Abstract
Introduction
The decline in mortality and increase in cure rates following distal gastrectomy have led to a growing focus on patient quality of life (QoL). This study aims to compare QoL outcomes between uncut Roux-en-Y (URY) and Billroth-II with Braun anastomosis (BIIB) reconstructions after totally laparoscopic distal gastrectomy (TLDG), and to identify the preferred surgical approach.
Materials and Methods
From May 2017 to May 2019, a prospective randomized controlled trial (RCT) enrolled 101 gastric cancer patients undergoing TLDG, who were randomly allocated to either URY or BIIB reconstruction. The primary endpoint evaluated the superiority of URY in postoperative QoL at 24 months, as quantified by composite scores from the validated QLQ-C30 version 3.0 and QLQ-STO22 instruments.
Results
After TLDG, compared with BIIB, the URY group had lower EORTC QLQ-C 30 3.0 nausea and vomiting scores at 12 (3.6 (3.3) vs 8.3 (5.3)) and 24 (2.7 (3.4) vs 7.5 (4.2), P < 0.001) months; and fewer reflux symptoms in QLQ-STO 22 at 12 (6.7 (8.9) vs 13.7 (16.1)), and 24 months (4.7 (5.9) vs 12.5 (10.7), P < 0.05). Meanwhile, patients who underwent URY also had better Gastrointestinal Symptom Rating Scale scores (P < 0.01), and shorter frequency and duration of bile reflux at 3, 12, and 24 months (P < 0.01); shorter Bilitec monitoring for bilirubin aspiration at 3 (P = 0.0032), 12 (P = 0.0004), and 24 months (P = 0.042); and lower rates of bile reflux gastritis at 3, 12, and 24 months (P < 0.001). The mean pH of morning gastric fluid was obviously lower in the URY group at 1∼7 days postoperatively (P < 0.001).
Conclusion
URY reconstruction after TLDG effectively reduced short-term bile reflux and improved patients’ QoL compared to BIIB reconstruction.
Plain Language Summary
Better Quality of Life After Stomach Surgery: Uncut Roux-en-Y Outperforms Billroth II with Braun.
Compared to Billroth II with Braun (BIIB), the Uncut Roux-en-Y (URY) method causes fewer digestive problems. Patients who underwent the URY procedure reported better long-term comfort and quality of life.
Keywords
Introduction
According to 2025 cancer statistics, approximately 1 million gastric cancer (GC) cases are newly diagnosed, and 769 000 of these deaths worldwide in 2025. 1 GC ranked fifth in incidence and fourth in mortality among all malignant tumors in the world, with especially high rates in eastern and south-central Asia, and eastern Europe. 2 A comprehensive treatment strategy that includes surgery remains the first-line treatment for GC. Depending on GC stage, patients require different degrees of lymph node dissection 3 followed by digestive tract reconstruction after gastrectomy. 3 The type of reconstruction is tightly associated with patients’ quality of life (QoL).4,5 Postoperative dumping syndrome, bile reflux symptoms, and malnutrition are considered risk factors for QoL in patients with GC.6,7 According to the 2018 Japanese GC treatment guidelines, distal gastrectomy + D1/D2 lymph node dissection is still the standard approach for distal GC. 8 However, with the increasing early diagnosis rate of GC and the growing minimally invasive surgery, the improvement of patients’ QoL is a project for us.
As one of the most optimal surgical procedures for early GC (EGC), LDG is primarily applied in cases where GC affects the middle or lower-third of the stomach. 9 The pyloric portion is removed in LDG, and increased gastrointestinal bile reflux can seriously reduce patient QoL and increase the risk of residual GC (RGC).10-12 There is still tremendous controversy regarding which digestive tract reconstruction procedure should be performed after LDG.4,13
Billroth I reconstruction is extensively used for physiological conformity and simplicity of procedure. 14 However, it is not appropriate for every patient due to hypertonic anastomosis and a relatively high GC recurrence rate.14,15 Conversely, the physiological anatomy of the stomach is altered during Billroth II (B-II) reconstruction, which predisposes patients to postoperative alkaline reflux gastritis. 16 B-II outcomes can be improved with the addition of a Braun anastomosis (BIIB), but treatment is still unsatisfactory. 17 Roux-en-Y reconstruction has advantages in preventing bile reflux gastritis, nevertheless patients may develop Roux retention syndrome caused by the blockage of nerve impulses emanating from the duodenal pacing point. 18 URY reconstruction is a modified BIIB procedure which was first previously reported by van Stiegman and Goff in 1988. 19 URY reconstruction is theoretically an advantageous surgical procedure because the jejunal input collaterals of the gastro-jejunal anastomosis are closed by URY reconstruction, so refluxed bile and pancreatic fluid are not allowed to penetrate the residual stomach. 20 Moreover, nerve impulses from the duodenal pacing point are uninterruptedly conducted by the jejunum, reducing the incidence rates of alkaline reflux gastritis and Roux retention syndrome. 21 Although the URY reconstruction has been broadly performed in clinical practice after TLDG, the procedure remains controversial, and no uniform standard has been developed. 22
Zhang et al 23 evaluated residual gastric function and QoL after URY reconstruction and conducted a within-group comparative study, and Wang et al 24 compared BIIB and URY reconstruction from the perspective of bile reflux. However, no study has directly compared patient QoL after LDG with BIIB vs URY. Based on the foregoing, we conjecture that URY reconstruction is superior to BIIB reconstruction in preventing bile reflux and improving patients’ QoL. This randomized clinical trial was performed to assess differences in bile reflux and patient QoL with the aim of identifying an optimal reconstruction procedure after LDG for GC.
Materials and Methods
Patients
This single-center prospective randomized controlled trial compared QoL outcomes between BIIB and URY reconstructions following TLDG for early gastric cancer. From May 2017 to May 2019, 192 consecutive patients undergoing TLDG at our institution were screened. After excluding 47 patients (34 due to altered surgical plans, 3 for unresectable disease, and 5 declining participation), 103 eligible participants were randomized 1:1 to either URY (n = 52) or BIIB (n = 51) using computer-generated block randomization (block sizes 4 and 6) by an independent statistician. Opaque sealed envelopes were opened by operating room nurses only after confirming resectability post-D2 lymphadenectomy to ensure allocation concealment.
Blinding and Outcome Assessment
Given the inherent impossibility of blinding surgeons or patients to surgical techniques, we implemented partial blinding: postoperative assessors, endoscopists, and data analysts remained masked to group allocation throughout follow-up. Patients self-completed QoL questionnaires without surgeon involvement to minimize observer bias. All questionnaires, data collection, and statistical analyses were performed by separate research teams to maintain methodological rigor. This design aligns with CONSORT guidelines for surgical RCTs where full blinding is impractical.
Preoperatively, all patients received an endoscopic biopsy for histological confirmation of gastric adenocarcinoma. Meanwhile, according to the Japanese Guidelines for the Treatment of GC, 2018, 5th Edition 25 their clinical stage was evaluated by endoscopic ultrasonography and abdominal enhancement computed tomography. With the endoscope, two titanium clips (EZ Clip, HX-610-135 L, Olympus, Japan) were positioned at the upper and lower 2 cm of the cancerous lesion for positioning, which facilitated tumor excision.
Inclusion criteria were: gastroscopy - and histopathological biopsy-confirmed adenocarcinoma of the stomach; patient age between 18 and 75 years; pre-operative physical status rating at 0/1 by the Eastern Cooperative Oncology Group (ECOG); American Society of Anaesthesiologists (ASA) grade I-III; tumor lesions in the antrum, angle or lower body of the stomach; no metastases remotely or peripheral organ invasion; no previous history of other malignant tumors and stomach surgery; and informed consent obtained from patients and/or their family members.
Exclusion criteria were: preoperative history of radiotherapy, chemotherapy, or stomach surgery; significant organ abnormalities; severe cardiopulmonary, renal, or hepatic disease with prolonged hospitalization; non-primary or recurrence of GC; patients with mental illness not suitable for study enrolment; and no informed consent.
This trial was registered with the China Clinical Trials Registry on 9 February 2017. Registration number: ChiCTR-INR-17010594. Furthermore, this trial was also approved by the Ethics Committee of the Lanzhou University Second Hospital (2017A-024). A preoperative informed consent form was signed by participating patients and reaffirmed at the follow-up within 24 months of surgery. The reporting of this study conforms to the CONSORT statements. 26
Surgery Procedure
TLDG with lymph node dissection
All operations were performed by two experienced gastrointestinal surgeons (each with >200 TLDG cases) following the 2018 Japanese GC Treatment Guidelines. 25 Standardized D2 lymphadenectomy (stations 1, 3, 4, 5, 6, 7, 8a, 9, 11, 12a ± 14v) was completed, with duodenal transection along the greater-to-lesser curvature using linear staplers. Approximately one-third of the proximal stomach was preserved during resection. To minimize learning curve effects, both surgeons had performed ≥20 URY and BIIB reconstructions pre-trial. In the BIIB group, we created a Billroth-II gastrojejunostomy with a 15-cm Braun enteroenterostomy distal to the anastomosis to reduce afferent loop syndrome and bile reflux. For URY reconstruction, we performed gastrojejunostomy and jejunojejunostomy while stapling (but not dividing) the afferent limb to prevent reflux, preserving myoneural continuity to avoid Roux stasis. All procedures adhered to institutional protocols for standardization.
URY reconstruction
Using the Treitz ligament as a marker, an appropriate length of silk thread (10 cm) was used to measure 25 cm towards the distal jejunum, where a hole was punched.
27
The greater curvature of the stomach was aligned opposite the jejunal input collaterals, where the jejunum and the residual stomach were lifted with the posterior wall aperture on the side of the greater curvature. And the posterior wall of the greater curvature of the residual stomach was incised, where the jejunum and the residual stomach were lifted. Using the entry hole internally, the jejunum and the proximal third of the remnant stomach were anastomosed with a linear stapling device (Echelon 60-3.5, Ethicon Endo-Surgery, Cincinnati, OH, USA). After that, the Common Entrance was closed (Figure 1A-B, Figure S1A-B). Using the silk thread, we measured 15 cm from the input side of the gastrointestinal anastomosis and 25 cm from the output side of the gastrojejunal anastomosis, where we performed a lateral (Braun) anastomosis of the distal and proximal jejunum with a linear stapling device (Figure 1C-D, Figure S1C-D). Finally, a proprietary closure device (Covidien GIA8038S, Medtronic, Minneapolis, MN, the United States of America) was used to close (not cut) the jejunal lumen at the 5-cm distance from the gastrojejunal anastomosis using a laparoscopic articulating head linear closure (Figure 1A-B, Figure S1B and D).
28
Reconstruction Procedures Following Totally Laparoscopic Distal Gastrectomy (TLDG). (A, B) Schematic (A) and Key Procedural Steps (B) of Uncut Roux-en-Y (URY) Reconstruction. (C, D) Schematic (C) and Key Steps (D) of Billroth II With Braun (BIIB) Reconstruction. Red Arrows Indicate Gastric Juice Flow, Blue Arrows Denote Intestinal Peristalsis, and Green Arrows Represent Bile/Pancreatic Juice Flow. The Red Clip Marks the Uncut Staple Line in URY
BIIB Reconstruction
The gastrojejunostomy and lateral jejunojejunostomy (Braun anastomosis) were performed as for the URY reconstruction, However, in the BIIB reconstruction, the Roux limb input at 5 cm from the gastrojejunum was not closed. (Figure 1B, Figure S1B).
Learning Curve Analysis by CUSUM Method
We performed cumulative sum (CUSUM) analysis to quantitatively evaluate the learning curves for total operative time, reconstruction time, and intraoperative blood loss. All cases were chronologically ordered from the first procedure performed. The CUSUM for each case (n) was calculated as follows:
QoL Assessment
Patients’ QoL data at preoperative, 3, 12, and 24 months postoperative were collected with the European Organisation for Research and Treatment of Cancer (EORTC) QLQ-C30 Chinese Version 3.0 (Table S1) and QLQ-STO22 (Table S2). The former consists of 30 items including 1 general health status scale, 3symptom scales (pain, fatigue, nausea, and vomiting), 5 functional scales (physical, role, cognitive, emotional, and social), and 6 individual items (dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties). Except for the final two items (29, 30), which were rated from 1 to 7, other items were scored from 1 to 4 (in the range of “never,” “a little,” “often,” “frequently”). The second scale consists of 9 domains including 4 single items (dry mouth, taste, body image, and hair loss) and 5 symptom scales (dysphagia, pain, restricted feeding, reflux symptoms, and anxiety) for a total of 20 indicators. Scores in the functional and general health status domains of the questionnaire were higher, indicated better functional status and QoL for patients with GC after TLDG. whereas for the symptom domain a higher score indicated increased symptoms or problems (poorer QoL). The preoperative 1-day QoL was the baseline. Patients’ QoL and disease progression were collected at 3, 12, and 24 months postoperatively by follow-up telephone, email, and outpatient review.
Endoscopic Assessment
We performed gastroscopies of participating patients preoperatively and postoperatively at 3, 12, and 24 months; the results were interpreted by experienced endoscopists. The endoscopic “residue, gastritis, bile” (RGB) classification proposed by Kubo et al 29 was used to assess residue, gastritis level, and bile reflux of the residual stomach (Table S3), with a higher RGB score indicating worse symptoms and signs.
Gastrointestinal Symptom Scoring
The Gastrointestinal Symptom Rating Scale (GSRS) was used to score patients’ gastrointestinal symptoms preoperatively and 3, 12, and 24 months postoperatively. The GSRS contains 5 major subsets of abdominal pain, diarrhea, constipation, dyspepsia and reflux, with 3 subsets for every item except reflex, which contained 4 items. The score was taken as the average of each subset and ranged from 1 to 7, with 1 indicating “not at all” and 7 meaning “extremely severe.” The participants in this survey were not aware of the final symptom scores (Table S4).
Monitoring of pH and Bile
To monitor pH changes and bile reflux in patients after TLDG, a portable pH detector (Digitrapper MK-III; Synectics Medical, Stockholm Sweden) with electrodes calibrated with standard buffers (pH = 7 and 1) was transversally inserted 5 cm above the lower esophageal sphincter (LES). The reference electrode and catheter were positioned below the glabellar area for fixation at the cheek, and a cassette pH recorder was connected with a computer to collect data with gastrointestinal symptoms before and after meals and during changes in body position (upright/lying). BioVIEW v.5.3.4 software (Sandhill Scientific, Highlands Ranch, CO, USA) was installed to estimate the lower esophagus 24-h reflux frequency and reflux duration (pH > 7). Bilitec 2000 probes were also placed 5 cm above the LES to monitor bile reflux and calculate the percentage of time with bilirubin absorbance > 0.25, which was analyzed by Polygram 98 software (Synectics Medical, Stockholm, Sweden). The pH values of patients’ morning gastric juice at 1 day preoperatively and 1-7 days postoperatively were measured by a calibrated pH pen (SX610, Shanghai Youyi Instruments Ltd, Shanghai, China).
Stomach Specimen Handling
Postoperatively, the residual stomach specimen with surrounding tissue structures was removed, and the lymph nodes were stripped and counted on the self-designed measuring board according to the gastric lymph node groups (Figure S2). The stomach wall was incised along the side of the greater curvature longitudinally to expose the tumor, which was measured with a scale. The gross specimens and lymph nodes were separately collected in specimen bags and sent to pathology. The specimens and lymph nodes were fixated in 4% formalin liquid, stained with hematoxylin and eosin, sectioned, and observed under the microscope. Tumor differentiation and positive lymph nodes were recorded.
Study Endpoints and Follow up
The primary endpoint was superiority in QoL at 24 months postoperatively, assessed using validated Chinese versions of the EORTC QLQ-C30 (v3.0) and QLQ-STO22 questionnaires. Trained blinded interviewers administered these surveys at baseline, 3, 6, 12, and 24 months. Secondary endpoints included: (1) endoscopic evaluation of reflux esophagitis/gastritis (RGB classification at 12/24 months); (2) 24-h esophageal pH monitoring (mean pH/reflux time percentage at 12 months); (3) nutritional parameters (body weight, albumin, hemoglobin); and (4) surgical outcomes (operative time, blood loss, 30-day morbidity/mortality per Clavien-Dindo criteria). Long-term oncologic outcomes (5-year DFS/OS) were tracked separately. Standardized follow-up at 3, 12, and 24 months included clinical examinations, laboratory tests, and symptom assessments, with protocol-mandated endoscopy at 12/24 months. To minimize attrition bias, patients lost to follow-up after ≥3 contact attempts were censored at their last visit, with their latest data carried forward for intention-to-treat (ITT) analysis.
Sample Size
With reference to the wang et al report, 30 we set the incidence of reflux gastritis in the control group (BII B) at 45%. A non-inferiority test was used with a 20% boundary (α = 0.05, 1-β = 0.80, means an efficacy of 80%). A sample size of 148 participants (74 participants per group) was calculated in PASS 15.0 (AQ5; NCSS, LLC, Kaysville, UT, USA). Given a 15%-20% drop-out rate and clinical statistical aspects, no less than 190 participants (95 participants per group) were recruited in total.
Statistical Analysis
Statistical analyses were performed using SPSS version 21.0 (IBM Corp, Armonk, North Castle, NY, USA) and R software (version 4.1.3; R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were reported as mean ± standard deviation or median (interquartile range) based on distribution normality, with comparisons made using Student's t-test or Mann-Whitney U test, respectively. Categorical variables were presented as counts (percentages) and analyzed using χ2 tests or Fisher’s exact tests for small cell sizes. Longitudinal quality-of-life measurements were evaluated through linear mixed-effects models incorporating subject-specific random effects, with Bonferroni-adjusted post hoc comparisons when significant main effects were identified. For repeated-measures analyses, we verified sphericity assumptions using Mauchly’s test (P < 0.05 indicating violation), applying Greenhouse-Geisser corrections when appropriate. Both intention-to-treat (with last observation carried forward for missing data) and per-protocol analyses were performed to ensure robustness. Learning curve proficiency was determined through CUSUM analysis, comparing linear, quadratic, and cubic regression models (selection based on significance P < 0.05 and maximal R 2 values) to identify inflection points in operative time and blood loss trajectories. A two-sided α level of 0.05 defined statistical significance throughout the study.
Results
Clinicopathological Characteristics
Among the 103 patients enrolled, 2 cases in group of BIIB had URY anastomosis due to personal choice. Additionally, 2 cases in the URY group ultimately underwent BIIB reconstruction because of financial and patient requirements. There was 1 recurrence in each of the BIIB and URY groups at 24 months postoperatively. Unfortunately, an individual in the BIIB group died of multi-organ dysfunction syndrome due to postoperative stress. A total of 103 patients (in groups BIIB 51 and URY 52) were recruited with complete clinicopathological, bile reflux, gastric pH, and QoL. Subsequently, both groups were followed for 2 years. During the first year, one patient in the BIIB group and two patients in the URY group were lost to external hospitalization and death due to tumor metastases. One patient was excluded from each group for refusing to fill out the QoL questionnaire during the second year. The remaining patients completed the study (Figure 2). Given the design of the trial project and experimental staffing, we followed the QoL of GC patients within 24 months after surgery. CONSORT 2025 Flow Diagram. QoL, Quality of Life
Clinicopathological Characteristics of Enrolled Patients

Values in parentheses are percentages unless indicated otherwise.
aPearson’s chi-square test or Fisher’s exact test, except.
bmedian (range).
cMann-Whitney U test.
dmean(s.d.).
eStudent's t test.
Surgical Background and Complications
Patient Surgical Information and Complications

Values in parentheses are percentages unless indicated otherwise.
amean(s.d.).
bStudent's t test.
cmedian (range).
dMann-Whitney U test.
ePearson’s chi-square test or Fisher’s exact test.
Learning Curve Analysis
Initial operative metrics revealed clinically significant differences between the two reconstruction techniques. The URY group required significantly longer total operative time (184.8 (25.8) vs 170.3 (21.4) minutes, P = 0.084) and digestive tract reconstruction time (43.9 (9.2) vs 35.9 (7.9) minutes, P = 0.0037) than the BIIB group, while maintaining comparable blood loss volumes (178 (43.3) vs 177.3 (47.1) mL, P = 0.763, Table 2). Furthermore, all three parameters exhibited progressive improvement with increasing surgical experience, as evidenced by the downward trends in operative time (Figure 3A), reconstruction time (Figure 3B) and blood loss (Figure 3C). These consistent patterns across multiple performance metrics suggest that both techniques demonstrate measurable learning effects, albeit with potentially different learning trajectories, which were subsequently quantified through CUSUM modelling. Learning Curve Analysis Comparing URY and BIIB Reconstruction Techniques. (A-C) Depict the Case-By-Case Operative Metrics (Total Surgery Time, Reconstruction Time, and Blood Loss) With Scatter Plots (Orange Circles = URY; Teal Triangles = BIIB) and Corresponding Trendlines. (D-F) Present the CUSUM-Derived Learning Curves, where Red Diamonds Mark the Transition Points Between Learning (Left of Marker) and Proficiency Phases (Right of Marker). Model Fits are Shown as: Translucent Lines (Linear), Dashed Lines (Quadratic), and Solid Lines (Cubic), With the Optimal Fit (Highest R
2
) Emphasized
Learning curve analysis revealed distinct proficiency thresholds between the URY and BIIB reconstruction techniques. Cubic regression models provided the best fit for total operative time (URY: y = 0.005x 3 − 0.867x 2 + 30.216x + 25.084, R 2 = 0.9006; BIIB: y = 0.005x 3 − 0.686x 2 + 21.254x + 24.211, R 2 = 0.934), with inflection points at 18 and 17 cases, respectively (Figure 3D). Similar patterns were observed for digestive tract reconstruction time, with 18 cases required for URY (y = 0.002x 3 − 0.227x 2 + 7.529x − 13.402, R 2 = 0.7576) and 14 cases for BIIB (y = 0.004x 3 − 0.322x 2 + 6.594x−22.761, R 2 = 0.8376) to reach proficiency (Figure 3E). Intraoperative blood loss demonstrated the greatest divergence: URY required 22 cases (y = 0.009x 3 − 1.264x 2 + 42.398x − 65.478, R 2 = 0.8405) to reach steady-state performance, compared to just 13 cases for BIIB (y = 0.012x 3 − 0.938x 2 + 15.672x + 27.326, R 2 = 0.6097) (Figure 3F). Collectively, these findings indicate that surgeons require approximately 22 cases to overcome the learning curve for URY reconstruction, whereas BIIB stabilization occurs earlier (at 17 cases), primarily due to its less technically demanding anastomotic configuration.
Our phase-specific analysis revealed significant improvements in operative efficiency for both techniques. For URY reconstruction, the learning phase showed substantially longer operative times compared to the proficiency phase: total time (200.2 (22.8) vs 173.5 (21.9) min, P < 0.001), reconstruction time (45.1 (10.2) vs 39.7 (7.8) min, P = 0.046) and blood loss (median 190 vs 177.3 millilitres, P = 0.006). Similarly, the BIIB technique showed reductions in operative time during the learning-to-proficiency phase (187.3 (31.7) vs 161.8 (17.9) min, P = 0.006, Table S5). Notably, URY reconstruction consistently required longer times than BIIB reconstruction during both the learning (45.1 (10.2) vs 36.8 (9.9) min, P = 0.014) and proficiency (39.7 (7.8) vs 33.8 (6.6) min, P = 0.002) phases. However, this did not result in differences in lymph node retrieval, hospital stay, or complication rates (all P > 0.05, Table S5). These findings confirm the technical complexity of URY while demonstrating its comparable safety profile to BIIB reconstruction throughout the learning continuum.
QoL and Clinical Course
The preoperative EORTC QLQ-C 30 3.0 questionnaires showed that the BIIB and the URY groups have no substantially different QoL. Although the URY group had better sleep outcomes before surgery (16.5 (10.3) vs 18.4 (18.1); P = 0.534), no differences were observed at the preoperative and 3, 12, 24-month postoperative stages (Table S6; Figure 4A; P > 0.05). Nausea and vomiting were significantly superior in the URY group vs the BIIB group at 12 months post-operatively, with an overall mean difference of 5 points (3.6 (3.3) vs 8.3 (5.3), P = 0.0001. Interestingly, this difference persisted at 24 months postoperatively (2.7 (3.4) vs 7.5 (4.2), P = 0.00001, Table S6, Figure 4A). Compared with the BIIB group, patients who underwent URY had higher overall health status scores at 24 months postoperatively, with an average overall difference of 3 points (83.6 (10.2) vs 80.3 (15.9), P = 0.213, Table S6, Figure 4A). Although the URY group exhibited favorable trends across multiple domains—including global health status, dyspnea, and fatigue—these differences failed to reach statistical significance (P > 0.05). The URY and BIIB groups did not differ significantly in terms of additional scales such as physical functioning, constipation, diarrhea, and economic hardship (Table S6; Figure 4A, Figure S3A, P > 0.05). Longitudinal Changes in Quality of Life (QoL) and Nutritional Indicators After Distal Gastrectomy With BIIB or URY Reconstruction. (A) Mean EORTC QLQ-C30 Scores (Chinese Version) Over 24 Months for Functional and Global Health Domains (Higher Scores = Better QoL). Symptom Scales and Single-Item Scores (Lower Scores Indicate a Better QoL). (B) EORTC QLQ-STO22 Symptom Domain Scores (Higher Scores Indicate Poorer QoL). (C) Nutritional Parameter Trends Over 24 Months. Data are Presented as Mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001.
The poster-operative QLQ-STO 22 questionnaire revealed that compared to the BIIB group, the URY group had apparently lower rates of reflux symptoms at 12 months (6.7 (8.9) vs 13.7 (16.1), P = 0.031), and 24 months (4.7 (5.9) vs 12.5 (10.7), P = 0.0001, Table S7, Figure 4B). However, other areas included dysphagia, pain, dietary restrictions, dry mouth, altered taste, anxiety, body image and hair loss were not greatly different between the two groups (Table S7, Figure 4B, Figure S3B, P > 0.05).
Clinical factors of patients in both groups were collected. Although the percentage of neutrophils in the postoperative period was higher in the URY group than in the BIIB group at 3 months (P = 0.399), 12 months (P = 0.908), and 24 months (P = 0.984), it was not statistically significant (Table S8; Figure 4C). Meanwhile, we found neither total protein, albumin, albumin-globulin ratio, nor total cholesterol, calcium, and hemoglobin differed between the two groups (Table S8; Figure 4C, Figure S3C, P > 0.05).
Endoscopic Scores
With the endoscopy, we found that gastric residues, gastritis (P = 0.49) or and bile reflux (P = 0.156) were not statistically different between the BIIB and URY groups preoperatively, as well as residues at 3 (P = 0.46), 12 (P = 0.417), 24 months (P = 0.891) postoperatively (Table S9; Figure 5). However, compared with the BIIB group, the URY group demonstrated significantly lower incidence rates of bile reflux at 3 months (P = 0.0009) and 12 months (P = 0.0138), as well as reduced gastritis at 12 months (P = 0.039) and 24 months (P = 0.042) postoperatively (Table S9; Figur 5B–D, Figure S4A-F). Repeated assessments consistently showed that the BIIB group had markedly higher overall incidence of postoperative gastritis and bile reflux across all time points (P < 0.05; Figure 5A–D). Endoscopy Findings and Statistics by Percentages of RGB Scores. (A) Preoperative and (B-D) Postoperative (3/12/24 Months) Endoscopic RGB Grading Percentages are Shown. Data Were Analyzed Using Pearson’s Chi-Square or Fisher’s Exact Test (*P < 0.05, **P < 0.01, ***P < 0.001).
Comparison of Reflux Esophagitis in the Postoperative Time Period

Values in parentheses are percentages.
aLos Angeles Classification System 31 .
bPearson’s chi-square test or Fisher’s exact test.
Additionally, Inter-rater reliability between endoscopists was robust across all assessed parameters. Cohen’s Kappa and intraclass correlation coefficient (ICC) analyses demonstrated moderate to substantial agreement for endoscopic findings: esophagitis grade (κ = 0.556, 77.7% agreement), gastric residue (κ = 0.707, 88.2% agreement), gastritis (κ = 0.637, 72.1% agreement), and bile reflux (κ = 0.721, 90.3% agreement). Notably, bile reflux exhibited the highest concordance (substantial agreement, κ > 0.70). These results confirm strong consistency in categorical assessments between independent evaluators (Table S10).
Comparison of Postoperative Clinical and Bile Reflux Factors

aValue is expressed as %, preoperative level adjusted to 100%.
bmean (s.d.).
cGastrointestinal symptom rating scale.
dStudent's t test.
eProbe was inserted into the lower esophagus.
fProbe was inserted into the lower esophagus with the percentage of time absorbance > 0.25.

Longitudinal Comparison of Gastrointestinal Symptoms and Reflux Parameters Between URY and BIIB Reconstruction. (A) Gastrointestinal Symptom Rating Scale (GSRS) Scores at 3, 12, and 24 Months Postoperatively. (B-C) 24-H pH Monitoring Parameters: Reflux Frequency (B) and Duration (C) at 3 and 12 Months. (D) Bilitec Monitoring Data at 3, 12, and 24 Months (*P < 0.05, **P < 0.01, ***P < 0.001).
Comparison of Postoperative Gastric Fluid pH Value
Morning gastric fluid was extracted 1 day preoperatively and 1-7 days postoperatively, and pH values were measured with the pH pen. Repeated measures ANOVA on pH at different time points revealed that the data did not pass the Mauchly sphericity test (W = 0.1343, P < 0.001), so multivariate tests were performed to compare the postoperative pH values of the two groups. The results suggested that the pH trends were not completely identical in the two groups on various days (Table S11, F = 35.152, P < 0.001).
The results of the ANOVA also showed that URY reconstruction was superior to BIIB reconstruction in terms of efficacy in reducing bile reflux (Table S12, F = 1639.098, P < 0.001), as the pH levels for the two reconstructions were substantially different between 1 day preoperatively and 1-7 days postoperatively.
A simple effect analysis and intergroup t-tests were performed on the pH values of gastric juice at different time points between the two groups. Except for the 1 day before surgery, the pH level of the URY reconstruction was appreciably lower than the BIIB reconstruction from 1 to 7 days after surgery (Table S13-S14, Figure 7, P < 0.001). Perioperative Stomach pH. Changes in the pH of Gastric Fluid in the Residual Stomach at 08:00 on the Day before Surgery and From One to Seven Days Postoperatively. Postoperatively, the pH of Gastric Juice was Below 7.00 in the URY Group. In Contrast, all Patients in the BIIB Group had pH Values Above 7.00. ^1 day Preoperatively
Afterwards, we performed two-by-two comparisons of overall and subgroup pH at each time point, which showed significant differences in pH between 1 day preoperatively and 1-7 days postoperatively (Table S15, P < 0.001). However, the differences in pH values among the groups at time points from 1 to 7 days after surgery were not statistically significant (Table S15, P > 0.05).
After a median follow-up of 5 years, patients undergoing URY reconstruction demonstrated comparable survival outcomes to those receiving conventional BIIB anastomosis. The Kaplan-Meier analysis revealed nearly identical overall survival curves (median OS: 2.1 vs 1.8 years; HR 0.93, 95% CI 0.59-1.46; log-rank P = 0.72, Figure 8A) and disease-free survival patterns (median DFS: 1.6 vs 1.3 years; HR 0.93, 95% CI 0.60-1.46; P = 0.765, Figure 8B) between the two groups. Importantly, the overlapping survival trajectories and consistent hazard ratios across all time points confirm that the technically distinct URY procedure maintains equivalent oncologic safety to the standard BIIB approach, providing surgeons with an effective alternative reconstruction option without compromising long-term outcomes. Kaplan-Meier Survival Analysis Comparing URY and BIIB Groups. (A) Overall Survival and (B) Progression-Free Survival Curves for 52 Patients Undergoing URY Reconstruction Versus 51 Patients Receiving Conventional BIIB Anastomosis, With Median Follow-Up of 5 Years. The Overlapping Curves (HR 0.93 for OS and PFS; Both P > 0.05 by Log-Rank Test) Demonstrate that the Two Procedures Have Comparable Long-Term Outcomes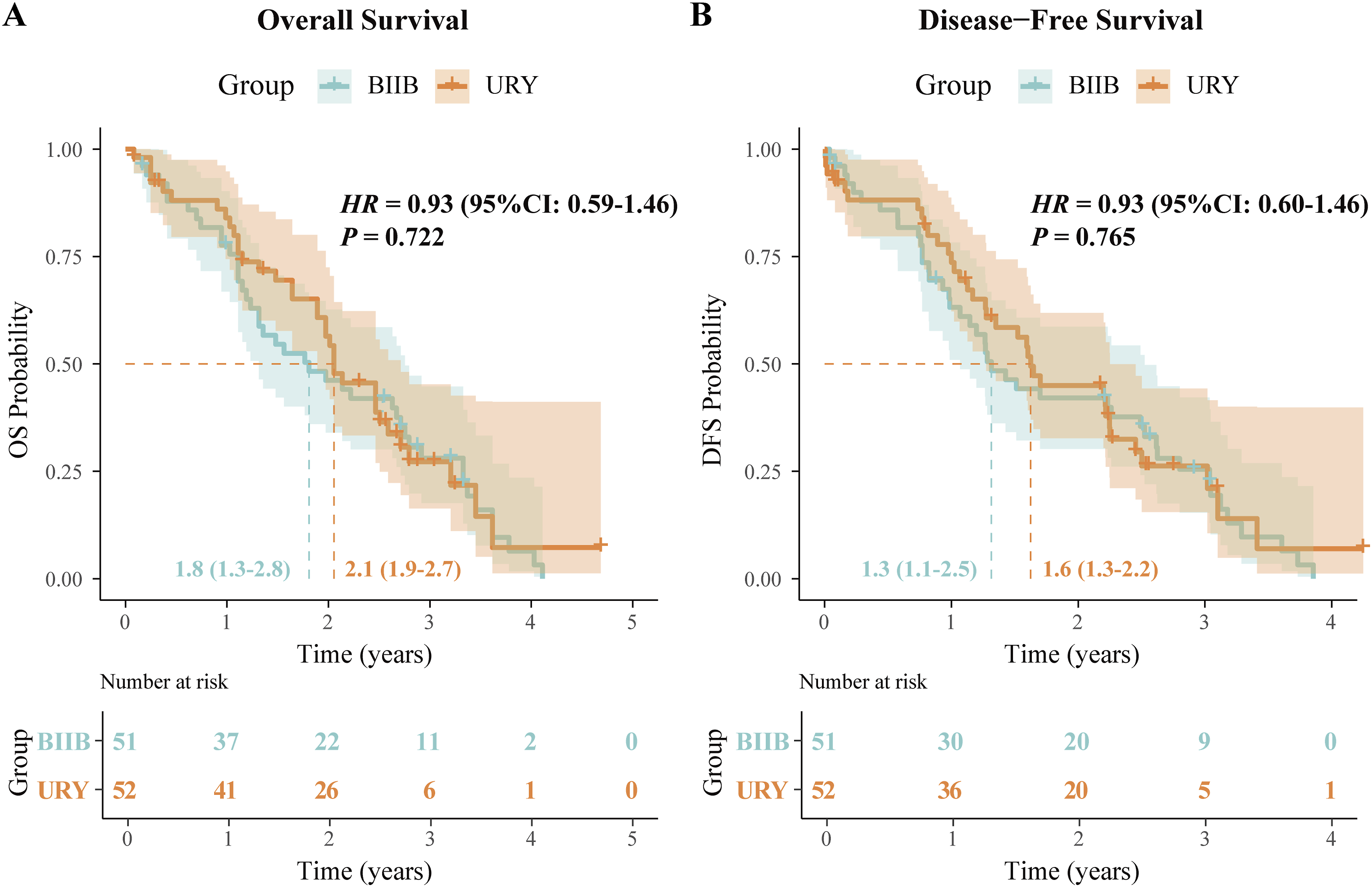
Discussion
With this single-center RCT, we found that patients with GC had significantly higher QoL after URY reconstruction compared to BIIB reconstruction. Additionally, there was less bile reflux into the esophagus and no increased complications after URY compared with BIIB reconstruction. Therefore, URY reconstruction is a reliable and effective surgical procedure.
Whether the patients’ preoperative clinicopathological information or the patients’ operative time, hospital stay, or the time of first exhaust (defecation), there was no statistically significant difference between the two reconstructive procedures. This is broadly consistent with the findings of Wang et al, 24 except for the significantly longer reconstruction time for URY compared to BIIB, which was probably related to the operator’s surgical skills and the patients’ underlying conditions. The complications after surgery showed less difference between the two reconstructive procedures, which corresponds with results reported by Wang et al 24 and Shen et al. 28 The URY reconstruction merely closes rather than severs the local proximal gastro-jejunal anastomosis without disrupting the gastric contents and duodenal secretion flows to the distal jejunum. Moreover, the uncut proximal jejunum retains the original, physical electrical conduction and muscle-stretching capacity without impairing jejunal circulation or neurological dysfunction. 22 In a study of gastrointestinal electromyographic activity and motility in patients after URY reconstruction, Zhang et al 32 found that compared with Roux-en-Y, the URY reconstruction reduced the surgical impact on electromyographic activity such as slow waves, spike potentials, and migrating motor complexes, which helped maintain gastrointestinal motility and c-kit mRNA expression. Subsequent clinical studies33,34 found no complications such as delayed gastric emptying or ileal obstruction in patients after URY reconstruction, which is in agreement with our results.
The QoL questionnaire responses showed that patients who underwent URY reconstruction had a higher QoL than those who received BIIB reconstruction within the short term (2 years), which was mainly reflected in gastrointestinal and reflux symptoms. A randomized controlled study 23 compared the consequences of conventional RY reconstruction with URY reconstruction in patients after laparoscopic total gastrectomy and found no difference in QoL scores 1 month postoperatively, but patients in the URY group had higher QoL scores at 6 and 12 months. However, this was a small trial with a relatively limited evidence level. Another randomized controlled trial by Zhang et al 23 assessed QoL after reconstruction with RY and URY and showed that patients with URY reconstruction had lower dietary restriction scores and higher anxiety scores at 1 month postoperatively. However, patients with URY had lower anxiety scores and higher dry mouth scores than the patients with RY at 3 and 6 months postoperatively. Although there were no significant differences in preoperative nausea and vomiting and reflux symptom scores between the two groups, patients with URY showed lower nausea and vomiting and reflux symptom scores than patients with BIIB at various time points during the 24-month follow-up. The GSRS questionnaire answers also supported this observation. It is possible that properly closed Roux limbs could clearly reduce bile reflux and the occurrence of alkaline reflux gastritis, consequently increasing patient QoL. However, Cui and colleagues 35 compared isoperistaltic URY (iso-URY) and antiperistaltic URY (anti-URY) reconstruction data after LDG and found no differences in short-term complications, nutritional status, or QoL at 1 year postoperatively between the two groups, but endoscopy showed that the anti-URY group had more severe alkaline reflux gastritis. Given that there were minimal differences between the URY and BIIB groups with regard to nutritional parameters in terms of body weight, total protein, albumin, calcium and hemoglobin, we speculate that URY reconstruction might not impact food digestion and absorption or the regulation of gastrointestinal hormones. Given the smaller sample sizes and shorter follow-up periods in most of the above-cited studies, larger investigations with longer follow-up might be required to elucidate any differences.
The RGB score and Los Angeles classification of esophagitis by endoscopy showed a marked improvement in bile reflux and esophagitis of the URY reconstruction. Besides, there was lower frequency and duration of reflux and bilirubin light absorption values in URY patients than in BIIB patients as confirmed by 24-h pH monitoring and Bilitec monitoring. This difference is probably due to the closure of the proximal bile reflux channel in the gastro-jejunal anastomosis during URY reconstruction, which diverted bile and pancreatic fluid from the jejune-jejunal anastomosis. Noh et al 36 suggested that since the URY reconstruction closed rather than severed the Roux limb, it preserved jejunal motility, reduced Roux stasis syndrome, and relieved alkaline reflux gastritis and esophagitis, all of which were extremely favorable to improve QoL and reduce Barrett’s esophagus after surgery.37,38 However, in a study of 20 minipigs with distal gastrectomy and URY reconstruction, Wu et al 39 found a higher occurrence of afferent limb recanalization. They suggested that closure of the afferent limb with a linear stapling device could not reduce the incidence of recanalization or decrease the incidence of alkaline reflux gastritis and esophagitis and subsequently improve QoL. As a corollary, they recommended against URY reconstruction after distal gastrectomy. Given the small number of minipigs and only 1 month of observation, their results require verification.
Interestingly, we found that morning gastric fluid pH was significantly lower in URY patients than in BIIB patients from 1 to 7 days postoperatively; the tendencies of pH changes at various time points also differed between the two groups as indicated by Mauchly’s sphericity test, which was generally consistent with previous studies.24,40 While these findings align with Wang et al 24 reports of preserved gastric acidity after successful Roux limb isolation, they contrast sharply with the more neutral pH range (6.2-6.6) reported by Tanaka et al 40 - a discrepancy likely attributable to technical variations in anastomotic technique. Our use of a reinforced 6-row linear anastomosis clutch, building on Cai et al 41 methodology, achieved complete Roux limb isolation without recanalization events, mirroring the success of Shibata et al 42 transmural reinforcement approach.
Notably, we revealed no statistically significant differences in oncological outcomes between URY and BIIB reconstruction groups, with comparable rates of tumor recurrence, distant metastasis, and survival outcomes (5-year OS; DFS, P > 0.05). The comparable safety profile of URY was achieved through standardized perioperative protocols including comprehensive preoperative optimization, systematic lymphadenectomy (median 32 nodes retrieved), and strict adherence to aseptic and no-touch techniques.
Although our randomized controlled design and complete patient datasets provide robust support for URY reconstruction following distal gastrectomy, there are several limitations that warrant consideration. As this is a single-center study with a modest sample size and a 24-month follow-up period, our findings may not capture long-term complications or QoL trajectories. Although the learning curve analysis showed no significant disparities, potential biases related to surgical technique or patient demographics could limit the generalizability of our results to other settings. Additionally, while valuable, QoL assessments via questionnaire remain susceptible to cultural, educational and emotional confounders. Meanwhile, our study also identified several important practical considerations. While URY reconstruction demonstrated clinical benefits, the procedure required significantly longer operative times, which may increase healthcare costs. Future health economic analyses should evaluate whether these additional investments translate to proportional improvements in long-term outcomes and patient-reported benefits. These limitations emphasize the need for multicenter RCTs with an extended follow-up period to validate our conclusions and evaluate long-term patient satisfaction.
Despite these limitations, our study strengthens the existing body of literature on this topic by directly comparing quality of life (QoL) outcomes following BIIB and URY reconstruction. This distinction has rarely been examined in previous reports. The randomized design and comprehensive data collection strengthen the internal validity of our findings. Future research should prioritize larger cohorts, longer observation periods (>5 years) and standardized QoL metrics to account for socioeconomic and cultural variability. Such efforts will clarify the role of reconstruction techniques in optimizing both oncological and functional outcomes.
Conclusion
The URY reconstruction achieved less bile reflux and better QoL in patients compared to BIIB reconstruction after TLDG. URY reconstruction is a remarkably safe and efficacious procedure.
Supplemental Material
Supplemental Material - Comparing Quality of Life for Billroth II With Braun Versus Uncut Roux-en-Y Reconstruction After Total Laparoscopic Distal Gastrectomy for Gastric Cancer: A Randomized Clinical Trial
Supplemental Material for Comparing Quality of Life for Billroth II With Braun Versus Uncut Roux-en-Y Reconstruction After Total Laparoscopic Distal Gastrectomy for Gastric Cancer: A Randomized Clinical Trial by Xiaojun Li, Qiuya Wei, Jianxing Ma, Luofei Chen, Minxue Chen, Cheng Wei, Boxiong Kang, and Changjiang Luo in Cancer Control
Supplemental Material
Supplemental Material - Comparing Quality of Life for Billroth II With Braun Versus Uncut Roux-en-Y Reconstruction After Total Laparoscopic Distal Gastrectomy for Gastric Cancer: A Randomized Clinical Trial
Supplemental Material for Comparing Quality of Life for Billroth II With Braun Versus Uncut Roux-en-Y Reconstruction After Total Laparoscopic Distal Gastrectomy for Gastric Cancer: A Randomized Clinical Trial by Xiaojun Li, Qiuya Wei, Jianxing Ma, Luofei Chen, Minxue Chen, Cheng Wei, Boxiong Kang, and Changjiang Luo in Cancer Control
Footnotes
Acknowledgments
The authors greatly thank for all of the nurses in the Operating Room, General Surgery Department of the Second Hospital of Lanzhou University for their support.
Ethical Consideration
Ethical approval was granted by the Ethics Committee of Lanzhou University Second Hospital (2017A-024).
Consent to Participate
Written informed consent was obtained from all participating patients.
Consent for Publication
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Authors’ Contributions
Xiaojun Li, Luofei Chen, Minxue Chen and Cheng Wei conducted the questionnaire and data collection. Boxiong Kang and Changjiang Luo performed the surgery and Changjiang Luo designed the research project. Xiaojun Li, Luofei Chen and Qiuya Wei carried out the data collation, software validation and data visualisation. Xiaojun Li wrote the original manuscript for the paper. Changjiang Luo provided methodological guidance and carried out the review and editing.
Funding
This study was supported by the Cuiying Programme of Lanzhou University Second Hospital (grant no. CY2021-BJ-A03), the Natural Science Foundation of Gansu Province (grant no. 24JRRA333), the Chengguan District Science and Technology Project of Lanzhou City (grant no. 2024SHFZ0018) and the Gansu Provincial Traditional Chinese Medicine Research Project (grant no. GZKG-2024-75).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data for this article are available upon request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

