Zhengnan Li, Wanxiang You, Junjie Zhao, Shiwei Xiao, Bo Yu, Hao Su, Dongbo Yuan, Jianguo Zhu, and Bo Yan in Cancer Control
Footnotes
Acknowledgement
We would like to thank the Institute for Health Metrics and Evaluation, the GBD Disease and Injury Collaborators, and all the staff who shared the data required for this study. We thank the Basic research project (natural science) of Department of Guizhou science and technology, zk[2023], general-210.
Ethical Considerations
This study utilized publicly available, de-identified data from the GBD database. As such, it did not involve direct contact with human participants or the collection of personal health information. Therefore, no ethical approval or informed consent was required. The study adhered to the principles outlined in the Declaration of Helsinki to ensure ethical behavior when using secondary data in public health research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic research project (natural science) of Department of Guizhou science and technology, zk [2023], general-210.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
