Abstract
Cancer screening is increasingly promoted as a public health intervention, yet its effective translation with demonstrable impact on disease burden remains limited in low resource settings due to the suboptimal implementation of guidelines. While evidence from high-income countries and settings have informed international guidelines, the direct application of these recommendations often overlooks the realities of under-resourced settings, where diagnostic infrastructure, referral pathways, and treatment capacity are fragmented and inadequate. Moreover, the societal and health belief systems in many societies are different from high income country settings where screening was well received. This perspective provides a critical look at cancer screening with a focus on breast, cervical, colorectal, and oral cancers in low- and middle-income countries (LMICs) and draws on global and national guidelines and capacity to highlight gaps in implementation and system preparedness. Considerable variation exists across countries in terms of eligible age groups, test modalities, and screening intervals, reflecting both contextual adaptation and the challenges of aligning evidence with feasibility. Emerging innovations, including digital technologies and artificial intelligence, offer potential benefits but raise important concerns related to validation, ethical use, and equity of access. Ultimately, cancer screening can serve as a “best buy” intervention only if countries invest in strengthening early diagnosis and treatment pathways, ensure system readiness, and adopt phased implementation strategies tailored to local contexts. Without such preparedness, large-scale screening risks misallocation of scarce resources without measurable impact on the disease burden.
Plain Language Summary
Cancer screening a is a key component of cancer control. Current implementation of screening programmes especially in low- and middle-income countries (LMICs) are suboptimal. There are challenges in translating global evidence. Formation of global evidence is often based on research in high income settings or controlled artificial settings in low income settings. Both are not generalisable. Capacity of the health system to manage the end-to-end screening programme is critical. We provide an informed guidance on implementing cancer screening guidelines, considerations in adapting them to LMICs, and a checklist to assess the health system readiness and a pathway to ensure that screening programmes work well and can produce impact.
Keywords
Introduction
As early as 1951, the United States Commission on Chronic Illness defined screening as ‘the presumptive identification of unrecognized disease or effect by the application of tests, examinations, or other procedures which can be applied rapidly. Screening tests sort out apparently well people who probably have a disease from those who probably do not. A screening test is not intended to be diagnostic. People with positive or suspicious findings must be referred to their physicians for diagnosis and necessary treatment’. 1 The wisdom of this definition and the elements specified are applicable even after 75 years.
WHO commissioned a paper which was brought in 1968 as the ‘Principles and practice of screening for disease’ by JMG Wilson and G Jungner which provided granular details of the diseases, test and screening process. 2 Concepts were made clear in the often referred to as ‘Wilson and Jungner criteria’ with ten principles. Many of these are relative and not absolute and tend to get used depending on the interest and bias of the investigators and implementors.
Screening is often considered as an element of national Noncommunicable diseases (NCD) disease control programmes, however due consideration should be placed on the population to be covered and national health system readiness. Evidence of screening impact often comes from controlled trials and the health system capacity implemented for the trial is not available in routine public health systems. Sustained large scale national programmes for resource limited settings are to be generated in a phased manner. 3
Screening as an intervention is appealing to all stakeholders and gives a notion that the disease can be screened, detected and cured as soon as a test is available. 4 Rapid advances in technology including now the use of Artificial Intelligence (AI) supported tests will add in more complexity.
This perspective brings out the challenges in the implementation of large-scale breast, colorectal, cervical, and oral cancers cancer screening programmes in resource limited settings and propose some suggestions for a phased approach.
Basis of Screening
The biological basis of screening is an important concept and must be understood clearly to make informed choices. WHO has presented an illustration for cervical cancer progression and the critical importance of a ‘pre-clinical detectable phase (PCDP)’.
5
This is the phase where biological onset of disease has started in the body but has not yet manifested as clinical signs and symptoms. During this ‘silent’ period if there are tests which can detect them reliably and if there are interventions which are easy to use and are effective, then the progression of the disease can be arrested. This is the abortion of the natural history of the biological process thereby saving the individual from the disease (Figure 1). Preclinical detectable phase in the natural history of the disease (Adapted from. National cancer control programmes: policies and managerial guidelines. – 2nd ed. WHO.) (Image by Autor)
The presence of PCDP is clearly identified in cervical cancer pathogenesis and tests to detect the phase along with safe and reliable interventions to abort the process at this stage are available. This makes cervical cancer as the poster child of screening.
There are 2 caveats that have to be kept in mind while evaluating screening. Length bias - tumours which are slowly growing are likely to spend more time in the PCDP and hence are more likely to be picked up. They might have a natural history which is less aggressive and hence such cancers might do well with a better survival outcome. This length bias may be contributing to the survival advantage in screen detected cancers.
Lead time bias is the process in which the difference in survival advantage from screen detected cancers is due to the labelling of the case earlier than clinically detected cancers. True difference can be obtained after adjusting for lead time bias.
Early diagnosis is different from screening as people with symptoms are being identified and checked to diagnose the presence of cancer. The ‘WHO guide to early detection’ provides more clarification on early diagnosis. 6
Evidence Based Guidelines for Cancer Screening
Evidence-based cancer screening guidelines are typically derived from systematically collated and graded evidence, most often from randomized controlled trials, diagnostic accuracy studies, or modelling predominantly conducted in HICs.7-9 These trials are performed in health systems with established laboratory services, reliable referral pathways, and capacity for timely treatment of precancers and cancers, meaning only incremental adjustments are needed before program initiation. In contrast, in resource limited settings, the foundational infrastructure for screening, diagnosis and management are even lacking at tertiary-level facilities.1,10 Consequently, the estimates of benefit and harm generated in controlled environments where an artificial health system was introduced for the trial, may not be directly transferable to settings where the health system is fragmented and under-resourced. Moreover, nuances in guideline wording such as recommendations graded as “conditional” or based on “moderate certainty” of evidence are often overlooked, and decisions to initiate screening programs may proceed without thorough assessment of system readiness or community preparedness.11,12
In practice, the translation of guideline recommendations into large-scale screening programs is constrained by systemic limitations. For example, while primary HPV testing for cervical cancer is recommended by WHO, 7 its successful implementation requires laboratory infrastructure, sample transport logistics, and timely linkage to triage and ablative or excisional treatment resources. 7 The term ‘conditional’ in guideline recommendations is a critical term to be considered in decisions to screen. The term, ‘conditional recommendation’ is more likely if there is uncertainty about the balance of benefits vs harms or when the anticipated net benefits are small, as indicated the WHO handbook. 13 Mammography for breast cancer, as advocated in high-income settings, may be infeasible due to equipment shortages, inadequate quality assurance systems, and insufficient trained personnel, leading some LMICs to adopt clinical breast examination as an interim measure. 8 Similarly, faecal immunochemical testing (FIT) or colonoscopy for colorectal cancer depends on endoscopy capacity that is frequently inadequate, 14 and visual oral inspection programs may falter without clear referral pathways or treatment availability. 15 These contextual gaps can result in low coverage, poor follow-up rates, and negligible or no mortality benefit despite adherence to the “recommended” screening test. Such programmes may end up in nonproductive use of the limited resources and may even lead to harm in resource limited settings.
To improve applicability, evidence-based guidelines would benefit from explicit articulation of minimum system readiness criteria, phased or context-specific pathways, and operational guidance for implementation in resource-constrained settings. Conditional or moderate-certainty recommendations should be accompanied by clear caveats outlining the health-system investments required for safe and effective roll-out. Incorporating evidence from pragmatic trials, pilot programmes, and implementation research rather than relying solely on trial efficacy data would enable countries to anticipate real-world barriers, adapt screening strategies to local realities, and avoid premature nationwide deployment of programmes that health systems cannot sustain. Such an approach would help bridge the persistent gap between “what works” in controlled conditions and “what can work” in routine practice in resource limited settings.
The experience of most patients navigating the challenging and fragmented cancer care journey in most resource limited settings is depicted in Figure 2 which portrays complex patient pathways, referral gaps, and treatment delays. Depending on the context, 6 to 8 months can lapse from the time of clinical presentation to the starting of definitive cancer directed treatment. Navigating the cancer care journey (Image by Autor)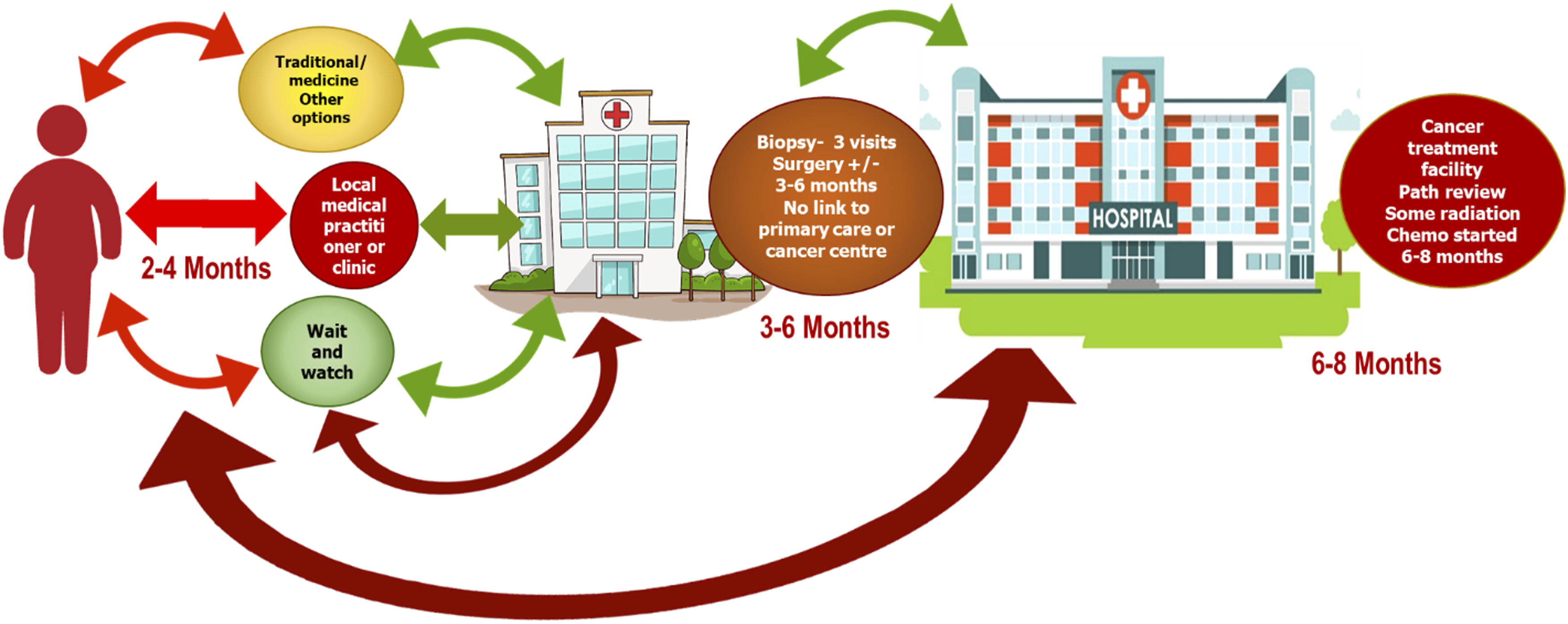
National Guidelines

Variations in age group, frequency and type of test for the same disease in different countries of the world is an illustration of the adaptation of evidence-based guidelines in different contexts.
National Capacity and System Response for Screening and Coverage in the WHO South-East Asia Region
National NCD country capacity survey for the South-East Asia Region countries published in 2025 indicate the situation in 11 countries. For cervical cancer, the age range of screening varied from 18 to 65 years. Visual inspection was the method in 6 of the 11 countries and, 2 used PAP smear and 3 had HPV testing. Coverage was less than 10% in 3 countries and only 1 country achieved more than 70% coverage.
Screening for breast cancer also showed wide variations. Age ranged from 15 to 69 years; clinical breast examination was the method used and only 2 countries had more than 70% coverage. 16 These variations reflect the challenges of decisions at the national level.
To Screen or Not to Screen?
Cancer is emerging as a public health priority in all LMICs. While efforts to reduce risk factors are important. Strong considerations should be given to operational feasibility and prevailing health system capacity readiness before embarking on initiation of cancer screening for the population.
The first step is to understand the burden of cancers, current stage at presentation, and the health system capacity to manage cancers. Priority is to have cancers detected in early stages through awareness, and capacity to diagnose at heath facilities closer to the person. Pathology is a critical factor, and often reliable and timely pathology diagnostic services are not available. Staging of the disease is essential in cancer care and imaging, and other modalities should be available to stage cancers. Surgery, radiotherapy and chemotherapy as needed should be available to offer optimal treatment without delay. All these must be available without Out of Pocket Expenditure. The delay in diagnosing and treating cancer will further exacerbate the increasing deaths from cancers in resource limited settings.
Country Readiness for Screening
‘WHO guide to cancer early diagnosis’ has provided guidance to assess the situation and to initiate screening in a phased manner Figure 3. Current capacity based planning for cancer early diagnosis and screening (Adapted from Guide to Cancer Early Diagnosis. World Health Organization 2027)
In most resource limited settings early diagnosis is limited and screening is often ineffective. In such situations, the optimal strategy will be to enhance capacity to diagnose cancer and to get them treated without delay. Mortality-to-incidence ratios from breast cancer decreased dramatically, even before the generalized use of mammography or adjuvant chemotherapy and antioestrogen therapy that commenced in the mid-to late 1970 in the United States of America. 17
Innovations and Digital Solutions
Over the past decades, there has been a growing use of Artificial Intelligence (AI) and digital technologies across cancer screening, diagnosis and care spectrum. 18 While this has generated a lot of interest particularly its potential to mitigate the HR capacity, logistics and health systems infrastructure gaps in resource limited settings, its use in most of these settings is limited to pilots and demonstration projects. The integration of AI technologies into national cancer screening programs requires health system augmentation and needs to address key areas like policy guidelines, regulatory compliance, data security, ethical considerations in addition to clinical validation. AI models are currently being used for interpreting screening test results, for example in cervical cancer, breast cancer, and it is to be noted that algorithmic bias may be introduced if the datasets from high income countries are extrapolated to low-income populations, leading to false results and misdiagnosis.19,20
Learning by Doing
Countries should learn the practical challenges of a screening programme by carrying out small scale programme including in well-defined populations. People who are invited can be offered screening and learn the elements needed in running large scale programmes. Before introducing screening to lower levels of health care delivery system, there should be a clear understanding of what can be done in such facilities. Availability of an end-to-end solution for the person who is invited or checked should be ensured.
Testing and precancer treatment may not be a linear process and sufficient capacity should be available in the system along with the necessary diagnostic and therapeutic capacity to ensure that every person who is screened benefits from the programme and that harms are minimized.
Screening can lead to harm which is often not mentioned. False positive and false negative results can be life-changing situations for the person. Disease Control Priorities (DCP 3) chapter on cancer screening have indicated similar challenges and comments. 21
WHO Initiatives on Cervical and Breast Cancer Control
WHO Cervical Cancer Elimination Initiative has provided good guidance and operational recommendations. Even with this detailed guidance, countries have to check their systems and their ability to justice to the complex process of screening and precancer management. Affordable and easily accessible clinically validated HPV testing and health system capacity to triage and manage test positives might need significant enhancements in the current health system capacity. Guidance on introducing HPV testing is a welcome effort in this domain. 22
The Global Breast Cancer Initiative (GBCI)
This initiative has provided guidance in a comprehensive manner and includes timely breast diagnosis as a pillar. An operational approach illustrating the steps and time taken from this initiative will help countries to plan their interventions in the national context. 23
Assessment of Prerequisites for Cancer Screening

What Will Success Look like?
Impactful screening programmes in the world are seen in high income countries with relatively small populations. In these countries screening programmes have been in place for over 50 years such as in Denmark, Finland, Norway and Sweden. 24 A lower middle income country with more than 50 million population need to check its readiness before taking the decision to screen.
Figure 4 illustrates the pathway of the screening program, starting from the community and moving through various steps, highlighting the rate-limiting factors at each stage. A similar systems approach may be considered to understand the pathway within the local context. Screening pathway with rate limiting steps (Image by Autor)
Cancer screening pathway should be planned as an ‘end to end’ solution and all steps should be ensured including the large numbers, to be handled. This is possible if enough resources are in place and the system is made efficient. Bhutan has shown very high coverage rates and detection of cancers and precancers with adequate management of all those who needs it. 25 Sustainability and quality assurance across the screening and treatment cascade remain important considerations of such programmes.
Conclusion
‘Screening for cancer’ is a ‘best buy’ intervention, provided the country is ready to implement the same effectively. Models of small-scale programmes directly under a cancer centre which has adequate capacity might be a start and will serve as a reality check. 26 Before making a decision to screen the entire population, countries can conduct an implementation research, identify what is required and proceed in a phased manner ‘learning by doing’.
Supplemental Material
Supplemental material - Application of Cancer Screening Guidelines in Resource Limited Settings
Supplemental material for Association of State and Local Social and Public Health Spending With Cancer Incidence and Mortality by Cherian Varghese, PhD, Baridalyne Nongkynrih, MD, Ayush Lohiya, MD, Ashmitha Prasad, MPH and Prebo Barango, MD in Cancer Control
Footnotes
Acknowledgments
The authors would like to thank their respective institutions for academic support during the preparation of this manuscript.
Author Contributions
CV conceptualized the manuscript and contributed to drafting, supervision, and critical revision of the manuscript. BN was involved in conceptualization, drafting, and reviewing edits. AL contributed to investigation, drafting of specific sections, and manuscript revision. AP was involved in data curation, content synthesis, drafting, and coordination of revisions. PB provided critical review and contributed to sections on current development. All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
