Abstract
We conducted a survey of primary care clinicians and gastroenterologists (n = 938) between 11/06/19–12/06/19 to assess knowledge and attitudes regarding colorectal cancer screening. We assessed clinicians’ attitudes toward lowering the colorectal cancer screening initiation age to 45 years, a topic of current debate. We also evaluated provider and practice characteristics associated with agreement. Only 38.1% of primary care clinicians endorsed colorectal cancer screening initiation at age 45 years, compared to 75.5% of gastroenterologists (p < .0001). Gastroenterologists were over 5 times more likely than primary care clinicians to endorse lowering the screening initiation age (OR = 5.30, 3.54-7.93). Other factors found to be independently associated with agreement with colorectal cancer screening initiation at age 45 years included seeing more than 25 patients per day (vs. fewer) and suburban (vs. urban) location. Results emphasize the need for collaboration between primary care clinicians and gastroenterologists to ensure that patients receive consistent messaging and evidence-based care.
Keywords
Introduction
In the US, age 50 years is the historic initiation threshold for average-risk colorectal cancer screening by most professional organizations, including the US Preventive Services Task Force (USPSTF) and the Multi-Specialty Task Force (MSTF). 1 -3 In 2018, in response to evidence of increasing colorectal cancer incidence and mortality rates in younger populations 4,5 and results of microsimulation modeling, 5,6 the American Cancer Society issued a qualified recommendation to begin average-risk colorectal cancer screening at age 45 years. 7 Of note, the recommendation made by ACS was made as a qualified recommendation due to the lack of randomized clinical trial evidence to support population benefit. On October 27, 2020, the USPSTF posted a draft version of their updated colorectal cancer screening recommendations for a 4-week public comment period prior to final publication of their updated recommendations; the draft version updates prior recommendations with inclusion of a B recommendation for colorectal cancer screening among adults aged 45-49. While efforts have been made to reconcile disparate recommendations to inform clinical practice, 8,9 uniform consensus among healthcare providers (HCPs) has not yet been achieved with respect to the relative benefits and risks of lowering drawbacks the age of initiation. 10,11 The ACS recommendation emerged as a departure from the previously unanimous recommendation of CRC screening initiation at age 50 of the USPSTF and the MSTF and thus, sparked debate regarding the benefits and risks of earlier CRC screening initiation, particularly among specialists. 12 -14 Furthermore, the qualified ACS recommendation was widely covered in the media, with varying attention to the nuances and qualified nature of the recommendation. 15 -17 Thus, clinicians are tasked with making clinical recommendations for their patients in the absence of direct clinical trial evidence, with evolving guidance from recommending bodies, and in the context of incomplete or potentially misleading information about recommendations being conveyed to the public through various media. 8,9
Clinician recommendation is consistently identified as an important factor associated with completion of colorectal cancer screening and patients look to their healthcare providers for guidance on appropriate cancer screening. 18 It is therefore important to understand clinician perspectives on the appropriate age for average-risk colorectal cancer screening initiation and to ascertain whether perspectives differ by clinician or practice characteristics. We conducted a survey of clinicians in the U.S. to assess knowledge, attitudes and behavior related to colorectal cancer screening and herein report results of an analysis of these data to characterize current perceptions about lowering the initiation age for colorectal cancer screening to age 45 among average risk adults.
Methods
We developed a questionnaire using previously validated items from the National Cancer Institute’s Physician Survey on Colorectal Cancer Screening in addition to drafting new items to assess knowledge, attitudes, and behaviors related to colorectal cancer screening among HCPs. 19 The questionnaire was implemented as a web survey by the National Opinion Research Center (NORC) at the University of Chicago (http://www.norc.org). The study was reviewed and deemed exempt by the NORC IRB (#19.09.15NF). NORC partnered with Dynata, (www.dynata.com) who maintains a national survey panel comprised of more than 200,000 HCPs in the US.verified through matching to comprehensive national professional membership organizations such as the American Hospital Association ant the American Medical Association. Pretest interviews were conducted with 11 clinicians to confirm the questionnaire length and verify the survey programming prior to the main survey field period which occurred between 11/06/19–12/06/19.
Questionnaires were sent by email to 3,299 primary care clinicians and 538 gastroenterologists with up to 2 reminders sent within a 21-day period to non-responders. Surveys were completed by 814 primary care clinicians and 159 gastroenterologists resulting in completion rates of 25.3% and 29.6%, respectively. Nominal remuneration was provided to respondents, consistent with established panel protocols. In addition to questions regarding provider and practice characteristics, clinicians were asked: “Should the age to begin colorectal cancer screening for patients at average risk of developing colorectal cancer be lowered from age 50 to age 45?”
We used SAS version 9.4 for all analyses. Respondents who indicated specialties other than primary care or gastroenterology (n = 13) and those who reported that they do not recommend colorectal cancer screening to their average risk patients or if they reported a colorectal cancer screening mode preference other than gFOBT, FIT, Cologuard, or colonoscopy (n = 42) were excluded from our analyses, resulting in a final total analytic sample of 938. We summarized the frequency of responses and evaluated bivariate (Chi-square) and multivariable (logistic regression controlling for age, sex, race and income) associations of clinician- and practice-level factors associated with agreement with colorectal cancer screening initiation at age 45 years.
Results
Table 1 summarizes sociodemographic characteristics of both primary care clinicians and gastroenterologists. Table 2 summarizes the percentage of primary care clinicians and gastroenterologists who agreed to colorectal cancer screening initiation at age 45 years overall and by clinician and practice characteristics. While only 38.1% of primary care clinicians agreed with lowering the colorectal cancer screening initiation age to 45 years, 75.5% of gastroenterologists agreed (p < .0001). In multivariable analyses, gastroenterologists were over 5 times (OR = 5.30; 95% CI 3.54-7.93) more likely than primary care clinicians to endorse lowering the colorectal cancer screening initiation age to 45 years. Other factors found to be independently associated with agreement with lowering the colorectal cancer screening initiation age to 45 years included seeing more than 25 patients per day (vs. fewer) and suburban (vs. urban) location of clinic (Table 2).
Sociodemographic Characteristics for the Total Sample and by Clinical Specialty (n = 938).
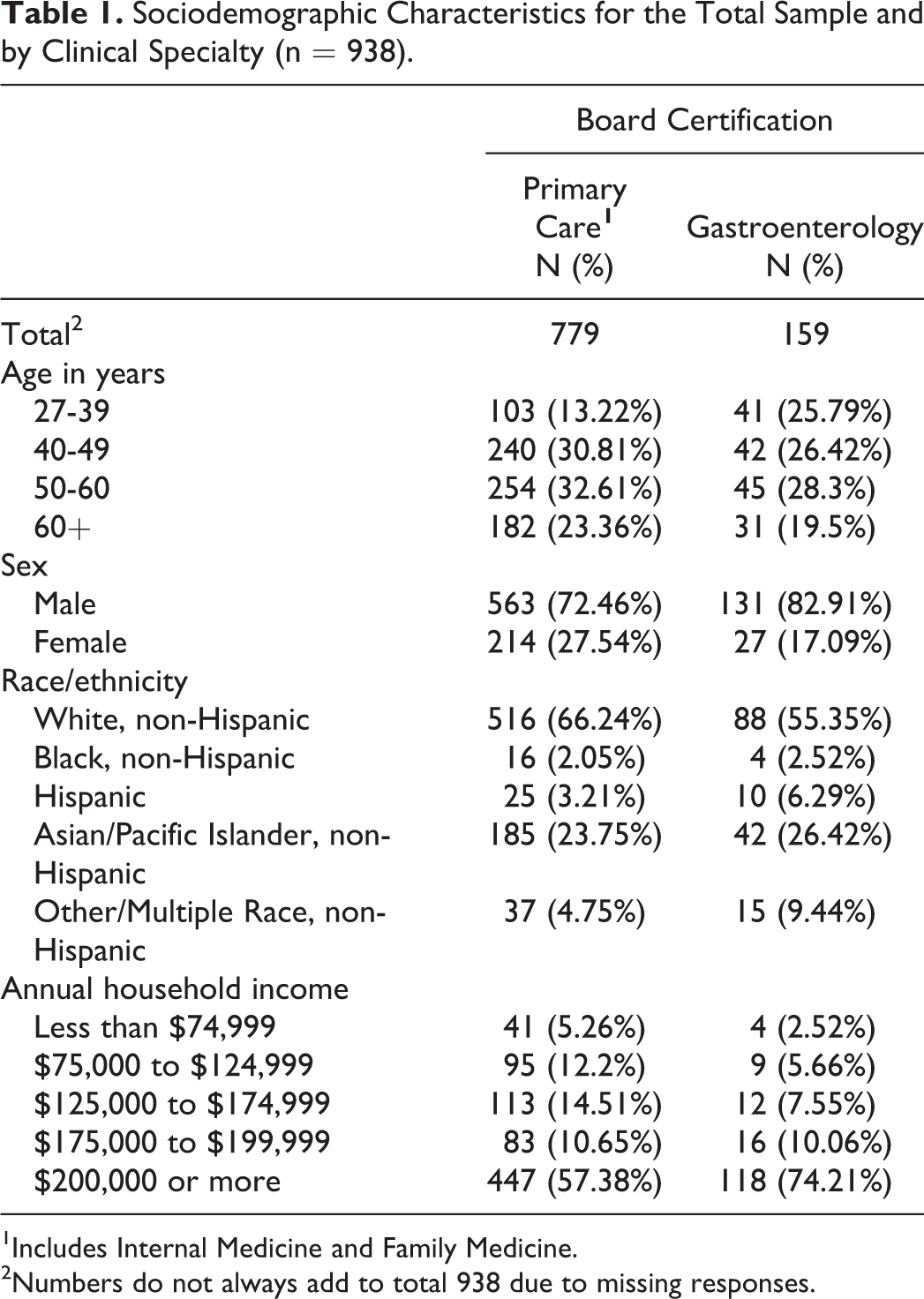
1Includes Internal Medicine and Family Medicine.
2Numbers do not always add to total 938 due to missing responses.
Primary Care Clinicians’ (PCCs) and Gastroenterologists’ (GIs) Preference for Reducing Screening Age for Average Risk Patients to Age 45 by Provider and Practice Characteristics.

1Adjusted for age (except in case of years of practice), sex, race, income, and type of provider.
Discussion
Novel data from this survey of PCC and GI clinicians drawn from a national panel of clinicians suggest that clinicians’ perspectives on the appropriate age for average-risk colorectal cancer screening initiation diverges by clinical specialty. Significantly greater support for initiation of colorectal cancer screening at aged 45 years was observed among gastroenterologists compared to primary care clinicians. The published literature suggests that gastroenterology specialists have been more actively involved in discussions around the pros and cons of earlier CRC screening initiation, 12 -14 which may explain, in part, the differences we observed. The associations observed also suggest that higher patient volume and non-urban practice location may influence the adoption of expanded colorectal cancer screening for average-risk patients. Clinician recommendation is a key driver of patient adherence to CRC screening recommendations 18 ; thus, said recommendations are likely to differ according to the clinician and practice factors we have identified which may result in greater CRC screening uptake among populations in non-urban areas receiving care in high-volume practices and from GI specialists.
Further research is needed to gain insights into the public health implications of expanding average-risk colorectal cancer screening to younger populations. As the evidence base to inform recommendations continues to evolve and as recommending bodies and clinicians continue to grapple with the lack of consensus and incomplete evidence upon which to make recommendations, it is vital to track provider preferences and practices, which strongly influence patients’ CRC screening decisions.
A limitation of this research is the relatively low survey completion rate. While consistent with the generally lower response rates observed in clinician surveys and overall declining response rates to clinician surveys, our low completion rates may introduce bias and limit the generalizability of our findings. 20 -22 Another limitation of our research is the cross sectional design which precludes making assessment of causal relationships.
Despite these limitations, our analyses provide timely insight into perspectives of clinicians on lowering the CRC screening initiation age for average risk adults to 45 years. Our data point to divergent perspectives by specialty practice and emphasize the need for strong collaboration between primary care clinicians (who order the majority of tests for CRC screening) and gastroenterologists (who have been more actively involved in discerning the pros and cons of earlier screening) to define and discuss the public health implications of earlier-onset colorectal cancer screening, to ensure that patients receive consistent messaging from their healthcare providers and optimal evidence-based care.
Footnotes
Acknowledgments
Author's Note
Philip Parks is now affiliated with Cardinal Health, Boston, MA, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lila J. Finney Rutten serves as a consultant to Exact Sciences through a contracted services agreement between Exact Sciences and Mayo Clinic; Philip Parks was employed by Exact Sciences at the time of manuscript development; Emily Weiser is employed by Exact Sciences; Joan M Griffin has a grant with Exact Sciences to consult on the development of a screening protocol for colorectal cancer screening; Paul J Limburg serves as Chief Medical Officer for Screening at Exact Sciences through a contracted services agreement with Mayo Clinic. Other authors have no conflicts to disclose.
Ethical Approval
The study was reviewed and deemed exempt by the National Opinion Research Center (NORC) IRB (#19.09.15NF).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Exact Sciences. The funding agreement ensured author independence when developing the survey questions, interpreting and analyzing the data, and writing and submitting the publication.
Statement of Human and Animal Rights
The study involved minimal risk to participants and was conducted in accordance with the National Opinion Research Center (NORC) IRB standards for ethical research.
Statement of Informed Consent
Consent was implied by completion of survey by respondents for their anonymized information to be published in this article.
