Abstract
Introduction
In Japan, while public health insurance ensures evidence-based-care, high-cost private medical practices offer advanced interventions—collectively referred to as “X” in this study—which have limited scientific evidence but have raised ethical and clinical concerns, particularly in oncology. This study explores oncologists’ perceptions and responses to X in practice to inform appropriate countermeasures.
Methods
Members of the Japanese Society of Medical Oncology (n = 6273) completed a questionnaire survey between April 24 and June 10, 2024. The survey explored the oncologists’ evaluations of X, their responses to patient inquiries about such interventions, and their personal preferences if they were personally diagnosed with cancer.
Results
Of 828 valid responses (response rate: 13.2%), 63.4% of oncologists reported receiving patient inquiries about X in the past year. Overall, 76.4% gave a negative evaluation of X, while over 90% provided negative ratings on 7 out of the 9 specific items. Despite this, many oncologists reported a neutral stance when discussing X with the patients. Notably, even without standard of care options, most indicated that they would not choose X for themselves. The findings reveal a considerable gap between oncologists’ evaluation of X and what they convey when communicating with the patients. Qualitative responses expressed concerns about insufficient follow-up care and potential exploitation of vulnerable patients.
Conclusion
This study highlights the need for more transparent, evidence-based communication between oncologists and patients regarding X. Enhancing such dialogue is essential for supporting informed decision-making and maintaining the integrity of cancer care in the face of unproven private medical interventions. Furthermore, considering X’s profound negative impact on patients, society, and the field of medicine, legitimate medical professionals must conduct clear and accurate evaluations of X to facilitate the understanding of the patients and general public.
Plain Language Summary
Public health insurance already covers proven cancer care in Japan and yet, a growing number of clinics sell costly cell, gene, or vaccine therapies that lack solid evidence, prompting patients to spend large sums in hope of a cure. To understand how front-line specialists perceive this trend, we invited all 6273 members of the Japanese Society of Medical Oncology to complete an anonymous online survey; 828 oncologists responded between April 24 and June 10, 2024. Nearly two-thirds (63.4%) answered that at least 1 patient had asked about these “X” treatments in the past year. Although three-quarters (76.4%) judged the therapies negatively and only 3.3% viewed them positively, many doctors softened their language during consultations: 30.9% explained neutrally while standard evidence-based options were still available, and 63.5% remained neutral even after those options had run out, citing patient autonomy or a wish to avoid conflict. Privately, however, most would refuse X for themselves: 93.5% while standard of care existed, and 73.3% even after all approved therapies failed. Their main worries centered on its scientific inefficacy, very high costs, weak follow-up care, misleading advertising, non-specialization of practitioners, and the risk that desperate patients are exploited. The findings reveal a hidden communication gap: doctors distrust these unproven treatments yet often hesitate to voice strong warnings, which could leave patients unclear about the real risks and benefits. Clearer, evidence-based conversations, supported by professional associations’ statements, are needed so that people with cancer can make truly informed decisions and avoid unnecessary financial and health harm.
Keywords
Introduction
In Japan, a well-established public health insurance system ensures the availability of safe and effective treatments. Therefore, patients have access to effective “standard of care” (ie, the best state-of-the-art medical care) involving a relatively low financial burden. In contrast, in “private medical practice,” medical care is not covered by public health insurance but paid for entirely by the patient. It involves advanced medical technologies—such as regenerative medicine, gene therapy, and immunotherapy—which, although lacking sufficient scientific evidence, are provided at the patient’s request and typically involve high out-of-pocket costs.
The Japan universal public health insurance system’s treatment coverage is determined through a centralized national process. New drugs and technologies are first reviewed by the Pharmaceuticals and Medical Devices Agency for safety and efficacy, then assessed by the Ministry of Health, Labour and Welfare for inclusion in the National Health Insurance (NHI) list. However, unlike in the UK’s National Institute of Health and Care Excellence, for Japan’s NHI list, explicit cost-effectiveness thresholds are not uniformly applied. Since 2019, Japan introduced the Health Technology Assessment framework; however, rather than for initial reimbursement decisions, it is mainly used for post-launch price adjustments. Through this framework, most evidence-based treatments are widely accessible, with patients paying only 10%-30% of costs depending on age and income. In contrast, unproven interventions outside this process are not covered by NHI and remain entirely patient-funded in private practice.
Caulfield introduced the concept of “scienceploitation” to describe the promotion of advanced medical technologies without robust supporting evidence. The term “scienceploitation” is coined by combining the words “science” and “exploitation.” Some physicians present themselves as experts, as if scientific evidence exists on their proposed therapies, and receive large sums of medical fees from patients, but in reality, the therapies are not recognized by the medical community and do not work. Such fraudulent activities by self-proclaimed experts cause the entire medical community to lose social trust.1-6 In response, researchers worldwide have reported on this problematic situation,7-11 with some studies focusing on oncology.6,12 In the United States, the Food and Drug Administration (FDA) has expressed concern about the increasing number of clinics offering unproven stem cell-based interventions. 13 In Japan, reports have highlighted issues such as exorbitant fees for gene therapy that appear ineffective for cancer, 14 serious adverse events (eg, septic infections) following regenerative medicine, 15 and legal cases in which patients have successfully sued the treating institutions.16,17 Moreover, cell-based interventions are classified as “regenerative medicine” in Japan and must be conducted under the Act on the Safety of Regenerative Medicine (ASRM). Although this law also applies to immuno-cell therapy—an unproven treatment commonly offered to cancer patients, involving the culturing and reinfusion of a patient’s own immune cells—serious concerns have been raised regarding how effectively these regulations are being implemented.18,19 Such problematic cases can be considered exploitative, profit‐driven activities that embody scienceploitation.
Therefore, we focused on oncology—a field with widespread private medical practices employing advanced interventions—to explore potential social countermeasures against the growing issue of such practices promoting advanced medical technologies without established scientific evidence. For instance, as of March 2021, there were 779 plans for “cancer immunotherapy,” classified as regenerative medicine under the ASRM and registered with the government, 20 making oncology one of the areas where regenerative medicine is most frequently employed.
In this study, we coined the placeholder “X” to denote advanced medical interventions for cancer patients (eg, cancer vaccines, regenerative medicine, gene therapy, and immunotherapy) that are relatively expensive, lack established scientific evidence, and are primarily offered in private medical practice outside the framework of clinical research. We introduced this shorthand due to the inadequacy of existing terminology: the commonly used term “unproven intervention” is overly broad, encompassing even traditional medicine, while limiting our focus to a predetermined list of interventions would risk overlooking emerging practices and unduly narrowing the scope of inquiry. Therefore, “X” serves as an analytic category that enables us to examine the perceptions of oncologists concerning these diverse, rapidly evolving practices without presupposing the related boundaries.
We then conducted a questionnaire survey of oncologists affiliated with the Japanese Society of Medical Oncology (JSMO) on their perceptions, evaluations, and responses regarding X in clinical practice. The aim was to elucidate oncologists’ evaluations of X, their responses to patients’ inquiries about X, and their personal treatment preferences if they were to develop cancer, thus highlighting the gap between expert judgment and actual clinical practice, and providing an empirical foundation for discussion on future ethical, legal, and social issues (ELSI).
Materials and Methods
Research Participants and Data Collection
We conducted a cross-sectional questionnaire survey targeting 6273 oncologists affiliated with the JSMO. This society was selected because many of the interventions covered in this survey involve internal medicine approaches—such as the administration of drugs and cells—and its members are well positioned to evaluate such treatments. Furthermore, the society as the principal body has published comprehensive guidelines pertaining to cancer immunotherapy, 21 including immune cell therapies frequently administered in private medical practice.
The survey was administered from April 24 to June 10, 2024. Participants were recruited and reminded via email and postcard from the society’s secretariat. The questionnaire was created using Google Forms and was designed to ensure respondent anonymity; thus, it did not include any questions that could identify individuals, and respondents were free to omit any questions regarding their attributes. Although the survey did not fall under the scope of the “Ethical Guidelines for Human Subjects in Life Science and Medical Research” in Japan and did not require institutional review board approval, it was conducted with the approval of the society. We provided explanations to the JSMO members (potential respondents), including the purpose of the survey (to clarify their views on cancer treatments related to X, publish research papers, disseminate information widely to society, and contribute to better treatments for patients), that privacy would be ensured (ie, respondents’ personal information would not be collected), that participation was voluntary, and that there was no remuneration for participating. Only those who ticked the online consent button after reading the explanation had access and could complete the questionnaire. The reporting of this study conforms to STROBE guidelines. 22
Questionnaire Items
The questionnaire was developed through a collaborative process: an initial draft was created by a legal sociologist (TI) and a psychometrician (TH), both drawing on their prior experience and research interests in conducting empirical studies on the ELSI of X.18,19,23 Its content validity was then examined through review by coauthors with relevant expertise—including 2 medical oncologists (NK, TS), a specialist in cancer communication (TT), and a bioethicist (MF)—who also provided feedback to refine the content so that it would appropriately capture issues commonly encountered in cancer care settings.
In this study, evaluation of the interventions referred to as “X” was based on a 27-item questionnaire, structured as follows: • Q1 (6 questions): Basic attributes of the respondents. • Q2 and Q3 (4 questions): Respondents’ experiences with cancer patients inquiring about X. • Q4 (9 questions) and Q5 (1 question): Items evaluating X. Respondents rated X on a 4-point Likert scale on 9 specific items: (1) scientific efficacy, (2) alleviation of psychological suffering, (3) price, (4) advertising, (5) mass media coverage, (6) patients’ understanding and self-determination, (7) the expertise of X providers, (8) patient support before receiving X, and (9) patient support after receiving X. They also provided an overall three-choice evaluation of X. • Q6 and Q7 (2 questions each): Respondents’ stance on X as a treatment option, designed to distinguish between available vs unavailable standard of care occurrences, and between scenarios involving patients vs respondents themselves. • Q8 (2 questions): Items addressing medical interventions that correspond to or are similar to X. The first was a multiple-choice format question with specific examples. The second was an open-ended question requesting free-form descriptions of them. • Q9: An open-ended question requesting free-form descriptions of memorable cases related to X.
For additional details, please refer to the Supplemental Material. The definition of “X” was displayed on each screen presenting the questions.
Data Analysis
The collected data were analyzed using descriptive statistics and cross-tabulations. A chi-square test was employed to assess the relevance and independence of the variables, at a significance level of P < 0.05. For the open-ended questions (Q6 and Q9), multiple authors (TI and TH) reviewed the responses, identified common characteristics across the cases and the underlying concerns expressed by the respondents, and categorized the descriptions accordingly. In particular, for the exploratory analysis of the open-ended responses to Q9, we coded the responses deductively based on the 9 items in Q4. These categorizations were then verified by all authors. IBM SPSS Statistics (version 27.0.1.0) was used for data processing and analysis.
Results
Respondent Characteristics
Respondent Characteristics (n = 828)

JSMO, Japanese Society of Medical Oncology; SD, standard deviation; Min, minimum; Max, maximum; Tokyo, the capital of Japan; Prefectures with government-designated cities, cities with a population of ≥500 000, granted special administrative authority under the law.
Respondents’ Experience of Being Consulted by Patients
Respondents’ Experiences of Cancer Patients’ Inquiries About X

aAmong the initial open-ended responses listed as “Other,” 29 responses indicated “1 patient,” 19 responses indicated “2 patients,” 11 responses indicated “3 patients,” 1 response indicated “4 patients,” and 1 response indicated “5 patients.” These 61 responses were categorized by the authors as “Yes, I think so.” In addition, 2 responses indicated uncertainty and were categorized as “I’m not sure.” The remaining 2 open-ended responses under “Other” were as follows: “None in the past year, but there were some in earlier years”; and “There are probably almost none, but those who ask tend to be terminally ill, and I have not been able to follow up with them sufficiently afterward.”
bAmong the initial open-ended responses listed as “Other,” there was 1 response indicating “0 patients,” 10 responses indicating “1 patient,” 4 responses indicating “2 patients,” 1 response indicating “3 patients,” and 11 responses indicating “Don’t know.” The single response of “0 patients” was categorized by the authors as “No, I don’t think so,” the 15 responses indicating 1 to 3 patients were categorized as “Yes, I think so,” and the 11 “Don’t know” responses were categorized as “I’m not sure.” The remaining 5 open-ended responses under “Other” were as follows: “It may have contributed to life prolongation”; “Treatment was declined”; “The patient only had a consultation but did not receive treatment”; “The patient had a consultation but did not undergo X”; and “I believe there were patients who gained psychological stability.”
Of the 525 oncologists, 383 (73.0%) believed that the patients had actually visited an institution offering X (Table 2, Q3-2). Among these 383, 30 (7.8%) responded affirmatively and 337 (88.0%) negatively regarding whether any patients had experienced a therapeutic benefit from X (Table 2, Q3-3).
Regarding their response to a patient who had visited another institution for X, 166 oncologists (43.3%) stated they would “not actively follow up but would respond if the patient visited the hospital.” However, 183 (47.8%) would “continue to follow up and encourage the patient to return to their own institution,” while 34 (8.9%) would “not follow up at all” (Table 2, Q3-4).
Oncologists’ Evaluation of X
More than 90% of respondents expressed negative views regarding all of the following 7 aspects: scientific efficacy, pricing, advertising, mass media coverage, patients’ understanding and self-determination, expertise of X providers, and patient support after receiving X (Figure 1). Conversely, some respondents expressed positive opinions regarding the alleviation of psychological suffering due to X, as well as the pre-treatment support provided by doctors. Evaluation of Nine Specific Aspects of X by Oncologists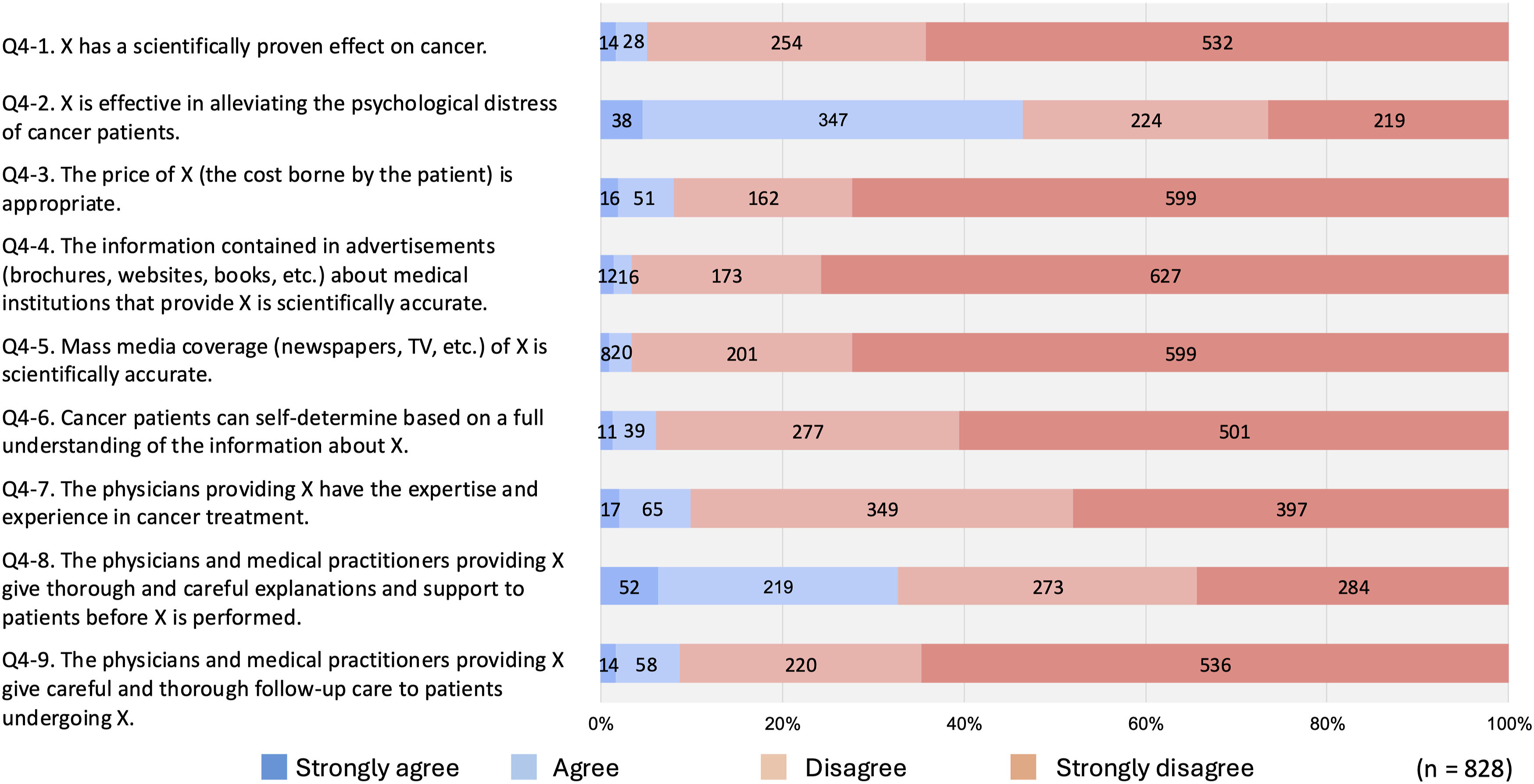
Respondents’ Evaluation and Attitudes Toward X (n = 828)

aFor both questions Q6-1 and Q6-2, responses categorized as “Other” were analyzed based on the content written in the free-text field. Excluding 2 and 4 entries from each question respectively that were unintelligible, it was inferred that respondents selected “Other” for 1 of 2 reasons: either they wished to express a clear negative, neutral, or positive stance along with a specific rationale, or they felt unable to take a general stance because their evaluation would depend on the details of X or the condition of the patient.

The Oncologists’ Evaluation of the Nine Specific Aspects of X, according to Their Overall Evaluation
Respondents’ Explanation and Attitude Toward X After Listening to the Patients Concerns, With or Without Standard of Care
When asked how they would explain X to a patient, as shown in Table 3 (Q6-1, 6-2), respondents differed significantly in their explanation stance based on the availability of standard of care. When standard of care treatment options remained, the respondents’ stance was mainly to discourage, whereas when the options were exhausted, the respondents’ stance was neutral (χ2(9) = 716.9, P < 0.01; Table 3).
Cross-Tabulation of Explanation/Attitude Toward the Patients and Overall Evaluation of X if the Standard of Care is Available as a Treatment Option (n = 828)

Cross-Tabulation of Explanation/Attitude Toward the Patients and Overall Evaluation of X if the Standard of Care is Unavailable as a Treatment Option (n = 828)

Percentages may not sum up to 100% due to rounding.
Preferences if the Oncologists Were the Cancer Patients
When asked to assume that they were the cancer patients, as shown in Table 3 (Q7-1, 7-2), most respondents would not choose X for themselves in either scenario, although reluctance lessened when standard of care was considered unavailable (χ2(4) = 201.9, P < 0.01; Table 3).
Cross-Tabulation of the Oncologists’ Attitudes Toward Explaining X and What Their Preferences as Patients Would be if the Standard of Care is Available as a Treatment Option (n = 828)

Percentages may not sum up to 100% due to rounding.
Cross-Tabulation of the Oncologists’ Attitudes Toward Explaining X and What Their Preferences as Patients Would Have Been if the Standard of Care is Unavailable as a Treatment Option (n = 828)

X Modalities and Therapies Similar to X Considered Problematic by the Oncologists
Modalities of X Considered Problematic by Respondents (n = 828)

NK, natural killer; NKT, natural killer T.
aThis question allowed multiple responses. 2 respondents selected both “None is problematic” and one or more other options. In such cases, “None is problematic” was not counted.
Furthermore, X was defined by 3 criteria: the promotion of advanced medical care, a lack of established scientific evidence, and relatively high treatment costs. When asked about “other problematic interventions lacking established scientific evidence that do not meet the criteria for X” (Q8-2), 391 free-text responses mentioned interventions such as high-dose radiation, Maruyama vaccine, low-dose immune checkpoint inhibitors, high-dose vitamin C, O-rings, complementary and alternative medicine, health foods, chemotherapy lacking evidence, and carbohydrate restriction.
Illustrative Cases From Open-Ended Responses
A total of 378 respondents to Q9 (45.7%) provided free-text responses describing notable cases or experiences related to X. Of these, 323 responses were deemed valid (ie, the content was understandable and coherent). The main trends observed in these responses are summarized below (note that the following classifications are not mutually exclusive; some responses overlapped across categories).
Of the 323 valid responses, 131 (40.6%) responses indicated that institutions providing X did not offer appropriate aftercare or take responsibility for managing adverse events following its administration, and that such care was left with the oncologists or institutions operating under the public health insurance. In contrast, only one response (0.3%) revealed that X-providing institutions managed aftercare and adverse events appropriately.
Second, 99 (30.7%) responses indicated that X lacked scientific evidence and/or had unclear efficacy, or mentioned the names of such interventions. In contrast, 17 (5.3%) responses mentioned successful cases involving X and expressed a positive view regarding its evidence or effectiveness.
Third, 62 (19.2%) responses described concerns about X in relation to standard of care and clinical research or trial. These responses fell into 2 categories: (1) cases in which the combination of X and standard of care negatively affected the implementation of the standard of care—such as the occurrence of adverse events, obstacles to the delivery of standard of care, and the loss of opportunities to receive standard of care or participate in clinical research or trials; and (2) cases in which the patients recovered after receiving both standard of care and X, but the institutions providing X claimed that the improvement was the result of X.
Fourth, 60 (18.6%) responses indicated that the cost of receiving X was extremely high.
Fifth, 31 (9.6%) responses included harsh expressions criticizing institutions providing X or describing the difficult circumstances of patients. These responses included phrases such as “deceiving patients,” “fraudulent business,” “money-driven,” “criminal,” “taking advantage of patients’ vulnerabilities,” “completely meaningless and malicious,” “patients suffered tragic consequences,” “physicians providing X coldly rejecting patients,” and “patients racked up debt and relationships with close relatives disintegrated.”
Discussion
Although several surveys have explored the attitudes of healthcare professionals toward complementary and alternative medicine, 24 to the best of our knowledge, no international survey has specifically targeted healthcare professionals’ views on cutting-edge medical interventions that are offered as therapies without supporting scientific evidence. Our survey revealed experts’ perspectives on X based on their real clinical experience with patients, with > 60% reporting patient consultations within the past year. More than three-quarters evaluated X negatively, while approximately 3% expressed positive views. Notably, some respondents offered explanations to patients that contradicted their personal evaluations of X. Our explorative analysis of the free-text revealed the seriousness with which the respondents viewed the challenges posed by X to both patients and healthcare providers. Based on the survey results, we aim to further examine experts’ evaluations of X and offer insights into how healthcare professionals may approach patient communication regarding X.
Negative Evaluation of X by the Oncologists
We positioned Questions 4 and 5, which directly examined oncologists’ evaluations of X, as central in this study. First, in 7 out of the 9 specific items assessing X, over 90% of oncologists evaluated X negatively (Figure 1). These results, considering the exploratory analysis of the open-ended responses to Q9, suggest that the respondents—experts in cancer treatment—perceived the following: (1) X, which lacks scientifically demonstrated therapeutic effects, is being administered by physicians who lack adequate expertise or experience, and without providing proper care for symptom exacerbation or adverse events experienced by patients; (2) the pricing of X is inappropriate; and (3) promotional materials issued by X-providing institutions, as well as media reports on X, are inaccurate, making it difficult for patients to understand it correctly or make appropriate autonomous decisions.
Caution is warranted in interpreting the results of the following 2 items. First, 271 respondents (32.7%) evaluated the pre-treatment explanations and communication by X-providing institutions as attentive and courteous (Figure 1, Q4-8). However, given the finding that these institutions do not offer adequate aftercare or manage adverse events (Figure 1, Q4-9), it is possible that their attentiveness is limited to the pre-treatment phase, serving as a strategy to recruit patients. This interpretation is supported by a Japanese court case in which a patient who had received X sued the hospital and won; the court’s findings described the hospital’s conduct in similar terms. 17
Second, 385 respondents (46.5%) believed that X had some relevance in alleviating the psychological distress of cancer patients (Figure 1, Q4-2). However, considering the widespread lack of patients’ understanding and autonomous decision-making regarding X (Figure 1, Q4-6), oncologists may regard this alleviation as a type of placebo effect. This interpretation is supported by the fact that most respondents rejected the scientific efficacy of X (Figure 1, Q4-1); moreover, of the free-text responses to Q9, not a single entry both affirmed the psychological benefit of X and supported the existence of scientific therapeutic efficacy. It can thus be inferred that if patients were aware of the expert evaluations revealed by this study—or if individual oncologists properly communicated the scientific assessment of X to their patients—then the placebo effect associated with psychological relief might be diminished. Furthermore, if even the placebo effect cannot be expected, it would no longer be justifiable to approve of X on the grounds that it provides “peace of mind” to patients who have exhausted standard of care options.
Notably, 633 respondents (76.4%) gave an overall negative evaluation of X (Table 3, Q5). Meanwhile, 168 respondents (20.3%) selected the neutral response, “Cannot evaluate/Unsure,” which should not be interpreted as the absence of a negative evaluation. Rather, as discussed in the next section, this response can be understood as a scientifically cautious stance—one that is withholding judgment on therapies whose efficacy remains unclear from a scientific perspective.
Evaluation of Specific Items on X by the Respondents
The detailed item-by-item assessments of X by each group—who evaluated X either positively, neutrally or undecidedly, or negatively—were also clarified (Figure 2).
In the positively evaluating group (n = 27), respondents were generally favorable toward X across all 9 items, clearly differing from the other 2 groups. In particular, 25 respondents (92.6%) in this group valued “the alleviation of psychological distress through X.” Furthermore, 21 respondents (77.8%) acknowledged each of “scientific therapeutic efficacy of X” and “the professional expertise of physicians administering X,” suggesting that their views on science, medicine, and clinical practice differ significantly from those of the other 2 groups.
In contrast, the negatively evaluating group (n = 633) exhibited a trend consistent with the overall findings described earlier. At least 607 respondents (95.9%) expressed negative evaluations on 7 of the 9 items, and approximately 60%-70% did so on the remaining 2 items. By contrast, although the neutral or attitude-reserved group (n = 168) expressed a neutral overall evaluation of X, their item-specific responses closely resembled those of the negatively evaluating group. This suggests that the neutral stance toward X indicated by respondents in this study is, in effect, nearly equivalent to a negative evaluation.
Discrepancy Between Internal Evaluations and Explanations to the Patients
These overall evaluations of X by oncologists did not necessarily manifest in their explanations or clinical attitudes toward individual patients. The findings also suggest that oncologists’ attitudes toward explaining may vary depending on whether standard of care options remain available to the patients. For example, among the 256 oncologists who reported adopting a neutral explanatory attitude toward X in cases where standard of care options were still available, more than half (141, or 55.1%) actually held a negative evaluation of X (Table 4). This tendency was even more pronounced when standard of care options were unavailable: among the 526 oncologists who adopted a neutral explanatory stance toward X, 362 (68.8%) evaluated X negatively (Table 5). In other words, when treatment options are exhausted, a negative evaluation of X may be perceived by oncologists as a form of “bad news” that is more difficult to communicate to patients. Delivering bad news is considered an emotional burden for oncologists themselves, according to Fallowfield and Jenkins, 25 and numerous studies have explored ways to improve communication skills to address this issue. 26 A similar emotional dynamic may be at play in the present context as well.
This result may reflect a number of considerations by oncologists, such as an intention to interact with patients from a professional and objective standpoint without inserting personal values, a wish to respect the autonomy of patients who hope to undergo X, or a concern about avoiding conflict that might lead to discontinuation of treatment. However, if oncologists’ neutral explanations and attitudes result in patients not fully understanding their professional evaluations of X, this poses a significant problem. This concern is supported by the finding that more than 70% of oncologists who had been consulted by patients about X later reported that the patients had actually visited an institution providing X (Table 2, Q3-2).
Akagi et al, in a questionnaire survey of bereaved families of cancer patients, 27 reported that patients who had discussed unapproved and unproven cancer treatments (UUCT) with their primary doctors were more likely to undergo UUCT. Contrary to expectations that patients lacking trust in their doctors would be more inclined to seek UUCT, the authors interpreted the finding as indicating that “doctors provided the correct information, but they could not deter the patients from undergoing UUCT.” However, we hypothesize that one of the factors contributing to the failure to dissuade patients from undergoing UUCT was that many doctors provided information about X with a “neutral attitude.” Akagi et al.’s survey found that among the patients who underwent UUCT, 61% had consulted with their primary doctors, and although 50% were told that it might not be effective, 76% responded that their doctors did not oppose their decision to undergo UUCT.
When patients consult their primary oncologists about X, they may be seeking not only objective facts such as the response rate to X but also value judgments about whether those facts make the therapy worth trying. Therefore, if oncologists merely convey facts from a neutral and objective stance, patients may interpret this as “support for receiving X” or “at least no opposition.” Such miscommunication has also been confirmed in interview studies involving cancer patients and oncologists. 28 Even when a treatment option lacks a scientific basis, failure to clearly deny its effectiveness can lead patients to form misguided hopes. Furthermore, in the framework of “shared decision making” proposed by Elwyn et al, 29 the physician’s role is to support patients in thoughtfully considering their options. If all responsibility for decision-making is transferred to the patient, they may feel “abandoned.” Thus, clearly conveying an oncologist’s own evaluation of X does not hinder patient autonomy but can in fact support it. If this is the case, then rather than intentionally maintaining a neutral stance, oncologists might communicate information about X more effectively by openly stating their own value or judgments. Moreover, if such communication can be achieved at a stage when standard of care options are still available, it may help reduce the psychological burden on oncologists.
Differences in Oncologists’ Stance When Considering X for Others vs Self
It was also revealed that oncologists’ stance toward X differs depending on whether they are explaining it to cancer patients or considering it as a treatment option for themselves as hypothetical cancer patients (Table 3). While differences were also observed depending on whether standard of care remained available, when the respondents imagined themselves as cancer patients, 774 (93.5%) and 605 (73.1%) oncologists said they would not undergo X whether standard of care options were available or not, respectively (Table 3, Q7-1 and Q7-2). These results are substantially higher than the number of oncologists who stated they would oppose X if consulted by a patient—535 (64.6%) and 250 (30.2%) for the respective conditions (Table 3, Q6-1 and Q6-2). Furthermore, among the 256 oncologists who reported that they would explain X in a neutral manner to patients when standard of care options were available, 220 (85.9%) would personally not undergo X if in the same situation (Table 6). Even when no standard of care options were available, 334 out of 526 oncologists (63.5%) who would explain X in a neutral manner still said they would not undergo X themselves (Table 7). In other words, most oncologists do not disclose to patients that they would not choose X for themselves, even though that is their personal position. This aligns with the earlier finding that oncologists’ overall evaluations of X are not necessarily reflected in their explanatory content and attitude toward patients.
This result may be understood as an expression of values emphasizing respect for the autonomy of patients as others, and such an attitude by oncologists should be regarded as admirable. At the same time, when patients ask the common question, “Doctor, if you were in my position, would you undergo X?” it may be helpful for oncologists to respond not only with their own personal views, but also by sharing the results of this study—namely, that an overwhelming majority of oncologists would not choose X as a treatment for themselves. Among the free-text responses to the questions about explanatory attitudes toward X when consulted by patients (Q6-1 and Q6-2), many stated that they would “review the evidence for X” or “explain that it lacks scientific evidence.” However, if such patient inquiries are seen as expressions of the deep distress of having to decide on a cancer treatment based on objective information about X, it may be more desirable for oncologists to offer responses or advice that include their own subjective stance—such as, “I wouldn’t undergo X myself, and it seems that most oncologists felt that way.”
Inadequate Follow-Up by X-Implementing Institutions and Increased Burden on Oncologists
Of the 828 respondents, 525 oncologists—approximately two-thirds—had actually encountered the issue of how to respond to patients with expectations about X within the past year (Table 2, Q2). When patients actually undergo X, oncologists are confronted not only with communication challenges but also with clinical management issues. Nevertheless, contrary to our expectations, fewer than 10% of oncologists chose not to provide follow-up care to patients who had visited institutions offering X (Table 2, Q3-4). Although there were differences in the degree of willingness to follow up such patients, more than 90% of oncologists demonstrated a willingness to address problems that arise when patients undergo X.
However, 756 of the 828 respondents (91.3%) negatively evaluated the follow-up care provided by X-offering institutions (Figure 1, Q4-9). Furthermore, according to the free-text responses to Q9, oncologists expressed concerns not only about the need to manage deterioration in the patient’s condition or adverse events following treatment with X, but also about the potential for such therapies to interfere with standard of care, and the possibility that the combination of such care and X may obscure the ineffectiveness of X. This perception of risk shares similarities with the approach taken by Stub et al, which distinguishes between direct and indirect risks associated with complementary therapies for cancer patients.
30
Some representative free-text comments are provided below. • They charge exorbitant fees and provide no follow-up whatsoever. They deliberately recommend combining X with standard of care, claim that any observed effects are due to X, and use such outcomes as promotional material to deceive the next patient. • There was a patient whose condition rapidly deteriorated and who passed away after undergoing X, but the institution claimed that the outcome was unrelated to the therapy and did not take any responsibility. In many cases, X is administered without any subsequent follow-up. • In cases where patients receiving standard of care also began X, any observed efficacy (likely attributable to the standard of care) was attributed to X, while adverse events were referred to our institution for management. (Facilities offering X refuse to treat anything beyond the X intervention.) • One of the key problems with providers of X is that, even if the patient’s condition worsens, they never take full responsibility through to the end. It seems that many institutions merely offer a therapy with no evidence and then take no further responsibility. • A patient with whom I had a good relationship began to reject standard of care when anticancer drugs were proving ineffective, and wished to transfer to a facility offering X. The patient later deteriorated and was transported back to our hospital via emergency services. The patient had developed deep mistrust toward medicine overall—likely influenced by the physician at the X-providing institution—including suspicions of collusion with pharmaceutical companies and profit motives, and passed away before we could rebuild our relationship. • One patient discontinued treatment at our hospital to undergo a cancer vaccine therapy as a private-pay medicine at another hospital; however, the therapy was ineffective and pain control was not provided. The patient returned to our hospital in distress. • In some cases, X was administered concurrently with chemotherapy, resulting in complications such as fever. This occasionally interfered with timely introduction to hospice care during the terminal stage. • Without obtaining permission from the attending physician or home-care doctor, a low dose of 5-fluorouracil (FU) was mixed into an at-home intravenous hyperalimentation (IVH) infusion for a terminally ill patient. Unexplained hepatic dysfunction suddenly appeared, and after pressing the patient for answers, the truth came to light. I still cannot forgive this. • In the case of a breast cancer patient, a 3-month course of high-dose vitamin C therapy was administered preoperatively (at the facility offering X). As expected, the tumor increased in size, and surgery was eventually performed. However, the patient subsequently refused to accept standard of care, and as a result, adequate postoperative adjuvant and recurrence treatments could not be provided.
Given such negative evaluations and concerns, the burden on oncologists who continue to provide follow-up care for patients who have undergone X is likely to be considerable. Insights into how oncologists who prefer that their patients not receive X can approach and communicate such to their patients were also obtained from the free-text responses to Q9. Several respondents reported that they explicitly informed patients that their institution would be unable to continue treating them if they proceeded with X, and that any adverse events or side effects resulting from X should be managed by the institution providing X. These responses suggest that, in some cases, patients reconsidered undergoing X after receiving such explanations.
Limitations and Significance of This Study
This study has 2 procedural limitations that might affect the validity of the findings. First, to encourage candid responses, we did not collect personally identifiable or demographic information from respondents, such as their JSMO membership numbers, age, and sex. Therefore, this study could not examine whether evaluations of X varied across these characteristics. Moreover, we cannot completely rule out the possibility that a single respondent may have submitted multiple responses to the online survey. However, we clearly stated in the survey request that only one response per person was allowed; therefore, we believe that the likelihood of multiple responses is low.
Second, despite repeated reminders, the valid response rate was relatively low at 13.2%. Nevertheless, it is noteworthy that 378 of the 828 respondents voluntarily provided memorable or noteworthy open-ended descriptions of cases involving X. This suggests that several respondents may have had particularly strong concerns or awareness regarding X. Although this potential for sampling bias should be taken into consideration when interpreting the findings―particularly the notably low evaluations of X―the study nevertheless provides valuable insights into real-world issues related to X in cancer treatment practice.
Conclusion
The results of this survey indicate that cancer oncologists hold strong concerns about X. More than three-quarters of those surveyed expressed negative evaluations of X, and even the approximately 20% who gave neutral evaluations could, in effect, be considered as largely holding negative views. In particular, over 90% of the respondents had negative evaluations on X regarding the scientific therapeutic efficacy, cost, promotional advertising, media coverage, patients’ understanding and capacity for autonomous decision-making, the expertise of the physicians administering X, and the patient care after the start of X. We hope that these findings will gain wide recognition as a professional evaluation of X by medical experts.
We also hope that patients and their close relatives will come to understand that oncologists’ attitudes and explanations about X during consultations may not always reflect their true personal evaluations of it. By learning how true cancer treatment specialists evaluate X, the challenges they face in communicating and providing medical care to cancer patients around X, as well as their private concerns about patients undergoing X, we hope that patients will be encouraged to reconsider or refrain from undergoing it.
In recent years, academic groups have expressed concern about medical interventions that fall under X. In May 2019, the JSMO expressed concern about the provision of unproven immune cell therapy and cancer vaccine therapy under private medical practice settings, without proof of efficacy or safety and without official insurance approval. 31 More recently, in the field of regenerative medicine, the Japan Medical Society of Spinal Cord Lesion issued a warning to patients and the public regarding the lack of evidence for regenerative medical treatments for spinal cord injuries offered as private medical practice. 32 Additionally, the International Society for Stem Cell Research issued a recommendation to Japan’s Ministry of Health, Labour and Welfare to strengthen regulations on private medical practice under the ASRM. 33 Considering the profound negative impact that X can have on patients, society, and the field of medicine, it is essential that legitimate medical professionals present clear and accurate evaluations of X in a way that is understandable to both patients and the general public.
Supplemental Material
Supplemental Material - Oncologists’ Views and Communication Practices Regarding Unproven High-Cost Cancer Interventions in Private Medical Practice: A Questionnaire Survey
Supplemental Material for Oncologists’ Views and Communication Practices Regarding Unproven High-Cost Cancer Interventions in Private Medical Practice: A Questionnaire Survey by Tsunakuni Ikka, PhD, Taichi Hatta, PhD Noriyuki Katsumata Tomoko Takayama Tatsunori Shimoi and Misao Fujita in Cancer Control
Footnotes
Acknowledgments
We would like to express our deepest gratitude to all members of Japanese Society of Medical Oncology for their cooperation in the survey.
Ethical Considerations
The survey did not fall under the scope of the “Ethical Guidelines for Human Subjects in Life Science and Medical Research” in Japan; hence an institutional review board approval was not required. However, it was conducted with the approval of the Japanese Society of Medical Oncology.
Consent to Participate
We provided explanations to the Japanese Society of Medical Oncology (JSMO) members (potential respondents), including the purpose of the survey, that privacy would be ensured (ie, respondents’ personal information would not be collected), that participation was voluntary, and that there was no remuneration for participating. Only those who ticked the online consent button after reading the explanation had access and could complete the questionnaire.
Author Contributions
Conceptualization, T.I.; Formal analysis, Methodology and Validation, T.I. and T.H.; Writing – original draft, T.I.; Investigation and Writing – review & editing, all authors; Supervision, M.F.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Scientific Research (grants numbers 21K10326 and 23K22074) and the Uehiro Foundation on Ethics and Education.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Noriyuki Katsumata, Tatsunori Shimoi and Tomoko Takayama are members of the Japanese Society of Medical Oncology.
Data Availability Statement
Data cannot be shared for ethical/privacy reasons.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
